Catalytic Coal Gasification Process Simulation with Alkaline Organic Wastewater in a Fluidized Bed Reactor Using Aspen Plus
Abstract
:1. Introduction
2. Method Description
2.1. Process Assumptions
- Gasification temperature remains stable.
- The raw material particles are mixed with the gasifying agents in the furnace quickly and uniformly.
- H, O, N, and S in the coal all changed into the gas phase, and C was assumed to be incompletely transformed according to the reaction.
- The char contains only fixed carbon and ash.
- Ash in coal is inert and does not participate in chemical reactions.
- The syngas consists of H2, CO, CO2, CH4, H2O, N2, and C6H6.
- The fluidized bed was divided into dense and dilute phases.
- The composition of the gas phase under the simulation conditions is regarded as an ideal gas, and it is applicable to the physical properties, methods, and models in the software.
- The catalytic effect was introduced into the gasification model of the fluidized bed using a correction factor.
2.2. Reactions
2.3. Process Modeling
2.3.1. Component Definitions and Method
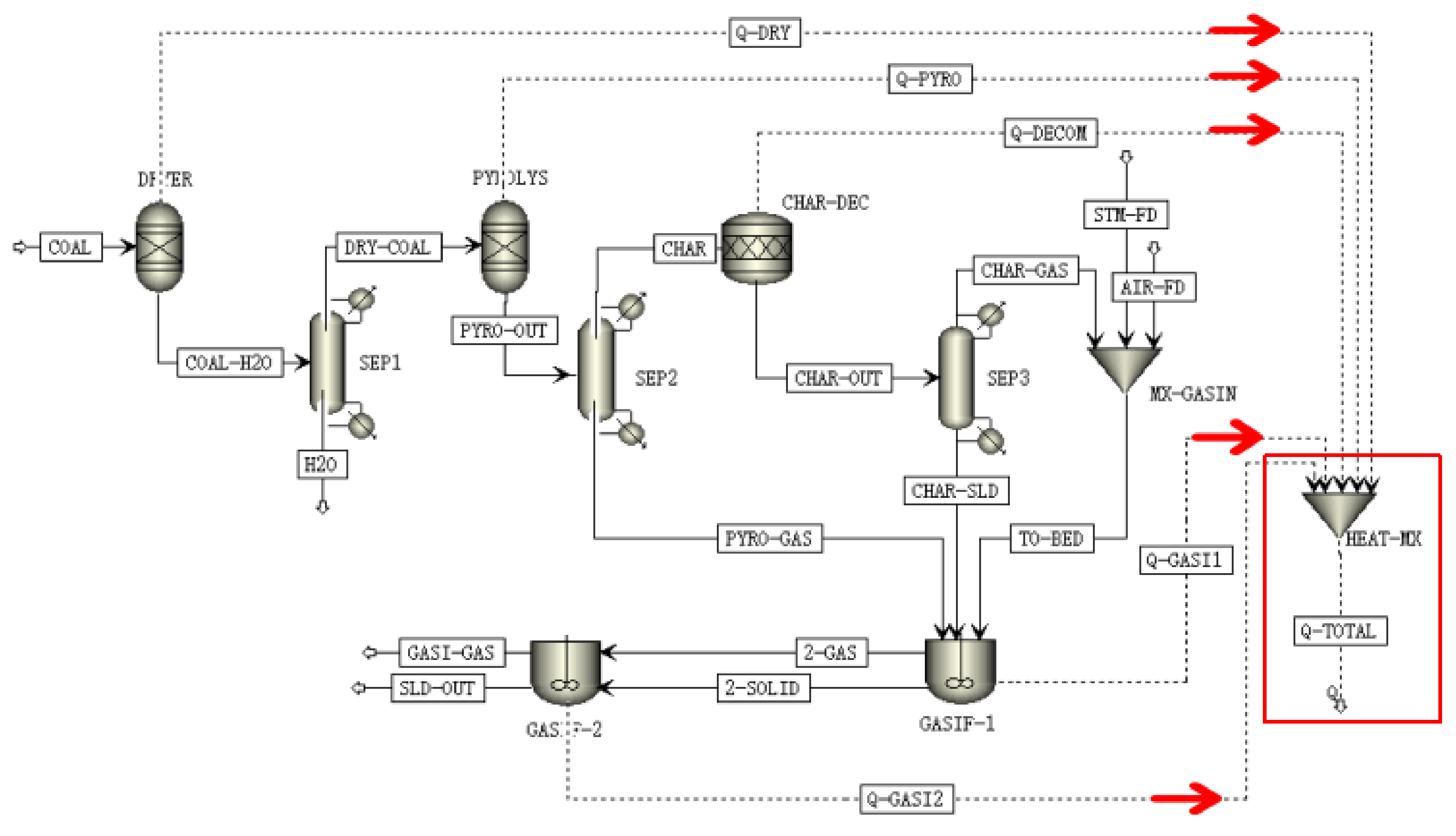
2.3.2. Process Description
- (1)
- Drying: A stream of coal, with a mass flow, was fed into the DRYER reactor in which the physical moisture bound was released completely into the gas phase. The amount of vaporized water was determined by the proximate analysis of the coal. We assumed that coal drying was instantaneous compared with that of the coal gasification process. Water content for the specific coal we used was set to x%, according to the proximate analysis. As a result the yield of dried coal was set to 1 − x%. The output was separated by a gas–solids separator called SEP-1.
- (2)
- Pyrolysis: The output of the drying process was called DRY-COAL, whereafter it entered the PYROLYS block. Pyrolysis is a complex thermochemical process that occurs during coal gasification which is difficult to simulate accurately using the database of Aspen Plus. Two ways are available to simulate coal pyrolysis. One is based on experiments and the other is to use functional group models based on a theoretical method [19]. Using experimental results will be simpler compared with using a functional group. Thus, the pyrolysis experimental data for a specific coal species was used to specify every component of the PYROLYS block. In this block, dried coal was broken down into CO, H2, CH4, CO2, N2, H2O, C6H6, and char. The gas–solids separator SEP-2 block separated the upstream product into two streams, namely CHAR and PYRO-GAS. This reaction was also considered instantaneous.
- (3)
- Decomposition: CHAR was downstream of the CHAR-DEC block. CHAR, as a NC component, must be transformed to participate in the gasification reaction. In this study, block RStoic was chosen to simulate the decomposition process. CHAR was decomposed into C, H2, O2, N2, S, and ash for the solid–gas reactions. The stoichiometric coefficients of the elements mentioned above were determined automatically according to char’s ultimate analysis in the CALCULATOR block. The output was separated by the gas–solids separator SEP-3 block into two streams of CHAR-GAS (including H2, O2, and N2) and CHAR-SLD.
- (4)
- Gasification: The air, steam and the stream CHAR-GAS gaseous products, were mixed in MIX-GASIN. Then, the stream CHAR-SLD together with MIX-GASIN and the stream PYRO-GAS from the upper block PYROLYS was reacted in the RCSTR reactor, named as block GASIF-1/2. We used two RCSTR reactors to simulate the dense and the dilute regions of the fluidized bed. The uncatalyzed kinetic parameters are shown in Table 1, which were written in an external FORTRAN subroutine and was linked to the simulation. The gaseous outputs were mainly CO2, CO, H2, CH4, and C6H6.
- (5)
- Catalytic effects: In fact, catalysts, such as alkali metals, will respectively increase the reaction rates of the chemical Equations (2)–(7), mentioned in Section 2.2, during the gasification process. Adding catalytic effects into the gasification model requires correcting of the kinetic parameters compared with the uncatalyzed simulation. We assumed that alkali metals merely increase the rate of the carbon-steam reaction (3), which is the step-determining step in the steam/air gasification process after the char is decomposed [31]. A correction factor is introduced based on the kinetics of catalytic coal gasification with alkaline organic wastewater in the fluidized bed. The correction factor was obtained via the following method: it equaled the ratio of kexp to kn, where kexp is the reaction rate constant from the catalytic gasification experiment, and kn is the reaction rate constant without catalytic effects calculated from Table 1 as used in Equation (8).
2.4. Characteristics of Feed Coal and Operation Parameters
2.5. Calculation of CCG, CGE, and LHV Values
3. Results and Discussion
3.1. Model Validation
3.1.1. Validation of Fluidized Bed Model without Catalytic Effect
3.1.2. Validation of the Fluidized Bed Model with a Catalytic Effect
3.1.3. Calculation of Root Mean Square Error (RMSE)
3.2. Influence of Operating Conditions on Gasification Performance
3.2.1. Effect of Gasification Temperature
3.2.2. Effect of the Steam to Coal (S/C) Ratio
3.2.3. Effect of the ER Ratio
3.3. Energy Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| Mad | moisture of the coal sample under air dry basis (%) |
| Vd | volatiles of the coal sample under dry basis (%) |
| Ad | ash of the coal sample under dry basis (%) |
| T | reaction temperature (K) |
| mass transfer coefficient for gas film diffusion (gm-mole/cm3·sec·atm) | |
| mass transfer coefficient for ash diffusion (gm-mole/cm3·sec·atm) | |
| porosity of ash | |
| average radius of shrinking unreacted coal particles (cm) | |
| average radius of feed coal (cm) | |
| [C] | concentration of char |
| C | mole concentration of the subscripted substance (mol/m3) |
| P | partial pressure of the subscripted substance (atm) |
| P* | back reaction equilibrium pressure of the subscripted substance (atm) |
| mole fraction of the subscripted substance (%) | |
| ash correction factor | |
| equilibrium constant of the water–gas shift reaction |
References
- Emamitaba, L.; Irfan, M.F.; Mohd, A.W.D.W.; Chakrabarti, M.H. Fuel blending effects on the co-gasification of coal and biomass—A review. Biomass Bioenergy 2013, 57, 249–263. [Google Scholar] [CrossRef]
- Na, S.H.; Shon, H.K.; Kim, J.B.; JuPark, H.; Kim, J.-H. Preparation and characterization of titania nanoparticle produced from Ti-flocculated sludge with paper mill wastewater. J. Ind. Eng. Chem. 2011, 17, 277–281. [Google Scholar] [CrossRef]
- Poulsen, T.G.; Hansen, J.A. Assessing the impacts of changes in treatment technology on energy and greenhouse gas balances for organic waste and wastewater treatment using historical data. Waste Manag. Res. J. Int. Solid Wastes Public Clean. Assoc. Iswa 2009, 27, 861. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Cai, J.; Chen, J.; Ying, P.; Li, A.; Yin, H. Further Study on Ash Deposits in a Large-Scale Wastewater Incineration Plant: Ash Fusion Characteristics and Kinetics. Energy Fuels 2016, 29, 1812–1822. [Google Scholar] [CrossRef]
- Ma, J.; Liu, D.; Chen, Z.; Chen, X. Agglomeration characteristics during fluidized bed combustion of salty wastewater. Powder Technol. 2014, 253, 537–547. [Google Scholar]
- Bie, R.; Zhao, Y.; Chen, Z.; Lu, J.; Yang, L. Formation Mechanism of Agglomeration Caused by Burning NSSC Black Liquor in a Fluidized Bed Incinerator. Energy Fuels 2009, 23, 683–689. [Google Scholar] [CrossRef]
- Li, S.Y. Study on Incineration of Organic Wastewater with High Concentration in Fluidized Bed. Doctoral Dissertation, Harbin Institute of Technology, Harbin, China, 2006. [Google Scholar]
- Zhao, J.; Ma, Z.; Chen, Y. Migration of alkali metal salts of high salinity organic wastewater in fluidized bed combustion. Energy Eng. 2016, 4, 51–55. [Google Scholar]
- Veraa, M.J.; Bell, A.T. Effect of alkali metal catalysts on gasification of coal char. Fuel 1978, 57, 194–200. [Google Scholar] [CrossRef]
- Tay, H.L.; Kajitani, S.; Wang, S.; Li, C.-Z. A preliminary Raman spectroscopic perspective for the roles of catalysts during char gasification. Fuel 2014, 121, 165–172. [Google Scholar] [CrossRef]
- Chen, Z.; Dun, Q.; Shi, Y.; Lai, D.; Zhou, Y.; Gao, S.; Xu, G. High quality syngas production from catalytic coal gasification using disposable Ca(OH)2, catalyst. Chem. Eng. J. 2017, 316, 842–849. [Google Scholar] [CrossRef]
- Gul-e-Rana, J.; Zhang, J.Y. Catalytic Activity of the Black Liquor and Calcium Mixture in CO2 Gasification of Fujian Anthracite. Cheminform 2007, 15, 670–679. [Google Scholar] [CrossRef]
- Jiang, Y.H.; Chen, Y.; Lin, C.; Zhang, J.-Y. Kinetics of catalytic gasification of anthracite by using organic sodium as catalysts. J. Combust. Sci. Technol. 2012, 18, 539–544. [Google Scholar]
- Lin, J.; Zhang, J.Y.; Zhong, X.Q. Kinetics and compensation effects during steam gasification of Fujian anthracite using viscose liquor as catalyst. J. Fuel Chem. Technol. 2009, 37, 398–404. [Google Scholar]
- Lin, J.; Zhang, J.Y.; Zhong, X.Q. Kinetics and compensation effects of steam gasification of Fujian anthracite using black liquor as catalyst. J. Chem. Ind. Eng. Soc. China 2009, 284, 15750–15761. [Google Scholar]
- Chen, Y.; Zhang, J.Y.; Feng, Y.F. Effects of dispersion of sodium ions on catalytic Fujian anthracite gasification. J. Fuel Chem. Technol. 2014, 11, 1302–1308. [Google Scholar]
- Kuang, J.P.; Zhou, J.H.; Zhou, Z.J.; Liu, J.-Z.; Cen, K. Research on alkali-catalyzed gasification of coal black liquor slurry cokes made up by five different coals. Asia Pac. J. Chem. Eng. 2010, 2, 152–157. [Google Scholar]
- Kuang, J.P.; Zhou, J.H.; Zhou, Z.J.; Liu, J.-Z.; Cen, K. Catalytic mechanism of sodium compounds in black liquor during gasification of coal black liquor slurry. Energy Convers. Manag. 2008, 49, 247–256. [Google Scholar] [CrossRef]
- Beheshti, S.M.; Ghassemi, H.; Shahsavan-Markadeh, R. Process simulation of biomass gasification in a bubbling fluidized bed reactor. Energy Convers. Manag. 2015, 94, 345–352. [Google Scholar] [CrossRef]
- Kong, X.D.; Zhong, W.M.; Du, W.L.; Qian, F. Three Stage Equilibrium Model for Coal Gasification in Entrained Flow Gasifiers Based on Aspen Plus. Chin. J. Chem. Eng. 2013, 21, 79–84. [Google Scholar] [CrossRef]
- Qiu, X.; Yan, W.P.; Sun, J.W.; Dong, J.L. Simulation of fluidized bed gasification of biomass based on minimization of gibbs free energy. Renew. Energy Resour. 2011, 29, 32–39. [Google Scholar]
- Begum, S.; Rasul, M.G.; Akbar, D.; Cork, D. An Experimental and Numerical Investigation of Fluidized Bed Gasification of Solid Waste. Energies 2013, 7, 43–61. [Google Scholar] [CrossRef]
- Tian, Y.; Zhou, X.; Lin, S.; Ji, X.; Bai, J.; Xu, M. Syngas production from air-steam gasification of biomass with natural catalysts. Sci. Total Environ. 2018, 645, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.; Engvall, K.; Yang, W. Model investigation of condensation behaviors of alkalis during syngas treatment of pressurized biomass gasification. Chem. Eng. Process. Process Intensif. 2018, 129, 28–36. [Google Scholar] [CrossRef]
- Preciado, J.E.; Ortizmartinez, J.J.; Gonzalezrivera, J.C.; Sierra-Ramirez, R.; Gordillo, G. Simulation of Synthesis Gas Production from Steam Oxygen Gasification of Colombian Coal Using Aspen Plus. Energies 2012, 5, 4924–4940. [Google Scholar] [CrossRef]
- Che, D.Y.; Han, N.N.; Li, S.H.; Liu, H. Simulation Study on Influence of Steam on Co-gasification of Biomass and Coal in a Fluidized Bed. Proc. CSEE 2013, 33, 40–45. [Google Scholar]
- Jang, D.H.; Kim, H.T.; Chan, L.; Kim, S.H. Kinetic analysis of catalytic coal gasification process in fixed bed condition using Aspen Plus. Int. J. Hydrog. Energy 2013, 38, 6021–6026. [Google Scholar] [CrossRef]
- Wen, C.Y.; Chen, H.; Onozaki, M. User’s Manual for Computer Simulation and Design of the Moving-Bed Coal Gasifier; Final Report; Deptement of Chemical Engineering, West Virginia University: Morgantown, WV, USA, 1982. [Google Scholar]
- Plus, A. Getting Started Modeling Processes with Solids; Aspen Technology, Inc.: Bedford, MA, USA, 2000. [Google Scholar]
- Xiong, J.M.; Yang, S.H. Instance Tutorial of Aspen Plus; Chemical Industry Press: Beijing, China, 2012. [Google Scholar]
- Lin, J.; Zhang, J.Y.; Zeng, C.; Zhu, Y.-Z. Apparent kinetics of catalytic gasification on pulverized Fujian anthracite with mixing gas in fluidized Beds. J. Fuel Chem. Technol. 2000, 28, 485–491. [Google Scholar]
- Lu, X.A.; Yan, W.P. Influence of blending ratios on fluidized bed co-gasification of biomass and coal. J. Power Eng. 2009, 29, 977–983. [Google Scholar]
- Wang, J.Q. System Integration Research on Coal Partial Gasification and Semicoke Combustion. Doctoral Dissertation, Zhejiang University, Hangzhou, China, 2008. [Google Scholar]
- Ding, L.; Yoshikawa, K.; Fukuhara, M.; Kowata, Y.; Nakamura, S.; Xin, D.; Muhan, L. Development of an ultra-small biomass gasification and power generation system: Part 2. Gasification characteristics of carbonized pellets/briquettes in a pilot-scale updraft fixed bed gasifier. Fuel 2018, 220, 210–219. [Google Scholar] [CrossRef]
- Fernandez-Lopez, M.; Pedroche, J.; Valverde, J.L.; Sanchez-Silva, L. Simulation of the gasification of animal wastes in a dual gasifier using Aspen Plus®. Energy Convers. Manag. 2017, 140, 211–217. [Google Scholar] [CrossRef]
- Pauls, J.H.; Mahinpey, N.; Mostafavi, E. Simulation of air-steam gasification of woody biomass in a bubbling fluidized bed using Aspen Plus: A comprehensive model including pyrolysis, hydrodynamics and tar production. Biomass Bioenergy 2016, 95, 157–166. [Google Scholar] [CrossRef]
- Li, G.; Liu, Z.; Li, J.; Fang, Y.; Liu, T.; Mei, Y.; Wang, Z. Application of general regression neural network to model a novel integrated fluidized bed gasifier. Int. J. Hydrog. Energy 2018, 43, 5512–5521. [Google Scholar] [CrossRef]
- Pala, L.P.R.; Wang, Q.; Kolb, G.; Hessel, V. Steam gasification of biomass with subsequent syngas adjustment using shift reaction for syngas production: An Aspen Plus model. Renew. Energy 2017, 101, 484–492. [Google Scholar] [CrossRef]















| Reaction | Reaction Rate | Remarks |
|---|---|---|
| 2 | ||
| 3 | ||
| 4 | ||
| 5 | ||
| 6 | ||
| 7 | - |
| Block Name | Block | Description |
|---|---|---|
| DRYER | RYield | Simulate coal drying based on the water content value in proximate analysis of coal |
| PYROLYS | RYield | Simulate coal pyrolysis based on pyrolysis experiment |
| CHAR-DEC | RStoic | Decompose char into C, H2, O2, N2, S, and ash |
| GASIFI-1/2 | RCSTR | Simulate char gasification |
| MX | MIXER | Mix the feed-in streams |
| SEP | SEP | Separate the gas and solid |
| Proximate Analysis | Ultimate Analysis (d) | ||||||
|---|---|---|---|---|---|---|---|
| Mad | Ad | Vd | C | H | N | S | O a |
| 5.69 | 72.04 | 17.88 | 75.86 | 1.26 | 1.33 | 1.78 | 0.77 |
| Types of Coal | Yongding Fujian Anthracite |
|---|---|
| Coal-feeding flow rate | 0.6 kg/h |
| Steam flow rate | Steam/coal = 0 to 5 |
| Air flow rate | ER = 0 to 1 |
| Gasification temperature | 550 to 900 °C |
| Pressure | 0.1 MPa |
| Fluidized bed volume | 0.001 + 0.001 |
| Void fraction | 0.25 to 0.65 (dense phase and dilute phase) |
| Temp. (K) | f | |
|---|---|---|
| 1023 | 0.0216 | 1593 |
| 1073 | 0.0266 | 699 |
| 1123 | 0.037 | 380 |
| 1173 | 0.0416 | 180 |
| Composition | CO | H2 | CO2 | CH4 | N2 |
|---|---|---|---|---|---|
| RMSE | 0.2714 | 0.1667 | 0.1526 | 0.2883 | 0.0728 |
| T (°C) | S/C | ER | CO (%) | CO2 (%) | H2 (%) | CH4 (%) | Q (W) | LHV (MJ/Nm3) | CCE (%) | CGE (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| 650 | 0.5 | 0.2 | 17.5 | 10.6 | 16.0 | 3.9 | 11.7 | 5.4 | 50.8 | 35.7 |
| 650 | 0.5 | 0.4 | 15.9 | 8.4 | 6.9 | 2.5 | 345.4 | 3.6 | 67.8 | 44.0 |
| 650 | 1 | 0.4 | 15.2 | 8.7 | 8.8 | 2.4 | 156.9 | 3.7 | 69.1 | 56.7 |
| 700 | 0.5 | 0.4 | 16.8 | 8.1 | 7.3 | 2.4 | 271.4 | 3.8 | 70.2 | 47.6 |
| 700 | 1.0 | 0.4 | 15.7 | 8.9 | 9.7 | 2.3 | 75.9 | 3.9 | 72.1 | 63.8 |
| 750 | 0.5 | 0.4 | 17.0 | 8.4 | 7.5 | 2.4 | 240.1 | 3.8 | 71.8 | 49.1 |
| 750 | 1 | 0.4 | 15.6 | 9.6 | 10.2 | 2.3 | 48.3 | 3.9 | 74.2 | 67.3 |
| 800 | 0.5 | 0.4 | 16.7 | 9.1 | 10.2 | 2.4 | 235.9 | 3.8 | 72.7 | 62.1 |
| 800 | 1.0 | 0.4 | 15.0 | 10.6 | 10.6 | 2.3 | 50.6 | 3.9 | 75.5 | 69.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, X.; Wang, X.; Zheng, Z.; Qin, W.; Zhou, Y. Catalytic Coal Gasification Process Simulation with Alkaline Organic Wastewater in a Fluidized Bed Reactor Using Aspen Plus. Energies 2019, 12, 1367. https://doi.org/10.3390/en12071367
Xiao X, Wang X, Zheng Z, Qin W, Zhou Y. Catalytic Coal Gasification Process Simulation with Alkaline Organic Wastewater in a Fluidized Bed Reactor Using Aspen Plus. Energies. 2019; 12(7):1367. https://doi.org/10.3390/en12071367
Chicago/Turabian StyleXiao, Xianbin, Xueying Wang, Zongming Zheng, Wu Qin, and Yumengqiu Zhou. 2019. "Catalytic Coal Gasification Process Simulation with Alkaline Organic Wastewater in a Fluidized Bed Reactor Using Aspen Plus" Energies 12, no. 7: 1367. https://doi.org/10.3390/en12071367




