1. Introduction
Drilling fluids are concentrated fluid–solid mixture whereas high density minerals such as barite with a density of 4.2 s.g. (specific gravity) are used to increase and adjust the density of drilling fluid. As density of the heavy minerals is much higher than the fluid density, gravitational force acts on these particles making them segregate and finally settle out of the fluid. A review of the phenomenon, better known as weighting material sag, and possible measurement methods is found by consulting Ofei et al. [
1]. The sagging effect occurs both in water-based and oil-based drilling fluids. However, it appears to happen faster in oil-based drilling fluids [
2,
3]. Saasen et al. [
4] showed that barite sag can be more intense in fluids that develop fragile gel structures when left static. They defined a fragile gel structure fluid as a fluid with high gel strength, but when the initial gel structure is ruptured, the remaining gel structures will easily break. Thus, a drilling fluid may have high gel strength, but when exposed to small shear rates, its solid particles can start to settle fast. Barite settling is known to cause problems during drilling operations with the most severe consequence leading to well control issues. This is due to the reduction of hydrostatic pressure exerted by the drilling fluid.
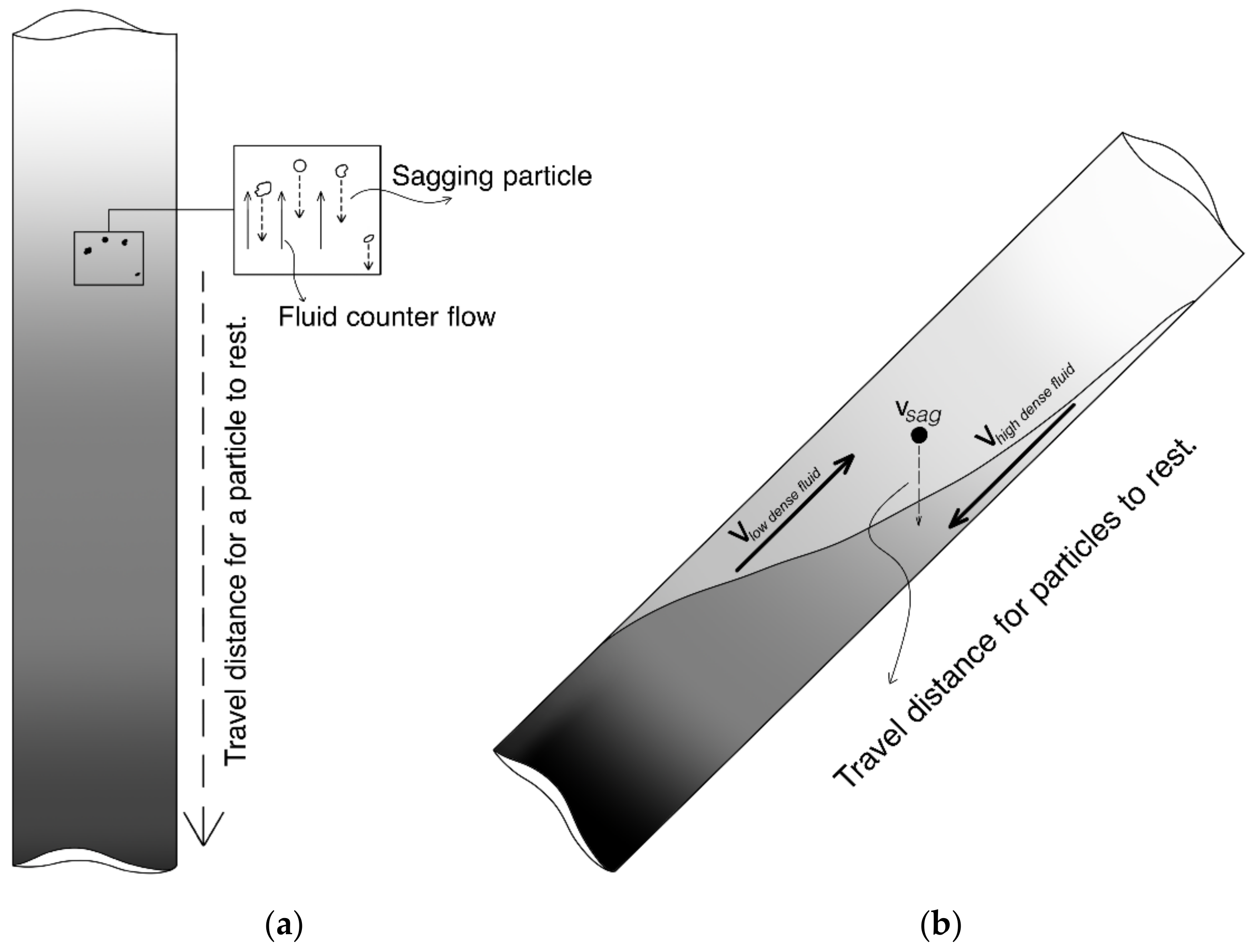
The sag phenomenon in a vertical, static drilling fluid column is controlled by viscosity and gel formation. As the barite particles move downwards because of gravity, a fluid motion is set up in the opposite direction to preserve the fluid mass balance. This counterflow may contain solid particles smaller than the sagging particles. Hence, the sag velocity is reduced. This is illustrated in
Figure 1a. If the static fluid column is inside a tilted pipe, the sag will be accelerated by the so-called Boycott effect [
5]. If the barite particles travel vertically downwards, a less dense fluid layer will be placed above a denser layer in a tilted tube. This will trigger a flow of light fluid upwards and a dense fluid downwards in the well, as illustrated in
Figure 1b, accelerating the settling process. This process continues until most weight material particles have reached the well bottom.
As barite starts to settle out from drilling fluid, the uniform distribution of barite decays resulting in a higher density fluid at the lower part and a lower density fluid at the higher part of the fluid column. This results in a static fluid system that no longer provides a constant hydrostatic pressure gradient throughout the fluid column. When the barite particles rest on a bed, the hydrostatic pressure of fluid column will be lost. This phenomenon can lead to the well bore pressure being in underbalance with respect to formation pressure, potentially causing influx of liquid or gas into the well.
The intensity of barite sedimentation is function of drilling fluid characteristics, such as particle size, concentration of particles, surface charge of particles, and operational circumstances such as flowrate or induced stresses caused by drilling activities, and distance between the particle and bedding distance [
4,
5,
6,
7]. Nguyen et al. [
8] showed that if the fluid is left in static conditions, the barite sag will not occur if the yield stress of the fluid is higher than approximately 6 Pa (12 lbf/100 ft
2). This statement, however, shows the difficulty in determination of the yield stress. By a force balance, it is easily shown that the barite particle should not move if it is exposed to a fluid with a real yield stress exceeding half a Pascal.
Previous studies on barite sedimentation mechanisms showed that in inclined pipes, from vertical to 20° and 40°, the contribution of gravity and Boycott effect on barite settling was observed. It was also observed that the hindered mechanism affected the settling time on the suspended barite particles in water-based drilling fluids as inclination was changed from 40° to vertical [
5,
9]. In drilling operations, there are circumstances where maintaining the hydrostatic pressure profile at the bottom of well or at a desired depth is necessary when the drilling operation is temporarily suspended.
A viscous pill has been tried as a liquid packer placed above the formation with influx potential, to collect the sagging weight material and thus provide proper pressure at this formation. However, a viscous fluid alone will be prone to Rayleigh–Taylor instability [
10] where the dense fluid will flow downward, and the lighter fluid will flow upward. The interface will be even more unstable if it is placed in a deviated section as longer disturbance lengths are less stable than the shorter; this also occurs in the case of polymeric liquids [
11,
12]. This was the reason for the use of the umbrella-like tool placed in the well for plug cementing purposes [
13].
A proper gel pill could be placed just above the formation with the potential of fluid influx, and would hold the settled barite while ideally still transmitting the pressure exerted by the fluid and barite above the pill to the formation below the pill. This pill needs to be properly gelled to prevent formation of Rayleigh–Taylor instability. Gel is defined as a soft colloid that is elastic, deformable, and solid. It is made of colloidal particles together with polymers, which together construct a nonliquid network. When considering the microstructure of gel at microscale, it is “liquid,” and when considering it at macroscale, it is “solid” [
14]. A viscous fluid is liquid with a relatively high viscosity. A viscous pill is usually used for special tasks such as cuttings transportation, lost circulation treatment, or removing filter cakes.
Fosso et al. [
15] emphasized that for a Bingham plastic with a proper yield stress of
in a pipe with diameter of
D, if the pressure gradient does not exceed
, the fluid above the gel pill will remain in stable condition and no flow will occur. A yield stress is the minimum gel strength of a material. Harestad et al. [
13] showed that the maximum pressure difference the gel ideally needs to hold is a density difference across the well cross-section if the fluid above the gel is carefully positioned. Hence, the necessary length of a gel pill is as short as possible to ensure proper placement over the entire pill length.
Inside a pipe, a proper gel pill will always hold back a pressure difference equal to the value shown in Equation (1):
where
is the gel strength,
D is the pipe diameter, and
is the pill length. This equation is found by conducting a simple force balance.
Settled barite may prevent the force transmission. For a layer of completely settled barite particles, any force transmission will be conducted through direct particle–particle contacts. Since these particles have different shapes and sizes, it is not possible to exactly predict the angle of attack between the particles. However, it is anticipated that this would be around 45°. Therefore, pressure transmission may be hindered when the height of the settled barite exceeds the pipe diameter. Any pressure increase after this condition has occurred will only apply more normal forces on the pipe wall.
The primary objective of this study was to evaluate a pressure transmitting gel pill that can act as a resting bed for settled barite particles. In addition, the gel pill should prevent any tendency for Rayleigh–Taylor instability or breakthrough of sagged barite staying on top of the gel pill. The gel pill should establish full pressure transmission of the overlying fluid column to maintain constant hydrostatic pressure underneath the pill. Subobjectives were to evaluate a gel pill with low viscosity during placement and high viscosity and gel strength after placement.
3. Discussion and Remarks
So-called viscous pills have been used extensively in the petroleum industry to prevent intermixing of a dense well fluid placed above a lighter fluid. The success rate has varied. The industry has not had a proper definition of the term viscous pill. Hence, viscous pill has been used for a large variety of limited well fluid volumes pumped into a well. In this work, the term viscous pill has been defined as a high viscosity material without any significant gel formation. If gel formation has been significant, then the term gel pill has been used. Based on the measurement and analysis in the previous section, use of a purely viscous pill cannot hinder intermixing of a high density fluid placed above a lighter fluid. This should therefore not be applied in practical operations.
Two bentonite-type-based gel pill systems were found to be efficient in hindering intermixing of a denser fluid placed above a lighter fluid. These fluids create a good gel structure with bonding to the surface. The analysis did not focus on finding the limit of the yield stress or gel formation required to maintain the desired function. An analysis can be performed based on Equation (1), presenting the necessary gel strength. However, the gel strength measurements that can be obtained following industrial standards recommend methods incapable of measuring the strength of a gelled particle network.
Both the successful gel systems presented in this work are particle gels. Earlier, a crosslinked polymer gel system provided similar effect, as shown by Ronæs et al. [
25]. In this case, the gel did not bond to hole walls. Hence, they transmitted pressure from the overlaying fluid column independent of the magnitude of the added pressure. For the current gel pill system, this transmission is not expected until a pressure difference given by Equation (1) is obtained. As can be seen from the measurements shown in
Figure 12, there is a significant pressure reduction in the region measured using differential pressure transducer number two, which includes the pressure drop over the gel pill. This pressure reduction was not observed by using the bottom hole pressure sensor. Hence, the gel pill was anticipated to trap the down hole pressure until the pressure difference over the gel pill exceeded the maximum pressure it could hold. Then, the gel would move slightly and re-establish pressure control.
During the time of the experiments, the pressure measurements dP1 and dP2 both demonstrated reduction in the differential pressure, resulting from settling of barite. Barite settled onto the gel pill surface without creating any changes in the down hole pressure. In addition to trapping the pressure because of the gel forces, the weight of the settled barite must also be lifted before the gelled pill moves in any direction. It is not yet known how the pressure will change if the barite occupies the whole pipe volume. In this case, it may be possible that the settled barite transfers parts of its weight to the tubing.