1. Introduction
Anthropogenic carbon dioxide (CO
2) emissions are the main cause of climate change [
1]. Despite ongoing efforts to deploy renewable energy generation technologies to replace fossil fuels [
2], the increase in energy demand has kept constant (at around 80%) the share of fossil fuels in the primary energy demand [
3]. As a consequence, global CO
2 emissions have continued to grow [
4] (albeit with a reduction seen during 2020 due to the decreased economic activity associated with the COVID-19 pandemic). Most future scenarios which comply with the Paris Agreement include a large shares of renewable energy such as from wind and solar power, but also a substantial role for carbon capture and storage (CCS) [
5] since there are many thermal conversion processes which for the foreseeable future will have to rely on carbon-based fuels or feedstocks. Several CCS technologies have been investigated and tested over the years, and there has been a special focus on post-combustion systems. The energy penalty associated with the operation of these processes remains, however, the major barrier to the commercial deployment of CCS [
6].
The expansion of renewable technologies during the past few decades relates primarily to wind and solar power, mainly driven by large reductions in the costs for implementation of these technologies. Due to the variability of wind and solar power generation, their market value decreases as their level of penetration in the system increases [
7]. In addition, they may provoke instabilities in the grid [
8]. Thus, to enable a higher level of penetration while maintaining the market value of wind and solar power, different forms of energy storage and flexibilization of operation need to be implemented.
Toward achieving the high penetration levels of wind and solar power generation, energy storage technologies that can handle variations in generation over several days or weeks are of particular importance. In this context, thermochemical energy storage (TCES) is attracting a lot of attention, since in contrast to other storage options, it offers the possibility for long-term storage and shipping with low losses [
9], and it has larger energy densities than thermal energy storage (TES) systems [
10]. Among the alternatives, some gas-solids cycles represent the most-promising TCES systems due to their high reversibility, stability, and enthalpy of reaction. Although packed beds (moving or stationary) have been typically used for investigations of TCES through solids cycling at the bench-, laboratory-, and pilot-scales [
11], fluidized beds are envisioned as the reactor technology of choice for commercial-scale usage due to their superior performance in terms of mass and thermal mixing and operational flexibility.
The calcium looping (CaL) process has been investigated both as a CCS and a TCES technology, as well as an integrated system ([
12,
13] with references therein). This indicates that CaL potentially has dual uses in the energy transition [
2]. The CaL process is based on the multicyclic calcination–carbonation of CaCO
3, which can be obtained from limestone, a cheap and abundant material. The process is based on the following reactions:
When applied for CO
2 capture, the CaL process represents a competitive capture technology in terms of both efficiency and costs [
14]. If implemented as TCES, it increases the dispatchability of renewable energy facilities that are able to provide high-temperature streams, such as concentrated solar power (CSP) plants [
15,
16]. Integrating both applications, the CaL process studied here can turn variable renewable energy (VRE) into dispatchable electricity while at the same time mitigating atmospheric CO
2 emissions from a nearby emitting plant.
A review of the technical implications of scale-up of the CaL process for both CO
2 capture and TCES applications has recently been published by Ortiz et al. [
17], including an analysis of different gas–solids reactor systems. Moreover, Ortiz et al. [
15] have published an in-depth review of the different process schemes, conditions, and materials that are advantageous for the operation of the TCES-CSP application. Bayon et al. [
18] have conducted a techno-economic comparison of the capital (excluding the reactors) and operational costs of 17 gas–solids TCES systems, arriving at a cost for CaL-TCES of 54
$/kWht (note that this cost is expressed per storage capacity), as compared to the cost of 56–59 €/MWhe estimated by Muto et al. [
19] for a CaL-TCES process using a synthetic sorbent. Nevertheless, detailed studies with cost estimations for the combined CaL-TCES process are scarce, due to the early stage of development of this technology [
20,
21]. This means that they are based on the more-abundant studies of the cost of the more-mature CaL process used for CO
2 capture (for an overview, see [
22]). Among the latter studies, it is worth mentioning the work of Michalski et al. [
23], in which a method for assessing the economic feasibility of CaL-CCS processes was proposed based on commercial technology appraisal tools. According to a study carried out by Mantripragada et al. [
24], the reactors, together with solids handling, represent the largest cost for the CaL plant.
In summary, although the economic feasibility of the CaL process has been widely studied for CO2 capture and some studies have assessed the TCES scheme, there is a gap in the knowledge regarding the economics of systems in which the CaL process is applied integrating both purposes. Additionally, there is a lack of research about the economic scalability of the process and the implications that the foreseen electricity selling price and carbon capture cost would have on the economic performance of the process.
The aim of the present work is to estimate the cost of the CaL process at different scales when it is deployed for TCES in a concentrated solar plant (CSP) as the renewable non-dispatchable energy source combined with capture of the CO
2 from a nearby emitter (i.e., not accounting for the transportation and storage of CO
2). The CaL process here studied is assumed to make profit from the sale of dispatchable electricity and from the CO
2 capture services provided to a nearby emitting plant. Such a case is motivated by: (i) The need for CO
2-emitting facilities to operate until the initial investment is paid off, which under a regime of increasing costs for CO
2 emissions, will require the capture of the emitted CO
2; (ii) the potential of biomass-based energy generation facilities to contribute to negative emissions by capturing their CO
2; and (iii) the suitability of CSP to drive heat-demanding conversion processes at high temperatures.
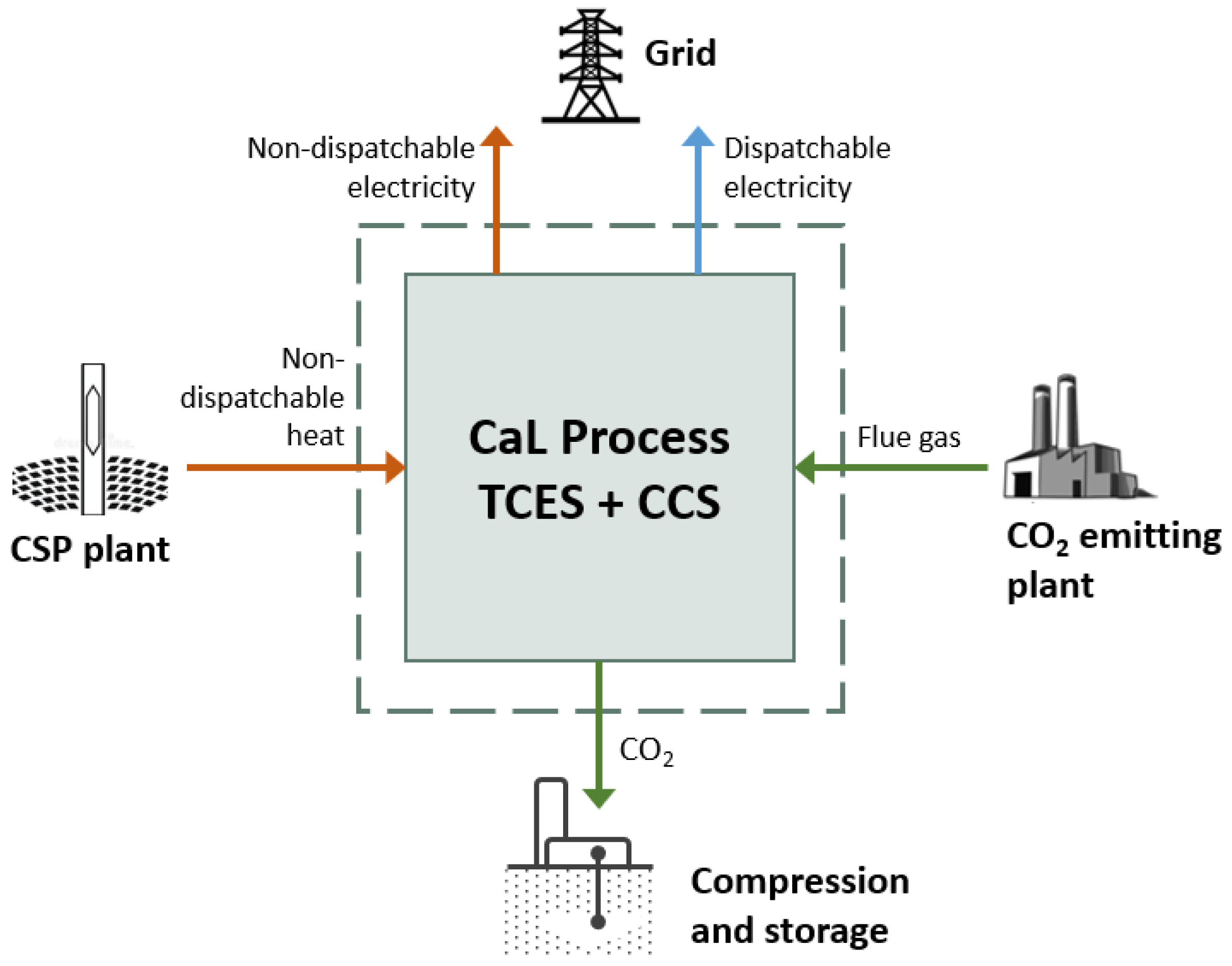
Figure 1 shows a schematic of the relationships between the process investigated in this work, the nearby CSP plant and CO
2-emitting process plant.
The process scheme presented here takes as its starting point a previous thermodynamic study of CaL for TCES [
25], thereafter adapting the process for the integration of carbon capture. The cost of the process is calculated using a bottom-up approach and presented in the forms of the breakeven electricity selling price (BESP), payback period (PBP), and the net capture cost (i.e., cost per CO
2 captured). Moreover, the sensitivity of the computed cost to process size, degree of conversion of solids, and income associated with the CO
2 captured and electricity sales, as well as to the economic parameters used for the assessment is investigated.
2. Process Description
This section describes the CaL process scheme used in the current investigation. The general process layout proposed by Chacartegui et al. [
25] has been used as the basis for the CaL scheme, while certain process conditions have been modified and additional assumptions have been made in line with the nature of the present work (i.e., combined TCES and CO
2 capture). Furthermore, the process scheme considers fluidized beds for both the carbonator and calciner reactors, which adds certain requirements related to the use of fluidization agents. The energy input to the process is assumed to originate from an intermittent renewable energy source that is capable of providing high-temperature (>700 °C) heat, i.e., a CSP plant. Note that the process presented here could also be supplied by other sources of renewable, non-dispatchable energy, such as wind power. However, that would require a power-to-heat process prior to the calciner that is capable of providing heat at high temperatures. In that case, the calciner should be designed to operate at the lowest possible temperature, given that power-to-heat technologies capable of providing high-temperature heat (e.g., direct bed irradiation or inserted thermal resistances) [
26] are still at early stages of development.
Figure 2 shows a schematic representation of the process studied in this work, consisting of charging and discharging sections that can be operated independently. Correspondingly, solids storage under ambient conditions is considered for both charged and discharged particles. Although this decreases the process efficiency, it allows for the potential introduction of shipping and make-up streams into the process, without altering the thermodynamic performance of the base case calculated here. Although the shipping of the solids would open up a wider range of markets for the process inputs and outputs, thereby enhancing the process economics, this alternative is outside the scope of the present work.
Energy in the form of heat at high temperature is used to run the charging section, which comprises the calcination reactor and a generator of superheated steam for fluidizing the reactor. As reported previously [
27], calcination in the presence of superheated steam decreases the temperature required for calcination and increases the conversion rate of the solids in the carbonation side. Lower calcination temperatures also offer the possibility to use simpler and cheaper energy collectors [
15]. In this work, the calciner conditions have been fixed at an intermediate temperature of 850 °C and pressure of 1 bar, following the conclusions of a previous study [
15] for calcination in a steam atmosphere, which corresponds to a steam/CO
2 molar ratio of 1. The gas stream leaving the calcination reactor (consisting of H
2O and CO
2) is used to preheat part of the total inflow of discharged solids (using the split fraction suggested previously [
25]), and is subsequently expanded in a turbine down to the condensing pressure (set by the cooling water temperature), enabling also separation of the condensate and leaving the CO
2 ready for compression. A water tank allows the condensate to be stored when the calciner is not in operation. The process incorporates two solids–solids heat exchangers that are used to partially preheat the input solids streams with the hot solids streams leaving the reactors, which is crucial to reduce energy losses when the storage is under ambient conditions.
Energy discharging is achieved through a Rankine cycle that runs the dispatchable steam turbine using the heat released in the carbonator reactor, which is fluidized with the flue gas from a nearby facility, setting the carbonation conditions of the investigated process to 650 °C and 1 bar. Since optimization of the process performance falls outside the scope of this work, a simplistic approach has been followed for the power cycle conditions. Steam at 550 °C and 120 bar is generated and expanded in a single step to the condensing pressure. It should be noted that the possibility of pre-heating the feed-water line is not considered in this work.
4. Results and Discussion
Figure 3 presents a simplified energy flow diagram of the process with the values corresponding to the base-case process (see
Table 1). The top panel illustrates the energy distribution when the charging side is operating (VRE source is available), while the bottom panel shows the energy flows when only the discharging side is operating. It is evident that the rather low efficiency of the base process (28%) is mainly attributable to heat losses in the heat exchangers (mostly in the condensers of the power blocks), parasitic losses connected to the solids conveying, and heat losses during solids storage. All the latter losses are high due to the low conversion efficiency in the carbonator (
xcarb = 0.25 for the base case), which renders a large proportion of the circulating solids inactive. Here, it must be pointed out that the process is far from optimal, with the two power blocks defined as basic Rankine cycles. Studies reported in the literature have shown that the efficiency of the optimized process (for the TCES scheme) can reach up to 45% [
25,
45]. Furthermore, the vast majority of the losses are in the form of low temperature heat, and this work does not consider the possibility to include heat streams in the product portfolio of the plant (in the form of district heating or industrial heating), in which case some portion of this loss would be transformed into additional revenues. It is worth pointing out that the use of superheated steam to fluidize the calciner implies an added energy loss in the steam generator that is partly recovered by the non-dispatchable turbine, which produces almost half of the energy when the charging side is in operation. Varying the percentage of steam in the calciner (50% is used in the base case) has an impact on the total energy output shares. A lower requirement for steam in the calciner increases the dispatchability of the process, i.e., the share of the output energy delivered by the dispatchable turbine, as well as the total efficiency of the process, while allowing for the capture of larger volumes of CO
2.
Figure 4 maps the total plant costs for different plant sizes expressed as different net energy inputs. It is clear that the reactors represent the major fraction of the total cost, especially for larger plant sizes (i.e., >80% of the total cost for the 1000-MW case versus 75% for the 50-MW case), which is in line with a previous study [
24]. Note that in the present work, the reactor costs also include the heat transfer surfaces, which function is fundamental both for transferring heat into the calciner as well as for steam generation and superheating in the carbonator. These results highlight the importance of costing of the reactors when assessing TCES processes, and this should be borne in mind when choosing a specific reactor type and design [
15]. It can also be seen in
Figure 4 that the cost of storage is negligible when compared to the reactors, indicating that the process could handle higher levels of flexibility at a similar cost. The fixed operational and maintenance (O&M) costs and the capital cost of the heat exchangers are the second and third largest expenses, with the relative importance of the former decreasing substantially with plant size.
As expected, the BESP decreases with the process size (see
Figure 5a), ranging from 141
$/MWh for the smaller (50-MW) reactor to −20
$/MWh for the 1-GW case (all other process parameters were fixed to the base case values). The curve for the variation of BESP with size is steeper for heat inputs in the range of 50–500 MW than between 500 and 1000 MW. Note that a negative BESP value indicates that the plant would already be profitable accounting only for the income connected to the carbon capture. The impact of the revenue stream related to CO
2 capture (
IncomeCC) is further explored by varying the base case assumption (50
$/tCO
2-captured) (see
Figure 5a). The results show that for an assumed carbon capture income of 10
$/tCO
2-captured, the BESP of the plant ranges from 229 to 68
$/MWh, whereas for the most optimistic assumption of 100
$/tCO
2-captured the computed BESP lies between 30 and −130
$/MWh. It can be seen that the net impact of the specific CO
2 capture income on the cost is constant with process size, due to the fact that the amount of CO
2 captured scales linearly with the net heat input. Consequently, the income associated with the CO
2 capture exerts a stronger influence on the total plant economics at larger sizes. It should be noted the importance of choosing an appropriate cost function for the reactors when assessing the economic performance of the CaL process, as the reactors are of major importance in the cost structure and underestimating their cost can yield misleadingly optimistic results. If, for instance, the cost functions of [
23] are used (see
Section 3.2), the computed BESP for the 100 MW base case would be lower down to −9
$/MWh, and down to −30
$/MWh if the cost equations from [
37] are used instead.
A similar trend is observed when the cost is expressed as
$/tCO
2-captured (
Figure 5b), with values ranging from 45 to −27
$/tCO
2-captured for the base case (plotted with orange bars). Here, a negative capturing cost indicates that the plant would be making a profit before the 20 years of the plant lifetime used in the analysis. The variations of the cost to the assumed electricity selling price (ESP) and carbon capture income (
IncomeCC, from 10 to 100
$/tCO
2) are also shown in
Figure 5b. The variations of the cost when varying only the assumed value for ESP (i.e.,
IncomeCC is fixed at the base case value) are plotted for each net heat input, whereas the vertical black lines show the sensitivity when varying
IncomeCC for each of the cases. These results can also be expressed as PBP, as shown in
Figure 5c. As indicated above, only cases with a negative cost in
Figure 5b are breakeven before 20 years, i.e., having a PBP < 20. The analysis shows that the PBP is most sensitive to the variations of
IncomeCC, with a PBP as low as 3.8 years for the most optimistic set of
IncomeCC and ESP assumptions.
The effects of varying some of the economic parameters on the computed BESP for the base case are shown in
Table 4. As expected, both the capacity factor and interest rate play a big role in the economic performance of the process. Note that the capacity factor is strongly linked to plant stops required for maintenance, which in the current process could be optimally planed due to the inherent intermittency expected in the operation. Regarding the limestone cost, it is shown in
Table 4 that its impact is negligible on the process performance.
Figure 6 presents the impacts on plant costs of varying the degree of conversion of the solids in the carbonator, both in terms of BESP and
$/tCO
2-captured. Note that the size of the process, the CO
2 capture income, and the electricity price assumptions have been set at their base case values (100 MW, 50
$/tCO
2, and 40
$/MWh, respectively). The relatively strong impact of
xcarb on the plant cost is confirmed by a reduction of the BESP of 24
$/MWh when
xcarb is increased from 0.15 to 0.25. Increased conversion in the carbonator enhances the efficiency of the process because the total amount of solids required to be cycled in order to generate a certain amount of electricity is decreased. This translates into lower heat losses during solids storage, lower parasitic losses, and smaller sizes of the reactors and heat exchangers. This impact becomes less prominent for
xcarb values > 0.5.
It is important to mention that the relevance of the material conversion for process performance has previously been identified by several groups [
17,
46], although not for a TCES-CCS combined system and not in terms of economic performance. Although important for most TCES solids–gas systems [
11], the solids conversion rate attains special relevance in the CaL process, as its value can change by a factor of 2.5 depending on the reactor conditions [
15] and on whether the process is designed for TCES only or for the TCES-CCS concept presented here. The conditions of the flue gases determine to a large extent the maximum achievable conversion rate and the capacity for CO
2 capture, the revenues from which have been shown to be important for the global feasibility of the process. These findings highlight once again the need for material developments for the successful deployment of the CaL process.
The cost of the energy supply facilities can also play a major role in the profitability of the process presented here. If the energy source is a solar receiver with the characteristics of a CSP plant as assumed in this work, its cost and design become additional aspects that need to be addressed in more detailed economic assessments.
Figure 7 shows the updated cost of the base case for a range of CSP costs, based on the predictions from [
47]. For instance, applying the total cost suggested by Ho [
48] (125
$/kWt for a large-scale particle receiver capable of reaching 1000 °C), the BESP increases from 83 to 102
$/MWh, while the capture cost increases from 21 to 30
$/tCO
2-captured. This is in line with the figures predicted by [
47], in which a CSP plant as the one here applied would represent an added cost of 20–70
$/MWh
th. If the conditions in the calciner are chosen so as to have lower temperatures than those used in this work (850 °C), less costly CSP equipment could be used. However, the impact of such a cost on the overall techno-economic performance is still unknown. Furthermore, if the process is intended to be coupled to a renewable energy facility other than solar CSP, an extra step would be required to convert the non-dispatchable power to heat at the required temperature, and the cost of this step would be added to the total cost.
The results of the current work can be compared on the one hand to other energy storage processes through the BESP values, and on the other hand to other CO
2 capture systems through the cost of capture expressed in
$/tCO
2-captured. As examples of the former, Ganwal et al. [
19] have reported a cost of 56–59
$/MWhe for a CaL storage process with a synthetic material of
xcarb = 0.4, which is comparable to the cost obtained in this work (see
Figure 6). Michalski et al. [
23] have reported a levelized cost of electricity (LCOE) of 80–95
$/MWhe, although their study considered retrofitting a coal power plant. Cormos [
49] also carried out a retrofitting study and concluded that the LCOE of a retrofitted CaL power plant would be in the range of 68–74
$/MWhe. As for cost for capture, some of the main publications that have addressed the cost of CaL-CCS [
22] have reported a cost range of 20–40
$/tCO
2-captured, which is comparable to the costs estimated in this work for carbonation reactivities higher than 0.5. Furthermore, the work of MacKenzie et al. [
50] proposes that the overall cost is primarily sensitive to the material deactivation and limestone cost. Although neglected in the present work, these factors could play important roles in the long-term, given the low degree of solids conversion in CCS applications [
17].