Identification of CT Values That Could Be Predictive of Necrosis (N-CTav) in Hepatocellular Carcinoma after Lenvatinib Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. CT Imaging
2.3. Evaluation of Therapeutic Response
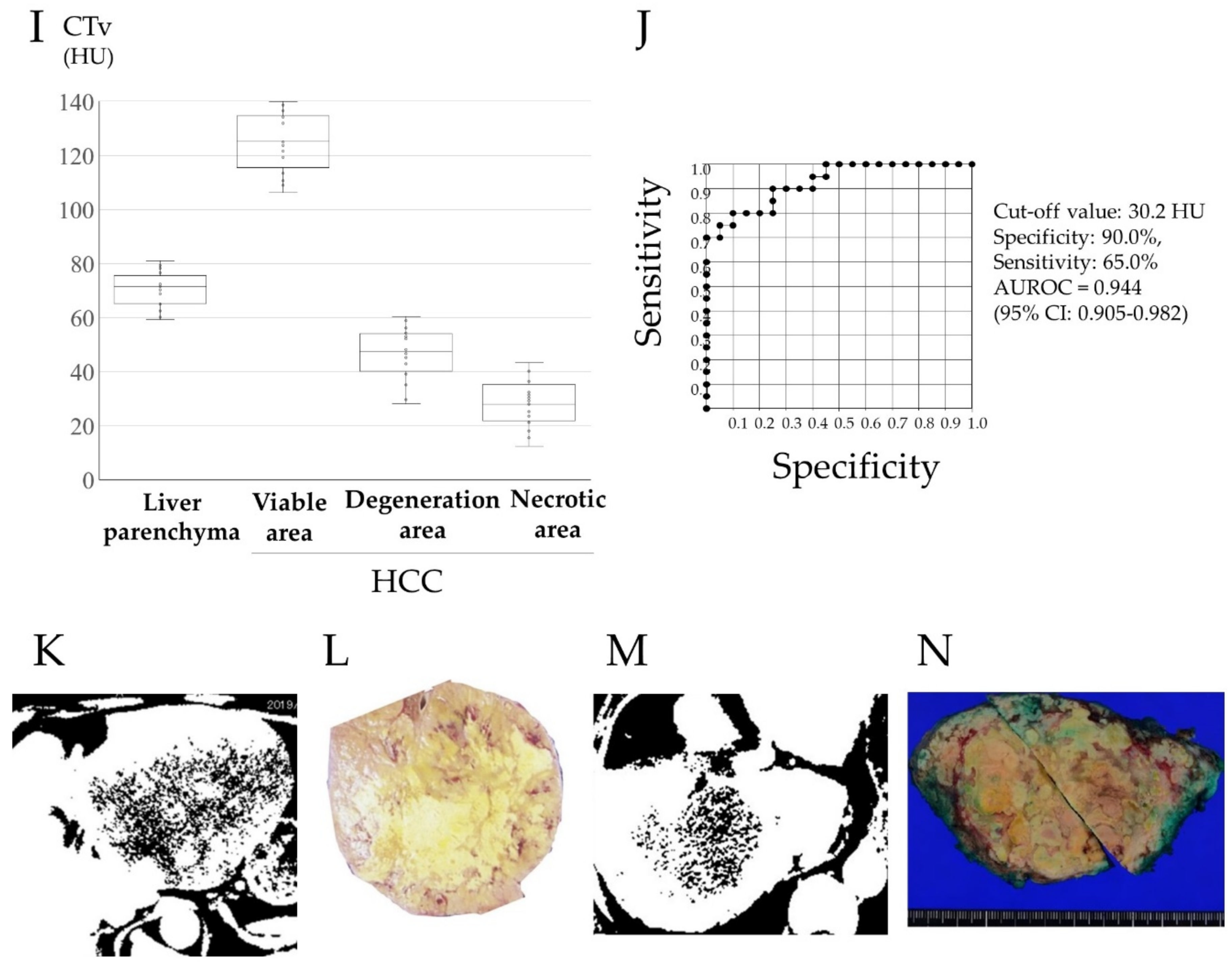
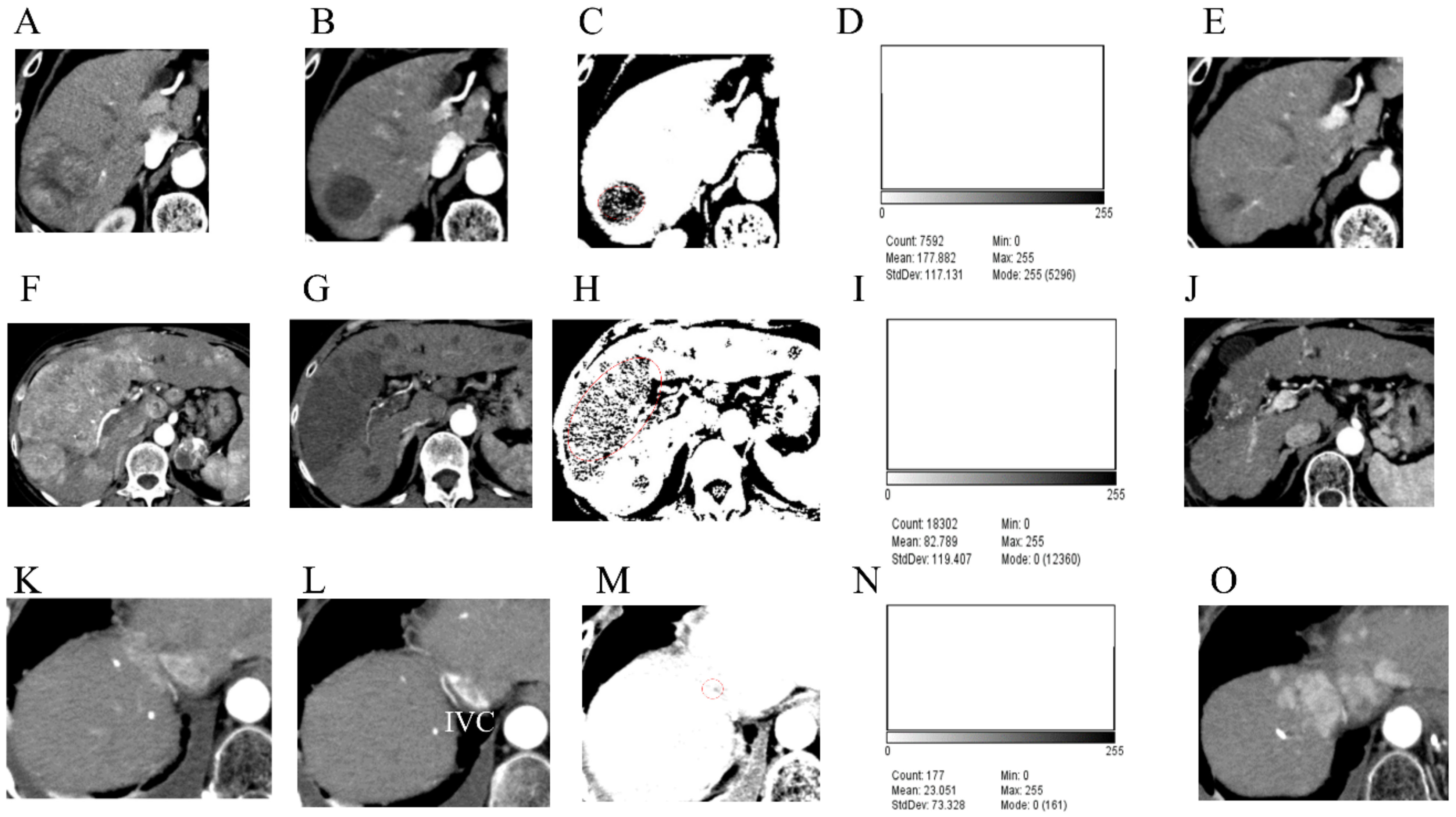
2.4. Image Analysis
2.5. Statistical Analysis
3. Results
3.1. Characteristics of HCC Patients Who Achieved a Complete Response following Lenvatinib Therapy
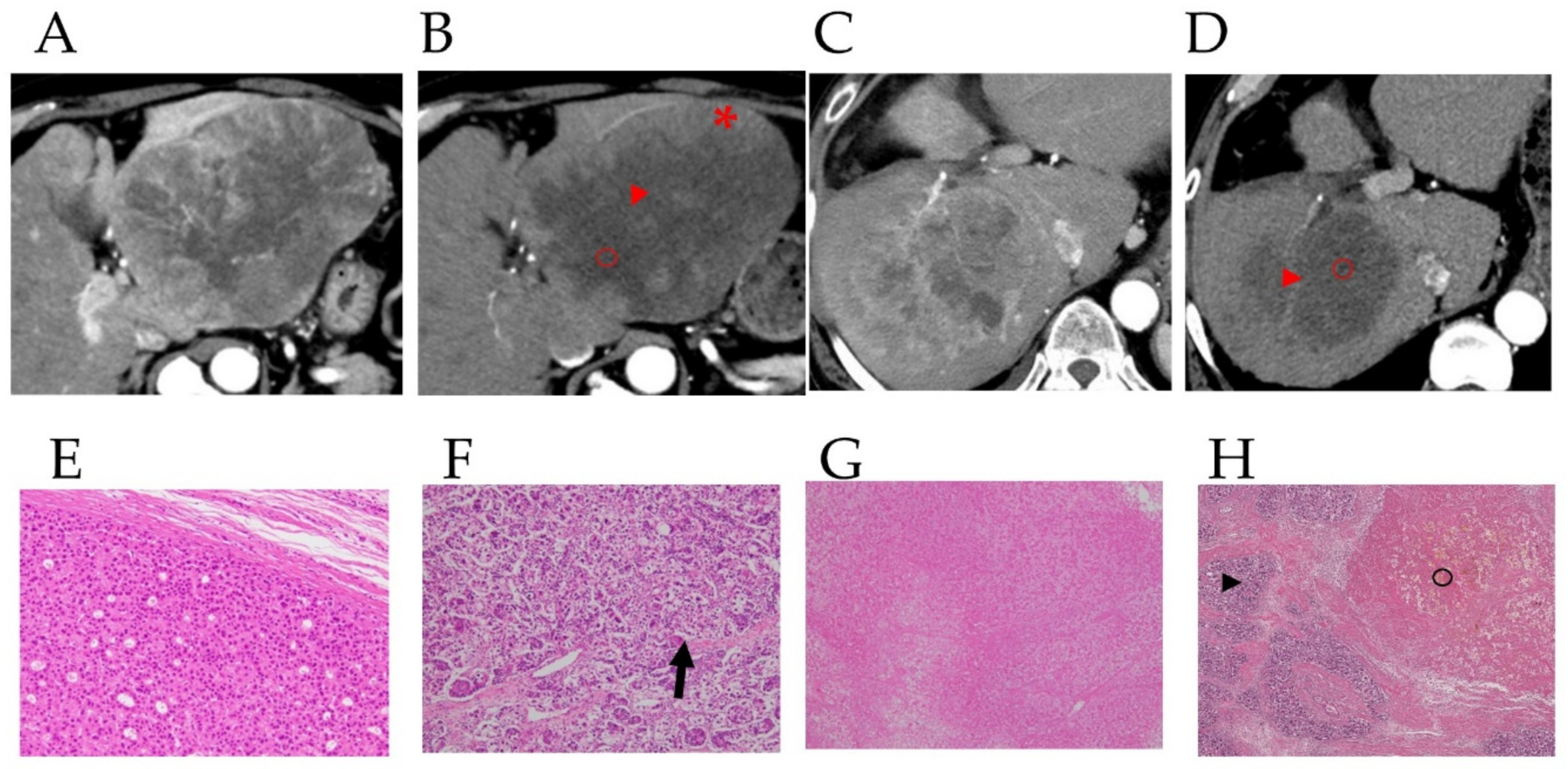
3.2. Preoperative Computed Tomography and Pathological Findings of the Resected Specimens
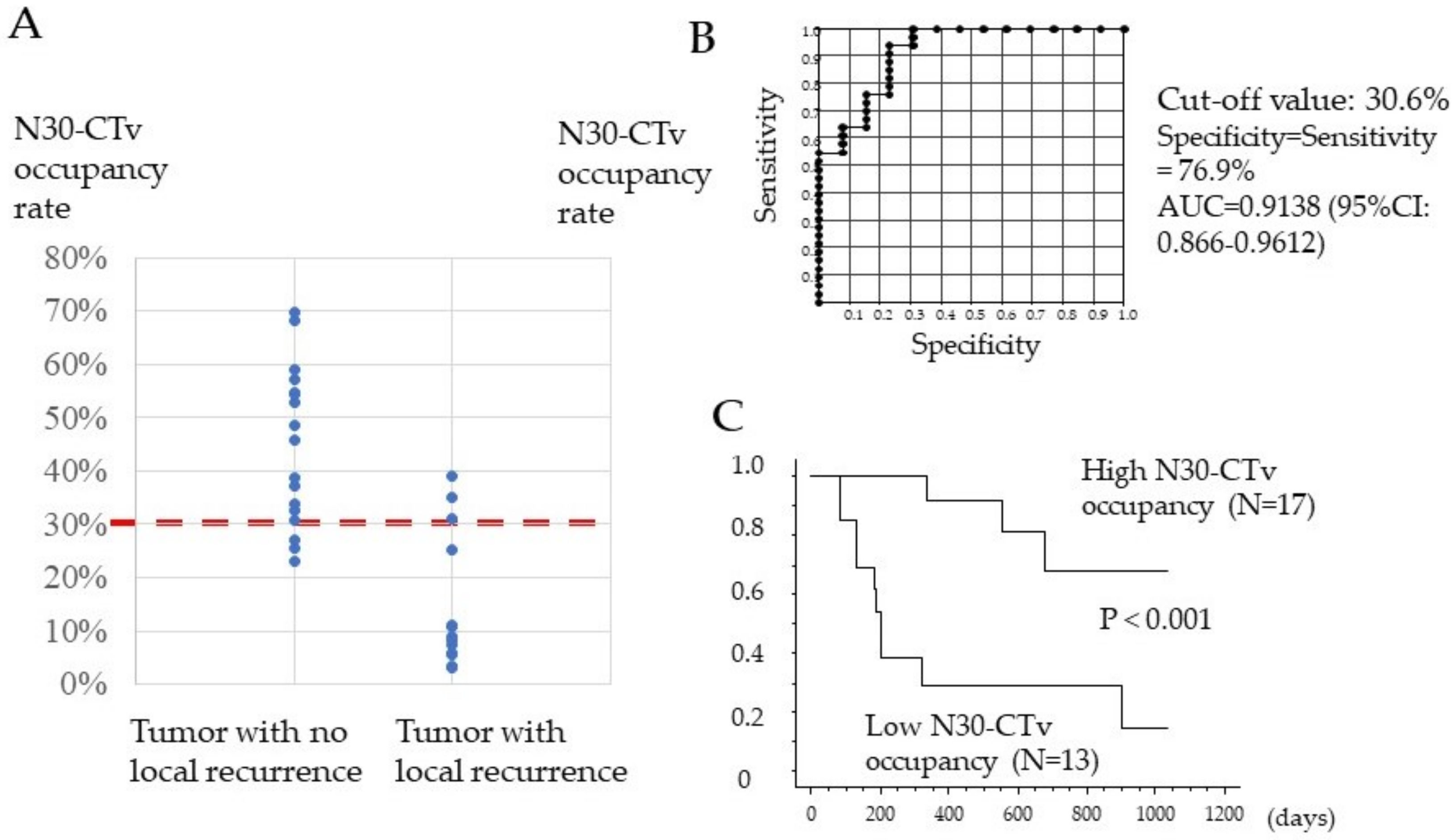
3.3. Association between N30-CTav Occupancy Rate of the Tumor and Local Tumor Progression
3.4. Case Presentations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, A.J. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordan, J.D.; Kennedy, E.B.; Abou-Alfa, G.K.; Beg, M.S.; Brower, S.T.; Gade, T.P.; Goff, L.; Gupta, S.; Guy, J.; Harris, W.P.; et al. Systemic Therapy for Advanced Hepatocellular Carcinoma: ASCO guideline. J. Clin. Oncol. 2020, 38, 4317–4345. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef] [Green Version]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.H.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Rizzo, A.; Dadduzio, V.; Ricci, A.D.; Massari, F.; Di Federico, A.; Gadaleta-Caldarola, G.; Brandi, G. Lenvatinib plus pembrolizumab: The next frontier for the treatment of hepatocellular carcinoma? Expert Opin. Investig. Drugs. 2022, 31, 371–378. [Google Scholar] [CrossRef]
- Rizzo, A.; Brandi, G. Biochemical predictors of response to immune checkpoint inhibitors in unresectable hepatocellular carcinoma. Cancer Treat. Res. Commun. 2021, 27, 100328. [Google Scholar] [CrossRef]
- Amioka, K.; Kawaoka, T.; Kosaka, M.; Johira, Y.; Shirane, Y.; Miura, R.; Murakami, S.; Yano, S.; Naruto, K.; Ando, Y.; et al. Analysis of Survival and Response to Lenvatinib in Unresectable Hepatocellular Carcinoma. Cancers 2022, 14, 320. [Google Scholar] [CrossRef]
- Ronot, M.; Bouattour, M.; Wassermann, J.; Bruno, O.; Dreyer, C.; Larroque, B.; Castera, L.; Vilgrain, V.; Belghiti, J.; Raymond, E.; et al. Alternative Criteria (Choi, European association for the study ofthe liver, and modified Response Evaluation Criteria in Solid Tumors [RECIST]) Versus RECIST 1.1 in patients with advanced hepatocellular carcinoma treated with sorafenib. Oncologist 2014, 19, 394–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver. Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Association for the Study of the Liver. EASL clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Llovet, J.M.; Lencioni, R. mRECIST for HCC: Performance and novel refinements. J. Hepatol. 2020, 72, 288–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lencioni, R.; Montal, R.; Torres, F.; Park, J.W.; Decaens, T.; Raoul, J.L.; Kudo, M.; Chang, C.; Ríos, J.; Boige, V.; et al. Objective response by mRECIST as a predictor and potential surrogate end-point of overall survival in advanced HCC. J. Hepatol. 2017, 66, 1166–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arslanoglu, A.; Chalian, H.; Sodagari, F.; Seyal, A.R.; Töre, H.G.; Salem, R.; Yaghmai, V. Threshold for Enhancement in Treated Hepatocellular Carcinoma on MDCT: Effect on Necrosis Quantification. Am. J. Roentgenol. 2016, 206, 536–543. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, W.J.; Lim, H.K.; Lim, J.H. Prediction of viable tumor in hepatocellular carcinoma treated with transcatheter arterial chemoembolization: Usefulness of attenuation value measurement at quadruple-phase helical computed tomography. J. Comput. Assist. Tomogr. 2007, 31, 198–203. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods. 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Miksch, R.C.; Hao, J.; Schoenberg, M.B.; Dötzer, K.; Schlüter, F.; Weniger, M.; Yin, S.; Ormanns, S.; D’Haese, J.G.; Guba, M.O.; et al. Development of a reliable and accurate algorithm to q.uantify the tumor immune stroma (QTiS) across tumor types. Oncotarget 2017, 8, 114935–114944. [Google Scholar] [CrossRef]
- Kudo, M.; Montal, R.; Finn, R.S.; Castet, F.; Ueshima, K.; Nishida, N.; Haber, P.K.; Hu, Y.; Chiba, Y.; Schwartz, M.; et al. Objective Response Predicts Survival in Advanced Hepatocellular Carcinoma treated with Systemic Therapies. Clin. Cancer Res. 2021, 3135. [Google Scholar] [CrossRef]
- Willett, C.G.; Boucher, Y.; di Tomaso, E.; Duda, D.G.; Munn, L.L.; Tong, R.T.; Chung, D.C.; Sahani, D.V.; Kalva, S.P.; Kozin, S.V.; et al. Direct evidence that the VEGF-specific antibody bevacizumab has antivascular effects in human rectal cancer. Nat. Med. 2004, 10, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Willett, C.G.; Duda, D.G.; di Tomaso, E.; Boucher, Y.; Czito, B.G.; Vujaskovic, Z.; Vlahovic, G.; Bendell, J.; Cohen, K.S.; Hurwitz, H.I.; et al. Complete pathological response to bevacizumab and chemoradiation in advanced rectal cancer. Nat. Clin. Pract. Oncol. 2007, 4, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Frentzas, S.; Simoneau, E.; Bridgeman, V.L.; Vermeulen, P.B.; Foo, S.; Kostaras, E.; Nathan, M.; Wotherspoon, A.; Gao, Z.H.; Shi, Y.; et al. Vessel co-option mediates resistance to anti-angiogenic therapy in liver metastases. Nat. Med. 2016, 22, 1294–1302. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| n = 12 | |
|---|---|
| Median age, years (range) | 73 (25–84) |
| Sex (Male/Female) | 7/5 |
| Cause of HCC (HBV/HCV/NBNC) | 4/6/2 |
| Child-Pugh grade (A/B) | 11/1 |
| Child-Pugh score (5/6/7) | 8/3/1 |
| mALBI (1/2a/2b) | 6/3/3 |
| ECOG PS (0/1) | 11/1 |
| BMI (kg/m2) (range) | 22.0 (19.9–31.8) |
| Extrahepatic metastasis, n (%) | 2 (16.7%) |
| MVI n (%) | 1 (8.3%) |
| BCLC stage (B/C) | 9/3 |
| TNM stage (II/III/IV) | 1/8/3 |
| MTA naïve, n (%) | 11 (91.7%) |
| Past history of TACE, n (%) | 8 (66.7%) |
| AFP (range) | 136 (1.0–1686) |
| DCP (range) | 239 (12–67,900) |
| Median observation period, days (range) | 831 (262–1104) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuma, M.; Yokoo, H.; Hiraoka, A.; Ueda, K.; Yokoyama, T.; Tsuji, K.; Shimada, N.; Uojima, H.; Kobayashi, S.; Hattori, N.; et al. Identification of CT Values That Could Be Predictive of Necrosis (N-CTav) in Hepatocellular Carcinoma after Lenvatinib Treatment. Curr. Oncol. 2022, 29, 3259-3271. https://doi.org/10.3390/curroncol29050266
Chuma M, Yokoo H, Hiraoka A, Ueda K, Yokoyama T, Tsuji K, Shimada N, Uojima H, Kobayashi S, Hattori N, et al. Identification of CT Values That Could Be Predictive of Necrosis (N-CTav) in Hepatocellular Carcinoma after Lenvatinib Treatment. Current Oncology. 2022; 29(5):3259-3271. https://doi.org/10.3390/curroncol29050266
Chicago/Turabian StyleChuma, Makoto, Hideki Yokoo, Atsushi Hiraoka, Kazuhiko Ueda, Takahiro Yokoyama, Kunihiko Tsuji, Noritomo Shimada, Haruki Uojima, Satoshi Kobayashi, Nobuhiro Hattori, and et al. 2022. "Identification of CT Values That Could Be Predictive of Necrosis (N-CTav) in Hepatocellular Carcinoma after Lenvatinib Treatment" Current Oncology 29, no. 5: 3259-3271. https://doi.org/10.3390/curroncol29050266
APA StyleChuma, M., Yokoo, H., Hiraoka, A., Ueda, K., Yokoyama, T., Tsuji, K., Shimada, N., Uojima, H., Kobayashi, S., Hattori, N., Okubo, T., Atsukawa, M., Ishikawa, T., Takaguchi, K., Tsutsui, A., Toyoda, H., Tada, T., Saito, Y., Hirose, S., ... Maeda, S. (2022). Identification of CT Values That Could Be Predictive of Necrosis (N-CTav) in Hepatocellular Carcinoma after Lenvatinib Treatment. Current Oncology, 29(5), 3259-3271. https://doi.org/10.3390/curroncol29050266








