Porphyridium cruentum Grown in Ultra-Filtered Swine Wastewater and Its Effects on Microalgae Growth Productivity and Fatty Acid Composition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strain and Culture Medium
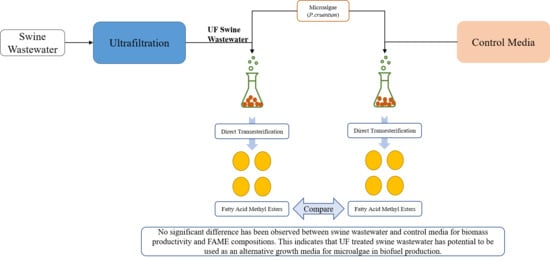
2.2. Swine Wastewater Preparation and Ultrafiltration
2.3. Microalgae Growth Experiments
2.4. Direct Transesterification
2.5. Gas Chromatography Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Growth and Productivity
3.2. FAME Composition
4. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Raheem, A.; Prinsen, P.; Vuppaladadiyam, A.K.; Zhao, M.; Luque, R.A. review on sustainable microalgae based biofuel and bioenergy production: Recent developments. J. Clean. Prod. 2018, 181, 42–59. [Google Scholar] [CrossRef]
- Van Beilen, J.B. Why microalgal biofuels won’t save the internal combustion machine. Biofuels Bioprod. Biorefining 2010, 4, 41–52. [Google Scholar] [CrossRef]
- Cheah, W.; Ling, T.; Show, P.L.; Juan, J.; Chang, J.; Lee, D. Cultivation in wastewaters for energy: A microalgae platform. Appl. Energy 2016, 179, 609–625. [Google Scholar] [CrossRef]
- Asgharpour, M. Eicosapentaenoic Acid (EPA) from Porphyridium Cruentum: Increasing Growth and Productivity of the Microalgae for Pharmaceutical Products; University of Arkansas: Fayetteville, AR, USA, 2015. [Google Scholar]
- Wang, L.; Min, M.; Li, Y.; Chen, Y.; Chen, Y.; Liu, Y.; Wang, Y.; Ruan, R. Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl. Biochem. Biotechnol. 2010, 162, 1174–1186. [Google Scholar] [CrossRef]
- Goldman, J.; Stanley, H. Relative growth of different species of marine algae in wastewater-seawater mixtures. Mar. Biol. 1974, 28, 17–25. [Google Scholar] [CrossRef]
- Kumazawa., S.; Mitsui, A. Characterization and optimization of hydrogen photoproduction by a saltwater blue-green alga, Oscillatoria sp. Miami BG7. I. Enhancement through limiting the supply of nitrogen nutrients. Int. J. Hydrogen. Energy 1981, 6, 339–348. [Google Scholar] [CrossRef]
- Solovchenko, A.; Verschoor, A.M.; Jablonowski, N.D.; Nedbal, L. Phosphorus from wastewater to crops: An alternative path involving microalgae. Biotechnol. Adv. 2016, 34, 550–564. [Google Scholar] [CrossRef] [PubMed]
- Vo Hoang Nhat, P.; Ngo, H.H.; Guo, W.S.; Chang, S.W.; Nguyen, D.D.; Nguyen, P.D.; Bui, X.T.; Zhang, X.B.; Guo, J.B. Can algae-based technologies be an affordable green process for biofuel production and wastewater remediation? Bioresour. Technol. 2018, 256, 491–501. [Google Scholar] [CrossRef]
- Pittman, J.K.; Dean, A.P.; Osundeko, O. The potential of sustainable algal biofuel production using wastewater resources. Bioresour. Technol. 2011, 102, 17–25. [Google Scholar] [CrossRef]
- Valderrama, L.T.; Del Campo, C.M.; Rodriguez, C.M.; de-Bashan, L.E.; Bashan, Y. Treatment of recalcitrant wastewater from ethanol and citric acid production using the microalga Chlorella vulgaris and the macrophyte Lemna minuscula. Water Res. 2002, 36, 4185–4192. [Google Scholar] [CrossRef]
- Kalhor, A.X.; Movafeghi, A.; Mohammadi-Nassab, A.D.; Abedi, E.; Bahrami, A. Potential of the green alga Chlorella vulgaris for biodegradation of crude oil hydrocarbons. Mar. Pollut. Bull. 2017, 123, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Acharya, K. Removal of trimethoprim, sulfamethoxazole, and triclosan by the green alga Nannochloris sp. J. Hazard. Mater. 2016, 315, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Perez, M.; Sanchez-Castillo, P.; Romera, O.; Fernandez-Moreno, D.; Perez-Martinez, C. Growth and nutrient removal in free and immobilized planktonic green algae isolated from pig manure. Enzym. Microb. Technol. 2004, 34, 392–398. [Google Scholar] [CrossRef]
- Barlow, E.; Boersma, L.; Phinney, H.K.; Miner, J. Algal growth in diluted pig waste. Agric. Environ. 1975, 2, 339–355. [Google Scholar] [CrossRef]
- Hu, B.; Min, M.; Zhou, W.; Du, Z.; Chen, P.; Zhu, J.; Cheng, Y.; Liu, Y.; Ruan, R. Enhanced mixotrophic growth of microalga Chlorella sp. on pretreated swine manure for simultaneous biofuel feedstock production and nutrient removal. Bioresour. Technol. 2012, 126, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Mulbry, W.; Kondrad, S.; Buyer, J. Treatment of dairy and swine manure effluents using freshwater algae: Fatty acid content and composition of algal biomass at different manure loading rates. J. Appl. Phycol. 2008, 20, 1079–1085. [Google Scholar] [CrossRef]
- Wu, P.-F.; Teng, J.; Lin, Y.-H.; Hwang, S.-C.J. Increasing algal biofuel production using Nannocholropsis oculata cultivated with anaerobically and aerobically treated swine wastewater. Bioresour. Technol. 2013, 133, 102–108. [Google Scholar] [CrossRef]
- Guihéneuf, F.; Stengel, D.B. Towards the biorefinery concept: Interaction of light, temperature and nitrogen for optimizing the co-production of high-value compounds in Porphyridium purpureum. Algal Res. 2015, 10, 152–163. [Google Scholar] [CrossRef]
- Sato, N.; Moriyama, T.; Mori, N.; Toyoshima, M. Lipid metabolism and potentials of biofuel and high added-value oil production in red algae. World J. Microbiol. Biotechnol. 2017, 33, 74. [Google Scholar] [CrossRef]
- Griffiths, M.J.; Harrison, S.T.L. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J. Appl. Phycol. 2009, 21, 493–507. [Google Scholar] [CrossRef]
- Sandefur, H.; Asgharpour, M.; Mariott, J.; Gottberg, E.; Vaden, J.; Matlock, M.; Hestekin, J. Recovery of nutrients from swine wastewater using ultrafiltration: Applications for microalgae cultivation in photobioreactors. Ecol. Eng. 2016, 94, 75–81. [Google Scholar] [CrossRef]
- Kim, H.M.; Oh, C.H.; Bae, H.-J. Comparison of red microalgae (Porphyridium cruentum) culture conditions for bioethanol production. Bioresour. Technol. 2017, 233, 44–50. [Google Scholar] [CrossRef]
- Luo, J.; Ding, L.; Qi, B.; Jaffrin, M.; Wan, Y. A two-stage ultrafiltration and nanofiltration process for recycling dairy wastewater. Bioresour. Technol. 2011, 102, 7437–7442. [Google Scholar] [CrossRef]
- Falsanisi, D.; Liberti, L.; Notarnicola, M. Ultrafiltration (UF) pilot plant for municipal wastewater reuse in agriculture: Impact of the operation mode on process. Water 2010, 1, 872–885. [Google Scholar] [CrossRef] [Green Version]
- Standard Methods for the Examination of Water and Wastewater, 22nd ed.; APHA American Public Health Association: Washington, DC, USA, 2012.
- Asgharpour, M.; Rodgers, B.; Hestekin, J.A. Eicosapentaenoic acid from Porphyridium cruentum: Increasing growth and productivity of microalgae for pharmaceutical products. Energy 2015, 8, 10487–10503. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Liu, Y.; Wang, H.; Xue, S. Direct transesterification of fresh microalgal cells. Bioresour. Technol. 2015, 176, 284–287. [Google Scholar] [CrossRef]
- Velasquez-Orta, S.B.; Lee, J.G.M.; Harvey, A.P. Evaluation of FAME production from wet marine and freshwater microalgae by in situ transesterification. Biochem. Eng. J. 2013, 76, 83–89. [Google Scholar] [CrossRef]
- Lee, E.Y.; Bazin, M.J. Environmental factors influencing photosynthetic efficiency of the micro red alga Porphyridium cruentum (Agardh) Nageli in light-limited cultures. New Phytol. 1991, 118, 513–519. [Google Scholar] [CrossRef]
- Cohen, Z.; Shiran, D.; Khozin, I.; Heimer, Y.M. Fatty acid unsaturation in the red alga Porphyridium cruentum. Is the methylene interrupted nature of polyunsaturated fatty acids an intrinsic property of the desaturases? Biochim. Et Biophys. Acta (BBA)–Lipids Lipid Metab. 1997, 1344, 59–64. [Google Scholar] [CrossRef]
- Li, T.; Xu, J.; Wu, H.; Jiang, P.; Chen, Z.; Xiang, W. Growth and biochemical composition of Porphyridium cruentum SCS-02 under different nitrogen concentrations. Mar. Drugs 2019, 17, 124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rezanka, T.; Doucha, J.; Mares, P.; Podojil, M. Effect of cultivation temperature and ligth intensity on fatty acid production in the red alga Porphyridium cruentum. J. Basic Microbiol. 1987, 5, 275–278. [Google Scholar] [CrossRef]
- Redel-Macías, M.D.; Pinzi, S.; Ruz, M.F.; Cubero-Atienza, A.J.; Dorado, M.P. Biodiesel from saturated and monounsaturated fatty acid methyl esters and their influence over noise and air pollution. Fuel 2012, 97, 751–756. [Google Scholar] [CrossRef]
- Gopinath, A.; Puhan, S.; Nagarajan, G. Effect of unsaturated fatty acid esters of biodiesel fuels on combustion, performance and emission characteristics of a DI diesel engine. Int. J. Energy Environ. 2010, 1, 411–430. [Google Scholar]
- Yin, Z.; Zhu, L.; Li, S.; Hu, T.; Chu, R.; Mo, F.; Hu, D.; Liu, C.; Li, D. A comprehensive review on cultivation and harvesting of microalgae for biodiesel production: Environmental pollution control and future directions. Bioresour. Technol. 2020, 301, 122804. [Google Scholar] [CrossRef]
- Iasimone, F.; Seira, J.; Quéméner, E.D.L.; Panico, A.; De Felice, V.; Pirozzi, F.; Steye, J.P. Bioflocculation and settling studies of native wastewater filamentous cyanobacteria using different cultivation systems for a low-cost and easy to control harvesting process. J. Environ. Manag. 2020, 256, 109957. [Google Scholar] [CrossRef]
- Suparmaniam, U.; Lam, M.K.; Uemura, Y.; Lim, J.W.; Lee, K.T.; Shuit, S.H. Insights into the microalgae cultivation technology and harvesting process for biofuel production: A review. Renew. Sustain. Energy Rev. 2019, 115, 109361. [Google Scholar] [CrossRef]



| Sample Name | Medium | Water Type | Contents |
|---|---|---|---|
| C-SEA | Control | Seawater | 95 mL seawater medium, 5 mL algae |
| SW-UF-SEA | Swine Waste | Seawater | 30 mL seawater, 65 mL swine waste, 5 mL algae |
| C-SALT | Control | Salt Water | 95 mL saltwater medium, 5 mL algae |
| SW-UF-SALT | Swine Waste | Salt Water | 30 mL deionized water, 65 mL swine waste, 1.6 g NaCl, 5 mL algae |
| Growth Conditions | t test | ANOVA + Tukey | ANOVA |
|---|---|---|---|
| C-SEA/SW-UF-SEA | 0.26 | 0.32 | 0.14 |
| C-SALT/SW-UF-SALT | 0.06 | 0.08 | 0.08 |
| C-SEA/C-SALT | 0.06 | 0.13 | |
| SW-UF-SEA/SW-UF-SALT | 0.05 | 0.56 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulusoy Erol, H.B.; Menegazzo, M.L.; Sandefur, H.; Gottberg, E.; Vaden, J.; Asgharpour, M.; Hestekin, C.N.; Hestekin, J.A. Porphyridium cruentum Grown in Ultra-Filtered Swine Wastewater and Its Effects on Microalgae Growth Productivity and Fatty Acid Composition. Energies 2020, 13, 3194. https://doi.org/10.3390/en13123194
Ulusoy Erol HB, Menegazzo ML, Sandefur H, Gottberg E, Vaden J, Asgharpour M, Hestekin CN, Hestekin JA. Porphyridium cruentum Grown in Ultra-Filtered Swine Wastewater and Its Effects on Microalgae Growth Productivity and Fatty Acid Composition. Energies. 2020; 13(12):3194. https://doi.org/10.3390/en13123194
Chicago/Turabian StyleUlusoy Erol, Humeyra B., Mariana Lara Menegazzo, Heather Sandefur, Emily Gottberg, Jessica Vaden, Maryam Asgharpour, Christa N. Hestekin, and Jamie A. Hestekin. 2020. "Porphyridium cruentum Grown in Ultra-Filtered Swine Wastewater and Its Effects on Microalgae Growth Productivity and Fatty Acid Composition" Energies 13, no. 12: 3194. https://doi.org/10.3390/en13123194