High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence
Abstract
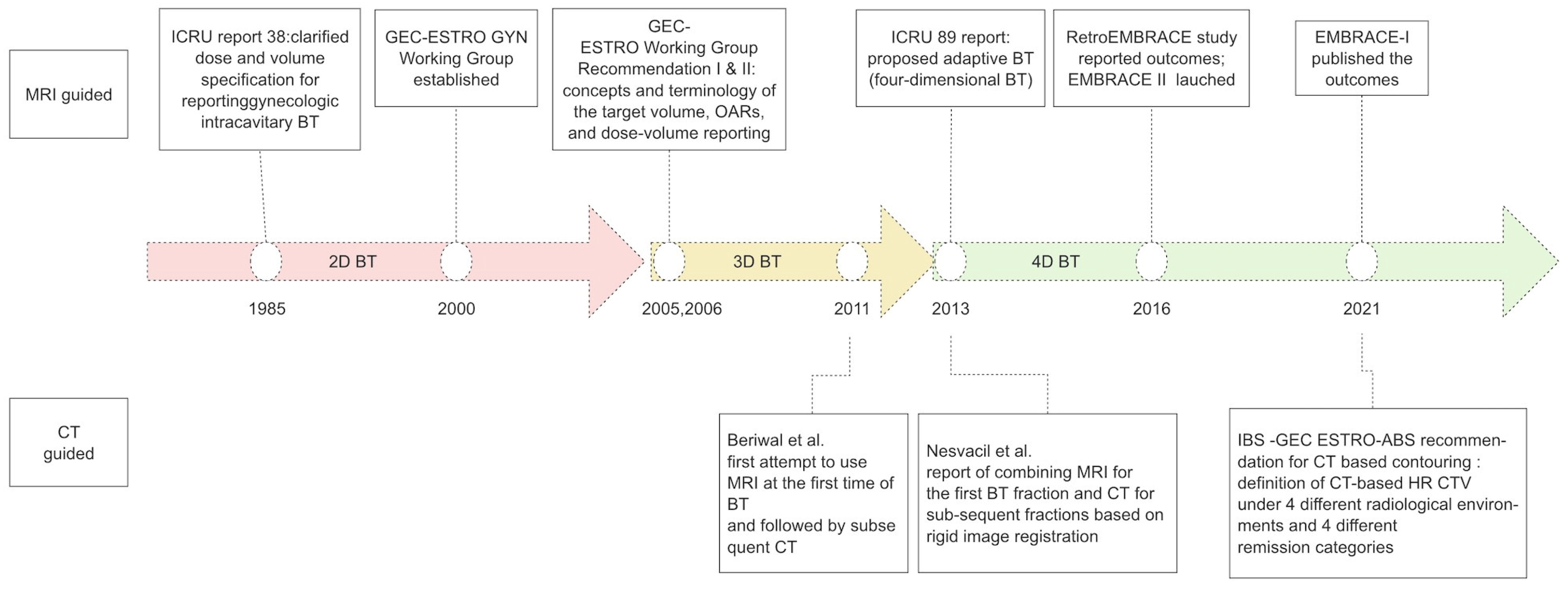
:1. Introduction
2. MRI-Guided BT: The Gold Standard
2.1. MRI-Guided Contouring and Planning
2.2. Clinical Outcomes and Prognostic Factors
3. CT-Guided BT: The Most Common Procedure
3.1. CT-Guided Contouring and Planning
3.2. Clinical Outcomes
| Imaging Modality for Planning | IC or IC/IS | Number of Patients | FIGO Stage | Median Follow-Up (m) | Local Control (%) | Disease-Free Survival (%) | Overall Survival (%) | Toxicity (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 y | 5 y | 3 y | 5 y | 3 y | 5 y | Any | ≥G3 | ||||||
| Mesko et al. [50] 2015 | CT | IS | 31 | IB–IVB | 19 | 90 * | 55 * | 61 * | |||||
| Cho et al. [47] 2016 | CT | IC | 128 | IA–IIB | 30 | 96 * | 88 *,a | 88 * | 16 c, 2 d | ||||
| Ohno et al. [51] 2017 | CT | IC, IC/ IS (17.5%) | 80 | I–IV | 60 | 94 | 90 b | 86 | 3.8 (GI) d 3.8 (GU) d | ||||
| Kawashima et al. [52] 2019 | CT | IC | 84 | IB–IVA | 36 | 89 | 81 a | 94 | 28.8 (GI) | 5 (GI) | |||
| Chan et al. [48] 2022 | CT | IC, IC/ IS (25.9%) | 135 | IB–IVA | 54 | 90.7 | 65.2 | 87.2 | 11.9 | 9.7 | |||
| Xiu et al. [53] 2022 | CT | IC, IC/ IS (31.3%) | 211 | IB2–IIIB | 69 | 89 | 67 | 78 | 25.1(GI) 1.9 (GU) | 12.3 (GI) | |||
| Uezono et al. [54] 2022 | CT | IC, IC/ IS (27%) | 171 | IB–IV | 33 | 86 | 75 | 5, 4 c 6 (GI) d, 2.9 (GU) d | |||||
| Beriwal et al. [22] 2011 | F1 MRI + serial CT | IC | 44 | IB–IIIB | 8 | 88 * | 86 * | 0 | |||||
| Choong et al. [55] 2015 | F1 HYBRID + serial CT | IC | 49 | IB–IVA | 47 | 92.6 | 78.8 a | 77.7 | 12 | ||||
| Van Dyk et al. [56] 2016 | single MR + serial US | IC | 191 | II–VB | 60 | 86 | 86 | 75 | 63 | 6 | |||
| Tharavichitkul et al. [57] 2018 | TAUS | IC | 92 | IB–III | 41 | 85.9 | 76.1 | 89.1 | |||||
| Tharavichitkul et al. [58] 2022 | CT (50.5%) TAUS (49.5%) | IC, IC/ IS (9.2%) | 295 | I–IV | 48 | 89.5 # | 74.9 a,# | 69.1 # | 1.7 (≥G2 GU) 4.1 (≥G2 GI) | ||||
| Kim et al. [59] 2022 | PET/CT | IC | 151 | I–IV | 57 | 89.2 | 64.1 | 76 | 8.6 c, 7.3 d | ||||
4. CT/MRI-Guided BT: Prominent Combination
| Publication, Year | Institution | CT/MRI Combination | Characterization of Workflow | Advantages and/or Limitations |
|---|---|---|---|---|
| Beriwal et al. [22] 2011 | Pittsburgh Cancer Institute | non-image fusion |
|
|
| Nesvacil et al. [23] 2013 | Vienna group | MRI at F1 |
|
|
| Trifiletti et al. [60] 2015 | Virginia University | asynchronous MRI image |
|
|
| Choong et al. [55] 2015 | St James’s University | F1 hybrid and F2-3 CT only |
|
|
5. PET-Guided BT: On the Way
6. US-Guided BT: Promising and Low-Cost
| Publication, Year | Institution | US-Guided BT Planning | Characterization of Workflow | Perspectives |
|---|---|---|---|---|
| Nesvacil et al. [73] 2016 | Vienna group | TRUS/CT |
|
|
| Van Dyk et al. [74] 2015 | Peter MacCallum Cancer Centre | single MR + serial TAUS |
|
|
| Tharavichitkul et al. [75] 2015 | Chiang Mai University | TAUS/orthogonal radiographs |
|
|
7. Emerging Techniques in Image Guidance
7.1. US-Guided Applicator Implantation
7.2. MRI-Guided Applicator Implantation
7.3. 3D Printing Personalized Applicators and Implant Templates
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Green, J.A.; Kirwan, J.M.; Tierney, J.F.; Symonds, P.; Fresco, L.; Collingwood, M.; Williams, C.J. Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix: A systematic review and meta-analysis. Lancet 2001, 358, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Sturdza, A.; Potter, R.; Fokdal, L.U.; Haie-Meder, C.; Tan, L.T.; Mazeron, R.; Petric, P.; Segedin, B.; Jurgenliemk-Schulz, I.M.; Nomden, C.; et al. Image guided brachytherapy in locally advanced cervical cancer: Improved pelvic control and survival in RetroEMBRACE, a multicenter cohort study. Radiother. Oncol. 2016, 120, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Potish, R.; Gerbi, B.; Engler, G. Dose Prescription, Dose Specification, and Applicator Geometry in Intracavitary Therapy; Medical Physics Publishing: Madison, WI, USA, 1995. [Google Scholar]
- Bethesda, M. ICRU report 38. Dose and volume specification for reporting intracavitary therapy in gynecology. Int. Comm. Radiat. Units Meas. 1985, 38, 1–20. [Google Scholar]
- Georgescu, M.; Tanase, A.; Dumitrache, M.; Ileanu, B.; Anghel, R. Dosimetric evaluation study on conventional and 3D conformal brachytherapy treatment of cervix cancer. Rom. Rep. Phys. 2017, 69, 608. [Google Scholar]
- Georgescu, M.; Moldoveanu, V.; Ileanu, B.; Anghel, R. Dosimetric influence of uterus position in cervix cancer high-dose-rate brachytherapy. Rom. J. Phys. 2016, 61, 1557–1566. [Google Scholar]
- Tanderup, K.; Nielsen, S.K.; Nyvang, G.B.; Pedersen, E.M.; Rohl, L.; Aagaard, T.; Fokdal, L.; Lindegaard, J.C. From point A to the sculpted pear: MR image guidance significantly improves tumour dose and sparing of organs at risk in brachytherapy of cervical cancer. Radiother. Oncol. 2010, 94, 173–180. [Google Scholar] [CrossRef]
- Fokdal, L.; Sturdza, A.; Mazeron, R.; Haie-Meder, C.; Tan, L.T.; Gillham, C.; Segedin, B.; Jurgenliemk-Schultz, I.; Kirisits, C.; Hoskin, P.; et al. Image guided adaptive brachytherapy with combined intracavitary and interstitial technique improves the therapeutic ratio in locally advanced cervical cancer: Analysis from the retroEMBRACE study. Radiother. Oncol. 2016, 120, 434–440. [Google Scholar] [CrossRef]
- Charra-Brunaud, C.; Harter, V.; Delannes, M.; Haie-Meder, C.; Quetin, P.; Kerr, C.; Castelain, B.; Thomas, L.; Peiffert, D. Impact of 3D image-based PDR brachytherapy on outcome of patients treated for cervix carcinoma in France: Results of the French STIC prospective study. Radiother. Oncol. 2012, 103, 305–313. [Google Scholar] [CrossRef]
- Hande, V.; Chopra, S.; Kalra, B.; Abdel-Wahab, M.; Kannan, S.; Tanderup, K.; Grover, S.; Zubizarreta, E.; Rubio, J.A.P. Point-A vs. volume-based brachytherapy for the treatment of cervix cancer: A meta-analysis. Radiother. Oncol. 2022, 170, 70–78. [Google Scholar] [CrossRef]
- Nag, S. Controversies and new developments in gynecologic brachytherapy: Image-based intracavitary brachytherapy for cervical carcinoma. Semin. Radiat. Oncol. 2006, 16, 164–167. [Google Scholar] [CrossRef]
- Haie-Meder, C.; Potter, R.; Van Limbergen, E.; Briot, E.; De Brabandere, M.; Dimopoulos, J.; Dumas, I.; Hellebust, T.P.; Kirisits, C.; Lang, S.; et al. Recommendations from Gynaecological (GYN) GEC-ESTRO Working Group (I): Concepts and terms in 3D image based 3D treatment planning in cervix cancer brachytherapy with emphasis on MRI assessment of GTV and CTV. Radiother. Oncol. 2005, 74, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Nag, S.; Cardenes, H.; Chang, S.; Das, I.J.; Erickson, B.; Ibbott, G.S.; Lowenstein, J.; Roll, J.; Thomadsen, B.; Varia, M. Proposed guidelines for image-based intracavitary brachytherapy for cervical carcinoma: Report from Image-Guided Brachytherapy Working Group. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.A.; Paulson, E.S.; van der Heide, U.A.; Fuller, C.D.; Raaymakers, B.W.; Lagendijk, J.J.W.; Li, X.A.; Jaffray, D.A.; Dawson, L.A.; Erickson, B.; et al. The transformation of radiation oncology using real-time magnetic resonance guidance: A review. Eur. J. Cancer 2019, 122, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Boldrini, L.; Piras, A.; Chiloiro, G.; Autorino, R.; Cellini, F.; Cusumano, D.; Fionda, B.; D’Aviero, A.; Campitelli, M.; Marazzi, F.; et al. Low Tesla magnetic resonance guided radiotherapy for locally advanced cervical cancer: First clinical experience. Tumori 2020, 106, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Potter, R.; Haie-Meder, C.; Van Limbergen, E.; Barillot, I.; De Brabandere, M.; Dimopoulos, J.; Dumas, I.; Erickson, B.; Lang, S.; Nulens, A.; et al. Recommendations from gynaecological (GYN) GEC ESTRO working group (II): Concepts and terms in 3D image-based treatment planning in cervix cancer brachytherapy-3D dose volume parameters and aspects of 3D image-based anatomy, radiation physics, radiobiology. Radiother. Oncol. 2006, 78, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Prescribing, Recording, and Reporting Brachytherapy for Cancer of the Cervix. J. ICRU 2013, 13, NP. [CrossRef]
- Pötter, R.; Kirisits, C.; Fidarova, E.F.; Dimopoulos, J.C.; Berger, D.; Tanderup, K.; Lindegaard, J.C. Present status and future of high-precision image guided adaptive brachytherapy for cervix carcinoma. Acta Oncol. 2008, 47, 1325–1336. [Google Scholar] [CrossRef]
- Pötter, R.; Tanderup, K.; Schmid, M.P.; Jürgenliemk-Schulz, I.; Haie-Meder, C.; Fokdal, L.U.; Sturdza, A.E.; Hoskin, P.; Mahantshetty, U.; Segedin, B.; et al. MRI-guided adaptive brachytherapy in locally advanced cervical cancer (EMBRACE-I): A multicentre prospective cohort study. Lancet Oncol. 2021, 22, 538–547. [Google Scholar] [CrossRef]
- Potter, R.; Tanderup, K.; Kirisits, C.; de Leeuw, A.; Kirchheiner, K.; Nout, R.; Tan, L.T.; Haie-Meder, C.; Mahantshetty, U.; Segedin, B.; et al. The EMBRACE II study: The outcome and prospect of two decades of evolution within the GEC-ESTRO GYN working group and the EMBRACE studies. Clin. Transl. Radiat. Oncol. 2018, 9, 48–60. [Google Scholar] [CrossRef]
- Beriwal, S.; Kannan, N.; Kim, H.; Houser, C.; Mogus, R.; Sukumvanich, P.; Olawaiye, A.; Richard, S.; Kelley, J.L.; Edwards, R.P.; et al. Three-dimensional high dose rate intracavitary image-guided brachytherapy for the treatment of cervical cancer using ahybrid magnetic resonance imaging/computed tomography approach: Feasibility and early results. Clin. Oncol. (R Coll. Radiol.) 2011, 23, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Nesvacil, N.; Potter, R.; Sturdza, A.; Hegazy, N.; Federico, M.; Kirisits, C. Adaptive image guided brachytherapy for cervical cancer: A combined MRI-/CT-planning technique with MRI only at first fraction. Radiother. Oncol. 2013, 107, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Derks, K.; Steenhuijsen, J.L.G.; van den Berg, H.A.; Houterman, S.; Cnossen, J.; van Haaren, P.; De Jaeger, K. Impact of brachytherapy technique (2D versus 3D) on outcome following radiotherapy of cervical cancer. J. Contemp. Brachytherapy 2018, 10, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Potter, R.; Georg, P.; Dimopoulos, J.C.; Grimm, M.; Berger, D.; Nesvacil, N.; Georg, D.; Schmid, M.P.; Reinthaller, A.; Sturdza, A.; et al. Clinical outcome of protocol based image (MRI) guided adaptive brachytherapy combined with 3D conformal radiotherapy with or without chemotherapy in patients with locally advanced cervical cancer. Radiother. Oncol. 2011, 100, 116–123. [Google Scholar] [CrossRef]
- Tanderup, K.; Fokdal, L.U.; Sturdza, A.; Haie-Meder, C.; Mazeron, R.; van Limbergen, E.; Jurgenliemk-Schulz, I.; Petric, P.; Hoskin, P.; Dorr, W.; et al. Effect of tumor dose, volume and overall treatment time on local control after radiochemotherapy including MRI guided brachytherapy of locally advanced cervical cancer. Radiother. Oncol. 2016, 120, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Schernberg, A.; Bockel, S.; Annede, P.; Fumagalli, I.; Escande, A.; Mignot, F.; Kissel, M.; Morice, P.; Bentivegna, E.; Gouy, S.; et al. Tumor Shrinkage During Chemoradiation in Locally Advanced Cervical Cancer Patients: Prognostic Significance, and Impact for Image-Guided Adaptive Brachytherapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Ke, T.; Wang, J.; Zhang, N.; Zhao, H.; Guo, X.; Zhao, Z.; Mao, Z.; Cheng, G. Dose-effect relationship between dose-volume parameters of residual gross tumor volume and clinical prognosis in MRI-guided adaptive brachytherapy for locally advanced cervical cancer: A single-center retrospective study. Strahlenther. Onkol. 2023, 199, 131–140. [Google Scholar] [CrossRef]
- Harkenrider, M.M.; Shea, S.M.; Wood, A.M.; Chinsky, B.; Bajaj, A.; Mysz, M.; Yacoub, J.H.; Goldberg, A.; Liotta, M.; Potkul, R.; et al. How one institution overcame the challenges to start an MRI-based brachytherapy program for cervical cancer. J. Contemp. Brachytherapy 2017, 9, 177–186. [Google Scholar] [CrossRef]
- Wills, R.; Lowe, G.; Inchley, D.; Anderson, C.; Beenstock, V.; Hoskin, P. Applicator reconstruction for HDR cervix treatment planning using images from 0.35 T open MR scanner. Radiother. Oncol. 2010, 94, 346–352. [Google Scholar] [CrossRef]
- Ohno, T.; Toita, T.; Tsujino, K.; Uchida, N.; Hatano, K.; Nishimura, T.; Ishikura, S. A questionnaire-based survey on 3D image-guided brachytherapy for cervical cancer in Japan: Advances and obstacles. J. Radiat. Res. 2015, 56, 897–903. [Google Scholar] [CrossRef]
- Grover, S.; Harkenrider, M.M.; Cho, L.P.; Erickson, B.; Small, C.; Small, W., Jr.; Viswanathan, A.N. Image Guided Cervical Brachytherapy: 2014 Survey of the American Brachytherapy Society. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Tang, Q.; Lv, G.; Zhao, F.; Jiang, X.; Zhu, X.; Li, X.; Yan, S. Comparison of computed tomography and magnetic resonance imaging in cervical cancer brachytherapy: A systematic review. Brachytherapy 2017, 16, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, A.N.; Dimopoulos, J.; Kirisits, C.; Berger, D.; Potter, R. Computed tomography versus magnetic resonance imaging-based contouring in cervical cancer brachytherapy: Results of a prospective trial and preliminary guidelines for standardized contours. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Krishnatry, R.; Patel, F.D.; Singh, P.; Sharma, S.C.; Oinam, A.S.; Shukla, A.K. CT or MRI for image-based brachytherapy in cervical cancer. JPN J. Clin. Oncol. 2012, 42, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Rai, B.; Patel, F.D.; Tomar, P.; Singh, O.A.; Simha, V.; Dhanireddy, B.; Sharma, S.C. A study to assess the feasibility of using CT (±diagnostic MRI) instead of MRI at brachytherapy in image guided brachytherapy in cervical cancer. J. Radiother. Pract. 2014, 13, 438–446. [Google Scholar] [CrossRef]
- Hricak, H.; Gatsonis, C.; Coakley, F.V.; Snyder, B.; Reinhold, C.; Schwartz, L.H.; Woodward, P.J.; Pannu, H.K.; Amendola, M.; Mitchell, D.G. Early invasive cervical cancer: CT and MR imaging in preoperative evaluation—ACRIN/GOG comparative study of diagnostic performance and interobserver variability. Radiology 2007, 245, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Swanick, C.W.; Castle, K.O.; Vedam, S.; Munsell, M.F.; Turner, L.M.; Rauch, G.M.; Jhingran, A.; Eifel, P.J.; Klopp, A.H. Comparison of computed tomography–and magnetic resonance imaging–based clinical target volume contours at brachytherapy for cervical cancer. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 793–800. [Google Scholar] [CrossRef]
- Yip, W.W.L.; Wong, J.S.Y.; Lee, V.W.Y.; Wong, F.C.S.; Tung, S.Y. Throwing the dart blind-folded: Comparison of computed tomography versus magnetic resonance imaging-guided brachytherapy for cervical cancer with regard to dose received by the ‘actual’ targets and organs at risk. J. Contemp. Brachytherapy 2017, 9, 446–452. [Google Scholar] [CrossRef]
- Kamran, S.C.; Manuel, M.M.; Cho, L.P.; Damato, A.L.; Schmidt, E.J.; Tempany, C.; Cormack, R.A.; Viswanathan, A.N. Comparison of outcomes for MR-guided versus CT-guided high-dose-rate interstitial brachytherapy in women with locally advanced carcinoma of the cervix. Gynecol. Oncol. 2017, 145, 284–290. [Google Scholar] [CrossRef]
- Swamidas, J.; Jain, J.; Nesvacil, N.; Tanderup, K.; Kirisits, C.; Schmid, M.; Agarwal, P.; Joshi, K.; Naga, P.; Ranjan Khadanga, C.; et al. Dosimetric impact of target definition in brachytherapy for cervical cancer—Computed tomography and trans rectal ultrasound versus magnetic resonance imaging. Phys. Imaging Radiat. Oncol. 2022, 21, 126–133. [Google Scholar] [CrossRef]
- Mahantshetty, U.; Naga Ch, P.; Khadanga, C.R.; Gudi, S.; Chopra, S.; Gurram, L.; Jamema, S.; Ghadi, Y.; Shrivastava, S. A Prospective Comparison of Computed Tomography with Transrectal Ultrasonography Assistance and Magnetic Resonance Imaging-Based Target-Volume Definition during Image Guided Adaptive Brachytherapy for Cervical Cancers. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1448–1456. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, A.N.; Erickson, B.; Gaffney, D.K.; Beriwal, S.; Bhatia, S.K.; Lee Burnett, O., 3rd; D’Souza, D.P.; Patil, N.; Haddock, M.G.; Jhingran, A.; et al. Comparison and consensus guidelines for delineation of clinical target volume for CT- and MR-based brachytherapy in locally advanced cervical cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T.; Wakatsuki, M.; Toita, T.; Kaneyasu, Y.; Yoshida, K.; Kato, S.; Ii, N.; Tokumaru, S.; Ikushima, H.; Uno, T.; et al. Recommendations for high-risk clinical target volume definition with computed tomography for three-dimensional image-guided brachytherapy in cervical cancer patients. J. Radiat. Res. 2017, 58, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Mahantshetty, U.; Poetter, R.; Beriwal, S.; Grover, S.; Lavanya, G.; Rai, B.; Petric, P.; Tanderup, K.; Carvalho, H.; Hegazy, N.; et al. IBS-GEC ESTRO-ABS recommendations for CT based contouring in image guided adaptive brachytherapy for cervical cancer. Radiother. Oncol. 2021, 160, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, N.; Potter, R.; Kirisits, C.; Berger, D.; Federico, M.; Sturdza, A.; Nesvacil, N. High-risk clinical target volume delineation in CT-guided cervical cancer brachytherapy: Impact of information from FIGO stage with or without systematic inclusion of 3D documentation of clinical gynecological examination. Acta Oncol. 2013, 52, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Cho, L.P.; Manuel, M.; Catalano, P.; Lee, L.; Damato, A.L.; Cormack, R.A.; Buzurovic, I.; Bhagwat, M.; O’Farrell, D.; Devlin, P.M.; et al. Outcomes with volume-based dose specification in CT-planned high-dose-rate brachytherapy for stage I–II cervical carcinoma: A 10-year institutional experience. Gynecol. Oncol. 2016, 143, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.L.; Cheng, M.H.; Wu, J.T.; Choi, C.W.; Tse, R.P.; Ho, P.P.; Cheung, E.E.; Cheung, A.; Test, K.Y.; Chan, K.K.; et al. Treatment Outcomes of Computer Tomography-Guided Brachytherapy in Cervical Cancer in Hong Kong: A Retrospective Review. Cancers 2022, 14, 545–551. [Google Scholar] [CrossRef]
- D’Cunha, P.; Gonzalez, Y.; Nwachukwu, C.; Hrycushko, B.; Medin, P.; Owrangi, A.; Jia, X.; Albuquerque, K. Change in image-guided planning strategies over time impacts oncologic and survival outcomes for intracavitary cervical cancer brachytherapy. Brachytherapy 2022, 21, 668–677. [Google Scholar] [CrossRef]
- Mesko, S.; Swamy, U.; Park, S.J.; Borja, L.; Wang, J.; Demanes, D.J.; Kamrava, M. Early clinical outcomes of ultrasound-guided CT-planned high-dose-rate interstitial brachytherapy for primary locally advanced cervical cancer. Brachytherapy 2015, 14, 626–632. [Google Scholar] [CrossRef]
- Ohno, T.; Noda, S.E.; Okonogi, N.; Murata, K.; Shibuya, K.; Kiyohara, H.; Tamaki, T.; Ando, K.; Oike, T.; Ohkubo, Y.; et al. In-room computed tomography-based brachytherapy for uterine cervical cancer: Results of a 5-year retrospective study. J. Radiat. Res. 2017, 58, 543–551. [Google Scholar] [CrossRef]
- Kawashima, A.; Isohashi, F.; Mabuchi, S.; Sawada, K.; Ueda, Y.; Kobayashi, E.; Matsumoto, Y.; Otani, K.; Tamari, K.; Seo, Y.; et al. A 3-year follow-up study of radiotherapy using computed tomography-based image-guided brachytherapy for cervical cancer. J. Radiat. Res. 2019, 60, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Xiu, Y.T.; Meng, F.X.; Wang, Z.; Zhao, K.K.; Wang, Y.L.; Chen, Z.S.; Sun, B.S. Prognostic factors for IB2-IIIB cervical cancer patients treated by radiation therapy with high-dose-rate brachytherapy in a single-institution study. J. Contemp. Brachytherapy 2022, 14, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Uezono, H.; Tsujino, K.; Inoue, Y.; Kajihara, A.; Marudai, M.; Bessho, R.; Sekii, S.; Kubota, H.; Yamaguchi, S.; Ota, Y. CT-based image-guided brachytherapy in uterine cervical cancer: Effect of tumor dose and volume on local control. Brachytherapy 2022, 21, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Choong, E.S.; Bownes, P.; Musunuru, H.B.; Rodda, S.; Richardson, C.; Al-Qaisieh, B.; Swift, S.; Orton, J.; Cooper, R. Hybrid (CT/MRI based) vs. MRI only based image-guided brachytherapy in cervical cancer: Dosimetry comparisons and clinical outcome. Brachytherapy 2016, 15, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Van Dyk, S.; Narayan, K.; Bernshaw, D.; Kondalsamy-Chennakesavan, S.; Khaw, P.; Lin, M.Y.; Schneider, M. Clinical outcomes from an innovative protocol using serial ultrasound imaging and a single MR image to guide brachytherapy for locally advanced cervix cancer. Brachytherapy 2016, 15, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Tharavichitkul, E.; Chakrabandhu, S.; Klunklin, P.; Onchan, W.; Jia-Mahasap, B.; Wanwilairat, S.; Tippanya, D.; Jayasvasti, R.; Sripan, P.; Galalae, R.M.; et al. Intermediate-term results of trans-abdominal ultrasound (TAUS)-guided brachytherapy in cervical cancer. Gynecol. Oncol. 2018, 148, 468–473. [Google Scholar] [CrossRef]
- Tharavichitkul, E.; Jia-Mahasap, B.; Muangwong, P.; Chakrabandhu, S.; Klunklin, P.; Onchan, W.; Tippanya, D.; Nobnop, W.; Watcharawipha, A.; Kittidachanan, K.; et al. Survival outcome of cervical cancer patients treated by image-guided brachytherapy: A ‘real world’ single center experience in Thailand from 2008 to 2018. J. Radiat. Res. 2022, 63, 657–665. [Google Scholar] [CrossRef]
- Kim, N.; Park, W.; Cho, W.K.; Cho, Y.S. Clinical outcomes after positron emission tomography/computed tomography-based image-guided brachytherapy for cervical cancer. Asia Pac. J. Clin. Oncol. 2022, 18, 743–750. [Google Scholar] [CrossRef]
- Trifiletti, D.M.; Libby, B.; Feuerlein, S.; Kim, T.; Garda, A.; Watkins, W.T.; Erickson, S.; Ornan, A.; Showalter, T.N. Implementing MRI-based target delineation for cervical cancer treatment within a rapid workflow environment for image-guided brachytherapy: A practical approach for centers without in-room MRI. Brachytherapy 2015, 14, 905–909. [Google Scholar] [CrossRef]
- Corriher, T.J.; Dutta, S.W.; Alonso, C.E.; Libby, B.; Romano, K.D.; Showalter, T.N. Comparison of initial computed tomography-based target delineation and subsequent magnetic resonance imaging-based target delineation for cervical cancer brachytherapy. J. Contemp. Brachytherapy 2020, 12, 279–282. [Google Scholar] [CrossRef]
- Kidd, E.A.; Siegel, B.A.; Dehdashti, F.; Rader, J.S.; Mutic, S.; Mutch, D.G.; Powell, M.A.; Grigsby, P.W. Clinical outcomes of definitive intensity-modulated radiation therapy with fluorodeoxyglucose-positron emission tomography simulation in patients with locally advanced cervical cancer. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Havrilesky, L.J.; Kulasingam, S.L.; Matchar, D.B.; Myers, E.R. FDG-PET for management of cervical and ovarian cancer. Gynecol. Oncol. 2005, 97, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Kidd, E.A. Imaging to optimize gynecological radiation oncology. Int. J. Gynecol. Cancer 2022, 32, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.; Huh, S.J.; Ju, S.G.; Park, W.; Lee, J.E.; Choi, J.Y.; Kim, B.T.; Kim, C.K.; Park, B.K. 18F-fluorodeoxyglucose positron emisson tomography/computed tomography guided conformal brachytherapy for cervical cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e29–e34. [Google Scholar] [CrossRef]
- Lin, L.L.; Mutic, S.; Low, D.A.; LaForest, R.; Vicic, M.; Zoberi, I.; Miller, T.R.; Grigsby, P.W. Adaptive brachytherapy treatment planning for cervical cancer using FDG-PET. Int. J. Radiat. Oncol. Biol. Phys. 2007, 67, 91–96. [Google Scholar] [CrossRef]
- Miller, T.R.; Grigsby, P.W. Measurement of tumor volume by PET to evaluate prognosis in patients with advanced cervical cancer treated by radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 353–359. [Google Scholar] [CrossRef]
- Lin, L.; Mutic, S.; Low, D.; LaForest, R.; Vicic, M.; Zoberi, I.; Miller, T.; Grigsby, P. Adaptive brachytherapy treatment planning for cervical cancer. Int. J. Radiat. Oncol. Biol. Phys. 2005, 63, S213–S214. [Google Scholar] [CrossRef]
- Oh, D.; Huh, S.J.; Park, W.; Ju, S.G.; Nam, H.; Lee, J.E. Clinical outcomes in cervical cancer patients treated by FDG-PET/CT-based 3-dimensional planning for the first brachytherapy session. Medicine 2016, 95, e3895. [Google Scholar] [CrossRef]
- Kim, N.; Park, W.; Cho, W.K.; Bae, D.-S.; Kim, B.-G.; Lee, J.-W.; Kim, T.-J.; Choi, C.H.; Lee, Y.-Y.; Cho, Y.S. Early metabolic response assessed using 18F-FDG-PET/CT for image-guided intracavitary brachytherapy can better predict treatment outcomes in patients with cervical cancer. Cancer Res. Treat. Off. J. Korean Cancer Assoc. 2021, 53, 803–812. [Google Scholar] [CrossRef]
- Lucia, F.; Bourbonne, V.; Dissaux, G.; Miranda, O.; Abgral, R.; Pradier, O.; Schick, U. Use of Baseline 18F-FDG PET/CT to Identify Initial Sub-Volumes with Local Failure after Concomitant Chemoradiotherapy in Locally Advanced Cervical Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, E329. [Google Scholar] [CrossRef]
- Lucia, F.; Bourbonne, V.; Gujral, D.; Dissaux, G.; Miranda, O.; Mauguen, M.; Pradier, O.; Abgral, R.; Schick, U. Impact of suboptimal dosimetric coverage of pretherapeutic 18F-FDG PET/CT hotspots on outcome in patients with locally advanced cervical cancer treated with chemoradiotherapy followed by brachytherapy. Clin. Transl. Radiat. Oncol. 2020, 23, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.P.; Nesvacil, N.; Potter, R.; Kronreif, G.; Kirisits, C. Transrectal ultrasound for image-guided adaptive brachytherapy in cervix cancer—An alternative to MRI for target definition? Radiother. Oncol. 2016, 120, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Van Dyk, S.; Kondalsamy-Chennakesavan, S.; Schneider, M.; Bernshaw, D.; Narayan, K. Assessing changes to the brachytherapy target for cervical cancer using a single MRI and serial ultrasound. Brachytherapy 2015, 14, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Tharavichitkul, E.; Tippanya, D.; Jayavasti, R.; Chakrabandhu, S.; Klunklin, P.; Onchan, W.; Wanwilairat, S.; Galalae, R.M.; Chitapanarux, I. Two-year results of transabdominal ultrasound-guided brachytherapy for cervical cancer. Brachytherapy 2015, 14, 238–244. [Google Scholar] [CrossRef]
- Schmid, M.P.; Potter, R.; Brader, P.; Kratochwil, A.; Goldner, G.; Kirchheiner, K.; Sturdza, A.; Kirisits, C. Feasibility of transrectal ultrasonography for assessment of cervical cancer. Strahlenther. Onkol. 2013, 189, 123–128. [Google Scholar] [CrossRef]
- Nesvacil, N.; Schmid, M.P.; Potter, R.; Kronreif, G.; Kirisits, C. Combining transrectal ultrasound and CT for image-guided adaptive brachytherapy of cervical cancer: Proof of concept. Brachytherapy 2016, 15, 839–844. [Google Scholar] [CrossRef]
- Smet, S.; Nesvacil, N.; Knoth, J.; Sturdza, A.; Najjari-Jamal, D.; Jelinek, F.; Kronreif, G.; Potter, R.; Widder, J.; Kirisits, C.; et al. Hybrid TRUS/CT with optical tracking for target delineation in image-guided adaptive brachytherapy for cervical cancer. Strahlenther. Onkol. 2020, 196, 983–992. [Google Scholar] [CrossRef]
- Van Dyk, S.; Kondalsamy-Chennakesavan, S.; Schneider, M.; Bernshaw, D.; Narayan, K. Comparison of measurements of the uterus and cervix obtained by magnetic resonance and transabdominal ultrasound imaging to identify the brachytherapy target in patients with cervix cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 860–865. [Google Scholar] [CrossRef]
- Van Dyk, S.; Narayan, K.; Fisher, R.; Bernshaw, D. Conformal Brachytherapy Planning for Cervical Cancer Using Transabdominal Ultrasound. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 64–70. [Google Scholar] [CrossRef]
- Rao, P.B.; Ghosh, S. Routine use of ultrasound guided tandem placement in intracavitary brachytherapy for the treatment of cervical cancer—A South Indian institutional experience. J. Contemp. Brachytherapy 2015, 7, 352–356. [Google Scholar] [CrossRef]
- Viswanathan, A.N.; Moughan, J.; Small, W., Jr.; Levenback, C.; Iyer, R.; Hymes, S.; Dicker, A.P.; Miller, B.; Erickson, B.; Gaffney, D.K. The quality of cervical cancer brachytherapy implantation and the impact on local recurrence and disease-free survival in radiation therapy oncology group prospective trials 0116 and 0128. Int. J. Gynecol. Cancer 2012, 22, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Sapienza, L.G.; Jhingran, A.; Kollmeier, M.A.; Lin, L.L.; Calsavara, V.F.; Gomes, M.J.L.; Baiocchi, G. Decrease in uterine perforations with ultrasound image-guided applicator insertion in intracavitary brachytherapy for cervical cancer: A systematic review and meta-analysis. Gynecol. Oncol. 2018, 151, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Pareek, V.; Barthwal, M.; Giridhar, P.; Patil, P.A.; Upadhyay, A.D.; Mallick, S. A phase III randomised trial of trans-abdominal ultrasound in improving application quality and dosimetry of intra-cavitary brachytherapy in locally advanced cervical cancer. Gynecol. Oncol. 2021, 160, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Mayr, N.A.; Montebello, J.F.; Sorosky, J.I.; Daugherty, J.S.; Nguyen, D.L.; Mardirossian, G.; Wang, J.Z.; Edwards, S.M.; Li, W.; Yuh, W.T. Brachytherapy management of the retroverted uterus using ultrasound-guided implant applicator placement. Brachytherapy 2005, 4, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.B.; Portelance, L.; de la Zerda, A.; Wolfson, A. Practical Considerations During Brachytherapy Applicator Placement for Locally Advanced Cervical Cancer. Pract. Radiat. Oncol. 2023, 13, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Schaner, P.E.; Caudell, J.J.; De Los Santos, J.F.; Spencer, S.A.; Shen, S.; Kim, R.Y. Intraoperative ultrasound guidance during intracavitary brachytherapy applicator placement in cervical cancer: The University of Alabama at Birmingham experience. Int. J. Gynecol. Cancer 2013, 23, 559–566. [Google Scholar] [CrossRef]
- Chino, J.; Annunziata, C.M.; Beriwal, S.; Bradfield, L.; Erickson, B.A.; Fields, E.C.; Fitch, K.; Harkenrider, M.M.; Holschneider, C.H.; Kamrava, M.; et al. Radiation Therapy for Cervical Cancer: Executive Summary of an ASTRO Clinical Practice Guideline. Pract. Radiat. Oncol. 2020, 10, 220–234. [Google Scholar] [CrossRef]
- Sapienza, L.G.; Ludwig, M.S.; Baiocchi, G. US-guided versus US-assisted tandem insertion for cervical cancer: A nomenclature proposal. Brachytherapy 2022, 21, 260–261. [Google Scholar] [CrossRef]
- Knoth, J.; Nesvacil, N.; Sturdza, A.; Kronreif, G.; Widder, J.; Kirisits, C.; Schmid, M.P. Toward 3D-TRUS image-guided interstitial brachytherapy for cervical cancer. Brachytherapy 2022, 21, 186–192. [Google Scholar] [CrossRef]
- Lin, Y.; Shi, D.; Li, H.; Cheng, G.; Wang, H. Application of transrectal ultrasound in guiding interstitial brachytherapy for advanced cervical cancer. J. Contemp. Brachytherapy 2020, 12, 375–382. [Google Scholar] [CrossRef]
- Rodgers, J.R.; Surry, K.; Leung, E.; D’Souza, D.; Fenster, A. Toward a 3D transrectal ultrasound system for verification of needle placement during high-dose-rate interstitial gynecologic brachytherapy. Med. Phys. 2017, 44, 1899–1911. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liu, Z.; Jiang, S.; Zeng, J.; Hu, Y.; Zhang, G. Verification of needle guidance accuracy in pelvic phantom using registered ultrasound and MRI images for intracavitary/interstitial gynecologic brachytherapy. J. Contemp. Brachytherapy 2020, 12, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Liu, Z.; Jiang, S.; Pang, Q.; Wang, P. Verification of Guiding Needle Placement by Registered Ultrasound Image during Combined Intracavitary/Interstitial Gynecologic Brachytherapy. Cancer Manag. Res. 2021, 13, 1917–1928. [Google Scholar] [CrossRef] [PubMed]
- Fields, E.C.; Hazell, S.; Morcos, M.; Schmidt, E.J.; Chargari, C.; Viswanathan, A.N. Image-Guided Gynecologic Brachytherapy for Cervical Cancer. Semin. Radiat. Oncol. 2020, 30, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, A.N.; Szymonifka, J.; Tempany-Afdhal, C.M.; O’Farrell, D.A.; Cormack, R.A. A prospective trial of real-time magnetic resonance-guided catheter placement in interstitial gynecologic brachytherapy. Brachytherapy 2013, 12, 240–247. [Google Scholar] [CrossRef] [PubMed]
- De Arcos, J.; Schmidt, E.J.; Wang, W.; Tokuda, J.; Vij, K.; Seethamraju, R.T.; Damato, A.L.; Dumoulin, C.L.; Cormack, R.A.; Viswanathan, A.N. Prospective Clinical Implementation of a Novel Magnetic Resonance Tracking Device for Real-Time Brachytherapy Catheter Positioning. Int. J. Radiat. Oncol. Biol. Phys. 2017, 99, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Dumoulin, C.L.; Viswanathan, A.N.; Tse, Z.T.; Mehrtash, A.; Loew, W.; Norton, I.; Tokuda, J.; Seethamraju, R.T.; Kapur, T.; et al. Real-time active MR-tracking of metallic stylets in MR-guided radiation therapy. Magn. Reson. Med. 2015, 73, 1803–1811. [Google Scholar] [CrossRef]
- Cunha, J.A.M.; Flynn, R.; Bélanger, C.; Callaghan, C.; Kim, Y.; Jia, X.; Chen, Z.; Beaulieu, L. Brachytherapy Future Directions. Semin. Radiat. Oncol. 2020, 30, 94–106. [Google Scholar] [CrossRef]
- Song, W.Y.; Robar, J.L.; Morén, B.; Larsson, T.; Carlsson Tedgren, Å.; Jia, X. Emerging technologies in brachytherapy. Phys. Med. Biol. 2021, 66, 94–106. [Google Scholar] [CrossRef]
- Jiang, P.; Qu, A.; Wei, S.; Sun, H.; Zhang, X.; Li, X.; Wang, J. The Preliminary Results of 3-Dimensional Printed Individual Template Assisted 192Ir High-Dose Rate Interstitial Brachytherapy for Central Recurrent Gynecologic Cancer. Technol. Cancer Res. Treat. 2020, 19. [Google Scholar] [CrossRef]
- Lindegaard, J.C.; Madsen, M.L.; Traberg, A.; Meisner, B.; Nielsen, S.K.; Tanderup, K.; Spejlborg, H.; Fokdal, L.U.; Nørrevang, O. Individualised 3D printed vaginal template for MRI guided brachytherapy in locally advanced cervical cancer. Radiother. Oncol. 2016, 118, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Cobussen, A.; Petric, P.; Wulff, C.N.; Buus, S.; Spejlborg, H.; Nielsen, S.K.; Traberg, A.; Meisner, B.; Hokland, S.; Lindegaard, J.C. Clinical outcomes using a 3D printed tandem-needle-template and the EMBRACE-II planning aims for image guided adaptive brachytherapy in locally advanced cervical cancer. Acta Oncol. 2023, 62, 1470–1478. [Google Scholar] [CrossRef] [PubMed]
- Marar, M.; Simiele, E.; Niedermayr, T.; Kidd, E.A. Applying 3D-Printed Templates in High-Dose-Rate Brachytherapy for Cervix Cancer: Simplified Needle Insertion for Optimized Dosimetry. Int. J. Radiat. Oncol. Biol. Phys. 2022, 114, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Serban, M.; Fokdal, L.; Nielsen, S.K.; Hokland, S.B.; Hansen, A.T.; Spejlborg, H.; Rylander, S.; Petric, P.; Lindegaard, J.C.; Tanderup, K. Characterization of combined intracavitary/interstitial brachytherapy including oblique needles in locally advanced cervix cancer. Brachytherapy 2021, 20, 796–806. [Google Scholar] [CrossRef]
- Logar, H.B.Z.; Hudej, R.; Šegedin, B. Development and assessment of 3D-printed individual applicators in gynecological MRI-guided brachytherapy. J. Contemp. Brachytherapy 2019, 11, 128–136. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, Z.; Jiang, S.; Zhou, L.; Zhou, Z.; Wang, W. Individualized and inverse optimized needle configuration for combined intracavitary-interstitial brachytherapy in locally advanced cervical cancer. J. Cancer Res. Ther. 2019, 15, 1589–1596. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, K.; Wang, J.; Jiang, P. High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence. Curr. Oncol. 2024, 31, 50-65. https://doi.org/10.3390/curroncol31010004
Wang K, Wang J, Jiang P. High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence. Current Oncology. 2024; 31(1):50-65. https://doi.org/10.3390/curroncol31010004
Chicago/Turabian StyleWang, Kaiyue, Junjie Wang, and Ping Jiang. 2024. "High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence" Current Oncology 31, no. 1: 50-65. https://doi.org/10.3390/curroncol31010004
APA StyleWang, K., Wang, J., & Jiang, P. (2024). High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence. Current Oncology, 31(1), 50-65. https://doi.org/10.3390/curroncol31010004