An Intensification of Biomass and Waste Char Gasification in a Gasifier
Abstract
:1. Introduction
2. Materials and Methods
2.1. Used Materials
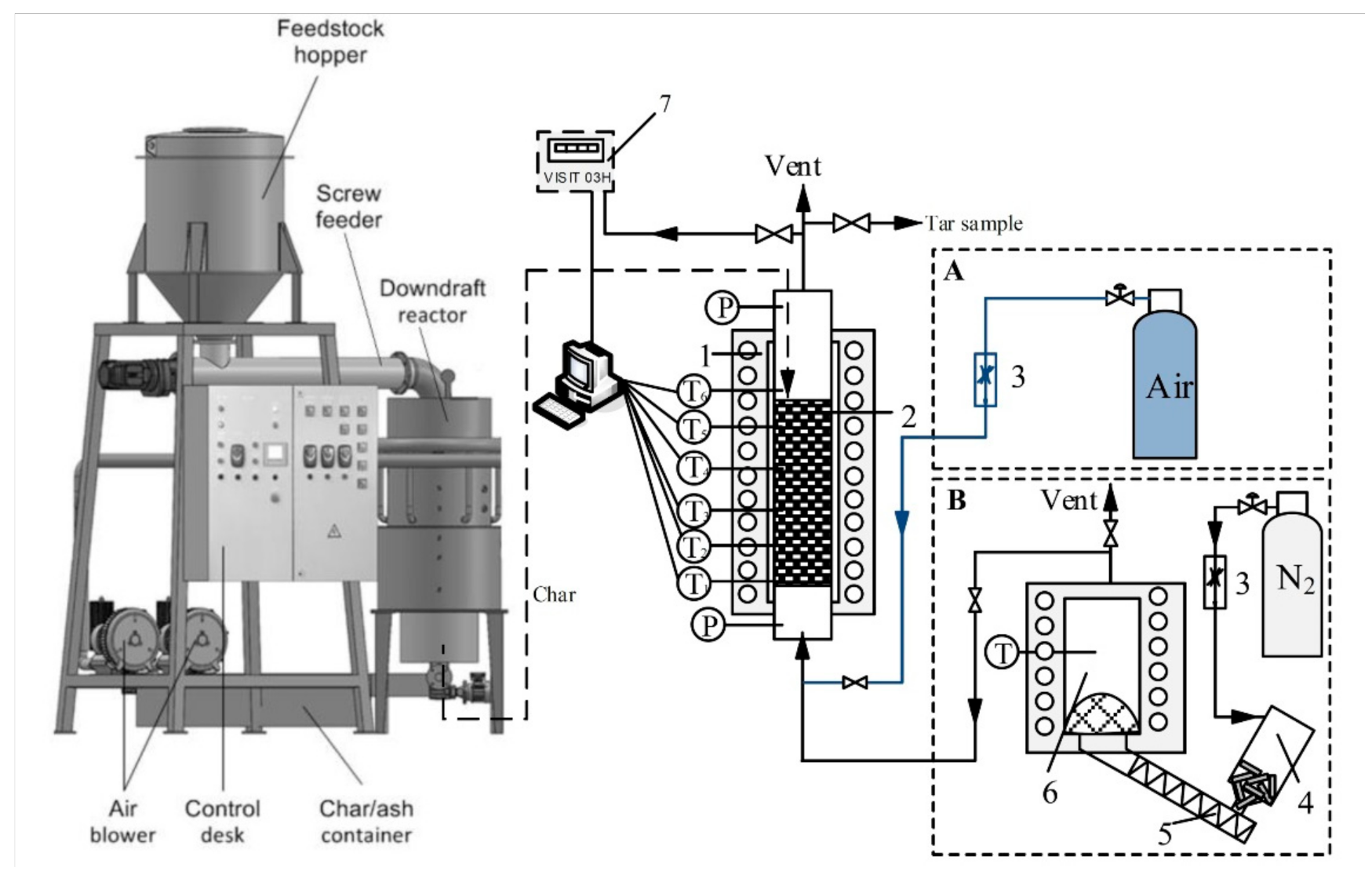
2.2. Experimental Setup and Procedure
3. Results and Discussion
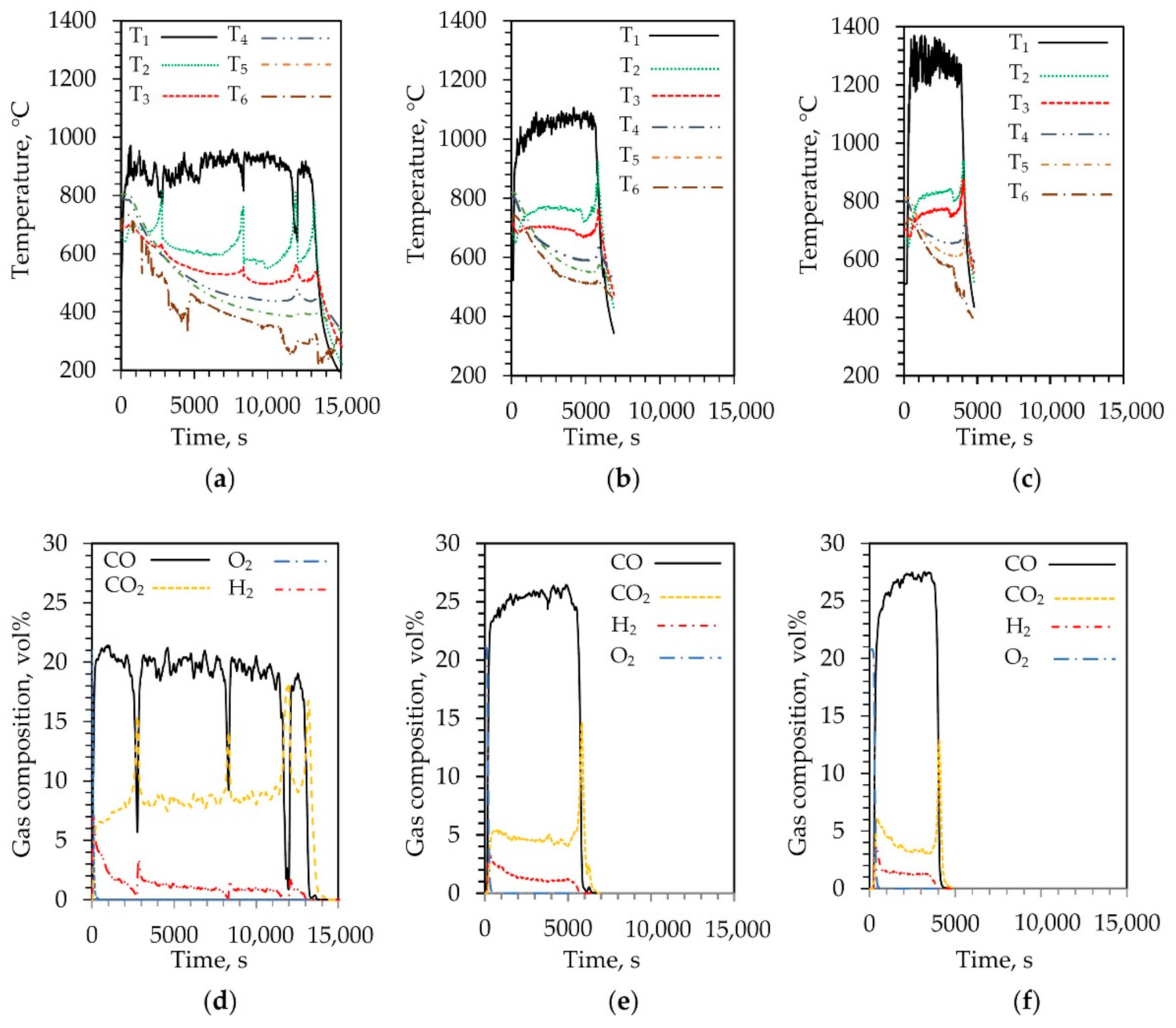
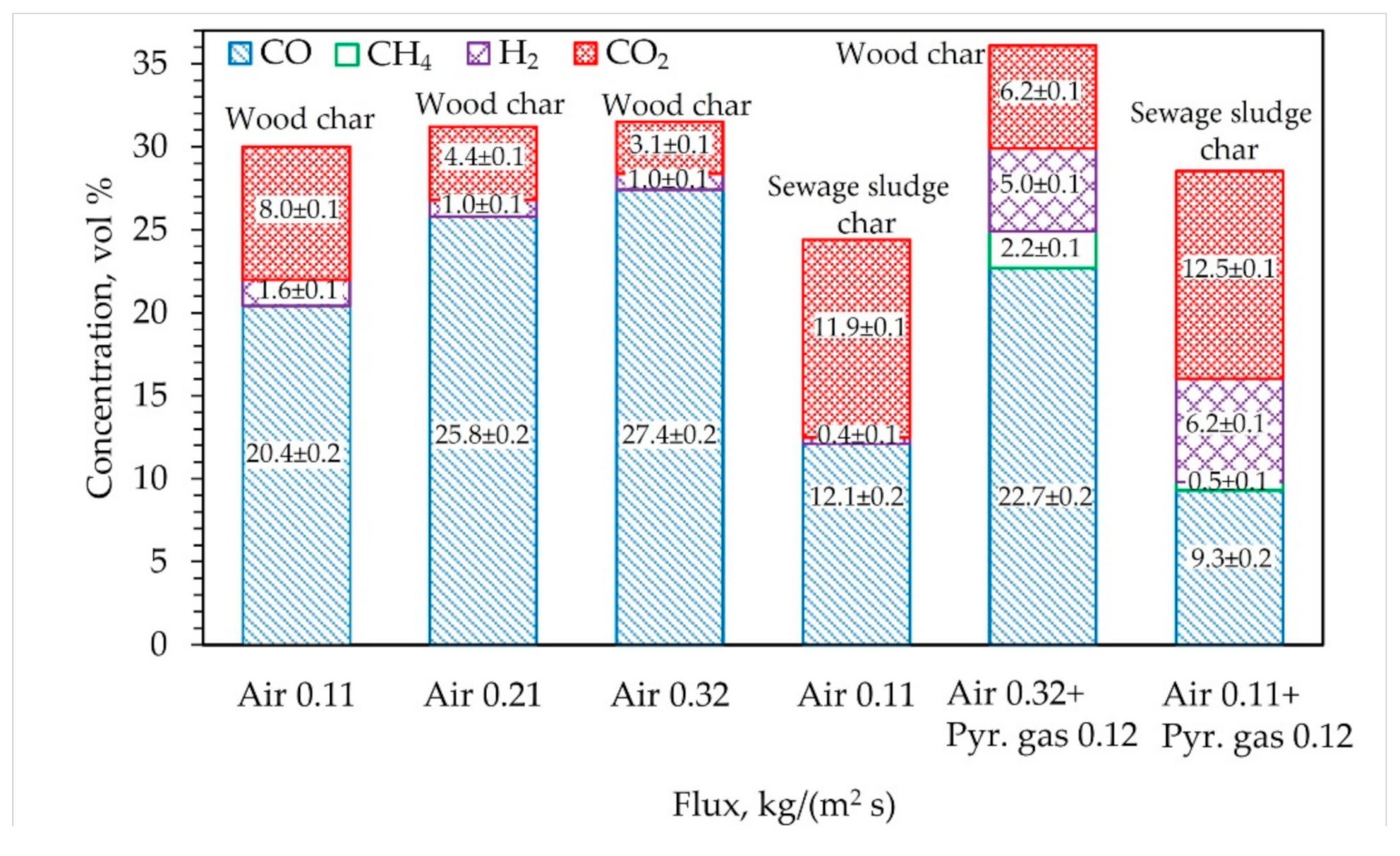
3.1. Analysis of Char Gasification under Air Conditions
3.2. Analysis of Char Gasification Supplying Air and Pyrolysis Gas
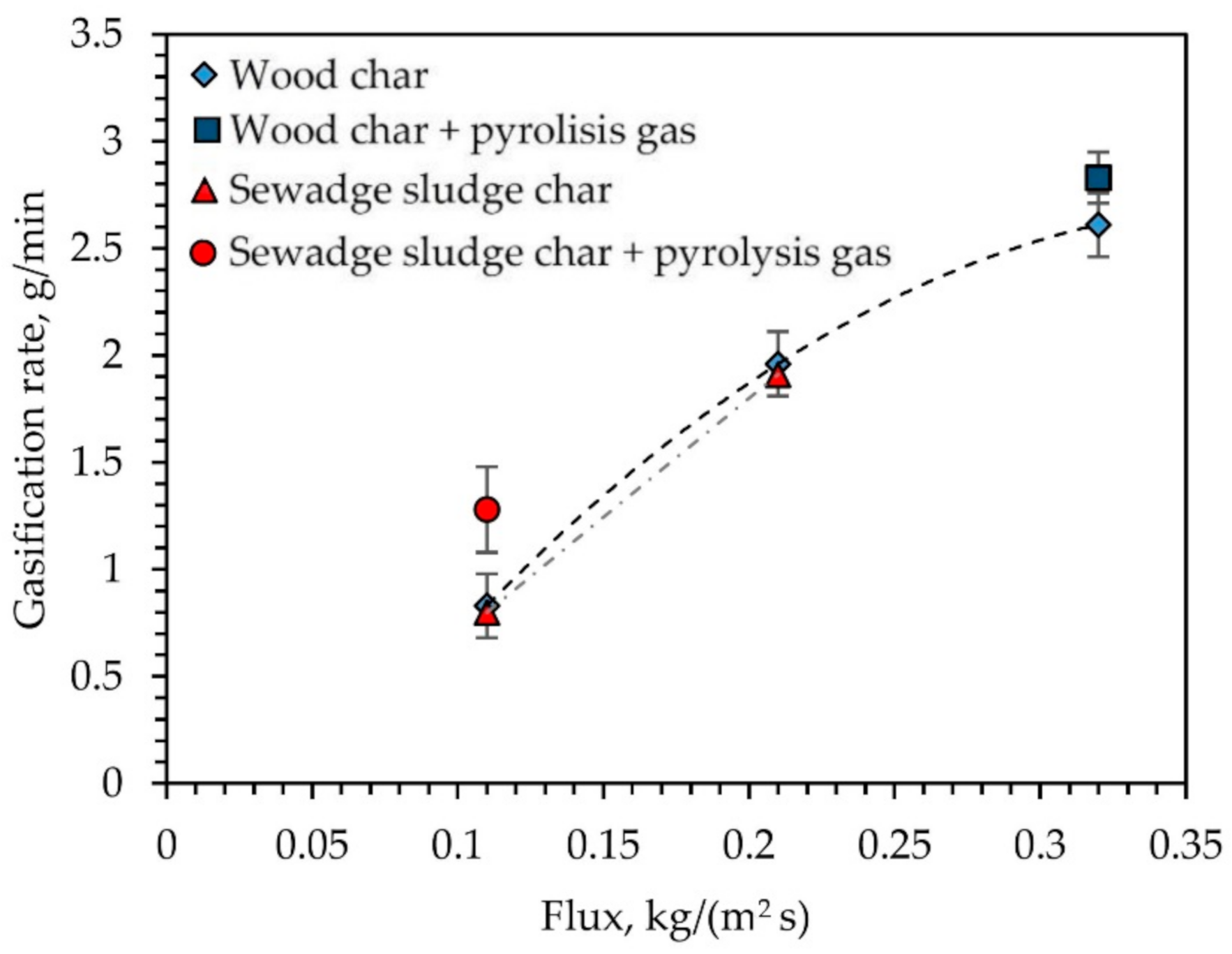
3.3. Air and Pyrolysis Gas Effect on Intensification of Char Gasification
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koniuszy, A.; Hawrot-Paw, M.; Podsiadło, C.; Sędłak, P.; Możdżer, E. Gasification of Cup Plant (Silphium perfoliatum L.) Biomass—ENERGY Recovery and Environmental Impacts. Energies 2020, 13, 4960. [Google Scholar] [CrossRef]
- Chojnacki, J.; Najser, J.; Rokosz, K.; Peer, V.; Kielar, J.; Berner, B. Syngas Composition: Gasification of Wood Pellet with Water Steam through a Reactor with Continuous Biomass Feed System. Energies 2020, 13, 4376. [Google Scholar] [CrossRef]
- Cerone, N.; Zimbardi, F.; Contuzzi, L.; Baleta, J.; Cerinski, D.; Skvorčinskienė, R. Experimental investigation of syngas composition variation along updraft fixed bed gasifier. Energy Convers. Manag. 2020, 221, 113116. [Google Scholar] [CrossRef]
- Ardolino, F.; Arena, U. Biowaste-to-Biomethane: An LCA study on biogas and syngas roads. Waste Manag. 2019, 87, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Michailos, S.; Walker, M.; Moody, A.; Poggio, D.; Pourkashanian, M. Biomethane production using an integrated anaerobic digestion, gasification and CO2 biomethanation process in a real waste water treatment plant: A techno-economic assessment. Energy Convers. Manag. 2020, 209, 112663. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, Z.; Soomro, A.; Ma, S.; Wu, M.; Sun, Z.; Xiang, W. Hydrogen-rich syngas production via sorption-enhanced steam gasification of sewage sludge. Biomass Bioenergy 2020, 138, 105607. [Google Scholar] [CrossRef]
- Lin, C.-L.; Chou, J.-D.; Iu, C.-H. Effects of second-stage bed materials on hydrogen production in the syngas of a two-stage gasification process. Renew. Energy 2020, 154, 903–912. [Google Scholar] [CrossRef]
- dos Santos, R.G.; Alencar, A.C. Biomass-derived syngas production via gasification process and its catalytic conversion into fuels by Fischer Tropsch synthesis: A review. Int. J. Hydrog. Energy 2020, 45, 18114–18132. [Google Scholar] [CrossRef]
- Speight, J.G. Gasifier Types. In Gasification of Unconventional Feedstocks; Elsevier: Amsterdam, The Netherlands, 2014; pp. 54–90. [Google Scholar]
- Sharma, P.; Gupta, B.; Pandey, M.; Singh Bisen, K.; Baredar, P. Downdraft biomass gasification: A review on concepts, designs analysis, modelling and recent advances. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- González-Vázquez, M.P.; García, R.; Gil, M.V.; Pevida, C.; Rubiera, F. Comparison of the gasification performance of multiple biomass types in a bubbling fluidized bed. Energy Convers. Manag. 2018, 176, 309–323. [Google Scholar] [CrossRef]
- Khan, A.A.; de Jong, W.; Jansens, P.J.; Spliethoff, H. Biomass combustion in fluidized bed boilers: Potential problems and remedies. Fuel Process. Technol. 2009, 90, 21–50. [Google Scholar] [CrossRef]
- Kumar, A.; Jones, D.; Hanna, M. Thermochemical Biomass Gasification: A Review of the Current Status of the Technology. Energies 2009, 2, 556–581. [Google Scholar] [CrossRef] [Green Version]
- Plis, P.; Wilk, R.K. Theoretical and experimental investigation of biomass gasification process in a fixed bed gasifier. Energy 2011, 36, 3838–3845. [Google Scholar] [CrossRef]
- Cetin, E.; Moghtaderi, B.; Gupta, R.; Wall, T. Influence of pyrolysis conditions on the structure and gasification reactivity of biomass chars. Fuel 2004, 83, 2139–2150. [Google Scholar] [CrossRef]
- Striugas, N.; Zakarauskas, K.; Džiugys, A.; Navakas, R.; Paulauskas, R. An evaluation of performance of automatically operated multi-fuel downdraft gasifier for energy production. Appl. Therm. Eng. 2014, 73, 1151–1159. [Google Scholar] [CrossRef]
- Khiari, B.; Jeguirim, M.; Limousy, L.; Bennici, S. Biomass derived chars for energy applications. Renew. Sustain. Energy Rev. 2019, 108, 253–273. [Google Scholar] [CrossRef]
- Assima, G.P.; Marie-Rose, S.; Lavoie, J.-M. Role of fixed carbon and metal oxides in char during the catalytic conversion of tar from RDF gasification. Fuel 2018, 218, 406–416. [Google Scholar] [CrossRef]
- Zhang, S.; Asadullah, M.; Dong, L.; Tay, H.-L.; Li, C.-Z. An advanced biomass gasification technology with integrated catalytic hot gas cleaning. Part II: Tar reforming using char as a catalyst or as a catalyst support. Fuel 2013, 112, 646–653. [Google Scholar] [CrossRef]
- Henriksen, U.; Ahrenfeldt, J.; Jensen, T.K.; Gøbel, B.; Bentzen, J.D.; Hindsgaul, C.; Sørensen, L.H. The design, construction and operation of a 75kW two-stage gasifier. Energy 2006, 31, 1542–1553. [Google Scholar] [CrossRef] [Green Version]
- Van de Steene, L.; Tagutchou, J.P.; Mermoud, F.; Martin, E.; Salvador, S. A new experimental Continuous Fixed Bed Reactor to characterise wood char gasification. Fuel 2010, 89, 3320–3329. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, G.; Van de Steene, L.; Martin, E.; Gelix, F.; Salvador, S. Gasification of char from wood pellets and from wood chips: Textural properties and thermochemical conversion along a continuous fixed bed. Fuel 2012, 102, 514–524. [Google Scholar] [CrossRef] [Green Version]
- Roncancio, R.; Gore, J.P. CO2 char gasification: A systematic review from 2014 to 2020. Energy Convers. Manag. X 2020, 100060. [Google Scholar] [CrossRef]
- Kim, D.; Park, D.; Lim, Y.; Park, S.; Park, Y.-S.; Kim, K. Combustion Melting Characterisation of Solid Fuel Obtained from Sewage Sludge. Energies 2021, 14, 805. [Google Scholar] [CrossRef]
- Abánades, A.; Ruiz, E.; Ferruelo, E.M.; Hernández, F.; Cabanillas, A.; Martínez-Val, J.M.; Rubio, J.A.; López, C.; Gavela, R.; Barrera, G.; et al. Experimental analysis of direct thermal methane cracking. Int. J. Hydrog. Energy 2011, 36, 12877–12886. [Google Scholar] [CrossRef] [Green Version]
- Striūgas, N.; Zakarauskas, K.; Džiugys, A.; Grigaitienė, V. Experimental modelling of tar destruction with addition of CO2 for application in the biomass gasification system. Appl. Therm. Eng. 2013, 61, 213–219. [Google Scholar] [CrossRef]



| Wood Char | Sewage Sludge Char | Tire Char | |
|---|---|---|---|
| Ash content, % | 2.2 | 50.1 | 10.3 |
| HHV, MJ/kg | 33.8 | 16.3 | 31.4 |
| Compound | Concentration |
|---|---|
| H2, vol% | 11.2 ± 0.1 |
| CO, vol% | 16.3 ± 0.2 |
| CH4, vol% | 9.1 ± 0.1 |
| CO2, vol% | 19.1 ± 0.1 |
| N2, vol% | 44.3 ± 0.4 |
| Tar, g/m3 | 21.1 ± 1.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paulauskas, R.; Zakarauskas, K.; Striūgas, N. An Intensification of Biomass and Waste Char Gasification in a Gasifier. Energies 2021, 14, 1983. https://doi.org/10.3390/en14071983
Paulauskas R, Zakarauskas K, Striūgas N. An Intensification of Biomass and Waste Char Gasification in a Gasifier. Energies. 2021; 14(7):1983. https://doi.org/10.3390/en14071983
Chicago/Turabian StylePaulauskas, Rolandas, Kęstutis Zakarauskas, and Nerijus Striūgas. 2021. "An Intensification of Biomass and Waste Char Gasification in a Gasifier" Energies 14, no. 7: 1983. https://doi.org/10.3390/en14071983
APA StylePaulauskas, R., Zakarauskas, K., & Striūgas, N. (2021). An Intensification of Biomass and Waste Char Gasification in a Gasifier. Energies, 14(7), 1983. https://doi.org/10.3390/en14071983





