Significance of Capping Agents of Colloidal Nanoparticles from the Perspective of Drug and Gene Delivery, Bioimaging, and Biosensing: An Insight
Abstract
:1. Introduction
2. Preparation and Biomedical Evaluation of Capped Nanoparticles
2.1. Physical and Chemical Synthesis
2.2. Biological/Green Synthesis
3. Effects of Capping Agents on Drug Delivery
4. Effects of Capping Agents on Gene Delivery
5. Effects of Capping Agents in Bioimaging
6. Effects of Capping Agents in Biosensing
7. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Salata, O. Applications of nanoparticles in biology and medicine. J. Nanobiotechnol. 2004, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Javed, R.; Ahmad, M.A.; Ao, Q. Theranostic Applications of Nanobiotechnology in Cancer. In Nanotheranostics; Rai, M., Jamil, B., Eds.; Springer: Cham, Swizherland, 2019. [Google Scholar] [CrossRef]
- Gulati, S.; Sachdeva, M.; Bhasin, K.K. Capping agents in nanoparticle synthesis: Surfactant and solvent system. AIP Conf. Proc. 2018, 1953, 030214. [Google Scholar] [CrossRef]
- Ahmad, M.A.; Yuesuo, Y.; Ao, Q.; Adeel, M.; Hui, Z.Y.; Javed, R. Appraisal of Comparative Therapeutic Potential of Undoped and Nitrogen-Doped Titanium Dioxide Nanoparticles. Molecules 2019, 24, 3916. [Google Scholar] [CrossRef] [PubMed]
- Javed, R.; Rais, F.; Fatima, H.; Haq, I.U.; Kaleem, M.; Naz, S.S.; Ao, Q. Chitosan encapsulated ZnO nanocomposites: Fabrication, characterization, and functionalization of bio-dental approaches. Mater. Sci. Eng. C 2020, 116, 111184. [Google Scholar] [CrossRef]
- Niu, Z.; Li, Y. Removal and Utilization of Capping Agents in Nanocatalysis. Chem. Mater. 2013, 26, 72–83. [Google Scholar] [CrossRef]
- Barman, S.R.; Nain, A.; Jain, S.; Punjabi, N.; Mukherji, S.; Satija, J. Dendrimer as a multifunctional capping agent for metal nanopar-ticles for use in bioimaging, drug delivery and sensor applications. J. Mater. Chem. B 2018, 6, 2368–2384. [Google Scholar] [CrossRef]
- Javed, R.; Usman, M.; Tabassum, S.; Zia, M. Effect of capping agents: Structural, optical and biological properties of ZnO nano-particles. Appl. Surf. Sci. 2016, 386, 319–326. [Google Scholar] [CrossRef]
- Anderson, S.D.; Gwenin, V.V.; Gwenin, C.D. Magnetic functionalized nanoparticles for biomedical, drug delivery and imaging applications. Nanoscale Res. Lett. 2019, 14, 188. [Google Scholar] [CrossRef]
- Jankiewicz, B.J.; Jamiola, D.; Choma, J.; Jaroniec, M. Silica–metal core–shell nanostructures. Adv. Colloid Interface Sci. 2012, 170, 28–47. [Google Scholar] [CrossRef]
- Restrepo, C.V.; Villa, C.C. Synthesis of silver nanoparticles, influence of capping agents, and dependence on size and shape: A review. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100428. [Google Scholar] [CrossRef]
- Latha, C.K.; Raghasudha, M.; Aparna, Y.; Ramchander, M.; Ravinder, D.; Jaipal, K.; Veerasomaiah, P.; Shridhar, D. Effect of Capping Agent on the Morphology, Size and Optical Properties of In2O3 Nanoparticles. Mater. Res. 2017, 20, 256–263. [Google Scholar] [CrossRef]
- Javed, R.; Zia, M.; Naz, S.; Aisida, S.O.; Ain, N.U.; Ao, Q. Role of capping agents in the application of nanoparticles in biomedicine and environmental remediation: Recent trends and future prospects. J. Nanobiotechnol. 2020, 18, 172. [Google Scholar] [CrossRef] [PubMed]
- Ajitha, B.; Reddy, Y.A.K.; Reddy, P.S.; Jeon, H.-J.; Ahn, C.W. Role of capping agents in controlling silver nanoparticles size, antibacterial activity and potential application as optical hydrogen peroxide sensor. RSC Adv. 2016, 6, 36171–36179. [Google Scholar] [CrossRef]
- Selvam, R.; Ramasamy, S.; Mohiyuddin, S.; Enoch, I.V.M.V.; Gopinath, P.; Filimonov, D. Molecular encapsulator–appended poly(vinyl alcohol) shroud on ferrite nanoparticles. Augmented cancer–drug loading and anticancer property. Mater. Sci. Eng. C 2018, 93, 125–133. [Google Scholar] [CrossRef]
- Popov, A.L.; Han, B.; Ermakov, A.M.; Savintseva, I.V.; Ermakova, O.N.; Popova, N.R.; Shcherbakov, A.B.; Shekunova, T.O.; Ivanova, O.S.; Kozlov, D.A.; et al. PVP-stabilized tungsten oxide nanoparticles: pH sensitive anti-cancer platform with high cytotoxicity. Mater. Sci. Eng. C 2020, 108, 110494. [Google Scholar] [CrossRef]
- Shalaby, T.I.; El-Refaie, W.M. Bioadhesive Chitosan-Coated Cationic Nanoliposomes with Improved Insulin Encapsulation and Prolonged Oral Hypoglycemic Effect in Diabetic Mice. J. Pharm. Sci. 2018, 107, 2136–2143. [Google Scholar] [CrossRef]
- Javed, R.; Ahmed, M.; Haq, I.U.; Nisa, S.; Zia, M. PVP and PEG doped CuO nanoparticles are more biologically active: Antibacteri-al, antioxidant, antidiabetic and cytotoxic perspective. Mater. Sci. Eng. C 2017, 79, 108–115. [Google Scholar] [CrossRef]
- Javed, R.; Rais, F.; Kaleem, M.; Jamil, B.; Ahmad, M.A.; Yu, T.; Qureshi, S.W.; Ao, Q. Chitosan capping of CuO nanoparticles: Facile chemical preparation, biological analysis, and applications in dentistry. Int. J. Biol. Macromol. 2020, 167, 1452–1467. [Google Scholar] [CrossRef]
- Ferreira, F.V.; Mariano, M.; Lepesqueur, L.S.S.; Pinheiro, I.F.; Santos, L.G.; BurgaSanchez, J. Silver nanoparticles coated with dodecan-ethiol used as fllers in non-cytotoxic and antifungal PBAT surface based on nanocomposites. Mater. Sci. Eng. C 2019, 98, 800–807. [Google Scholar] [CrossRef]
- Radini, I.A.; Hasan, N.; Malik, M.A.; Khan, Z. Biosynthesis of iron nanoparticles using Trigonella foenum-graecum seed extract for photocatalytic methyl orange dye degradation and antibacterial applications. J. Photochem. Photobiol. B Biol. 2018, 183, 154–163. [Google Scholar] [CrossRef]
- Javed, R.; Ghonaim, R.; Shathili, A.; Khalifa, S.A.M.; El-Seedi, H.R. Phytonanotechnology: A greener approach for biomedical ap-plications. In Micro and Nano Technologies: Biogenic Nanoparticles for Cancer Theranostics; Elsevier: Amsterdam, The Netherlands, 2021; pp. 43–86. [Google Scholar]
- Naz, S.; Islam, M.; Tabassum, S.; Fernandes, N.F.; Carcache de Blanco, E.J.; Zia, M. Green synthesis of hematite (α-Fe2O3) nanoparti-cles using Rhus punjabensis extract and their biomedical prospect in pathogenic diseases and cancer. J. Mol. Struct. 2019, 1185, 1–7. [Google Scholar] [CrossRef]
- Ravichandran, V.; Vasanthi, S.; Shalini, S.; Shah, S.A.A.; Tripathy, M.; Paliwal, N. Green synthesis, characterization, antibacterial, antioxidant and photocatalytic activity of Parkia speciosa leaves extract mediated silver nanoparticles. Results Phys. 2019, 15, 102565. [Google Scholar] [CrossRef]
- Wypij, M.; Jedrzejewski, T.; Trzcinska-Wencel, J.; Ostrowski, M.; Rai, M.; Golinska, P. Green synthesized silver nanoparticles: An-tibacterial and anticancer activities, biocompatibility, and analyses of surface-attached proteins. Front. Microbiol. 2021, 12, 632505. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Saravanakumar, K.; Jin, T.; Wang, M.-H. Mycosynthesis, characterization, anticancer and antibacterial activity of silver nanoparticles from endophytic fungus Talaromyces purpureogenus. Int. J. Nanomed. 2019, 14, 3427–3438. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Kim, S.-K.; Shim, M.S. Antimicrobial, Antioxidant, and Anticancer Activities of Biosynthesized Silver Nanoparticles Using Marine Algae Ecklonia cava. Nanomaterials 2016, 6, 235. [Google Scholar] [CrossRef]
- Patil, S.; Chandrasekaran, R. Biogenic nanoparticles: A comprehensive perspective in synthesis, characterization, application and its challenges. J. Genet. Eng. Biotechnol. 2020, 18, 67. [Google Scholar] [CrossRef]
- Naz, S.; Gul, A.; Zia, M. Toxicity of copper oxide nanoparticles: A review study. IET Nanobiotechnol. 2019, 14, 1–13. [Google Scholar] [CrossRef]
- Gu, F.; Zhang, L.; Teply, B.A.; Mann, N.; Wang, A.; Radovic-Moreno, A.F.; Langer, R.; Farokhzad, O.C. Precise engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers. Proc. Natl. Acad. Sci. USA 2008, 105, 2586–2591. [Google Scholar] [CrossRef]
- Paul, W.; Sharma, C.P. Inorganic nanoparticles for targeted drug delivery. In Biointegration of Medical Implant Materials; Elsevier: Amsterdam, The Netherlands, 2020; pp. 333–373. [Google Scholar]
- Mandal, B.; Bhattacharjee, H.; Mittal, N. Core–shell-type lipid–polymer hybrid nanoparticles as a drug delivery platform. Na-Nomed. Nanotechnol. Biol. Med. 2013, 9, 474–491. [Google Scholar] [CrossRef]
- Giménez, C.; de la Torre, C.; Gorbe, M.; Aznar, E.; Sancenón, F.; Murguía, J.R.; Martínez-Máñez, R.; Marcos, M.D.; Amorós, P. Gated Mesoporous Silica Nanoparticles for the Controlled Delivery of Drugs in Cancer Cells. Langmuir 2015, 31, 3753–3762. [Google Scholar] [CrossRef]
- Ajnai, G.; Chiu, A.; Kan, T.; Cheng, C.-C.; Tsai, T.-H.; Chang, J. Trends of Gold Nanoparticle-based Drug Delivery System in Cancer Therapy. J. Exp. Clin. Med. 2014, 6, 172–178. [Google Scholar] [CrossRef]
- Cole, A.J.; David, A.E.; Wang, J.; Galbán, C.J.; Hill, H.L.; Yang, V.C. Polyethylene glycol modified, cross-linked starch-coated iron oxide nanoparticles for enhanced magnetic tumor targeting. Biomaterials 2011, 32, 2183–2193. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Simchi, A.; Imani, M. Cytotoxicity of Uncoated and Polyvinyl Alcohol Coated Superparamagnetic Iron Oxide Nanoparticles. J. Phys. Chem. C 2009, 113, 9573–9580. [Google Scholar] [CrossRef]
- Paul, K.G.; Frigo, T.B.; Groman, J.Y.; Groman, E.V. Synthesis of ultrasmall superparamagnetic iron oxides using reduced polysac-charides. Bioconjug. Chem. 2004, 15, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.D.; Nativo, P.; Smith, J.-A.; Stirling, D.; Edwards, P.R.; Venugopal, B.; Flint, D.J.; Plumb, J.A.; Graham, D.; Wheate, N.J. Gold Nanoparticles for the Improved Anticancer Drug Delivery of the Active Component of Oxaliplatin. J. Am. Chem. Soc. 2010, 132, 4678–4684. [Google Scholar] [CrossRef]
- Katas, H.; Moden, N.Z.; Lim, C.S.; Celesistinus, T.; Chan, J.Y.; Ganasan, P.; Abdalla, S.S.I. Biosynthesis and potential applications of silver and gold nanoparticles and their chi-tosan-based nanocomposites in nanomedicine. J. Nanotechnol. 2018, 2018, 4290705. [Google Scholar]
- Fatrekar, A.P.; Morajkar, R.; Krishnan, S.; Dusane, A.; Madhyastha, H.; Vernekar, A.A. Delineating the Role of Tailored Gold Nanostructures at the Biointerface. ACS Appl. Bio Mater. 2021, 4, 8172–8191. [Google Scholar] [CrossRef]
- Ramalingam, V.; Varunkumar, K.; Ravikumar, V.; Rajaram, R. Target delivery of doxorubicin tethered with PVP stabilized gold nanoparticles for effective treatment of lung cancer. Sci. Rep. 2018, 8, 3815. [Google Scholar] [CrossRef]
- Yonezawa, Y.; Kawabata, I.; Sato, T. Photochemical formation of colloidal gold particles in chitosan films. Ber. Der Bunsenges. Für Phys. Chem. 1996, 100, 39–45. [Google Scholar] [CrossRef]
- Esumi, K.; Takei, N.; Yoshimura, T. Antioxidant-potentiality of gold–chitosan nanocomposites. Colloids Surf. B Biointerfaces 2003, 32, 117–123. [Google Scholar] [CrossRef]
- Ishizuki, N.; Torigoe, K.; Esumi, K.; Meguro, K. Characterization of precious metal particles prepared using chitosan as a pro-tective agent. Colloid Surf. 1991, 55, 15–21. [Google Scholar] [CrossRef]
- Mahalunkar, S.; Yadav, A.S.; Gorain, M.; Pawar, V.; Braathen, R.; Weiss, S.; Bogen, B.; Gosavi, S.W.; Kundu, G.C. Functional design of pH-responsive folate-targeted polymer-coated gold nano-particles for drug delivery and in vivo therapy in breast cancer. Int. J. Nanomed. 2019, 14, 8285. [Google Scholar] [CrossRef] [PubMed]
- Manju, S.; Sreenivasan, K. Conjugation of curcumin onto hyaluronic acid enhances its aqueous solubility and stability. J. Colloid Interface Sci. 2011, 359, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Golubeva, O.Y.; Shamova, O.V.; Orlov, D.S.; Yamshchikova, E.V.; Boldina, A.S.; Kokryakov, V.N. Synthesis and investigation of sil-ver-peptide bioconjugates and investigation in their antimicrobial activity. In Materials Challenges and Testing for Supply of Energy and Resources; Springer: Berlin/Heidelberg, Germany, 2012; pp. 163–171. [Google Scholar]
- Ravindra, S.; Mulaba-Bafubiandi, A.F.; Rajinikanth, V.; Varaprasad, K.; Reddy, N.N.; Raju, K.M. Development and Characterization of Curcumin Loaded Silver Nanoparticle Hydrogels for Antibacterial and Drug Delivery Applications. J. Inorg. Organomet. Polym. Mater. 2012, 22, 1254–1262. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Elechiguerra, J.L.; Burt, J.L.; Morones, J.R.; Camacho-Bragado, A.; Gao, X.; Lara, H.H.; Yacaman, M.J. Interaction of silver nanoparticles with HIV-1. J. Nanobiotechnol. 2005, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Singh, A.K.; Vig, K.; Pillai, S.R.; Singh, S.R. Silver nanoparticles inhibit replication of respiratory syncytial virus. J. Biomed. Nanotechnol. 2008, 4, 149–158. [Google Scholar] [CrossRef]
- Santos, M.; Seabra, A.; Pelegrino, M.; Haddad, P. Synthesis, characterization and cytotoxicity of glutathione- and PEG-glutathione-superparamagnetic iron oxide nanoparticles for nitric oxide delivery. Appl. Surf. Sci. 2016, 367, 26–35. [Google Scholar] [CrossRef]
- Kumar, R.; Inbaraj, B.S.; Chen, B. Surface modification of superparamagnetic iron nanoparticles with calcium salt of poly(γ-glutamic acid) as coating material. Mater. Res. Bull. 2010, 45, 1603–1607. [Google Scholar] [CrossRef]
- Rahayu, L.B.H.; Wulandari, I.O.; Santjojo, D.H.; Sabarudin, A. Synthesis and characterization of Fe3O4 nanoparticles using polyvi-nyl alcohol (PVA) as capping agent and glutaraldehyde (GA) as crosslinker. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2018; p. 012062. [Google Scholar]
- Dorniani, D.; Kura, A.U.; Hussein-Al-Ali, S.H.; Bin Hussein, M.Z.; Fakurazi, S.; Shaari, A.H.; Ahmad, Z. In Vitro Sustained Release Study of Gallic Acid Coated with Magnetite-PEG and Magnetite-PVA for Drug Delivery System. Sci. World J. 2014, 2014, 416354. [Google Scholar] [CrossRef] [Green Version]
- Franco, P.; De Marco, I. The Use of Poly(N-vinyl pyrrolidone) in the Delivery of Drugs: A Review. Polymers 2020, 12, 1114. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.A.; Praseetha, P.K.; Bhagat, M.; Alexander, P.; Abdeen, S.; Chavali, M. Drug Embedded PVP Coated Magnetic Nanoparticles for Targeted Killing of Breast Cancer Cells. Technol. Cancer Res. Treat. 2013, 12, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.; Bigham, A.; Hassanzadeh-Tabrizi, S.A.; Ahangar, H.A. Synthesis and characterization of Cu0.3Zn0.5Mg0.2Fe2O4 nanoparticles as a magnetic drug delivery system. J. Magn. Magn. Mater. 2017, 439, 67–75. [Google Scholar] [CrossRef]
- Waqas, H.; Khan, T.A.; Hameed, A.; Abbasi, R.; Naz, S.; Ahmed, M.J.; Shah, Z.H.; Hassan, S.M.; Qureshi, A.H.; Ahmed, M.B. In vitro cytotoxicity study of virgin, ethylenediaminetetraacetic acid-and hexameth-ylenetetramine-capped silica particles synthesized by precipitation method. Chem. Pap. 2020, 74, 1779–1789. [Google Scholar] [CrossRef]
- Song, Y.; Huang, Z.; Song, Y.; Tian, Q.; Liu, X.; She, Z.; Jiao, J.; Lu, E.; Deng, Y. The application of EDTA in drug delivery systems: Doxorubicin liposomes loaded via NH4EDTA gradient. Int. J. Nanomed. 2014, 9, 3611. [Google Scholar]
- Rajbongshi, H.; Bhattacharjee, S.; Datta, P. Shell thickness dependent photocatalytic activity of EDTA-assisted Ag/ZnO core-shell nanoparticles under sunlight irradiation. Mater. Res. Express 2018, 6, 045022. [Google Scholar] [CrossRef]
- Ferrero, M.E. Rationale for the Successful Management of EDTA Chelation Therapy in Human Burden by Toxic Metals. BioMed Res. Int. 2016, 2016, 8274504. [Google Scholar] [CrossRef]
- De-Deus, G.; Paciornik, S.; Mauricio, M.H.P. Evaluation of the effect of EDTA, EDTAC and citric acid on the microhardness of root dentine. Int. Endod. J. 2006, 39, 401–407. [Google Scholar] [CrossRef]
- Aghazadeh, M.; Karimzadeh, I.; Ganjali, M.R. Ethylenediaminetetraacetic acid capped superparamagnetic iron oxide (Fe3O4) nanoparticles: A novel preparation method and characterization. J. Magn. Magn. Mater. 2017, 439, 312–319. [Google Scholar] [CrossRef]
- Mishra, S.K.; Teotia, A.K.; Kumar, A.; Kannan, S. Mechanically tuned nanocomposite coating on titanium metal with integrated properties of biofilm inhibition, cell proliferation, and sustained drug delivery. Nanomed. Nanotechnol. Biol. Med. 2016, 13, 23–35. [Google Scholar] [CrossRef]
- Nguyen, N.; Le, C.H. Synthesis of PVA encapsulated silver nanoparticles as a drug delivery system for doxorubicin and cur-cumin. Int. J. High Sch. Res. 2021, 3, 41–47. [Google Scholar] [CrossRef]
- Sethuram, L.; Thomas, J.; Mukherjee, A.; Chandrasekaran, N. Effects and formulation of silver nanoscaffolds on cytotoxicity dependent ion release kinetics towards enhanced excision wound healing patterns in Wistar albino rats. RSC Adv. 2019, 9, 35677–35694. [Google Scholar] [CrossRef] [PubMed]
- Sakr, T.M.; Khowessah, O.M.; Motaleb, M.A.; Abd El-Bary, A.; El-Kolaly, M.T.; Swidan, M. I-131 doping of silver nanoparticles plat-form for tumor theranosis guided drug delivery. Eur. J. Pharm. Sci. 2018, 122, 239–245. [Google Scholar] [CrossRef]
- Alipour, R.; Khorshidi, A.; Shojaei, A.F.; Mashayekhi, F.; Moghaddam, M.J.M. Skin wound healing acceleration by Ag nanoparti-cles embedded in PVA/PVP/Pectin/Mafenide acetate composite nanofibers. Polym. Test. 2019, 79, 106022. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Tashkhourian, J.; Ana, S.F.N. Topical delivery of chitosan-capped silver nanoparticles speeds up heal-ing in burn wounds: A preclinical study. Carbohydr. Polym. 2018, 200, 82–92. [Google Scholar] [CrossRef]
- Prabha, G.; Raj, V. Sodium alginate–polyvinyl alcohol–bovin serum albumin coated Fe3O4 nanoparticles as anticancer drug delivery vehicle: Doxorubicin loading and in vitro release study and cytotoxicity to HepG2 and L02 cells. Mater. Sci. Eng. C 2017, 79, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Shah, E.; Upadhyay, P.; Singh, M.; Mansuri, M.S.; Begum, R.; Sheth, N.; Soni, H.P. EDTA capped iron oxide nanoparticles magnetic micelles: Drug delivery vehicle for treatment of chronic myeloid leukemia and T1–T2 dual contrast agent for magnetic resonance imaging. New J. Chem. 2016, 40, 9507–9519. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, D.; Ma, Y.; Zhang, Z.; Che, H.; Mu, J.; Zhang, X.; Zhang, Z. Synthesis and characterization of polymer-coated manganese ferrite nanoparticles as con-trolled drug delivery. Appl. Surf. Sci. 2018, 428, 258–263. [Google Scholar] [CrossRef]
- Sampath, M.; Pichaimani, A.; Kumpati, P.; Sengottuvelan, B. The remarkable role of emulsifier and chitosan, dextran and PEG as capping agents in the enhanced delivery of curcumin by nanoparticles in breast cancer cells. Int. J. Biol. Macromol. 2020, 162, 748–761. [Google Scholar] [CrossRef]
- Choi, J.Y.; Gupta, B.; Ramasamy, T.; Jeong, J.H.; Jin, S.G.; Choi, H.G.; Yong, C.S.; Kim, J.O. PEGylated polyaminoacid-capped mesoporous silica nanoparticles for mitochon-dria-targeted delivery of celastrol in solid tumors. Colloids Surf. B Biointerfaces 2018, 165, 56–66. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, G.; Zhang, Y.; Liu, D.; Xu, B.; Zhou, J. Near-infrared light triggered and separable microneedles for transdermal de-livery of metformin in diabetic rats. J. Mater. Chem. B 2017, 5, 9507–9513. [Google Scholar] [CrossRef] [PubMed]
- Khutale, G.V.; Casey, A. Synthesis and characterization of a multifunctional gold-doxorubicin nanoparticle system for pH triggered intracellular anticancer drug release. Eur. J. Pharm. Biopharm. 2017, 119, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Luong, D.; Sau, D.; Kesharwani, P.; Iyer, A.K. Polyvalent folate-dendrimer-coated iron oxide theranostic nanoparticles for sim-ultaneous magnetic resonance imaging and precise cancer cell targeting. Biomacromolecules 2017, 18, 1197–1209. [Google Scholar] [CrossRef] [PubMed]
- Murawala, P.; Tirmale, A.; Shiras, A.; Prasad, B.L.V. In situ synthesized BSA capped gold nanoparticles: Effective carrier of anti-cancer drug methotrexate to MCF-7 breast cancer cells. Mater. Sci. Eng. C 2014, 34, 158–167. [Google Scholar] [CrossRef]
- Chen, G.; Xie, Y.; Peltier, R.; Lei, H.; Wang, P.; Chen, J.; Hu, Y.; Wang, F.; Yao, X.; Sun, H. Peptide-Decorated Gold Nanoparticles as Functional Nano-Capping Agent of Mesoporous Silica Container for Targeting Drug Delivery. ACS Appl. Mater. Interfaces 2016, 8, 11204–11209. [Google Scholar] [CrossRef]
- Zhao, J.; Li, X.; Wang, X. Fabrication of Hybrid Nanostructures Based on Fe3O4 Nanoclusters as Theranostic Agents for Magnetic Resonance Imaging and Drug Delivery. Nanoscale Res. Lett. 2019, 14, 200. [Google Scholar] [CrossRef]
- Narkhede, A.A.; Sherwood, J.A.; Antone, A.; Coogan, K.R.; Bolding, M.S.; Deb, S.; Bao, Y.; Rao, S.S. Role of Surface Chemistry in Mediating the Uptake of Ultrasmall Iron Oxide Nanoparticles by Cancer Cells. ACS Appl. Mater. Interfaces 2019, 11, 17157–17166. [Google Scholar] [CrossRef]
- Küp, F.; Çoşkunçay, S.; Duman, F. Biosynthesis of silver nanoparticles using leaf extract of Aesculus hippocastanum (horse chestnut): Evaluation of their antibacterial, antioxidant and drug release system activities. Mater. Sci. Eng. C 2019, 107, 110207. [Google Scholar] [CrossRef]
- Patra, S.; Mukherjee, S.; Barui, A.K.; Ganguly, A.; Sreedhar, B.; Patra, C.R. Green synthesis, characterization of gold and silver na-noparticles and their potential application for cancer therapeutics. Mater. Sci. Eng. C 2015, 53, 298–309. [Google Scholar] [CrossRef]
- Sathiyaseelan, A.; Saravanakumar, K.; Mariadoss, A.V.A.; Wang, M.H. Antimicrobial and wound healing properties of FeO fabri-cated chitosan/PVA nanocomposite sponge. Antibiotics 2021, 10, 524. [Google Scholar] [CrossRef]
- Kalantari, K.; Mostafavi, E.; Saleh, B.; Soltantabar, P.; Webster, T.J. Chitosan/PVA hydrogels incorporated with green synthesized cerium oxide nanoparticles for wound healing applications. Eur. Polym. J. 2020, 134, 109853. [Google Scholar] [CrossRef]
- Akbarian, M.; Mahjoub, S.; Elahi, S.M.; Zabihi, E.; Tashakkorian, H. Green synthesis, formulation and biological evaluation of a novel ZnO nanocarrier loaded with paclitaxel as drug delivery system on MCF-7 cell line. Colloids Surf. B Biointerfaces 2019, 186, 110686. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.J.; Jasim, S.A.; Taban, T.Z.; Bokov, D.O.; Shalaby, M.N.; Al-Gazally, M.E.; Kzar, H.H.; Qasim, M.T.; Mustafa, Y.F.; Khatami, M. Anticancer Drug-Loading Capacity of Green Synthesized Porous Magnetic Iron Nanocarrier and Cytotoxic Effects Against Human Cancer Cell Line. J. Clust. Sci. 2022, 1–11. [Google Scholar] [CrossRef]
- Kang, L.; Gao, Z.; Huang, W.; Jin, M.; Wang, Q. Nanocarrier-mediated co-delivery of chemotherapeutic drugs and gene agents for cancer treatment. Acta Pharm. Sin. B 2015, 5, 169–175. [Google Scholar] [CrossRef]
- Pack, D.W.; Hoffman, A.S.; Pun, S.; Stayton, P.S. Design and development of polymers for gene delivery. Nat. Rev. Drug Discov. 2005, 4, 581–593. [Google Scholar] [CrossRef]
- Morille, M.; Passirani, C.; Vonarbourg, A.; Clavreul, A.; Benoit, J.-P. Progress in developing cationic vectors for non-viral systemic gene therapy against cancer. Biomaterials 2008, 29, 3477–3496. [Google Scholar] [CrossRef]
- Fedlheim, D.L.; Foss, C.A. Metal Nanoparticles: Synthesis, Characterization, and Applications; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Fernandes, A.R.; Baptista, P.V. Gene Silencing Using Multifunctionalized Gold Nanoparticles for Cancer Therapy. In Cancer Nanotechnology; Humana Press: New York, NY, USA, 2017; pp. 319–336. [Google Scholar] [CrossRef]
- Baptista, P.V. Gold nanoprobe-based non-crosslinking hybridization for molecular diagnostics: An update. Expert Rev. Mol. Diagn. 2018, 18, 767–773. [Google Scholar] [CrossRef]
- Lee, S.; Yoon, S.-M.; Shin, H.-J.; Joo, W.-J.; Yi, D.K.; Choi, J.-Y.; A Amarnath, C.; Paik, U. Hierarchical organization of Au nanoparticles in a poly(vinyl carbazole) matrix for hybrid electronic devices. Nanotechnology 2008, 19, 075606. [Google Scholar] [CrossRef]
- Prakash, A.; Ouyang, J.; Lin, J.-L.; Yang, Y. Polymer memory device based on conjugated polymer and gold nanoparticles. J. Appl. Phys. 2006, 100, 054309. [Google Scholar] [CrossRef]
- Chen, C.; Wang, W.; Ge, J.; Zhao, X.S. Kinetics and thermodynamics of DNA hybridization on gold nanoparticles. Nucleic Acids Res. 2009, 37, 3756–3765. [Google Scholar] [CrossRef] [Green Version]
- Javier, D.J.; Nitin, N.; Levy, M.; Ellington, A.; Richards-Kortum, R. Aptamer-Targeted Gold Nanoparticles As Molecular-Specific Contrast Agents for Reflectance Imaging. Bioconjugate Chem. 2008, 19, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Yonezawa, T.; Onoue, S.-Y.; Kimizuka, N. Metal Coating of DNA Molecules by Cationic, Metastable Gold Nanoparticles. Chem. Lett. 2002, 31, 1172–1173. [Google Scholar] [CrossRef]
- Sandström, P.; Boncheva, M.; Åkerman, B. Nonspecific and Thiol-Specific Binding of DNA to Gold Nanoparticles. Langmuir 2003, 19, 7537–7543. [Google Scholar] [CrossRef]
- Rosi, N.L.; Giljohann, D.A.; Thaxton, C.S.; Lytton-Jean, A.K.R.; Han, M.S.; Mirkin, C.A. Oligonucleotide-Modified Gold Nanoparticles for Intracellular Gene Regulation. Science 2006, 312, 1027–1030. [Google Scholar] [CrossRef]
- Barron, N.; Piskareva, O.; Muniyappa, M. Targeted genetic modification of cell lines for recombinant protein production. Cytotechnology 2007, 53, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.-H.; Huang, Y.-F.; Zhang, C.-Z.; Niu, J.; Chen, Y.; Chu, Y.; Jiang, Z.-H.; Gao, J.-Q.; Mao, Z.-W. Integration of antimicrobial peptides with gold nanoparticles as unique non-viral vectors for gene delivery to mesenchymal stem cells with antibacterial activity. Biomaterials 2016, 103, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Riley, M.K.; Vermerris, W. Recent Advances in Nanomaterials for Gene Delivery—A Review. Nanomaterials 2017, 7, 94. [Google Scholar] [CrossRef]
- Peng, J.; Liang, X. Progress in research on gold nanoparticles in cancer management. Medicine 2019, 98, e15311. [Google Scholar] [CrossRef]
- Wong, J.K.; Mohseni, R.; Hamidieh, A.A.; MacLaren, R.E.; Habib, N.; Seifalian, A.M. Will nanotechnology bring new hope for gene delivery? Trends Biotechnol. 2017, 35, 434–451. [Google Scholar] [CrossRef]
- Mendes, R.; Fernandes, A.R.; Baptista, P.V. Gold Nanoparticle Approach to the Selective Delivery of Gene Silencing in Cancer—The Case for Combined Delivery? Genes 2017, 8, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinhas, R.; Mendes, R.; Fernandes, A.R.; Baptista, P.V. Nanoparticles—emerging potential for managing leukemia and lympho-ma. Front. Bioeng. Biotechnol. 2017, 5, 79. [Google Scholar] [CrossRef] [PubMed]
- Abrica-Gonzalez, P.; Zamora-Justo, J.A.; Sotelo-Lopez, A.; Vázquez-Martínez, G.R.; Balderas-López, J.A.; Muñoz-Diosdado, A.; Ibáñez-Hernández, M. Gold nanoparticles with chitosan, N-acylated chitosan, and chi-tosan oligosaccharide as DNA carriers. Nanoscale Res. Lett. 2019, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Newell, B.; Irudayaraj, J. Folic Acid Protected Silver Nanocarriers for Targeted Drug Delivery. J. Biomed. Nanotechnol. 2012, 8, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Soumya, R.S.; Hela, P.G. Nano silver based targeted drug delivery for treatment of cancer. Der Pharm. Lett. 2013, 5, 189–197. [Google Scholar]
- Jin, L.; Wang, Q.; Chen, J.; Wang, Z.; Xin, H.; Zhang, D. Efficient Delivery of Therapeutic siRNA by Fe3O4 Magnetic Nanoparticles into Oral Cancer Cells. Pharmaceutics 2019, 11, 615. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Zhang, M.; Lu, J.-J.; Xu, F.; Chen, B.-A.; Xing, L.; Jiang, H.-L. PK11195-chitosan-graft-polyethylenimine-modified SPION as a mitochondria-targeting gene carrier. J. Drug Target. 2015, 24, 457–467. [Google Scholar] [CrossRef]
- Rahme, K.; Guo, J.; Holmes, J.D.; O’Driscoll, C.M. Evaluation of the physicochemical properties and the biocompatibility of pol-yethylene glycol-conjugated gold nanoparticles: A formulation strategy for siRNA delivery. Colloids Surf. B Biointerfaces 2015, 135, 604–612. [Google Scholar] [CrossRef]
- Jabir, M.; Sahib, U.I.; Taqi, Z.; Taha, A.; Sulaiman, G.; Albukhaty, S.; Al-Shammari, A.; Alwahibi, M.; Soliman, D.; Dewir, Y.H.; et al. Linalool-loaded glutathione-modified gold nanoparticles conjugated with CALNN peptide as apoptosis inducer and NF-κB translocation inhibitor in SKOV-3 cell line. Int. J. Nanomed. 2020, 15, 9025. [Google Scholar] [CrossRef]
- Zhang, L.; Jean, S.R.; Li, X.; Sack, T.; Wang, Z.; Ahmed, S.; Chan, G.; Das, J.; Zaragoza, A.; Sargent, E.H.; et al. Programmable metal/semiconductor nanostructures for mRNA-modulated molecular delivery. Nano Lett. 2018, 18, 6222–6228. [Google Scholar] [CrossRef]
- Abdel-Rashid, R.S.; Omar, S.M.; Teiama, M.S.; Khairy, A.; Magdy, M.; Anis, B. Fabrication of gold nanoparticles in absence of sur-factant as in vitro carrier of plasmid DNA. Int. J. Nanomed. 2019, 14, 8399. [Google Scholar] [CrossRef]
- Lee, C.-S.; Kim, H.; Yu, J.; Yu, S.H.; Ban, S.; Oh, S.; Jeong, D.; Im, J.; Baek, M.J.; Kim, T.H. Doxorubicin-loaded oligonucleotide conjugated gold nanoparticles: A promising in vivo drug delivery system for colorectal cancer therapy. Eur. J. Med. Chem. 2017, 142, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.A.; Rahme, K.; Guo, J.; Holmes, J.D.; O’Driscoll, C.M. Anisamide-targeted gold nanoparticles for siRNA delivery in prostate cancer–synthesis, physicochemical characterisation and in vitro evaluation. J. Mater. Chem. B 2016, 4, 2242–2252. [Google Scholar] [CrossRef] [PubMed]
- Azlan, A.Y.H.N.; Katas, H.; Habideen, N.H.; Busra, M.F.M. Dual-action of thermoresponsive gels containing DsiRNA-loaded gold nanoparticles for diabetic wound therapy: Characterization, in vitro safety and healing efficacy. Saudi Pharm. J. 2020, 28, 1420–1430. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Roessl, E.; Schlomka, J.P.; Caruthers, S.D.; Senpan, A.; Scott, M.J.; Allen, J.S.; Zhang, H.; Hu, G.; Gaffney, P.J.; et al. Computed tomography in color: NanoK-enhanced spectral CT molecular imaging. Angew. Chem. Int. Ed. 2020, 122, 9829–9833. [Google Scholar] [CrossRef]
- Kunjachan, S.; Ehling, J.; Storm, G.; Kiessling, F.; Lammers, T. Noninvasive Imaging of Nanomedicines and Nanotheranostics: Principles, Progress, and Prospects. Chem. Rev. 2015, 115, 10907–10937. [Google Scholar] [CrossRef] [PubMed]
- Ni, D.; Zhang, J.; Bu, W.; Xing, H.; Han, F.; Xiao, Q.; Yao, Z.; Chen, F.; He, Q.; Liu, J.; et al. Dual-targeting upconversion nanoprobes across the blood–brain barrier for magnetic reso-nance/fluorescence imaging of intracranial glioblastoma. ACS Nano 2014, 8, 1231–1242. [Google Scholar] [CrossRef]
- Rieffel, J.; Chen, F.; Kim, J.; Chen, G.; Shao, W.; Shao, S.; Chitgupi, U.; Hernandez, R.; Graves, S.A.; Nickles, R.J.; et al. Hexamodal Imaging with Porphyrin-Phospholipid-Coated Upconversion Nanoparticles. Adv. Mater. 2015, 27, 1785–1790. [Google Scholar] [CrossRef]
- Cai, E.; Ge, P.; Lee, S.H.; Jeyifous, O.; Wang, Y.; Liu, Y.; Wilson, K.M.; Lim, S.J.; Baird, M.A.; Stone, J.E.; et al. Stable Small Quantum Dots for Synaptic Receptor Tracking on Live Neurons. Angew. Chem. 2014, 126, 12692–12696. [Google Scholar] [CrossRef]
- Motiei, M.; Dreifuss, T.; Betzer, O.; Panet, H.; Popovtzer, A.; Santana, J.; Abourbeh, G.; Mishani, E.; Popovtzer, R. Differentiating between cancer and inflammation: A metabolic-based method for func-tional computed tomography imaging. ACS Nano 2016, 10, 3469–3477. [Google Scholar] [CrossRef]
- Cheng, H.-B.; Li, Y.; Tang, B.Z.; Yoon, J. Assembly strategies of organic-based imaging agents for fluorescence and photoacoustic bioimaging applications. Chem. Soc. Rev. 2019, 49, 21–31. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, T.; Liu, Z. Metal-Organic Frameworks for Bioimaging: Strategies and Challenges. Nanotheranostics 2022, 6, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Gao, H.; Lei, Y. Guest-host doped strategy for constructing ultralong-lifetime near-infrared organic phosphores-cence materials for bioimaging. Nat. Commun. 2022, 13, 186. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, H.; Liu, J.; Haley, K.N.; Treadway, J.; Larson, J.P.; Ge, N.; Peale, F.; Bruchez, M. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat. Biotechnol. 2002, 21, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Hadjipanayis, C.G.; Bonder, M.J.; Balakrishnan, S.; Wang, X.; Mao, H.; Hadjipanayis, G.C. Metallic iron nanoparticles for MRI con-trast enhancement and local hyperthermia. Small 2008, 4, 1925–1929. [Google Scholar] [CrossRef]
- Mi, P.; Kokuryo, D.; Cabral, H. A pH-activatable nanoparticle with signal-amplification capabilities for non-invasive imaging of tumour malignancy. Nat. Nanotechnol. 2016, 11, 724–730. [Google Scholar] [CrossRef]
- Lee, N.; Choi, Y.; Lee, Y.; Park, M.; Moon, W.K.; Choi, S.H.; Hyeon, T. Water-Dispersible Ferrimagnetic Iron Oxide Nanocubes with Extremely High r2 Relaxivity for Highly Sensitive in Vivo MRI of Tumors. Nano Lett. 2012, 12, 3127–3131. [Google Scholar] [CrossRef]
- Chen, R.; Christiansen, M.G.; Sourakov, A.; Mohr, A.; Matsumoto, Y.; Okada, S.; Jasanoff, A.; Anikeeva, P. High-Performance Ferrite Nanoparticles through Nonaqueous Redox Phase Tuning. Nano Lett. 2016, 16, 1345–1351. [Google Scholar] [CrossRef]
- Kim, D.; Park, S.; Lee, J.H.; Jeong, Y.Y.; Jon, S. Antibiofouling Polymer-Coated Gold Nanoparticles as a Contrast Agent for in Vivo X-ray Computed Tomography Imaging. J. Am. Chem. Soc. 2007, 129, 7661–7665. [Google Scholar] [CrossRef]
- Hainfeld, J.F.; Slatkin, D.N.; Focella, T.M.; Smilowitz, H.M. Gold nanoparticles: A new X-ray contrast agent. Br. J. Radiol. 2006, 79, 248–253. [Google Scholar] [CrossRef]
- Ai, K.; Liu, Y.; Liu, J.; Yuan, Q.; He, Y.; Lu, L. Large-Scale Synthesis of Bi2S3Nanodots as a Contrast Agent for In Vivo X-ray Computed Tomography Imaging. Adv. Mater. 2011, 23, 4886–4891. [Google Scholar] [CrossRef]
- Kinsella, J.M.; Jimenez, R.E.; Karmali, P.P.; Rush, A.M.; Kotamraju, V.R.; Gianneschi, N.C.; Ruoslahti, E.; Stupack, D.; Sailor, M.J. X-ray computed tomography imaging of breast cancer by using targeted pep-tide-labeled bismuth sulfide nanoparticles. Angew. Chem. Int. Ed. 2011, 50, 12308–12311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonitatibus, P.J.; Torres, A.S.; Goddard, G.D.; FitzGerald, P.F.; Kulkarni, A.M. Synthesis, characterization, and computed tomogra-phy imaging of a tantalum oxide nanoparticle imaging agent. Chem. Comm. 2010, 46, 8956–8958. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.H.; Lee, N.; Kim, H.; Park, S.P.; Piao, Y.; Lee, J.; Jun, S.W.; Moon, W.K.; Choi, S.H.; Hyeon, T.; et al. Large-scale synthesis of bioinert tantalum oxide nanoparticles for X-ray computed tomogra-phy imaging and bimodal image-guided sentinel lymph node mapping. J. Am. Chem. Soc. 2021, 133, 5508–5515. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Cui, Y.; Levenson, R.M.; Chung, L.W.K.; Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lim, Y.T.; Soltesz, E.G.; De Grand, A.M.; Lee, J.; Nakayama, A.; Parker, J.A.; Mihaljevic, T.; Laurence, R.G.; Dor, D.M.; et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat. Biotechnol. 2003, 22, 93–97. [Google Scholar] [CrossRef]
- Tsoi, K.M.; Dai, Q.; Alman, B.A.; Chan, W.C.W. Are Quantum Dots Toxic? Exploring the Discrepancy between Cell Culture and Animal Studies. Acc. Chem. Res. 2012, 46, 662–671. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.-M.; Yan, X.-P. Self-Assembly of Folate onto Polyethyleneimine-Coated CdS/ZnS Quantum Dots for Targeted Turn-On Fluorescence Imaging of Folate Receptor Overexpressed Cancer Cells. Anal. Chem. 2012, 85, 228–234. [Google Scholar] [CrossRef]
- Li, J.; Chang, X.; Chen, X.; Gu, Z.; Zhao, F.; Chai, Z.; Zhao, Y. Toxicity of inorganic nanomaterials in biomedical imaging. Biotechnol. Adv. 2014, 32, 727–743. [Google Scholar] [CrossRef]
- Liu, L.; Lin, G.; Yin, F.; Law, W.C.; Yong, K.T. Near-infrared fluorescent peptide probes for imaging of tumor in vivo and their biotoxicity evaluation. J. Biomed. Mater. Res. A. 2016, 104, 910–916. [Google Scholar] [CrossRef]
- Guo, C.; Irudayaraj, J. Fluorescent Ag Clusters via a Protein-Directed Approach as a Hg(II) Ion Sensor. Anal. Chem. 2011, 83, 2883–2889. [Google Scholar] [CrossRef]
- Liu, R.; Wang, Y.-L.; Cui, Y.-Y.; Sun, Z.-P.; Wei, Y.-T.; Gao, X.-Y. Recent Development of Noble Metal Clusters for Bioimaging and In vitro Detection. Acta Agron. Sin. 2013, 40, 977. [Google Scholar] [CrossRef]
- Diez, I.; Ras, R.H. Fluorescent silver nanoclusters. Nanoscale 2011, 3, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Mathew, A.; Pradeep, T. Noble Metal Clusters: Applications in Energy, Environment, and Biology. Part. Part. Syst. Charact. 2014, 31, 1017–1053. [Google Scholar] [CrossRef]
- Reyes-Esparza, J.; Martínez-Mena, A.; Gutiérrez-Sancha, I.; Rodríguez-Fragoso, P.; De La Cruz, G.G.; Mondragon, R.; Rodríguez-Fragoso, L. Synthesis, characterization and biocompatibility of cadmium sulfide nanoparticles capped with dextrin for in vivo and in vitro imaging application. J. Nanobiotechnol. 2015, 13, 83. [Google Scholar] [CrossRef]
- Peng, C.; Li, K.; Cao, X.; Xiao, T.; Hou, W.; Zheng, L.; Guo, R.; Shen, M.; Zhang, G.; Shi, X. Facile formation of dendrimer-stabilized gold nanoparticles modified with diatrizoic acid for enhanced computed tomography imaging applications. Nanoscale 2012, 4, 6768–6778. [Google Scholar] [CrossRef]
- Liu, H.; Shen, M.; Zhao, J.; Zhu, J.; Xiao, T.; Cao, X.; Zhang, G.; Shi, X. Facile formation of folic acid-modified dendrimer-stabilized gold–silver alloy nanoparticles for potential cellular computed tomography imaging applications. Analyst 2013, 138, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, H.; Guo, R.; Cao, X.; Zhao, J.; Luo, Y.; Shen, M.; Zhang, G.; Shi, X. Size-controlled synthesis of dendrimer-stabilized silver nanoparticles for X-ray computed to-mography imaging applications. Polym. Chem. 2010, 1, 1677–1683. [Google Scholar] [CrossRef]
- Liu, H.; Xu, Y.; Wen, S.; Chen, Q.; Zheng, L.; Shen, M.; Zhao, J.; Zhang, G.; Shi, X. Targeted Tumor Computed Tomography Imaging Using Low-Generation Dendrimer-Stabilized Gold Nanoparticles. Chem. A Eur. J. 2013, 19, 6409–6416. [Google Scholar] [CrossRef]
- Gong, T.; Goh, D.; Olivo, M.; Yong, K.-T. In vitro toxicity and bioimaging studies of gold nanorods formulations coated with biofunctional thiol-PEG molecules and Pluronic block copolymers. Beilstein J. Nanotechnol. 2014, 5, 546–553. [Google Scholar] [CrossRef]
- Moghimi, H.; Zohdiaghdam, R.; Riahialam, N.; Behrouzkia, Z. The Assessment of Toxicity Characteristics of Cellular Uptake of Paramagnetic Nanoparticles as a New Magnetic Resonance Imaging Contrast Agent. Iran. J. Pharm. Res. IJPR 2019, 18, 2083–2092. [Google Scholar] [CrossRef]
- Mishra, S.K.; Kumar, B.H.; Khushu, S.; Tripathi, R.P.; Gangenahalli, G. Increased transverse relaxivity in ultrasmall superpara-magnetic iron oxide nanoparticles used as MRI contrast agent for biomedical imaging. Contrast Media Mol. Imaging 2016, 11, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Cheheltani, R.; Ezzibdeh, R.M.; Chhour, P.; Pulaparthi, K.; Kim, J.; Jurcova, M.; Hsu, J.C.; Blundell, C.; Litt, H.I.; Ferrari, V.A.; et al. Tunable, biodegradable gold nanoparticles as contrast agents for computed tomography and photoacoustic imaging. Biomaterials 2016, 102, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Hainfeld, J.F.; Ridwan, S.M.; Stanishevskiy, Y.; Smilowitz, N.R.; Davis, J.; Smilowitz, H.M. Small, long blood half-life iodine nano-particle for vascular and tumor imaging. Sci. Rep. 2018, 8, 13803. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Zhao, L.; Zhu, J.; Li, Y.; Song, N.; Xing, Y.; Qiao, W.; Huang, H.; Zhao, J. 131I-labeled polyethylenimine-entrapped gold nanoparticles for targeted tumor SPECT/CT im-aging and radionuclide therapy. Int. J. Nanomed. 2019, 14, 4367. [Google Scholar] [CrossRef]
- Chen, J.; Yang, X.-Q.; Qin, M.-Y.; Zhang, X.-S.; Xuan, Y.; Zhao, Y.-D. Hybrid nanoprobes of bismuth sulfide nanoparticles and CdSe/ZnS quantum dots for mouse computed tomography/fluorescence dual mode imaging. J. Nanobiotechnol. 2015, 13, 76. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.B.; Kumar, D.; Li, Y.; Lee, I.-K.; Cho, S.J.; Kim, S.K.; Jeong, S.Y.; Lee, J.; Jeon, Y.H. PEGylated crushed gold shell-radiolabeled core nanoballs for in vivo tumor imaging with dual positron emission tomography and Cerenkov luminescent imaging. J. Nanobiotechnol. 2018, 16, 41. [Google Scholar] [CrossRef]
- Cui, L.; Xiong, C.; Zhou, M.; Shi, S.; Chow, D.S.; Li, C. Integrin αvβ3-targeted [64cu] cus nanoparticles for pet/ct imaging and pho-tothermal ablation therapy. Bioconjug. Chem. 2018, 29, 4062–4071. [Google Scholar] [CrossRef]
- Sun, I.-C.; Ahn, C.-H.; Kim, K.; Emelianov, S. Photoacoustic imaging of cancer cells with glycol-chitosan-coated gold nanoparticles as contrast agents. J. Biomed. Opt. 2019, 24, 121903–121905. [Google Scholar] [CrossRef]
- Prodi, L.; Biffi, S.; Petrizza, L.; Garrovo, C.; Rampazzo, E.; Andolfi, L.; Giustetto, P.; Nikolov, I.; Kurdi, G.; Danailov, M.B.; et al. Multimodal near-infrared-emitting PluS Silica nanoparticles with fluorescent, photoacoustic, and photothermal capabilities. Int. J. Nanomed. 2016, 11, 4865–4874. [Google Scholar] [CrossRef]
- Yang, Z.; Song, J.; Dai, Y.; Chen, J.; Wang, F.; Lin, L.; Liu, Y.; Zhang, F.; Yu, G.; Zhou, Z.; et al. Self-Assembly of Semiconducting-Plasmonic Gold Nanoparticles with Enhanced Optical Property for Photoacoustic Imaging and Photothermal Therapy. Theranostics 2017, 7, 2177–2185. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, H.; Li, X.; Duan, N.; Hu, S.; Liu, Y.; Yue, Y.; Song, L.; Zhang, Y.; Li, D.; et al. Plectin-1 Targeted Dual-modality Nanoparticles for Pancreatic Cancer Imaging. eBioMedicine 2018, 30, 129–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Runjun, S.; Kumar, D.M.; Lakshi, S.; Anand, R.; Ratul, S. Conjugation of curcumin with Ag nanoparticle for improving its bioa-vailability and study of the bioimaging response. Наносистемы Физика Химия Математика 2021, 12, 528–535. [Google Scholar]
- El-Sayed, N.; Trouillet, V.; Clasen, A.; Jung, G.; Hollemeyer, K.; Schneider, M. NIR-emitting gold nanoclusters–modified gelatin nanoparticles as a bioimaging agent in tissue. Adv. Healthcare Mater. 2019, 8, 1900993. [Google Scholar] [CrossRef] [PubMed]
- Pandey, G.; Marimuthu, M.; Kanagavalli, P.; Ravichandiran, V.; Balamurugan, K.; Veerapandian, M. Chitosanylated MoO3–Ruthenium(II) Nanocomposite as Biocompatible Probe for Bioimaging and Herbaceutical Detection. ACS Biomater. Sci. Eng. 2019, 5, 3606–3617. [Google Scholar] [CrossRef]
- Kotcherlakota, R.; Nimushakavi, S.; Roy, A.; Yadavalli, H.C.; Mukherjee, S.; Haque, S.; Patra, C.R. Biosynthesized gold nanoparticles: In vivo study of near-infrared fluores-cence (NIR)-based bio-imaging and cell labeling applications. ACS Biomater. Sci. Eng. 2019, 5, 5439–5452. [Google Scholar] [CrossRef]
- Yaraki, M.T.; Pan, Y.; Hu, F.; Yu, Y.; Liu, B.; Tan, Y.N. Nanosilver-enhanced AIE photosensitizer for simultaneous bioimaging and photodynamic therapy. Mater. Chem. Front. 2020, 4, 3074–3085. [Google Scholar] [CrossRef]
- Geng, X.; Li, Z.; Hu, Y.; Liu, H.; Sun, Y.; Meng, H.; Wang, Y.; Qu, L.; Lin, Y. One-pot green synthesis of ultrabright N-doped fluorescent silicon nanoparticles for cellular im-aging by using ethylenediaminetetraacetic acid disodium salt as an effective reductant. ACS Appl. Mater. Interfaces 2018, 10, 27979–27986. [Google Scholar] [CrossRef]
- Kim, T.E.; Jang, H.J.; Park, S.W.; Wei, J.; Cho, S.; Park, W.I.; Lee, B.R.; Yang, C.D.; Jung, Y.K. Folic Acid Functionalized Carbon Dot/Polypyrrole Nanoparticles for Specific Bioimaging and Photothermal Therapy. ACS Appl. Bio Mater. 2021, 4, 3453–3461. [Google Scholar] [CrossRef]
- Malekzad, H.; Zangabad, P.S.; Mirshekari, H.; Karimi, M.; Hamblin, M.R. Noble metal nanoparticles in biosensors: Recent studies and applications. Nanotechnol. Rev. 2016, 6, 301–329. [Google Scholar] [CrossRef]
- Jianrong, C.; Yuqing, M.; Nongyue, H.; Xiaohua, W.; Sijiao, L. Nanotechnology and biosensors. Biotechnol. Adv. 2004, 22, 505–518. [Google Scholar] [CrossRef]
- Tombelli, S.; Minunni, M.; Mascini, M. Analytical applications of aptamers. Biosens. Bioelectron. 2005, 20, 2424–2434. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Hsu, A.R.; Li, Z.B.; Chen, X. Are quantum dots ready for in vivo imaging in human subjects? Nanoscale Res. Lett. 2007, 2, 265–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchison, J.E. Greener nanoscience: A proactive approach to advancing applications and reducing implications of nano-technology. ACS Nano 2008, 2, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Chen, X. Nanoplatforms for Targeted Molecular Imaging in Living Subjects. Small 2007, 3, 1840–1854. [Google Scholar] [CrossRef]
- Cai, W.; Hong, H. In a “nutshell”: Intrinsically radio-labeled quantum dots. Am. J. Nucl. Med. Mol. Imaging 2012, 2, 136–140. [Google Scholar]
- Lacerda, L.; Bianco, A.; Prato, M.; Kostarelos, K. Carbon nanotubes as nanomedicines: From toxicology to pharmacology. Adv. Drug Deliv. Rev. 2006, 58, 1460–1470. [Google Scholar] [CrossRef]
- Hong, H.; Gao, T.; Cai, W. Molecular imaging with single-walled carbon nanotubes. Nano Today 2009, 4, 252–261. [Google Scholar] [CrossRef]
- Hirsch, L.R.; Gobin, A.M.; Lowery, A.R.; Tam, F.; Drezek, R.A.; Halas, N.J.; West, J.L. Metal nanoshells. Ann. Biomed. Eng. 2006, 34, 15–22. [Google Scholar] [CrossRef]
- Pumera, M.; Sánchez, S.; Ichinose, I.; Tang, J. Electrochemical nanobiosensors. Sens. Actuators B Chem. 2007, 123, 1195–1205. [Google Scholar] [CrossRef]
- Xiao, Y.; Li, C.M. Nanocomposites: From fabrications to electrochemical bioapplications. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2008, 20, 648–662. [Google Scholar] [CrossRef]
- Yakimova, R.; Selegard, L.; Khranovskyy, V.; Pearce, R.; Lloyd Spetz, A.; Uvdal, K. ZnO materials and surface tailoring for bio-sensing. Front. Biosci. 2012, 4, 254–278. [Google Scholar] [CrossRef] [PubMed]
- Tonga, G.Y.; Saha, K.; Rotello, V.M. 25th anniversary article: Interfacing nanoparticles and biology: New strategies for biomedi-cine. Adv. Mater. 2014, 26, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Howes, P.D.; Chandrawati, R.; Stevens, M.M. Colloidal nanoparticles as advanced biological sensors. Science 2014, 346, 1247390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomczak, N.; Liu, R.; Vancso, J.G. Polymer-coated quantum dots. Nanoscale 2013, 5, 12018–12032. [Google Scholar] [CrossRef]
- Nel, A.E.; Mädler, L.; Velegol, D.; Xia, T.; Hoek, E.M.V.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding biophysicochemical interactions at the nano–bio interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef]
- Corbierre, M.K.; Cameron, N.S.; Lennox, R.B. Polymer-Stabilized Gold Nanoparticles with High Grafting Densities. Langmuir 2004, 20, 2867–2873. [Google Scholar] [CrossRef]
- DeLong, R.K.; Reynolds, C.M.; Malcolm, Y.; Schaeffer, A.; Severs, T.; Wanekaya, A. Functionalized gold nanoparticles for the bind-ing, stabilization, and delivery of therapeutic DNA, RNA, and other biological macromolecules. Nanotechnol. Sci. Appl. 2010, 3, 53. [Google Scholar] [CrossRef]
- Szeitner, Z.; András, J.; Gyurcsányi, R.E.; Mészáros, T. Is less more? Lessons from aptamer selection strategies. J. Pharm. Biomed. Anal. 2014, 101, 58–65. [Google Scholar] [CrossRef]
- Wang, L.; Wen, W.; Xiong, H.; Zhang, X.; Gu, H.; Wang, S. A novel amperometric biosensor for superoxide anion based on super-oxide dismutase immobilized on gold nanoparticle-chitosan-ionic liquid biocomposite film. Anal. Chim. Acta. 2013, 758, 66–71. [Google Scholar] [CrossRef]
- Xu, T.; Jia, X.; Chen, X.; Ma, Z. Simultaneous electrochemical detection of multiple tumor markers using metal ions tagged im-munocolloidal gold. Biosens. Bioelectron. 2014, 56, 174–179. [Google Scholar] [CrossRef]
- Mir, T.A.; Akhtar, M.H.; Gurudatt, N.; Kim, J.-I.; Choi, C.S.; Shim, Y.-B. An amperometric nanobiosensor for the selective detection of K+-induced dopamine released from living cells. Biosens. Bioelectron. 2015, 68, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Liu, J.; Wang, T.; Li, H.; Miao, Y.; Liu, Y.; Wang, J.; Wang, E. A simple and rapid electrochemical strategy for non-invasive, sensitive and specific detection of cancerous cell. Talanta 2013, 104, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Labeb, M.; Sakr, A.-H.; Soliman, M.; Abdel-Fattah, T.M.; Ebrahim, S. Effect of capping agent on selectivity and sensitivity of CdTe quantum dots optical sensor for detection of mercury ions. Opt. Mater. 2018, 79, 331–335. [Google Scholar] [CrossRef]
- Munir, T.; Mahmood, A.; Imran, M.; Sohail, A.; Fakhar-E-Alam, M.; Sharif, M.; Masood, T.; Bajwa, S.Z.; Shafiq, F.; Latif, S. Quantitative analysis of glucose by using (PVP and MA) capped silver nanoparticles for biosensing applications. Phys. B Condens. Matter 2020, 602, 412564. [Google Scholar] [CrossRef]
- Kapan, B.; Kurbanoglu, S.; Esenturk, E.N.; Soylemez, S.; Toppare, L. Electrochemical catechol biosensor based on β-cyclodextrin capped gold nanoparticles and inhibition effect of ibuprofen. Process Biochem. 2021, 108, 80–89. [Google Scholar] [CrossRef]
- Wang, H.; He, Y.; Chai, Y.; Yuan, R. A super intramolecular self-enhanced electrochemiluminescence immunosensor based on polymer chains grafted on palladium nanocages. Nanoscale 2014, 6, 10316–10322. [Google Scholar] [CrossRef]
- Xiong, C.; Wang, H.; Yuan, Y.; Chai, Y.; Yuan, R. A novel solid-state Ru(bpy)32+ electrochemiluminescence immunosensor based on poly(ethylenimine) and polyamidoamine dendrimers as co-reactants. Talanta 2014, 131, 192–197. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, H.; Zhang, S. New bienzymatic strategy for glucose determination by immobilized-gold nanoparticle-enhanced chemiluminescence. Sci. China Ser. B Chem. 2009, 52, 196–202. [Google Scholar] [CrossRef]
- Lu, Y.; Yang, M.; Qu, F.; Shen, G.; Yu, R. Enzyme-functionalized gold nanowires for the fabrication of biosensors. Bioelectrochemistry 2007, 71, 211–216. [Google Scholar] [CrossRef]
- Qu, F.; Yang, M.; Shen, G.; Yu, R. Electrochemical biosensing utilizing synergic action of carbon nanotubes and platinum nan-owires prepared by template synthesis. Biosens. Bioelectron. 2007, 22, 1749–1755. [Google Scholar] [CrossRef]
- Ramanavičius, A.; Kaušaitė, A.; Ramanavičienė, A. Polypyrrole-coated glucose oxidase nanoparticles for biosensor design. Sens. Actuators B Chem. 2005, 111–112, 532–539. [Google Scholar] [CrossRef]
- Li, J.; Liu, Q.; Liu, Y.; Liu, S.; Yao, S. DNA biosensor based on chitosan film doped with carbon nanotubes. Anal. Biochem. 2005, 346, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wu, S.; Zhang, L.; Lu, J.; Verproot, F.; Liu, Y.; Xing, Z.; Li, J.; Song, X.-M. Fabrication of polymeric ionic liquid/graphene nanocomposite for glucose oxidase immobi-lization and direct electrochemistry. Biosens. Bioelectron. 2011, 26, 2632–2637. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Yang, H.; Han, D.; Zhang, Q.; Ivaska, A.; Niu, L. Water-Soluble Graphene Covalently Functionalized by Biocompatible Poly-l-lysine. Langmuir 2009, 25, 12030–12033. [Google Scholar] [CrossRef]
- Shan, C.; Yang, H.; Song, J.; Han, D.; Ivaska, A.; Niu, L. Direct Electrochemistry of Glucose Oxidase and Biosensing for Glucose Based on Graphene. Anal. Chem. 2009, 81, 2378–2382. [Google Scholar] [CrossRef]
- Endo, T.; Yanagida, Y.; Hatsuzawa, T. Quantitative determination of hydrogen peroxide using polymer coated Ag nanoparti-cles. Measurement 2008, 41, 1045–1053. [Google Scholar] [CrossRef]
- Tagad, C.K.; Kim, H.U.; Aiyer, R.C.; More, P.; Kim, T.; Moh, S.H.; Kulkarni, A.; Sabharwal, S.G. A sensitive hydrogen peroxide optical sensor based on polysaccharide stabilized silver nanoparticles. RSC Adv. 2013, 3, 22940–22943. [Google Scholar] [CrossRef]
- Shamsipur, M.; Rajabi, H.R. Pure zinc sulfide quantum dot as highly selective luminescent probe for determination of haz-ardous cyanide ion. Mater. Sci. Eng. C 2014, 36, 139–145. [Google Scholar] [CrossRef]
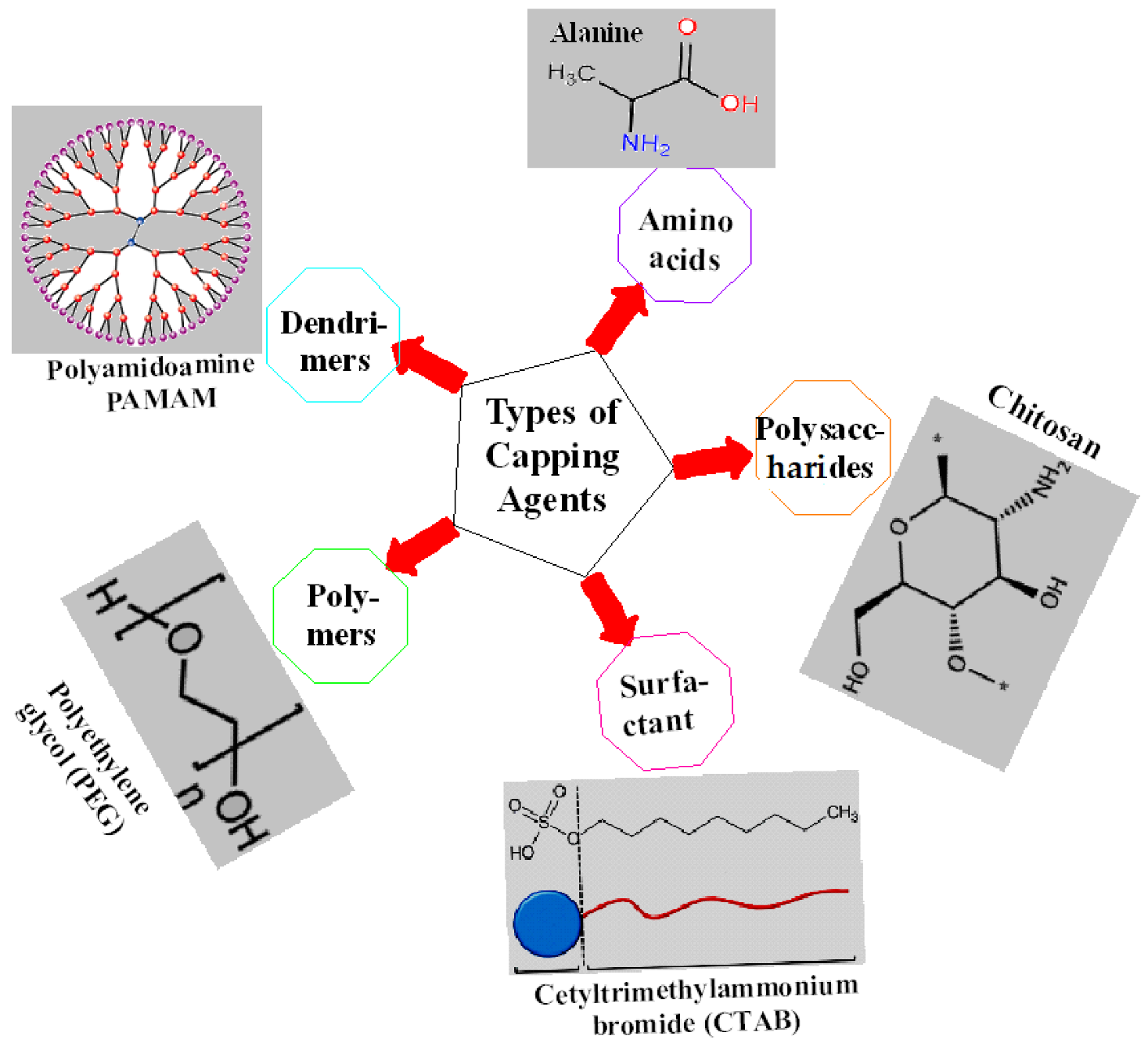
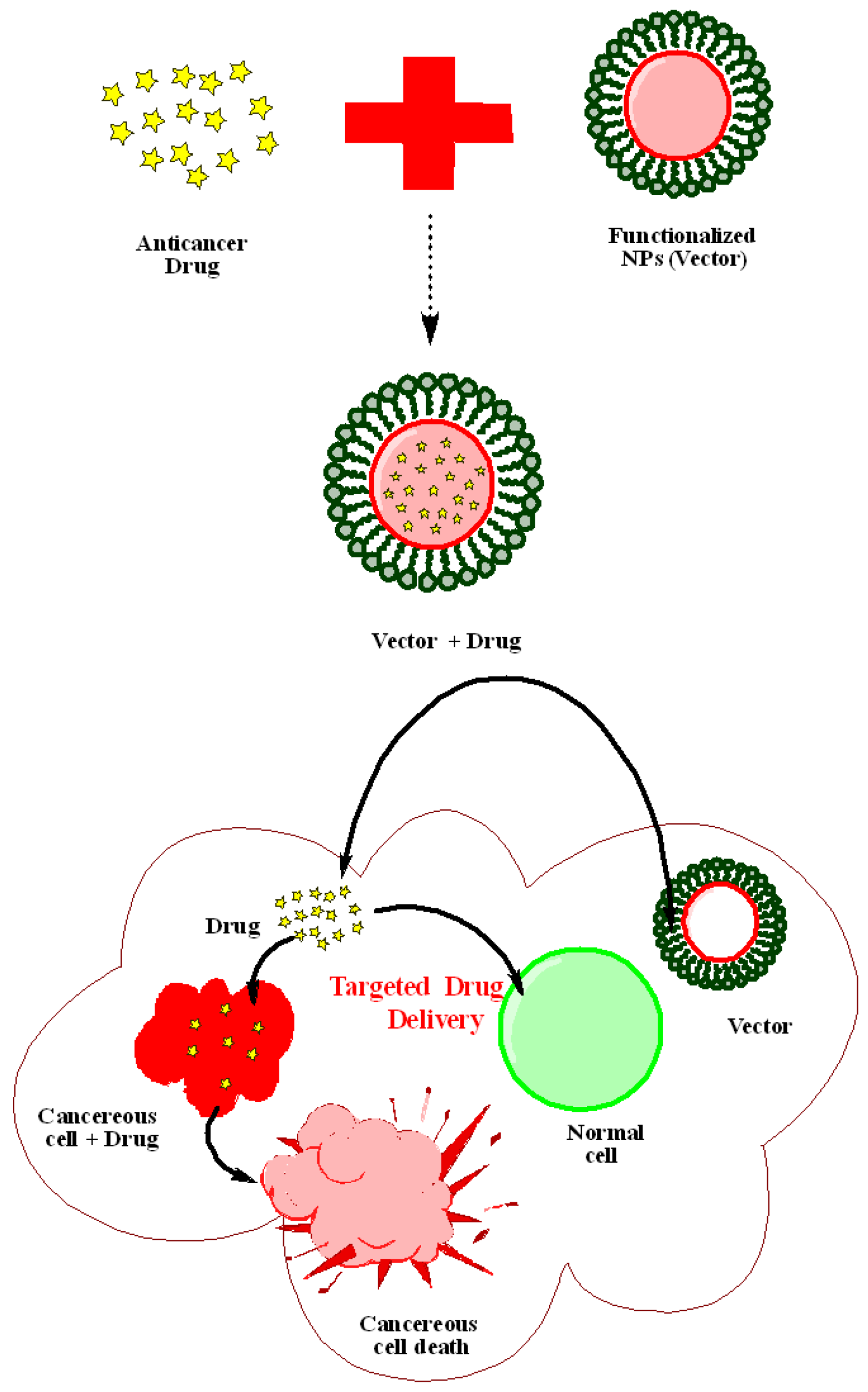




| Sr. No. | Nanoparticles (NPs) | Capping Agents | Drug | Targeting Disease/Cell Line Type | Mechanism of Action/Effect | Reference |
|---|---|---|---|---|---|---|
| 1. | Ag NPs | PVA & Chitosan | Naproxen | Saos-2 cells | Strong response of Saos-2 cells with a higher level of adhesion, proliferation, and mineralization | [65] |
| 2. | Ag NPs | PVA | DOX, Curcumin | Bacillus cereus, E. coli | Significant antibacterial activity | [66] |
| 3. | Ag NPs | PVA, PVP | PVA-Ag NPs, PVP-Ag NPs | Skin wound | PVA-Ag NPs: Exhibit a dominant antibacterial efficacy and showed positive effects through their anti-inflammatory and angiogenic properties, with a nearly 95% healing effect within 9 days; PVP-Ag NPs: Potential antimicrobial efficacy and wound healing properties | [67] |
| 4. | Ag NPs | PEG | I-131 radionuclide | WI-38 cells, solid tumor sarcoma bearing mice | High in-vitro and in-vivo stability, with no cytotoxic effect on normal cells at a lower concentration, high radioactivity accumulation in tumor tissues of mice | [68] |
| 5. | Ag NPs | PVA/PVP/Pectin | Mafenide acetate | Skin wound | Remarkable effect on wound healing | [69] |
| 6. | Ag NPs | Chitosan | Ag-Chitosan | Skin wound | Accelerated the healing of a burn wound by decreasing the inflammatory reaction; subsequently, decreasing the duration of the repair phase | [70] |
| 7. | Fe3O4 NPs | PVA, SA, BSA | DOX | HepG2, L02 cell lines | Fe3O4-SA-DOX-PVA-BSA toxic to HepG2 cell lines and non-toxic to L02 cell lines | [71] |
| 8. | Fe3O4 NPs | EDTA | Imatinib | Bone marrow cell line (K562) | Drug loaded NPs display lower liver accumulation compared to a bared drug, prolonged circulation time | [72] |
| 9. | MnFe2O4 NPs | PVP | DOX | HeLa Cells | No cytotoxicity of PVP-coated MnFe2O4 nanoparticles. Controlled drug delivery with pH-dependent release behavior. | [73] |
| 10. | PLGA NPs | Chitosan, PEG and Dextran | Curcumin | Breast cancer cells (MCF-7) | Effective in arresting cancer cell growth, induce apoptosis | [74] |
| 11. | Mesoporous silica nanoparticles (MS NPs) | PEG | DOX | HeLa cells | Decrease in cancer cell viability | [33] |
| 12. | MS NPs | PEGylated polyaminoacids | Celastrol (CST) | Cancer cells, and SCC-7 xenograft tumor-bearing mice | CMSN-PEG exhibited high in vitro cytotoxicity in different cancer cells, effectively used as a mitochondrial targeting system for efficient inhibition of solid tumors | [75] |
| 13. | MNs loaded with PB NPs | PVA/PVP | Metformin | Skin | The effective decline in the BGLs of diabetic rats. | [76] |
| 14. | Au NPs | PAMAM–COOH (G4) | DOX | Cancer | Enhanced permeation and retention (EPR) mediated drug targeting followed by the lysosomal drug release | [77] |
| 15. | FeO NPs | PAMAM–NH2 (G4) | 3,4-difluorobenz ylidene- curcumin | SKOV3 cells | Multivalent theranostic nanoparticles for simultaneous imaging and precise cancer cell targeting | [78] |
| 16. | Au NPs | BSA | Methotrexate (MTX) | MCF-7 | Inhibitory action on the growth of MCF-7 cell line induces apoptosis | [79] |
| 17. | MS NPs | Peptide coated Au NPs | DOX | U87 MG cells and HEK 293 cells | NPs-mediated apoptosis of αvβ3 integrin over-expressing cancer cells | [80] |
| 18. | Fe3O4 NC (nano-composite) | PAH/PSS | DOX | A549 cell lines | pH-responsive drug release and higher cytotoxicity towards human lung cancer (A549) cells in vitro in a dose-dependent manner | [81] |
| 19. | Ultrasmall iron oxide nanoparticles (USIONPs) | Tannic acid (TA) and Quinic acid (QA) | Quinic acid (QA) and its derivatives | U87 cells and metastatic (MDA-MB-231Br cells) | Higher cellular uptake of QA-coated USIONPs compared to TA-coated USIONPs | [82] |
| 20. | Ag NPs | Aesculus hippocastanum (horse chestnut) | Aqueous A. hippocastanum leaf extract, resveratrol | Bacterial agents, in vitro drug release | Significant antioxidant and antimicrobial activities, drug release from AgNPs exhibited pH dependency; the release was significant (45.6%) under acidic conditions (pH 5.2) | [83] |
| 21. | Au & Ag NPs | B. monosperma (BM) leaf extract | DOX | B16F10 & MCF-7 cancer cells | Significant inhibition of cell proliferation in a dose-dependent manner (0.06–0.25 μM w. r. t DOX) | [84] |
| 22. | PD-FeO NPs | CS, PVA | Leaf extract of Pinus densiflora (PD) | Diabetic and anemia-associated diabetic wounds | Enhanced cell proliferation and augmented angiogenesis, leading to wound contraction and reduction in cytotoxicity | [85] |
| 23. | CeO2 NPs | CS/PVA | Zingiber officinale extract | Human dermal fibroblasts cells | Significantly decreased wound infections without the use of antibiotics | [86] |
| 24. | ZnO NPs | Chitosan | Camellia sinensis/Paclitaxel | MCF-7 | High cytotoxic effect on the breast cancer cell line | [87] |
| 25. | α-Fe2O3 NPs | Nepeta cataria leaves extract | Doxorubicin | Melanoma cell line (A375) | Significant cytotoxic effect against the melanoma cancer cell line | [88] |
| Sr. No. | Nanoparticles (NPs) | Description/Capping Agent | Disease/Cell Lines | In Vitro/In Vivo Implication | Reference |
|---|---|---|---|---|---|
| 1. | Au NPs | Synthesis of Au NPs capped with l-cysteine methyl ester hydrochloride conjugated to PEG | Hep G2, Caco-2, B16F10, and CT26 | Bioengineered Au NPs with different sizes, shapes, structures, chemistry, and synthetic strategies have shown potential to enhance siRNA delivery in vitro and in vivo | [114] |
| 3. | Au NPs | Linalool-loaded glutathione-modified Au NPs conjugated with CALNN peptide | SKOV-3 | LG and LGC were selectively toxic in cancer cells and induced apoptosis by activating caspase-8, the p53 protein, and various proteins involved in apoptosis. | [115] |
| 5. | Au NPs | Central core Au NPs encapsulated by a layer of DNA-capped QDs used | A pair of human ovarian carcinoma cell lines-A2780, and DOX-resistant cell line A2780 ADR | Programmable hybrid nanostructures engage with the target MRP1 mRNA, reduces the MRP1 expression, results in a detectable turn-on fluorescence signal, and Dox release. The Dox-anti-MRP1 hybrid is significantly more cytotoxic against MDR cancer cells. | [116] |
| 6. | Au NPs | Au NPs fabricated with L-Cystine methyl ester hydrochloride as a capping agent, then loaded with plasmid DNA encoded p53 gene | WI38 and A549 | The high percentage of cell viability in WI 38 proved the safety of L-cysteine methyl ester functionalized Au NPs. Additionally, the apoptotic effect due to the expression of p53 gene loaded on Au NPs was only prominent in A549. | [117] |
| 7. | Au NPs | Doxorubicin loaded oligonucleotides (ONTs) attached to Au NPs (DOA) | SW480 and a xenograft mouse model | Successful cellular uptake of DOA by SW480, with significant cytotoxicity at reasonably low concentrations. In vivo, DOA could significantly suppress cancer growth in a mouse xenograft than free DOX | [118] |
| 8. | Au nanosphere | Gold nanosphere coated with poly(ethylenimine) (PEI), conjugated with the targeting ligand anisamide (Au-PEI-AA) | PC3 prostate cancer cells | Au-PEI-AA mediated siRNA uptake into PC3 prostate cancer cells via binding to the sigma receptor, anisamide-labeled Au NPs can target the sigma receptor | [119] |
| 9. | IONPs | Iron Oxide NPs to deliver siRNA targeting BCL-2 in oral cancer cells | Ca9-22 cell line | Reduced cell viability and relative cell migration in Ca9-22 cell line | [112] |
| 10. | Au NPs | Biosynthesis of Au NPs using cold and hot sclerotium of Lignosus rhinocerotis, capped with chitosan | Human dermal fibroblasts (HDF) | DsiRNA-AuNPs incorporated into thermo-responsive pluronic gels demonstrated high cell viability, proliferation, and cell migration rate via in vitro cultured cells of HDF, indicating their non-cytotoxicity and wound healing properties | [120] |
| Sr. No. | Nanoparticles (NPs) | Capping Agent | Effect of Capping Agent | Bioimaging Application | Reference |
|---|---|---|---|---|---|
| 1. | CdS QDs | Dextrin | Reduced toxicity of innate cadmium sulfide (CdS) | Used as fluorescent agent in in vitro and in vivo studies where maximum fluorescence was observed in kidney, liver, and brain | [151] |
| 2. | Au NPs & DTA | Polyamidoamine (PAMAM) dendrimer | Improved X-ray attenuation, stability, biocompatibility, and enhanced blood circulation time | CT Imaging | [152] |
| 3. | Bimetallic Au-Ag NPs | Folic acid (FA)-modified PAMAM dendrimer | 25% higher X-ray attenuation than Omnipaque. In in vitro better CT imaging of cancer cells overexpressing FA receptors showed 2.3 to 2.7-fold higher uptake than the cells possessing low level of FA expression. | In vitro CT imaging in cancer cells | [153] |
| 4. | Ag NPs | Acetylated-PAMAM dendrimer | Extended blood circulation time led to prolonged enhancement | X-ray CT contrast agent | [154] |
| 5. | Au NPs | Acetylated-PAMAM dendrimer | Improved biocompatibility, 1.6 times higher X-ray attenuation compared to Omnipaque, specific targeting through receptor-mediated endocytosis | In vivo CT imaging | [155] |
| 6. | Au NPs | Thiolated PEG & pluronic triblock copolymer (PEO–PPO–PEO) | Improved colloidal and optical stability and biocompatibility | Used as scattering probes for dark-field imaging of cancer cells under both in vitro and in vivo conditions | [156] |
| 7. | Gd2O3 & PMNPs | Diethylene glycol polymer & Liposomes | No in vitro cytotoxic effects, sensitive contrast agent | MRI contrast agent and marker for cell tracking | [157] |
| 8. | IONPs | Dextran | Biocompatible, superior T2 relaxation rate and high relaxivities led to clear distinguished signal imaging intensity of specific organ, tumor, and whole-body | MRI contrast agents | [158] |
| 9. | Au NPs | Poly di(carboxylatophenoxy)phosphazene | Biocompatible and biodegradable | Can be used as contrast agents for photoacoustic imaging | [159] |
| 10. | INPs | PEG | Biocompatible, extended high contrast vascular imaging and stability, selectively accumulated in tumor | Vascular and tumor imaging by Micro-CT | [160] |
| 11. | Iodine-131 labeled Au NPs | Polyethyleneimine (PEI) | Improved X-ray attenuation coefficient, colloidal stability, cytocompatibility, and radiochemical stability in vitro | Single-photon emission computed tomography/computed tomography (SPECT/CT) imaging and radionuclide therapy | [161] |
| 12. | Bi2S3 NPs & QDs | PEG-phospholipid bilayer | Enhanced CT contrast and fluorescence imaging capability, longer circulation time (>4 h) than iobitridol, biocompatibility, and safety. | Used for combined CT/fluorescence imaging | [162] |
| 13. | Radioactive iodide-124 labeled Au NPs | PEG | Non-toxic, high stability, and sensitivity in various pH, serum, and in vivo conditions | In vivo tumor imaging through combined positron emission tomography and cerenkov luminescent imaging (PET/CLI). | [163] |
| 14. | CuS [c(RGDfK)] | PEG | High efficacy and minimal side effects | Promising platform for image guided ablation therapy | [164] |
| 15. | Au NPs | Glycol-chitosan | Simplest nanocomposite did not require antibodies or complex surface modification | Photoacoustic contrast agent | [165] |
| 16. | Silica NPs | PEG & doping with cyanine 5.5 (Cy5.5) & cyanine 7 (Cy7) dyes | High colloidal stability in water and in biological environment, with absorption and fluorescence emission in the NIR field | Used to achieve optical and photoacoustic imaging | [166] |
| 17. | Au NPs | Poly(perylene diimide) (PPDI) & PEG | Greater photothermal effect and a stronger photoacoustic signal | Used as photoacoustic (PA) agents under in vivo imaging and therapeutic evaluation | [167] |
| 18. | Plectin-SPION-Cy7 or SPION-Cy7 | DSPE-PEG-NH2 | Highly accumulated in tumor, MIAPaCa2 and XPA-1 carcinoma cells but not in normal pancreatic tissues, liver, and kidney | Optical imaging and MRI | [168] |
| 19. | Curcumin-Ag NPs complex | Polyvinylpyrrolidone (PVP) | Enhanced water solubility and bioavailability in a biological system without effecting its therapeutic potential. Fluorescence efficiency in cancer cellular medium is ∼2.37 times higher | Used as fluorescent probe in CT imaging | [169] |
| 20. | Au NPs doped with silver | Gelatin | Improved stability, quantum yield, and fluorescence lifetime. Remarkable biocompatibility | Promising approach for imaging in a challenging tissue as skin | [170] |
| 21. | MoO3 mixed with optoelectrochemically active dye complex (Ru(II)) | Chitosan (CS) | Biocompatible | Used in intracellular imaging | [171] |
| 22. | Au NPs | Zinnia elegans plant extract | Highly biocompatible and do not use any targeted ligand | Used as imaging agent in NIR region | [172] |
| 23. | Ag NPs | 4-mercaptobenzoic acid-capped | Enhanced fluorescent brightness, improved photostability, and low cytotoxicity | Used for simultaneous cellular imaging and photodynamic therapy | [173] |
| 24. | N-doped fluorescent Si NPs with an ultra-high quantum yield | EDTA-2Na | Water dispersibility, higher stability, and biocompatibility | Used in cellular imaging | [174] |
| 25. | Carboxylated PPy-NPs | Folic acid functionalized carbon dots | Photostability, specific targeting, biocompatible | Used as PTT imaging agent | [175] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Javed, R.; Sajjad, A.; Naz, S.; Sajjad, H.; Ao, Q. Significance of Capping Agents of Colloidal Nanoparticles from the Perspective of Drug and Gene Delivery, Bioimaging, and Biosensing: An Insight. Int. J. Mol. Sci. 2022, 23, 10521. https://doi.org/10.3390/ijms231810521
Javed R, Sajjad A, Naz S, Sajjad H, Ao Q. Significance of Capping Agents of Colloidal Nanoparticles from the Perspective of Drug and Gene Delivery, Bioimaging, and Biosensing: An Insight. International Journal of Molecular Sciences. 2022; 23(18):10521. https://doi.org/10.3390/ijms231810521
Chicago/Turabian StyleJaved, Rabia, Anila Sajjad, Sania Naz, Humna Sajjad, and Qiang Ao. 2022. "Significance of Capping Agents of Colloidal Nanoparticles from the Perspective of Drug and Gene Delivery, Bioimaging, and Biosensing: An Insight" International Journal of Molecular Sciences 23, no. 18: 10521. https://doi.org/10.3390/ijms231810521






