Cadmium Addition Effects on Anaerobic Digestion with Elevated Temperatures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Anaerobic Fermentation Experiment Set-Up
2.3. Measurements
2.4. Data Analysis
3. Results and Discussion
3.1. Fate of Cd during the Digestion Process
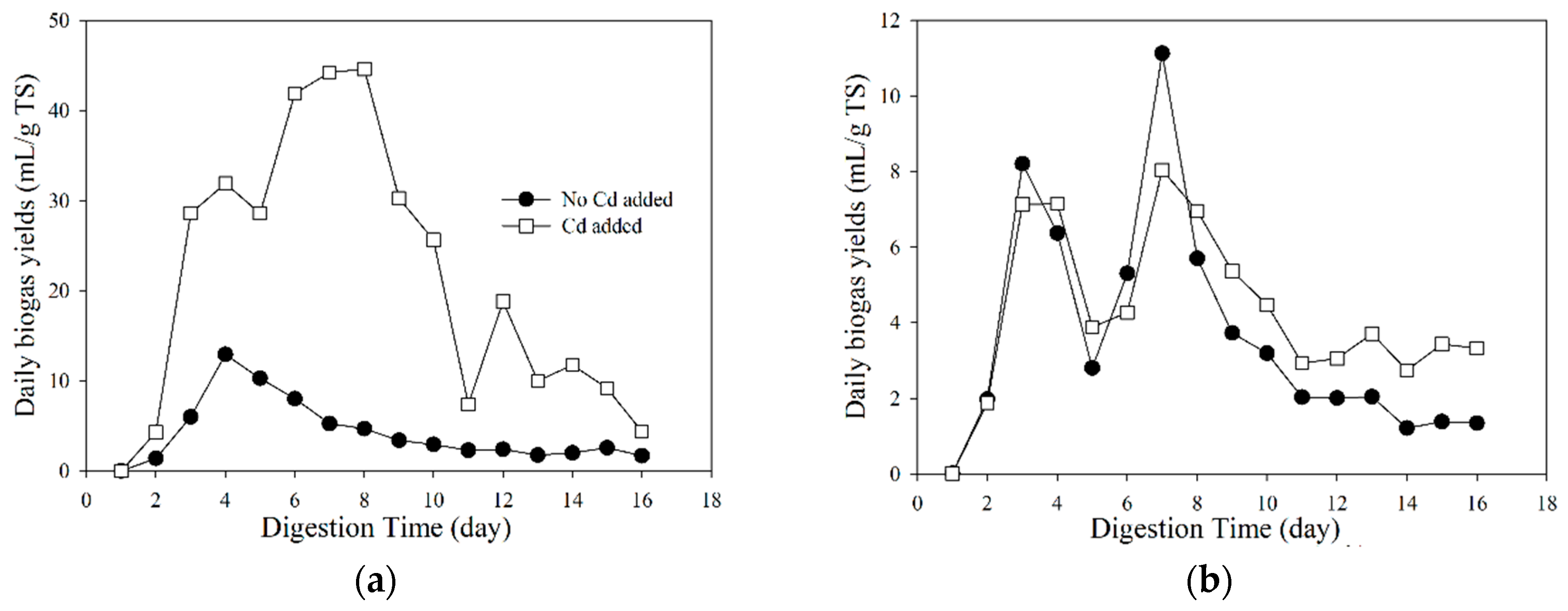
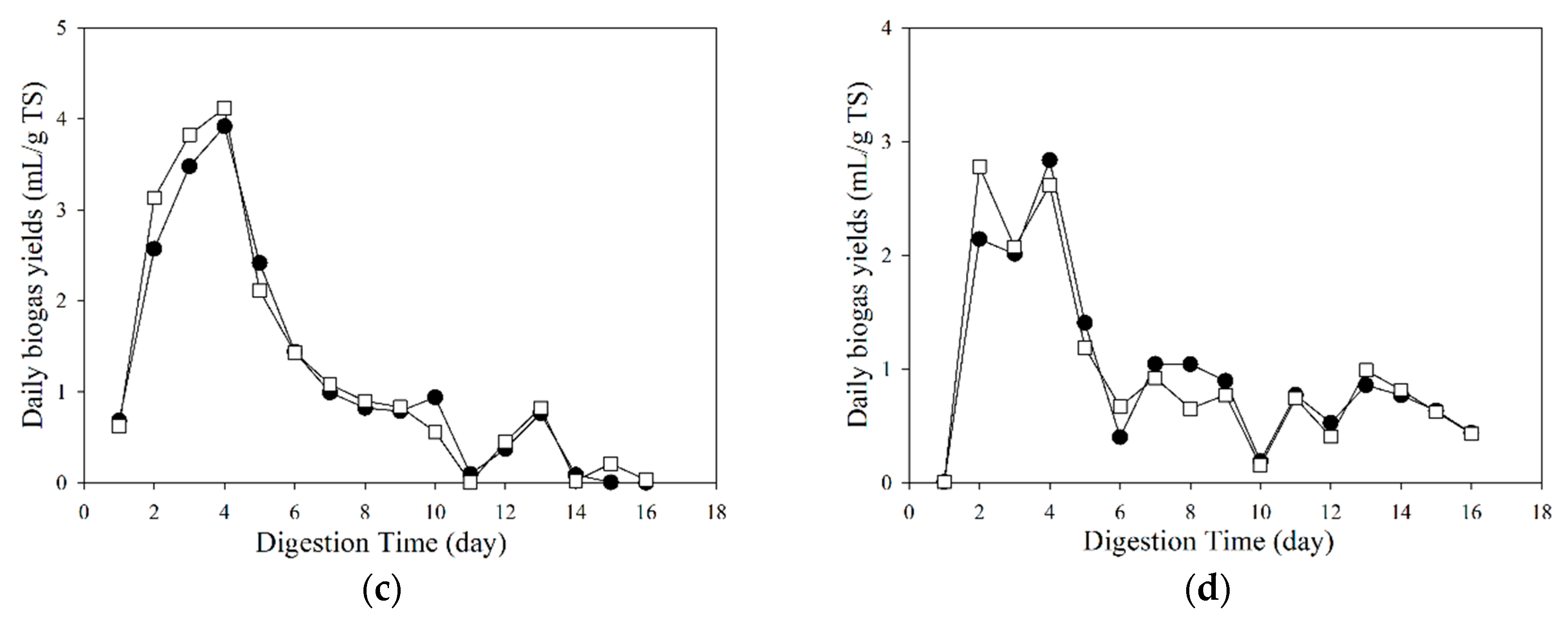
3.2. Biogas Properties under Different Temperatures
3.3. Substrate Biodegradation
3.3.1. Responses of VFAs
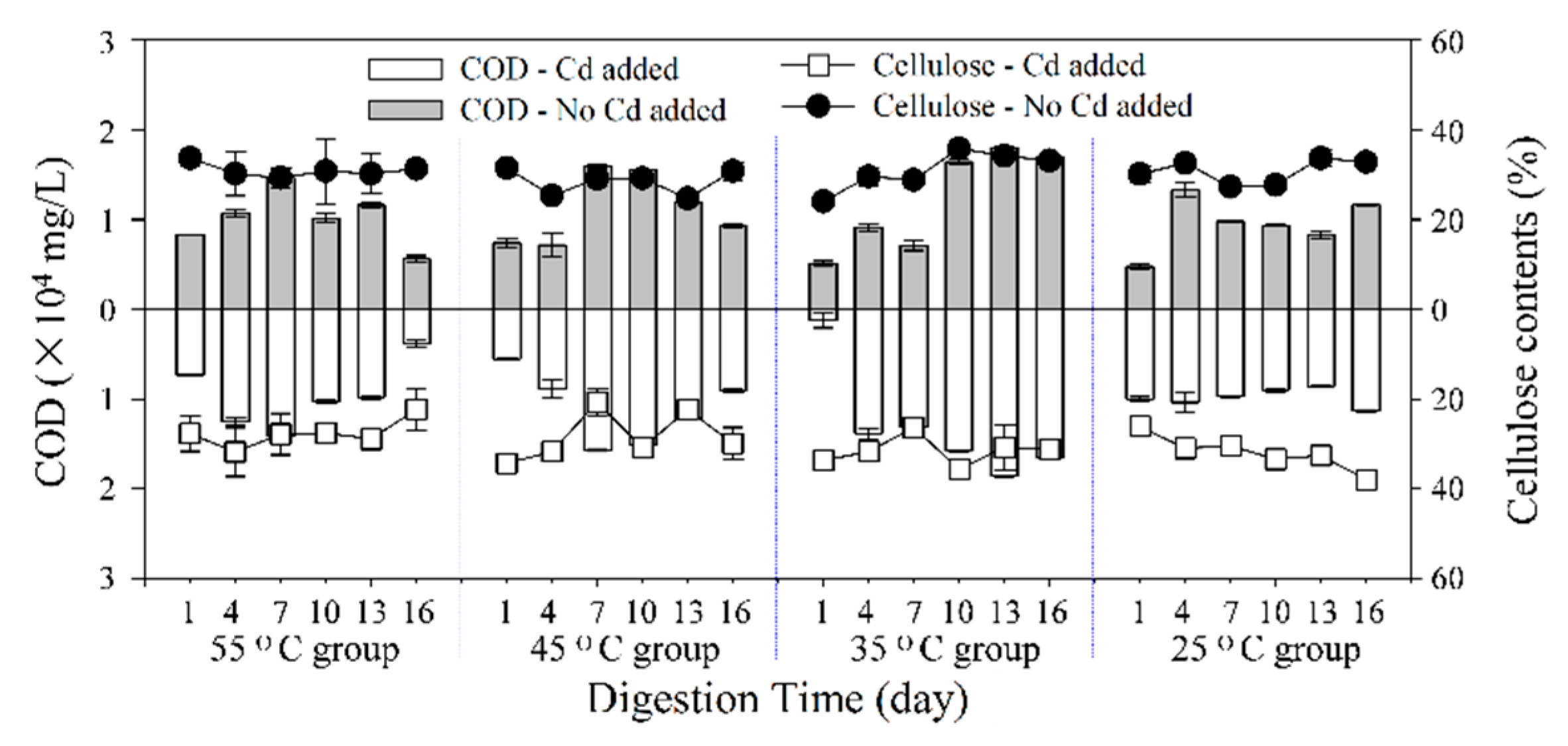
3.3.2. COD
3.3.3. Degradation of Cellulose
3.4. Responses of Enzyme Activity to Varied Temperatures under Cd Stress
3.4.1. Cellulase
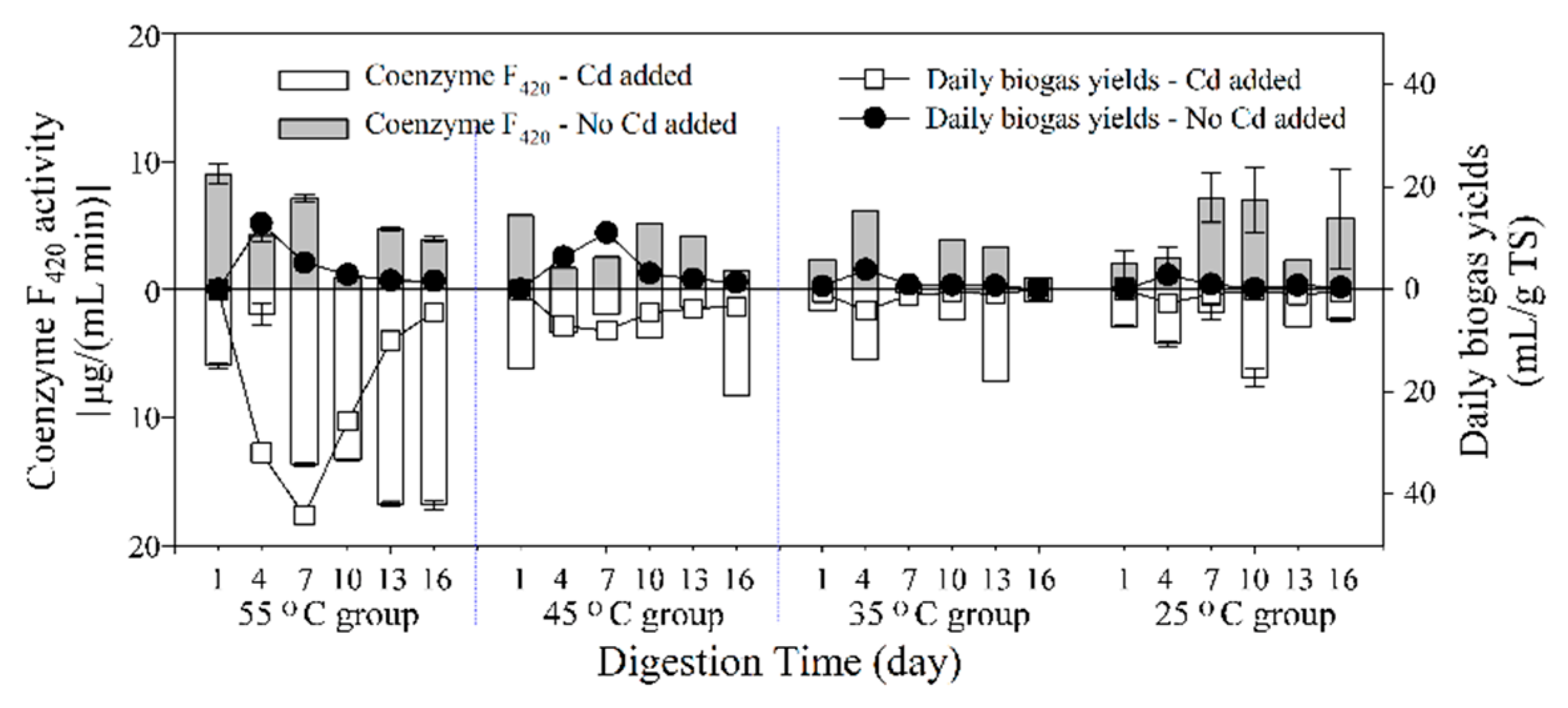
3.4.2. Coenzyme F420
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Uma, S.; Thalla, A.K.; Devatha, C.P. Co-digestion of food waste and switchgrass for biogas potential: Effects of process parameters. Waste Biomass Valorization 2018, 1–13. [Google Scholar] [CrossRef]
- Fernandes, K.D.; Cañote, S.J.B.; Ribeiro, E.M.; Thiago Filho, G.L.; Fonseca, A.L. Can we use Cd-contaminated macrophytes for biogas production? Environ. Sci. Pollut. Res. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bhui, I.; Mathew, A.K.; Chaudhury, S.; Balachandran, S. Influence of volatile fatty acids in different inoculum to substrate ratio and enhancement of biogas production using water hyacinth and salvinia. Bioresour. Technol. 2018, 270, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Manyiloh, C.E.; Mamphweli, S.N.; Meyer, E.L.; Okoh, A.I.; Makaka, G.; Simon, M. Microbial anaerobic digestion (bio-digesters) as an approach to the decontamination of animal wastes in pollution control and the generation of renewable energy. Int. J. Environ. Res. Public Health 2013, 10, 4390–4417. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Gujral, G.S.; Jha, N.K.; Vasudevan, P. Production of biogas from Azolla pinnata R.Br and Lemna minor L.: Effect of heavy metal contamination. Bioresour. Technol. 1992, 41, 273–277. [Google Scholar] [CrossRef]
- Lin, C.-Y. Effect of heavy metals on volatile fatty acid degradation in anaerobic digestion. Water Res. 1992, 26, 177–183. [Google Scholar] [CrossRef]
- Šotnar, M.; Mareček, J.; Máchal, P.; Koutný, T.; Geršl, M.; Krčálová, E.; Korenko, M. Biogas production of phytoremediation plants contaminated with cadmium. Научни Tpyдoве Pyceнския Университет 2014, 53, 174. [Google Scholar]
- Zhang, H.; Tian, Y.; Wang, L.; Zhang, L.; Dai, L. Ecophysiological characteristics and biogas production of cadmium-contaminated crops. Bioresour. Technol. 2013, 146, 628–636. [Google Scholar] [CrossRef]
- Bo, M.; Florczak, I.; Zdanowska, P.; Wojdalski, J. An analysis of metal concentrations in food wastes for biogas production. Renew. Energy 2015, 77, 467–472. [Google Scholar]
- Abdel-shafy, H.I.; Mansour, M.S.M. Biogas production as affected by heavy metals in the anaerobic digestion of sludge. Egypt. J. Pet. 2014, 23, 409–417. [Google Scholar] [CrossRef] [Green Version]
- Keshri, J.; Mankazana, B.B.J.; Momba, M.N.B. Profile of bacterial communities in South African mine-water samples using Illumina next-generation sequencing platform. Appl. Microbiol. Biotechnol. 2015, 99, 3233–3242. [Google Scholar] [CrossRef] [PubMed]
- Matheri, A.N.; Belaid, M.; Seodigeng, T.; CatherineNgila, J. The role of trace elements on anaerobic co-digestion in biogas production. In Proceedings of the World Congress on Engineering, London, UK, 29 June–1 July 2016. [Google Scholar]
- Yu, H.Q.; Fang, H.H.P. Inhibition by chromium and cadmium of anaerobic acidogenesis. Water Sci. Technol. 2001, 43, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Zhang, H. Producing biogas from agricultural residues generated during phytoremediation process: Possibility, threshold, and challenges. Int. J. Green Energy 2016, 13, 1556–1563. [Google Scholar] [CrossRef]
- Lira-Silva, E.; Santiago-Martínez, M.G.; Hernández-Juárez, V.; García-Contreras, R.; Moreno-Sánchez, R.; Jasso-Chávez, R. Activation of methanogenesis by cadmium in the marine archaeon Methanosarcina acetivorans. PLoS ONE 2012, 7, e48779. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Han, X.; Tian, Y.; Li, Y.; Yang, K.; Hao, H.; Chai, Y.; Xu, X. Process analysis of anaerobic fermentation of Phragmites australis straw and cow dung exposing to elevated chromium (VI) concentrations. J. Environ. Manag. 2018, 224, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Tian, Y.; Zhang, H.; Chai, Y. Copper stressed anaerobic fermentation: Biogas properties, process stability, biodegradation and enzyme responses. Biodegradation 2017, 28, 369–381. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, H.; Chai, Y.; Wang, L.; Mi, X.; Zhang, L.; Ware, M.A. Biogas properties and enzymatic analysis during anaerobic fermentation of Phragmites australis straw and cow dung: Influence of nickel chloride supplement. Biodegradation 2017, 28, 15–25. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, H.; Mi, X.; Wang, L.; Zhang, L.; Ai, Y. Research on anaerobic digestion of corn stover enhanced by dilute acid pretreatment: Mechanism study and potential utilization in practical application. J. Renew. Sustain. Energy 2016, 8, 023103. [Google Scholar] [CrossRef]
- Su, Y. Biogas Fermentation Detection Technology; Metallurgical Industry Press: Beijing, China, 2011. [Google Scholar]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Leyva-Ramos, R.; Bernal-Jacome, L.A.; Acosta-Rodriguez, I. Adsorption of cadmium (II) from aqueous solution on natural and oxidized corncob. Sep. Purif. Technol. 2005, 45, 41–49. [Google Scholar] [CrossRef]
- Zhu, Z.; Cheng, G.; Zhu, Y.; Zeng, H.; Wei, R.; Wei, C. The effects of different anaerobic fermentation temperature on biogas fermentation of swine manure. In Proceedings of the 2011 International Conference on Computer Distributed Control and Intelligent Environmental Monitoring, Changsha, China, 19 February 2011. [Google Scholar]
- Jayaweera, M.W.; Dilhani, J.A.; Kularatne, R.K.; Wijeyekoon, S.L. Biogas production from water hyacinth (Eichhornia crassipes (Mart.) Solms) grown under different nitrogen concentrations. J. Environ. Sci. Health Part A Toxic/Hazardous Subst. Environ. Eng. 2007, 42, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Amarneh, F.F.K. Quantitative and Qualitative Assessment of Macronutrients and Micronutrients of Digestate from Biogas Units under Different Feedstock Loadings. Master’s Thesis, An-Najah National University, Bloomington, IN, USA, 2014. [Google Scholar]
- Yu, H.Q.; Fang, H.H.P.; Tay, J.H. Effects of Fe2+ on sludge granulation in upflow anaerobic sludge blanket reactors. Water Sci. Technol. 2000, 41, 199–206. [Google Scholar] [CrossRef]
- Chakraborty, N.; Chatterjee, M.; Sarkar, G.M.; Lahiri, S.C. Inhibitory effects of the divalent metal ions on biomethanation by iolated mesophilic methanogen in AC21 medium in presence or absence of juices from water hyacinth. Bioenergy Res. 2010, 3, 314–320. [Google Scholar] [CrossRef]
- Zhang, H.; Tian, Y.; Wang, L.; Mi, X.; Chai, Y. Effect of ferrous chloride on biogas production and enzymatic activities during anaerobic fermentation of cow dung and Phragmites straw. Biodegradation 2016, 27, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Harper, S.R.; Pohland, F.G. Recent developments in hydrogen management during anaerobic biological wastewater treatment. Biotechnol. Bioeng. 1986, 28, 585–602. [Google Scholar] [CrossRef] [PubMed]
- Nettmann, E.; Bergmann, I.; Pramschu, S.; Mundt, K.; Plogsties, V.; Herrmann, C.; Klocke, M. Polyphasic analyses of methanogenic archaeal communities in agricultural biogas plants. Appl. Environ. Microbiol. 2010, 76, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Tripp, B.C.; Rd, B.C.; Cruz, F.; Krebs, C.; Ferry, J.G. A role for iron in an ancient carbonic anhydrase. J. Biol. Chem. 2004, 279, 6683. [Google Scholar] [CrossRef] [PubMed]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, Y.; Li, S.; Li, Y.; Zhang, H.; Mi, X.; Huang, H. Cadmium Addition Effects on Anaerobic Digestion with Elevated Temperatures. Energies 2019, 12, 2367. https://doi.org/10.3390/en12122367
Tian Y, Li S, Li Y, Zhang H, Mi X, Huang H. Cadmium Addition Effects on Anaerobic Digestion with Elevated Temperatures. Energies. 2019; 12(12):2367. https://doi.org/10.3390/en12122367
Chicago/Turabian StyleTian, Yonglan, Shusen Li, Ying Li, Huayong Zhang, Xueyue Mi, and Hai Huang. 2019. "Cadmium Addition Effects on Anaerobic Digestion with Elevated Temperatures" Energies 12, no. 12: 2367. https://doi.org/10.3390/en12122367
APA StyleTian, Y., Li, S., Li, Y., Zhang, H., Mi, X., & Huang, H. (2019). Cadmium Addition Effects on Anaerobic Digestion with Elevated Temperatures. Energies, 12(12), 2367. https://doi.org/10.3390/en12122367





