Modulating the Gut Microbiota with Alginate Oligosaccharides In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrates and Strains
2.2. Basal Colon Media
2.3. Single-Strain Growth Experiment
2.4. Faecal Sample Preparation
2.5. In Vitro Gut Model CoMiniGut
2.6. Fermentation Conditions
2.7. SCFA Analysis
2.8. DNA Extraction
2.9. Amplicon Sequencing
2.10. High Throughput Sequencing and Data Treatment
3. Results
3.1. Single-Strain Growth Analysis
3.2. SCFA Production during In Vitro Simulated Colon Passage
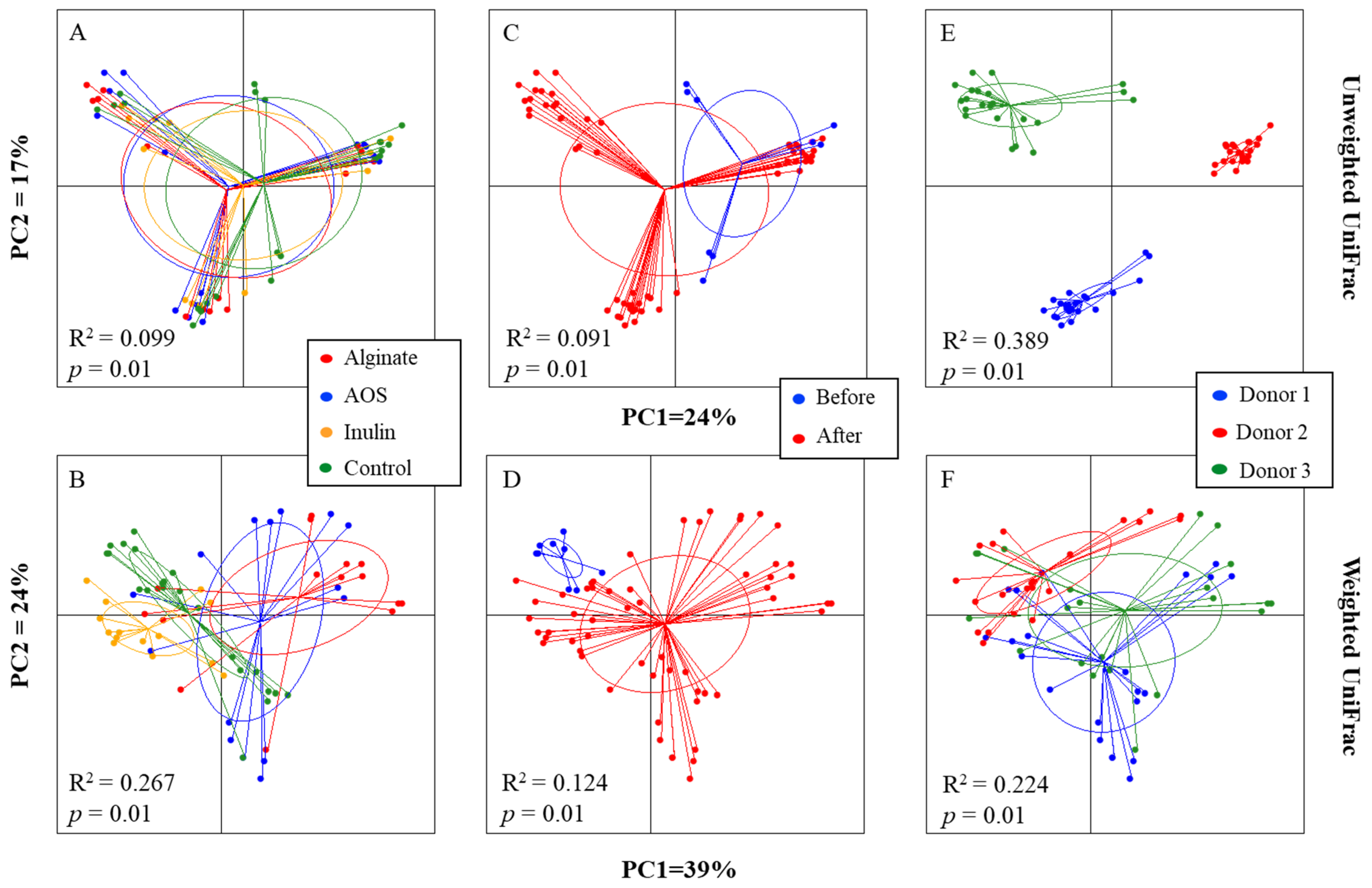
3.3. Changes in the Simulated GM Composition during Gut Model CoMiniGut
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Topolska, K.; Florkiewicz, A.; Filipiak-Florkiewicz, A. Functional Food-Consumer Motivations and Expectations. Int. J. Environ. Res. Public Health 2021, 18, 5327. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Attaye, I.; Warmbrunn, M.V.; Boot, A.; van der Wolk, S.C.; Hutten, B.A.; Daams, J.G.; Herrema, H.; Nieuwdorp, M. A Systematic Review and Meta-analysis of Dietary Interventions Modulating Gut Microbiota and Cardiometabolic Diseases-Striving for New Standards in Microbiome Studies. Gastroenterology 2022, 162, 1911–1932. [Google Scholar] [CrossRef] [PubMed]
- Deschasaux, M.; Bouter, K.E.; Prodan, A.; Levin, E.; Groen, A.K.; Herrema, H.; Tremaroli, V.; Bakker, G.J.; Attaye, I.; Pinto-Sietsma, S.J.; et al. Depicting the composition of gut microbiota in a population with varied ethnic origins but shared geography. Nat. Med. 2018, 24, 1526–1531. [Google Scholar] [CrossRef]
- Neri-Numa, I.A.; Pastore, G.M. Novel insights into prebiotic properties on human health: A review. Food Res. Int 2020, 131, 108973. [Google Scholar] [CrossRef]
- Wang, Z.; Tauzin, A.S.; Laville, E.; Tedesco, P.; Letisse, F.; Terrapon, N.; Lepercq, P.; Mercade, M.; Potocki-Veronese, G. Harvesting of Prebiotic Fructooligosaccharides by Nonbeneficial Human Gut Bacteria. mSphere 2020, 5, e00771-19. [Google Scholar] [CrossRef]
- Stephen, A.M.; Phillips, G.O. Food Polysaccharides and Their Applications; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- August, A.D.; Kong, H.J.; Mooney, D.J. Alginate hydrogels as biomaterials. Macromol. Biosci. 2006, 6, 623–633. [Google Scholar] [CrossRef]
- Brownlee, I.A.; Allen, A.; Pearson, J.P.; Dettmar, P.W.; Havler, M.E.; Atherton, M.R.; Onsoyen, E. Alginate as a source of dietary fiber. Crit. Rev. Food Sci. Nutr. 2005, 45, 497–510. [Google Scholar] [CrossRef]
- Qin, Y.; Jiang, J.; Zhao, L.; Zhang, J.; Wang, F. Applications of Alginate as a Functional Food Ingredient. In Biopolymers for Food Design; Handbook of Food Bioengineering; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: London, UK, 2018; pp. 409–429. [Google Scholar]
- Liu, J.; Yang, S.; Li, X.; Yan, Q.; Reaney, M.J.T.; Jiang, Z. Alginate Oligosaccharides: Production, Biological Activities, and Potential Applications. Compr. Rev. Food Sci Food Saf. 2019, 18, 1859–1881. [Google Scholar] [CrossRef]
- Jonathan, M.; Souza da Silva, C.; Bosch, G.; Schols, H.; Gruppen, H. In vivo degradation of alginate in the presence and in the absence of resistant starch. Food Chem. 2015, 172, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Jonathan, M.C.; Bosch, G.; Schols, H.A.; Gruppen, H. Separation and identification of individual alginate oligosaccharides in the feces of alginate-fed pigs. J. Agric. Food Chem. 2013, 61, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Han, F.; Hu, B.; Li, J.; Yu, W. In vivo prebiotic properties of alginate oligosaccharides prepared through enzymatic hydrolysis of alginate. Nutr. Res. 2006, 26, 597–603. [Google Scholar] [CrossRef]
- Ramnani, P.; Chitarrari, R.; Tuohy, K.; Grant, J.; Hotchkiss, S.; Philp, K.; Campbell, R.; Gill, C.; Rowland, I. In vitro fermentation and prebiotic potential of novel low molecular weight polysaccharides derived from agar and alginate seaweeds. Anaerobe 2012, 18, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.L.; Yang, M.; Fu, X.D.; Chen, M.; Su, Q.; Zhao, Y.H.; Mou, H.J. Evaluation of Prebiotic Potential of Three Marine Algae Oligosaccharides from Enzymatic Hydrolysis. Mar. Drugs 2019, 17, 173. [Google Scholar] [CrossRef]
- Li, M.; Li, G.; Shang, Q.; Chen, X.; Liu, W.; Pi, X.; Zhu, L.; Yin, Y.; Yu, G.; Wang, X. In vitro fermentation of alginate and its derivatives by human gut microbiota. Anaerobe 2016, 39, 19–25. [Google Scholar] [CrossRef]
- Wiese, M.; Khakimov, B.; Nielsen, S.; Sorensen, H.; van den Berg, F.; Nielsen, D.S. CoMiniGut-a small volume in vitro colon model for the screening of gut microbial fermentation processes. PeerJ 2018, 6, e4268. [Google Scholar] [CrossRef]
- Bouillon, G.A.; Gåserod, O.; Rattay, F.P. Evaluation of the inhibitory effect of alginate oligosaccharide on yeast and mould in yoghurt. Int. Dairy J. 2019, 99, 104544. [Google Scholar] [CrossRef]
- Mäkeläinen, H.; Saarinen, M.; Stowell, J.; Rautonen, N.; Ouwehand, A.C. Xylo-oligosaccharides and lactitol promote the growth of Bifidobacterium lactis and Lactobacillus species in pure cultures. Benef. Microbes 2010, 1, 139–148. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Tiihonen, K.; Saarinen, M.; Putaala, H.; Rautonen, N. Influence of a combination of Lactobacillus acidophilus NCFM and lactitol on healthy elderly: Intestinal and immune parameters. Br. J. Nutr. 2009, 101, 367–375. [Google Scholar] [CrossRef]
- Krych, L.; Kot, W.; Bendtsen, K.M.B.; Hansen, A.K.; Vogensen, F.K.; Nielsen, D.S. Have you tried spermine? A rapid and cost-effective method to eliminate dextran sodium sulfate inhibition of PCR and RT-PCR. J. Microbiol. Methods 2018, 144, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UNOISE2: Improved error-correction for Illumina 16S and ITS amplicon sequencing. bioRxiv 2016. [Google Scholar] [CrossRef]
- McDonald, D.; Price, M.N.; Goodrich, J.; Nawrocki, E.P.; DeSantis, T.Z.; Probst, A.; Andersen, G.L.; Knight, R.; Hugenholtz, P. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 2012, 6, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Meth. 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.; Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Van Treuren, W.; White, R.A.; Eggesbo, M.; Knight, R.; Peddada, S.D. Analysis of composition of microbiomes: A novel method for studying microbial composition. Microb. Ecol. Health Dis. 2015, 26, 27663. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Endo, T.; Nakakita, R.; Murata, K.; Yonemoto, Y.; Okayama, K. Effect of depolymerized alginates on the growth of bifidobacteria. Biosci. Biotechnol. Biochem. 1992, 56, 355–356. [Google Scholar] [CrossRef] [PubMed]
- El Kaoutari, A.; Armougom, F.; Gordon, J.I.; Raoult, D.; Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 2013, 11, 497–504. [Google Scholar] [CrossRef]
- Xu, X.; Iwamoto, Y.; Kitamura, Y.; Oda, T.; Muramatsu, T. Root growth-promoting activity of unsaturated oligomeric uronates from alginate on carrot and rice plants. Biosci. Biotechnol. Biochem. 2003, 67, 2022–2025. [Google Scholar] [CrossRef]
- Yamasaki, Y.; Yokose, T.; Nishikawa, T.; Kim, D.; Jiang, Z.; Yamaguchi, K.; Oda, T. Effects of alginate oligosaccharide mixtures on the growth and fatty acid composition of the green alga Chlamydomonas reinhardtii. J. Biosci. Bioeng. 2012, 113, 112–116. [Google Scholar] [CrossRef]
- Roy, C.C.; Kien, C.L.; Bouthillier, L.; Levy, E. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Li, D.; Wang, J.; Wu, H.; Xia, X.; Bi, W.; Guan, H.; Zhang, L. Effects of polymannuronate on performance, antioxidant capacity, immune status, cecal microflora, and volatile fatty acids in broiler chickens. Poult. Sci. 2015, 94, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Li, D.; Wu, H.; Li, J.; Chen, Y.; Guan, H.; Zhang, L. Effects of purified polymannuronate on the performance, immune status, antioxidant capacity, intestinal microbial populations and volatile fatty acid concentrations of weaned piglets. Anim. Feed Sci. Technol. 2016, 216, 161–168. [Google Scholar] [CrossRef]
- Fu, T.; Zhou, L.; Fu, Z.; Zhang, B.; Li, Q.; Pan, L.; Zhou, C.; Zhao, Q.; Shang, Q.; Yu, G. Enterotype-Specific Effect of Human Gut Microbiota on the Fermentation of Marine Algae Oligosaccharides: A Preliminary Proof-of-Concept In Vitro Study. Polymers 2022, 14, 770. [Google Scholar] [CrossRef] [PubMed]
- Murakami, R.; Hashikura, N.; Yoshida, K.; Xiao, J.Z.; Odamaki, T. Growth-promoting effect of alginate on Faecalibacterium prausnitzii through cross-feeding with Bacteroides. Food Res. Int. 2021, 144, 110326. [Google Scholar] [CrossRef] [PubMed]



| Species | Source * | Strain Name | Glucose a | AOS a | Alginate b | Inulin a |
|---|---|---|---|---|---|---|
| Bifidobacterium breve | BCCM/LMG | LMG 13208 | +++ | - | - | - |
| Bifidobacterium longum | IFF | Bl-05 | +++ | - | - | - |
| Bifidobacterium lactis | IFF | Bl-04 | +++ | - | - | - |
| Bifidobacterium bifidum | IFF | Bb-06 | +++ | - | - | +++ |
| Bifidobacterium adolescentis | DSMZ | DSM 20083 | +++ | + | + | +++ |
| Bacteroides vulgatus | BCCM/LMG | LMG 17767 | +++ | - | - | - |
| Bacteroides acidifaciens | DSMZ | DSM 15896 | +++ | - | - | - |
| Bacteroides thetaiotaomicron | DSMZ | DSM 2079 | +++ | - | - | - |
| Bacteroides fragilis | DSMZ | DSM 2151 | +++ | - | - | - |
| Bacteroides ovatus | DSMZ | DSM 1896 | +++ | - | ++ | - |
| Escherichia coli | ATCC | ATCC 43888 | +++ | - | - | +++ |
| Enterobacter cloacae | UCPH | NTCT 11572 | +++ | - | - | - |
| Klebsiella pneumoniae | UCPH | c132-98 WT | +++ | - | - | - |
| Salmonella enterica Typhimurium | UCPH | SML 27C | +++ | - | - | - |
| Cronobacter sakazakii | DSMZ | DSM 4485 | +++ | - | + | - |
| Lactobacillus acidophilus | BCCM/LMG | LMG 9433 | +++ | - | - | - |
| Lacticaseibacillus rhamnosus | DSMZ | DSM 20021 | +++ | - | - | - |
| Lacticaseibacillus casei | DSMZ | DSM 20011 | +++ | + | + | - |
| Lacticaseibacillus paracasei | NCIMB | NCFB 151 | +++ | + | + | +++ |
| Enterococcus faecium | DSMZ | DSM 2146 | +++ | + | - | ++ |
| Enterococcus hirae | DSMZ | DSM 3320 | +++ | + | + | ++ |
| Clostridium clostridioforme | DSMZ | DSM 933 | +++ | - | - | - |
| Anaerostipes hardus | DSMZ | DSM 3319 | +++ | - | - | - |
| Donor | Substrate | Concentration of Fatty Acids (µmol mL−1) | |||
|---|---|---|---|---|---|
| Acetic Acid | Butyric Acid | Propionic Acid | Total | ||
| 1 | Control | 14.4 ± 0.4 | 0.9 ± 0.0 | 1.6 ± 0.0 | 16.90 |
| AOS | 26.4 ± 0.5 *** | 0.6 ± 0.1 *** | 1.6 ± 0.1 | 28.60 * | |
| Alginate | 25.9 ± 3.2 ** | 1.0 ± 0.2 | 2.6 ± 0.6 * | 29.50 * | |
| Inulin | 60.5 ± 5.6 ** | 5.1 ± 1.8 * | 17.9 ± 5.4 ** | 83.50 ** | |
| 2 | Control | 23.4 ± 0.8 | 2.7 ± 0.3 | 2.0 ± 0.1 | 28.10 |
| AOS | 28.3 ± 5.7 | 2.3 ± 0.2 | 2.6 ± 0.4 | 33.20 | |
| Alginate | 23.3 ± 6.2 | 1.0 ± 0.6 ** | 2.0 ± 0.5 | 26.30 | |
| Inulin | 44.5 ± 6.9 ** | 4.1 ± 7.7 | 1.8 ± 0.7 | 50.40 ** | |
| 3 | Control | 13.3 ± 3.9 | 1.9 ± 0.5 | 2.0 ± 0.3 | 17.20 |
| AOS | 29.2 ± 5.7 ** | 2.3 ± 0.6 | 5.0 ± 1.7 * | 36.50 * | |
| Alginate | 21.9 ± 4.2 * | 1.0 ± 0.2 * | 2.8 ± 0.2 ** | 25.70 * | |
| Inulin | 30.7 ± 8.0 * | 22.6 ± 4.2 ** | 4.7 ± 1.1 * | 58.00 ** | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouillon, G.; Gåserød, O.; Krych, Ł.; Castro-Mejía, J.L.; Kot, W.; Saarinen, M.T.; Ouwehand, A.C.; Nielsen, D.S.; Rattray, F.P. Modulating the Gut Microbiota with Alginate Oligosaccharides In Vitro. Nutraceuticals 2023, 3, 26-38. https://doi.org/10.3390/nutraceuticals3010003
Bouillon G, Gåserød O, Krych Ł, Castro-Mejía JL, Kot W, Saarinen MT, Ouwehand AC, Nielsen DS, Rattray FP. Modulating the Gut Microbiota with Alginate Oligosaccharides In Vitro. Nutraceuticals. 2023; 3(1):26-38. https://doi.org/10.3390/nutraceuticals3010003
Chicago/Turabian StyleBouillon, Grégoire, Olav Gåserød, Łukasz Krych, Josué L. Castro-Mejía, Witold Kot, Markku T. Saarinen, Arthur C. Ouwehand, Dennis S. Nielsen, and Fergal P. Rattray. 2023. "Modulating the Gut Microbiota with Alginate Oligosaccharides In Vitro" Nutraceuticals 3, no. 1: 26-38. https://doi.org/10.3390/nutraceuticals3010003
APA StyleBouillon, G., Gåserød, O., Krych, Ł., Castro-Mejía, J. L., Kot, W., Saarinen, M. T., Ouwehand, A. C., Nielsen, D. S., & Rattray, F. P. (2023). Modulating the Gut Microbiota with Alginate Oligosaccharides In Vitro. Nutraceuticals, 3(1), 26-38. https://doi.org/10.3390/nutraceuticals3010003







