Abstract
Naphtha is an important distillation product of crude oil, and is used as a raw material for first-generation products such as ethylene, propylene, gasoline, xylene (BTX), and others. However, due to the different sources of crude oil, differences in naphtha composition impact the quality of conversion processes. Parameters such as pressure, charge flow, and temperature need to be adjusted for conversion efficiency. This work aims to compare naphtha samples from different origins, through the analysis of distillation curve (ASTM D86), density (ASTM D4052), total sulfur (ASTM D4294), and n-paraffins, iso-paraffins, olefins, naphthene, and aromatics (PIONA, ASTM D5134). Among these parameters evaluated in naphtha, the ones that showed the greatest correlation with the type of oil and its origin was the amount of total sulfur, number of aromatics, and paraffins. The three imported evaluated naphtha presented values greater than 200 mg/kg of total sulfur, aromatics above 9%w, and paraffins (P + I) below 76%w, while the national naphtha presented sulfur contents of at most 141 mg/kg, aromatics below 7%w, and paraffins (P + I) above 78%w. Finally, the study of this type of hydrocarbon enables the understanding of the needs of Latin American refineries and the world in relation to its treatment. National petrochemical companies have more difficulty in processing this product, causing an increase in naphtha importation by 108.51% from 2020/2021 in Brazil. Given this scenario, the Brazilian government should invest more in its petrochemical plants to reduce these imports, which, in the long term, would have a positive impact on the quality and value of naphtha byproducts.
1. Introduction
The petrochemical industry is based on raw materials from the energy industry, mainly naphtha, natural gas, liquified natural gas, and coal. Naphtha is the main raw material for the petrochemical industries of China, Japan, and Europe, occupying 50% of the global cracking raw materials [1]. Specifically, in South America, naphtha is the main raw material in the petrochemical chain for Brazil in (92%), followed by natural gas (8%). Based in capacity and production, Brazilian companies represent an interest of study compared with other samples in South America and the world. The Petrobras company is practically the only naphtha and natural gas producer in Brazil, meeting part of the national demand with its production and imports in the region [2].
The origins of naphtha describes multiple properties in relation to molecule structure and chemical characteristics, especially the relevant composition of aromatics and olefins which define the origins. In this way, different methodologies and sets in petrochemical refineries were improved with the knowledge and characterization of naphtha in the specific PIONA components presented. This raw material undergoes a cracking process, which results in basic petrochemicals such as olefins (ethylene and propylene) and aromatics (gasoline, benzene, toluene, and xylenes). These chemicals can be sold to third parties or, to create value, they can be polymerized in a process where they become thermoplastic resins. The resins are sold to processing industries, which convert them into a final product for sale on the market [3].
This work presents an experimental study applied in 1849 different samples of naphtha obtained in Latin America and their comparison with imported naphtha to quantify properties. The novelty results present the evaluated properties and their relationships with the quality of raw material and subproducts obtained to improve the knowledge of naphtha applications and processes for obtention.
1.1. Naphtha
Naphtha is produced by several methods, which include (1) fractionation of straight run, cracked, reforming distillates, and even crude petroleum; (2) solvent extraction; (3) hydrogenation of cracked distillates; (4) polymerization of unsaturated compounds (olefins); and (5) alkylation, direct distillation, and/or catalytic reforming processes [3,4].
The typical composition of naphtha is constituted of 15–30% crude oil by weight and boils at 30–200 °C. This complex mixture consists of hydrocarbon molecules with 4–12 carbon atoms, mainly including paraffins (P), iso-paraffins (I), olefins (O), naphthene (N), and aromatics (A) (PIONA). Some compounds contain sulfur, nitrogen, and oxygen as heteroatoms, while metallic derivatives (e.g., vanadium, nickel, and silicon) can also exist [5]. Besides the complexity of crude oil composition, the naphtha fraction properties vary according to the reservoir location. Since there is variability in the organic matter (aquatic plants and animals) and decomposition variables (layer deposits, climatic conditions (pressure, low oxygenation, and bacteria)), the crude oil derivatives also have different compositions [6,7].
The composition variability of naphtha is related to its origin and locality, directly impacting its conversion processes, as shown in Table 1 [8,9].

Table 1.
Naphtha: average composition according to reservoir locality, adapted from References [8,9].
In Table 1, it is possible to see that there is a significant difference between naphtha from different origins. Brazilian naphtha had a higher amount of sulfur and paraffin compared with Norway, but compared to Argentina, it had a lower amount of sulfur and a smaller number of aromatics, whereas paraffins are in the same range for PIONA.
Naphtha available on the market is characterized by some properties defined at the time of contract. Among these properties, density, distillation curve, PIONA composition, and sulfur content are the main evaluated properties [1].
The naphtha destination in the petrochemical industry depends on the content of paraffinic hydrocarbons, which is why the precision and detailing of naphtha composition is essential for the optimization of the process since the ethylene yield in the pyrolysis furnace is based on the quality of naphtha that will be used as feedstock [10].
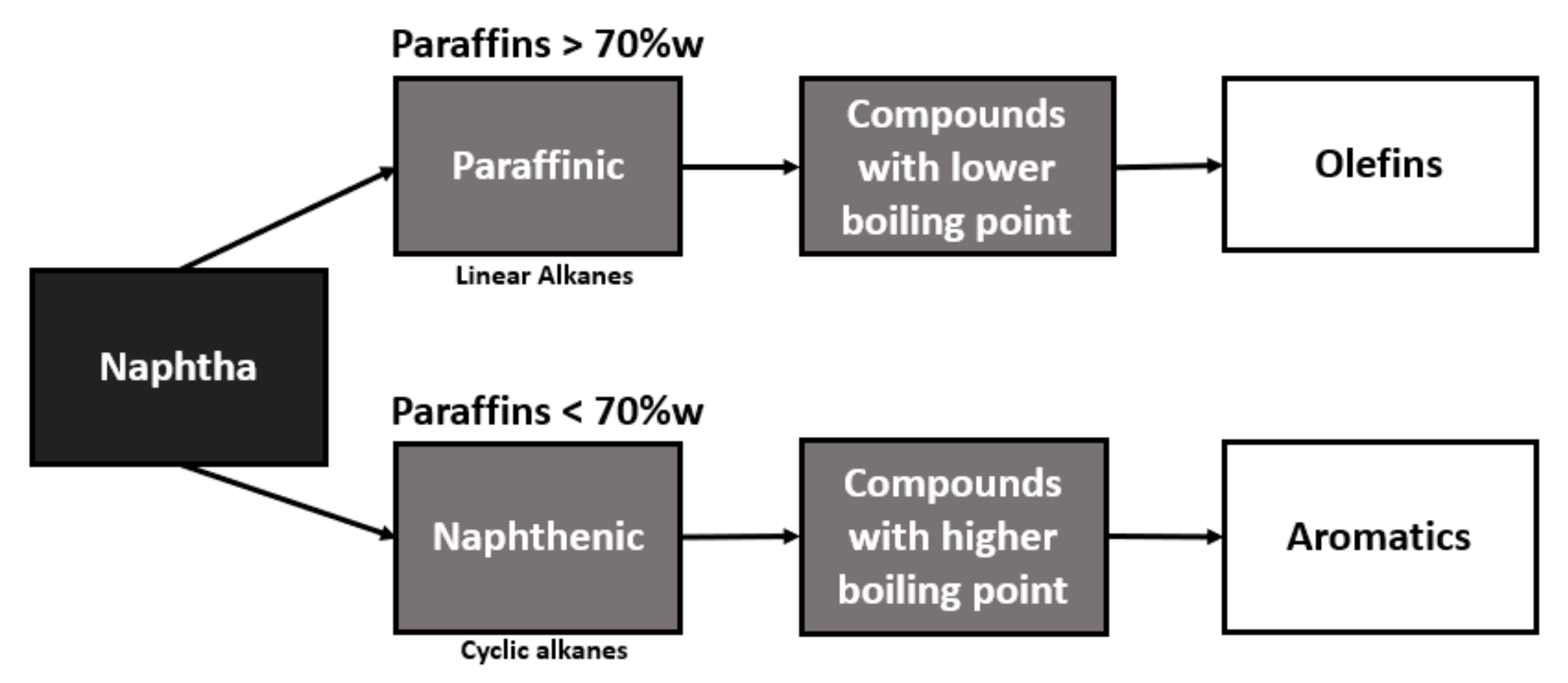
Naphtha with a higher content of paraffin, in its conversion, will present a higher yield in the production of olefins, while naphtha with a higher content of naphthenic will be better used for the production of aromatics, as can be seen in the flowchart in Figure 1.
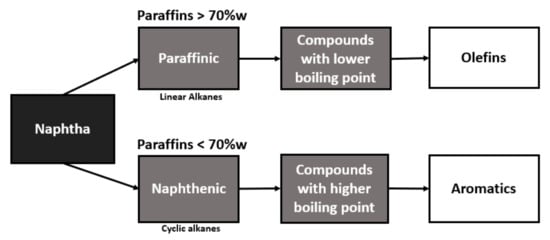
Figure 1.
Flowchart of naphtha conversion into basic inputs.
1.2. Conversion Processes
Petrochemical naphtha is used in processes to obtain various inputs to produce polymers and elastomers, among others [11]. The main processes are:
- a.
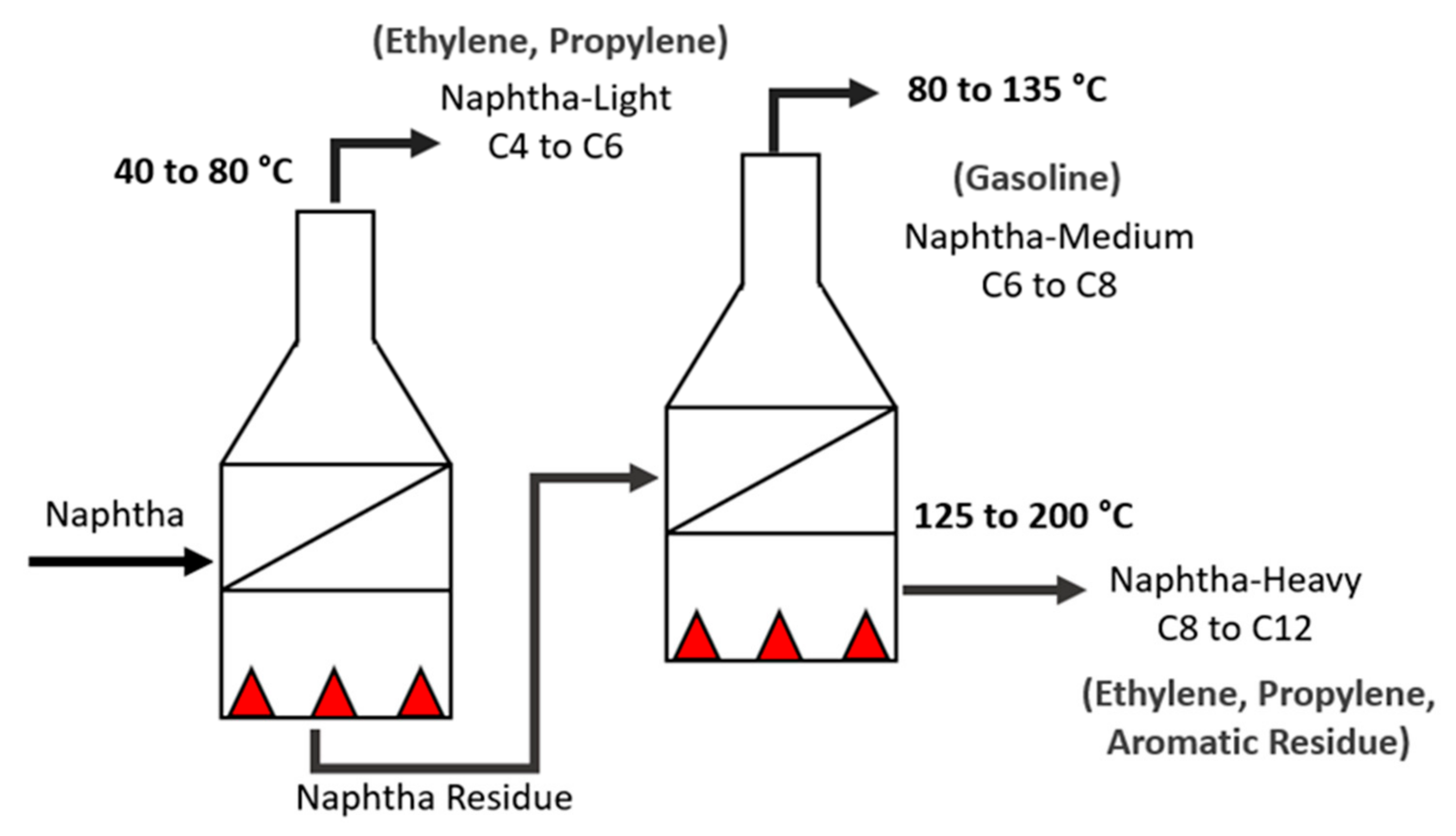
- Pyrolysis: In this process, the naphtha is cracked in several pyrolysis ovens, arranged in series, and is divided into three main fractions, as shown in Table 2 [12]. Each fraction will be better used to obtain certain byproducts, some of which are illustrated in Figure 2.
 Table 2. Naphtha fractions.
Table 2. Naphtha fractions.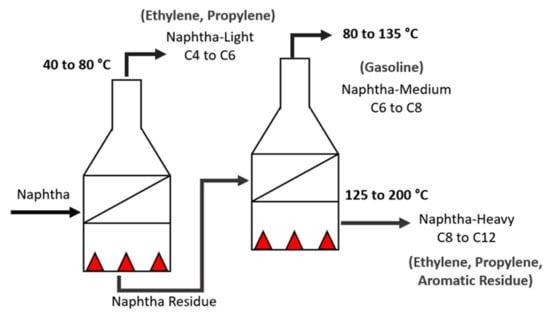 Figure 2. Naphtha cracking—pyrolysis furnaces.The furnaces are the most important and expensive pieces of equipment in the unit and they are considered the heart of the ethylene plant [13].Thermal cracking in the pyrolysis furnace consists of three sections: convection, radiation, and transfer line exchanger (TLE). Naphtha enters the convection zone at a certain temperature, flow rate, and pressure. Steam is added to the feed and serves as a diluent to lower the partial pressure of hydrocarbons (DS/HC) and to reduce coke deposits. The mixture is preheated and after that, it goes to the radiation section, which is where the endothermic reactions take place along the coils. Leaving the radiation zone, the products are transferred by the TLE [14].Considering that the furnace geometry and configuration are already defined to receive a certain type of raw material (distillation range, PIONA quantity, density, and contaminants), the parameters to be adjusted to improve cracking efficiency according to the quality of the naphtha are DS/HC flow rate and residence time, operating temperature in the radiation zone (coil outlet temperature—COT), and pressure at the coil outlet (coil outlet pressure—COP) [15].The COT is what determines the kinetics of the reactions. The higher the temperature, the greater the speed of the reactions and the decrease in the residence time. If the unit receives naphtha with a distillation range greater than the configured one, this means that there are heavier molecules that will need a longer residence time, and in turn, an increase in the residence time favors secondary reactions, that is, the production of aromatic compounds and coke [16].Higher DS/HC ratios favor a higher yield in the production of olefins and minimize the formation of coke in the coils. However, the excessive increase in flow due to the composition and density of the naphtha can cause erosion in coil curves [17].When cracking efficiency is not achieved, more coke is formed in the coils. The coke makes the reactions not occur efficiently within that residence period, making it necessary to increase the temperature of the radiation zone and change the coil outlet pressure. All these factors contribute to the useful coil lifetime, and by reducing it, the pyrolysis byproducts have a lower quality than expected.Naphtha with high levels of unsaturated hydrocarbons (olefins) and aromatics are coke precursors due to their high reactivity at high temperatures. Naphtha with high sulfur contents deactivates catalysts in processes subsequent to pyrolysis.For all these factors, it is extremely important to know the characteristics of the naphtha stream received before processing;
Figure 2. Naphtha cracking—pyrolysis furnaces.The furnaces are the most important and expensive pieces of equipment in the unit and they are considered the heart of the ethylene plant [13].Thermal cracking in the pyrolysis furnace consists of three sections: convection, radiation, and transfer line exchanger (TLE). Naphtha enters the convection zone at a certain temperature, flow rate, and pressure. Steam is added to the feed and serves as a diluent to lower the partial pressure of hydrocarbons (DS/HC) and to reduce coke deposits. The mixture is preheated and after that, it goes to the radiation section, which is where the endothermic reactions take place along the coils. Leaving the radiation zone, the products are transferred by the TLE [14].Considering that the furnace geometry and configuration are already defined to receive a certain type of raw material (distillation range, PIONA quantity, density, and contaminants), the parameters to be adjusted to improve cracking efficiency according to the quality of the naphtha are DS/HC flow rate and residence time, operating temperature in the radiation zone (coil outlet temperature—COT), and pressure at the coil outlet (coil outlet pressure—COP) [15].The COT is what determines the kinetics of the reactions. The higher the temperature, the greater the speed of the reactions and the decrease in the residence time. If the unit receives naphtha with a distillation range greater than the configured one, this means that there are heavier molecules that will need a longer residence time, and in turn, an increase in the residence time favors secondary reactions, that is, the production of aromatic compounds and coke [16].Higher DS/HC ratios favor a higher yield in the production of olefins and minimize the formation of coke in the coils. However, the excessive increase in flow due to the composition and density of the naphtha can cause erosion in coil curves [17].When cracking efficiency is not achieved, more coke is formed in the coils. The coke makes the reactions not occur efficiently within that residence period, making it necessary to increase the temperature of the radiation zone and change the coil outlet pressure. All these factors contribute to the useful coil lifetime, and by reducing it, the pyrolysis byproducts have a lower quality than expected.Naphtha with high levels of unsaturated hydrocarbons (olefins) and aromatics are coke precursors due to their high reactivity at high temperatures. Naphtha with high sulfur contents deactivates catalysts in processes subsequent to pyrolysis.For all these factors, it is extremely important to know the characteristics of the naphtha stream received before processing; - b.
- Catalytic Reforming: This is an important process for producing high octane gasoline, aromatic feedstock, and hydrogen in the petroleum refining and petrochemical industries [18]. In addition, it produces catalytic reformate, which is also a primary source of aromatics used in the petrochemical industry (benzene, toluene, and xylenes—BTX) [19,20].
1.3. Naphtha Characterization
Several analytical methods have been proposed to determine naphtha composition. Most methods are based on chromatographic techniques, such as gas chromatography, multidimensional chromatography, and gas chromatography coupled with mass spectrometry (GC/MS) and intensified separation [3,21].
Gas chromatography (GC) is considered to be the most appropriate method for determining the detailed molecular composition of naphtha fractions [22]. The sample is injected into the chromatograph, and at the same time, the obtained chromatogram is compared with a reference database called Detailed Hydrocarbon Analysis (DHA), where the peaks are categorized by group type and carbon number.
The ASTM D5134 method, based on gas chromatography with a flame ionization detector (GC/FID), is commonly used to determine PIONA composition by petrochemical industry laboratories [23]. GC/MS, for example, can only be used for fractions up until 300 °C, because the higher the boiling point, the greater the number of carbons, which exponentially increases the number of isomers [24]. GC/MS analysis also has limitations in terms of analysis time, components, complexity, and cost, thus, not making it inconvenient for industrial applications. An example of the PIONA analysis result is described in Table 3 [22,25,26,27].

Table 3.
Example of the PIONA analysis result, adapted from references [22,25,26,27].
Other important parameters in naphtha’s characterization are the distillation curve and density values. Such information combined with PIONA analysis enables estimation of naphtha quality, besides the parameter adjustments for the conversion processes. The petrochemical naphtha specification depends on the place where it is sold, while national naphtha specifications are defined by negotiation between suppliers and users in the same country. In Brazil, the National Petroleum Agency (Agência Nacional de Petróleo—ANP) regulates the performed analysis and the respective method to be used [25].
Therefore, the main techniques for measuring the most relevant properties to describe the quality of naphtha are described in Table 4 [12].

Table 4.
Parameters usually evaluated for naphtha characterization [12].
Naphtha quality is superior for the fractions with the absence of some contaminants, such as catalyst poisoning agents (metals such as copper, lead, and iron) as well as environmentally aggressive (chlorides) and corrosive materials (sulfur derivatives) [28].
This work presents the characterization and comparison between naphtha from different origins to correctly qualify them and correlate their composition according to the crude oil reservoir locality.
Although naphtha is the main raw material of petrochemical industries in the world, little is known about its characterization and its impacts on conversion processes. The large quantities of work on the subject are related to the catalysts involved in their production processes and not to their qualification.
The petrochemical industries have faced the challenge of adapting to each type of naphtha received to guarantee the quality of manufactured products. The naphtha cracking process in a pyrolysis furnace consists of a sequence of endothermic reactions that occur in a serpentine (tubular reactor) heated in the presence of water vapor. The average coil’s useful lifetime is around six years, with cleaning intervention every 45 to 60 days, for naphtha with low contaminant levels (sulfur, paraffins, and aromatics, within the specified temperature range of oven operation). However, due to the lack of knowledge about the quality of received naphtha, these interventions lasted less than 25 days, consequently reducing the coil’s useful lifetime, which considerably increases the cost of petrochemicals, generates waste, and impacts the products’ quality, due to loss of reaction efficiency.
Therefore, prior knowledge of the characteristics of naphtha is extremely important for the rapid parameterization of its conversion processes, reducing losses, and minimizing environmental impacts, combined with an economic approach.
2. Experimental Section
2.1. Materials
Data from 1849 samples of naphtha from 6 different origin sources were evaluated and distributed as below:
- Brazil-1 (SP) = 64 samples;
- Brazil-2 (RJ) = 204 samples;
- Brazil-3 (SP) = 50 samples;
- Import-1 (Peru) = 342 samples;
- Import-2 (Argentina) = 691 samples;
- Import-3 (USA) = 523 samples.
These data were obtained from analysis of the samples over 6 years (2014–2020). A novelty evaluation and comparative scope of samples in South America and Northway samples in the main study, in addition with one analysis per sample, describes information to improve the commercial transformation processes.
2.2. Methodology
The summarized analyses performed to characterize the naphtha samples, methodologies, and equipment used are in Table 5. A study of samples from different origins, through the analysis of distillation curve (ASTM D86), density (ASTM D4052), total sulfur (ASTM D4294), and n-paraffins, iso-paraffins, olefins, naphthene, and aromatics (PIONA, ASTM D5134) was applied for 1849 samples.

Table 5.
Analyses performed for naphtha characterization.
According to previous methodology references, the analysis performed for naphtha characterization with South American and Northway markets took these characterizations to determine the capacity of PIONA Oil to remain and understand the current value in the market. Variables considered for research are concentration of aromatics, naphthenic, paraffinity, density, and sulfur; the variables observed on the origin of samples.
3. Results and Discussion
3.1. PIONA (ASTM D5134)
Lighter oils are more paraffinic, such as light Arab oil, which generates more paraffinic naphtha, while heavier oils are more aromatic. This is a crucial point for the formation of a refinery’s inlet load, depending on its market target [31,32].
Analysis of naphtha’s composition by gas chromatography showed more than 198 molecules, whose molecules were grouped into PIONA portions according to their mass fraction.
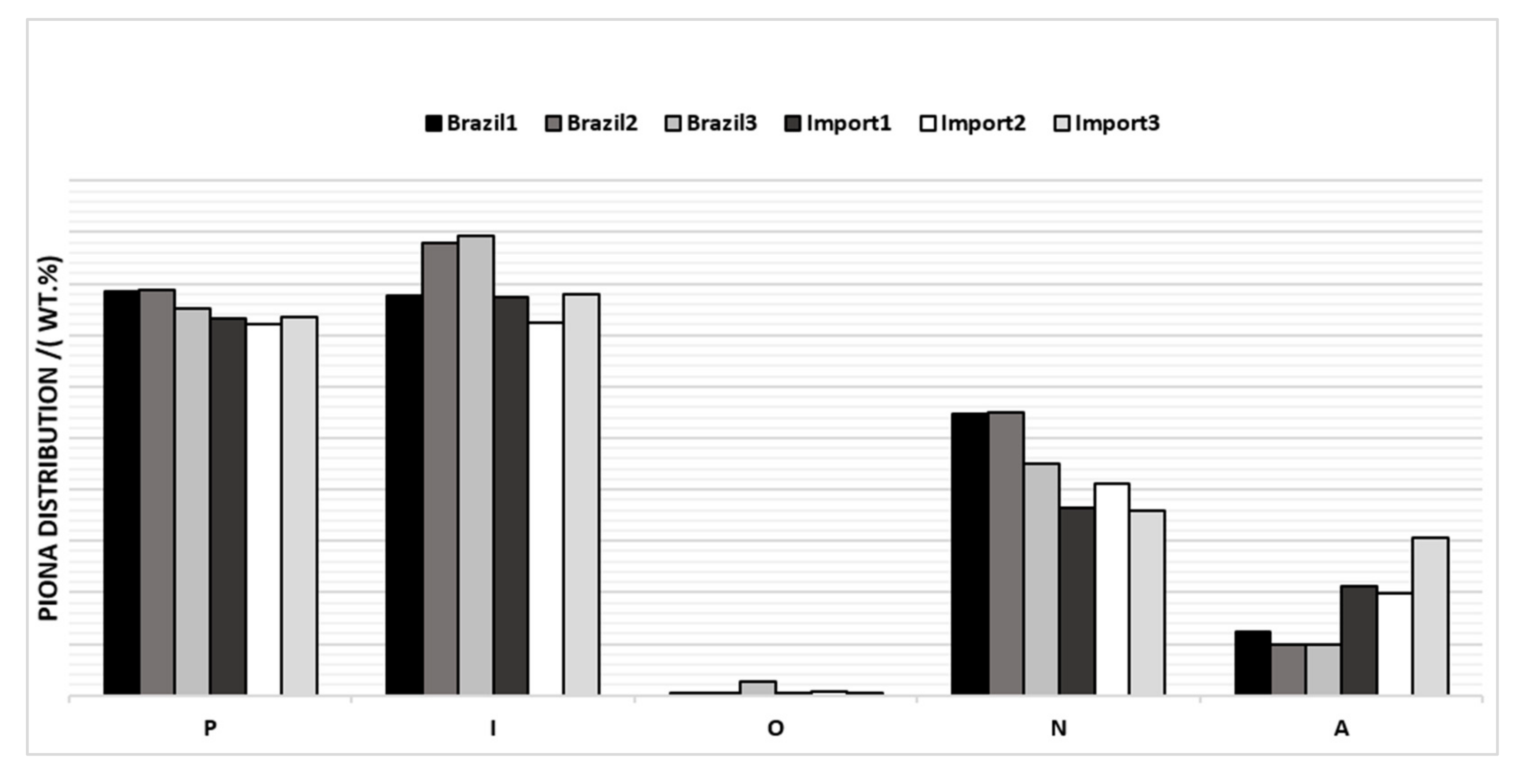
The average PIONA values of the six evaluated samples were plotted (Figure 3), where it was possible to verify differences in the composition between them.
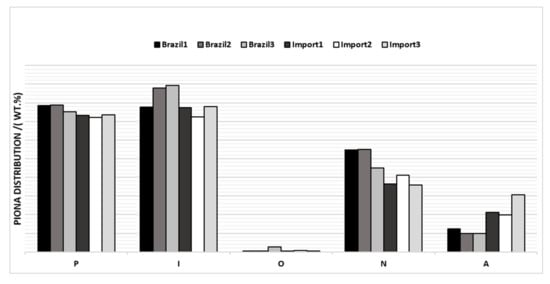
Figure 3.
PIONA of naphtha’s samples by origin.
From the graph above, it is possible to verify that Brazilian samples have an average value of aromatics < 7%w and imported samples have an average value of > 9%w. The number of naphthenics was higher in the Brazilian naphtha with mean values > 22%w, while the number of naphthenics was lower in the imported ones.
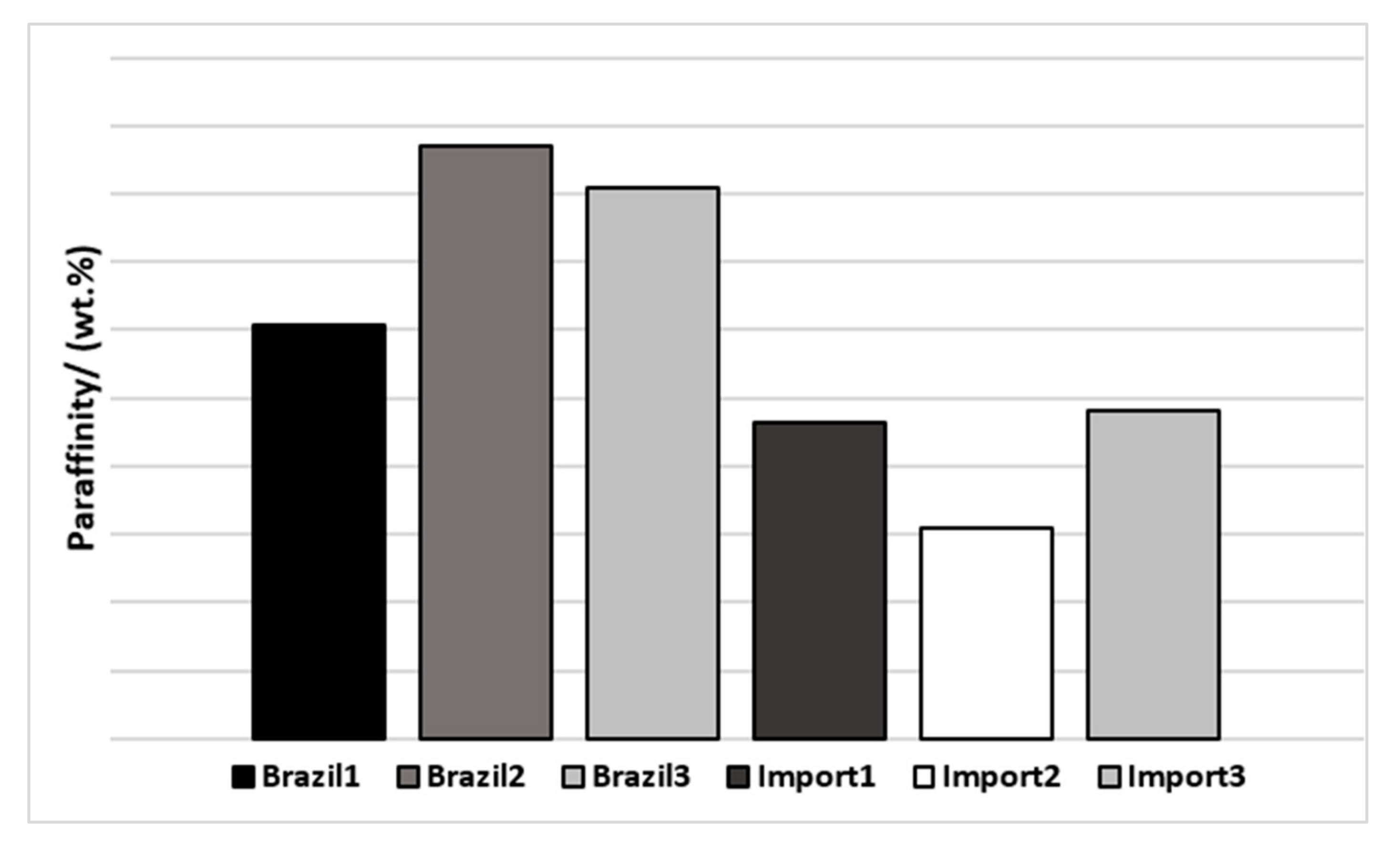
As paraffinity (P + I) is an important factor for qualifying naphtha, a graph with this sum was plotted to facilitate the visualization of differences (Figure 4).
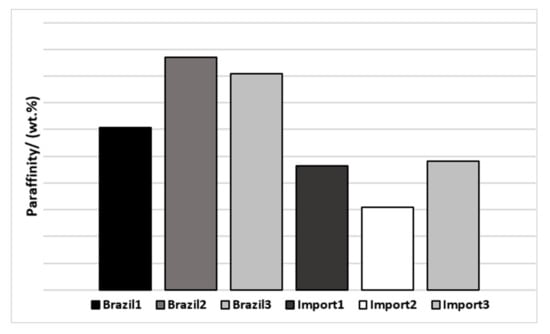
Figure 4.
Paraffinity of naphtha’s samples by origin.
It is possible to verify that the Brazilian naphtha dissipated a paraffinity greater than 78%w, while the imported ones had lower values, less than 72%w, which might be related to heavier compounds presented in the imported naphtha.
Paraffinity directly impacts the conversion yield of paraffins into olefins (ethylene and propylene) that will be used as raw material in second-generation industries (polymers); the higher the paraffinity, the higher the yield of olefins and polymers too. Therefore, predicting the paraffinity of naphtha facilitates the adjustment of process parameters to receive this current, especially the coil outlet temperature (COT), which is the temperature that controls pyrolysis in cracking furnaces [13,33,34].
There is also a factor that makes up the naphtha quality specification list, which is the ratio between the percentage of isoparaffins and n-paraffins (I/P). The lower this ratio, the more ethylene and hydrogen that the naphtha will tend to produce. The higher the I/P ratio, the naphtha will have a greater potential to generate propylene and methane. Comparing these types of naphtha, Brazilian naphtha present a higher ratio than the imported ones, showing that the former would be better used for propylene production [17].
3.2. Density (ASTM D4052)
A typical classification for crude oils is related to the American Petroleum Institute (API) grade, or API°, which measures the relative density of petroleum-derived liquids to water. The API grade is higher when the oil is lighter, which is important information, as it reflects, in general terms, oil classification, as shown in Table 6 [6,31].

Table 6.
Petroleum classification per API° adapted from references [6,31].
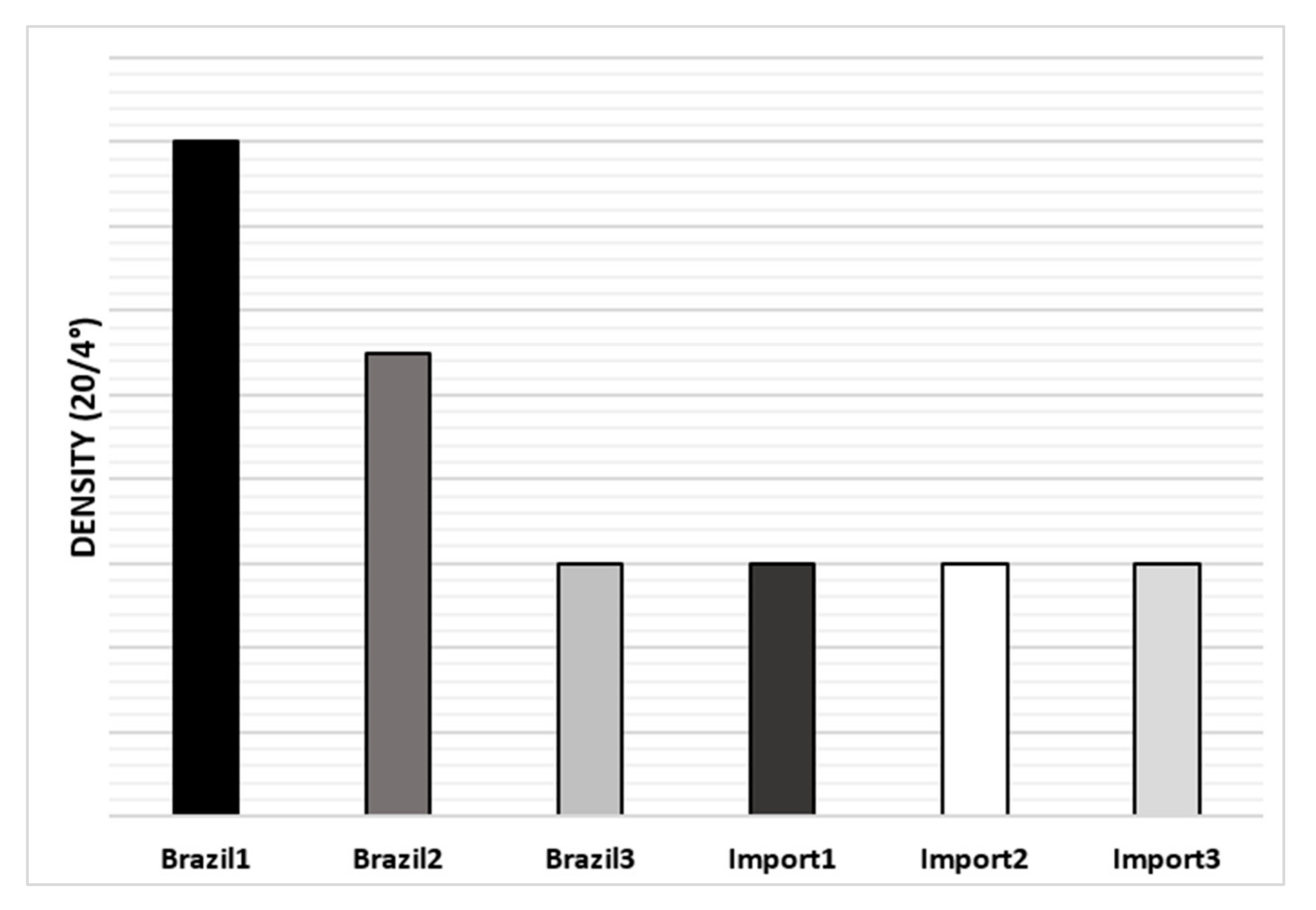
Regarding naphtha density analysis, the one with the highest density is Brazil-1, however, in general, there were no significant differences between them, as can be seen in Figure 5.
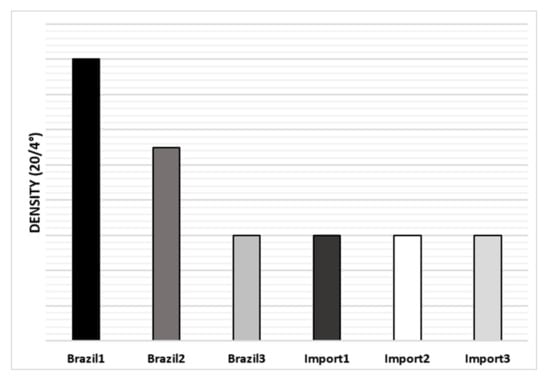
Figure 5.
The average density of naphtha’s samples.
3.3. Sulfur (ASTM D4294)
Sulfur compounds are undesirable because they increase the stability of water in crude oil emulsions, as they increase the polarity of natural surfactants present in the dispersed droplet interfacial layer, cause corrosion, contaminate refining process catalysts, and determine the color and smell of final products [6]. They produce Sox, and when present in final products, affect their environmental quality. Therefore, in addition to paraffinity, the amount of sulfur also determines the quality of naphtha. Most of the extracted oil contains high levels of sulfur, also called sour petroleum ([S] > 2.5%w). On the other hand, crude oil with a lower sulfur content can be called sweet petroleum (<0.5%) or “semi-sweet” or “semi-acid” which are in the intermediate range. Sour petroleum can be related to heavier petroleum, consequently, naphtha from sour petroleum has a higher amount of sulfur. For example, Venezuelan oil with an API° of 11 has a total sulfur content of 2.78%w/w, whereas Yemeni oil with an API° of 42.1 has a total sulfur content of 0.16%w/w [12,32,35].
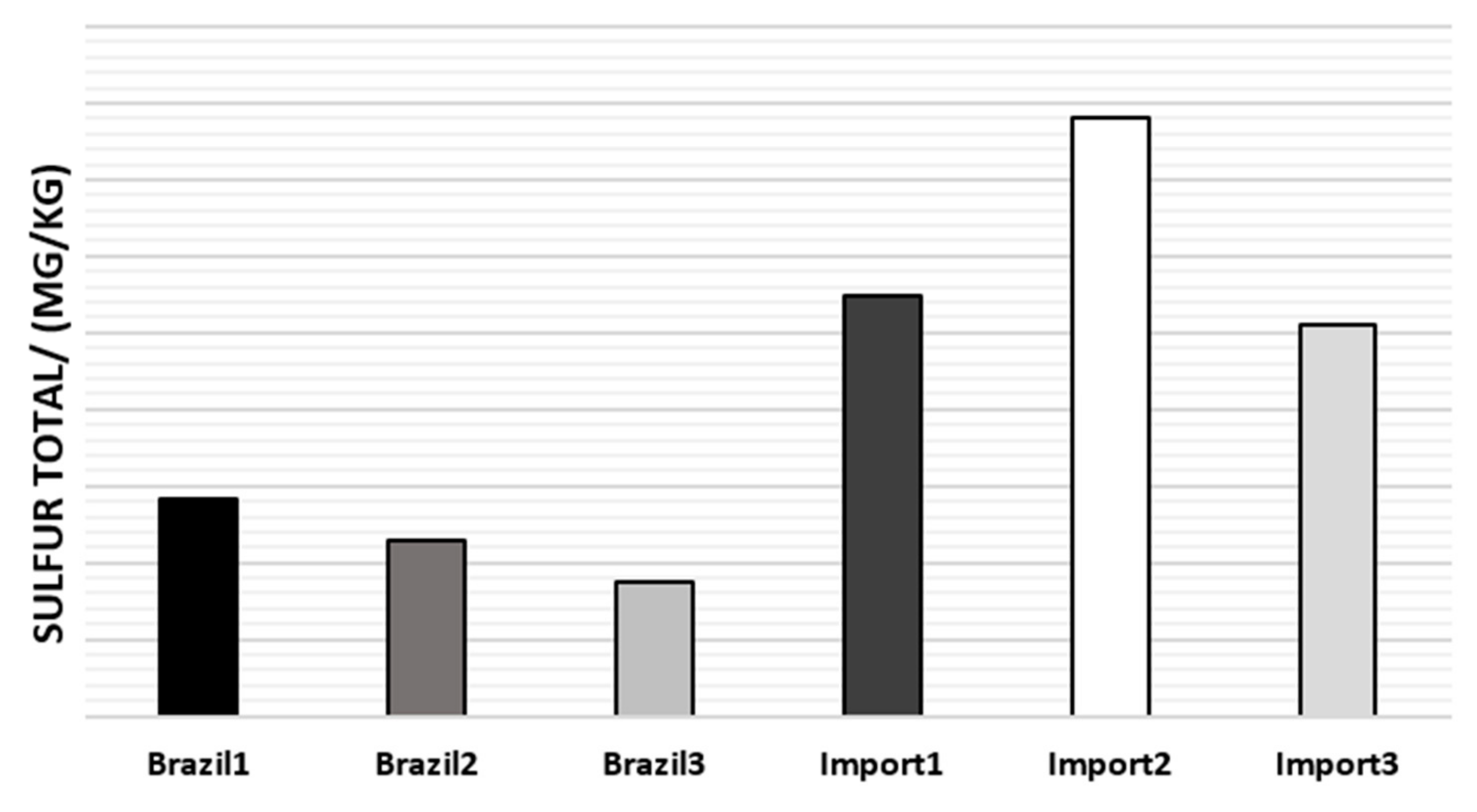
The total sulfur average of naphtha samples is reported in Figure 6.
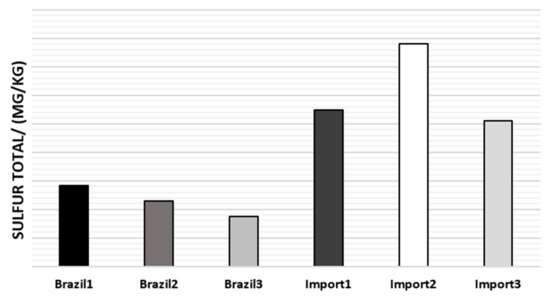
Figure 6.
Average total sulfur content in the naphtha samples.
As can be seen in Figure 6, Brazilian naphtha has a lower sulfur content, (<200 mg/kg), thus being considered low sulfur (semi-sweet) [31], which implies no intensive sweetening of this oily stream, minimizing the steps of downstream processes.
Sulfur, in addition to reducing the life of metallic catalysts, deactivating them corrodes the coils of pyrolysis furnaces which can cost around R$ 10 million for a coil exchange campaign. From the moment that the industry is aware of the type of naphtha that will be received, it manages to get around the situation, directing the high sulfur content to specific pyrolysis ovens or passing the naphtha stream through adsorbents that can help in its abatement [21,36,37].
The pyrolysis furnaces are already prepared for an acceptable amount of sulfur in the naphtha, but when this value becomes much greater than specified, it can be treated, passing the naphtha stream through an adsorption column before entering the ovens, but for this purpose, it is necessary to monitor this quantity in the long term, in order to dimension the column.
3.4. Distillation Curve (ASTM D86)
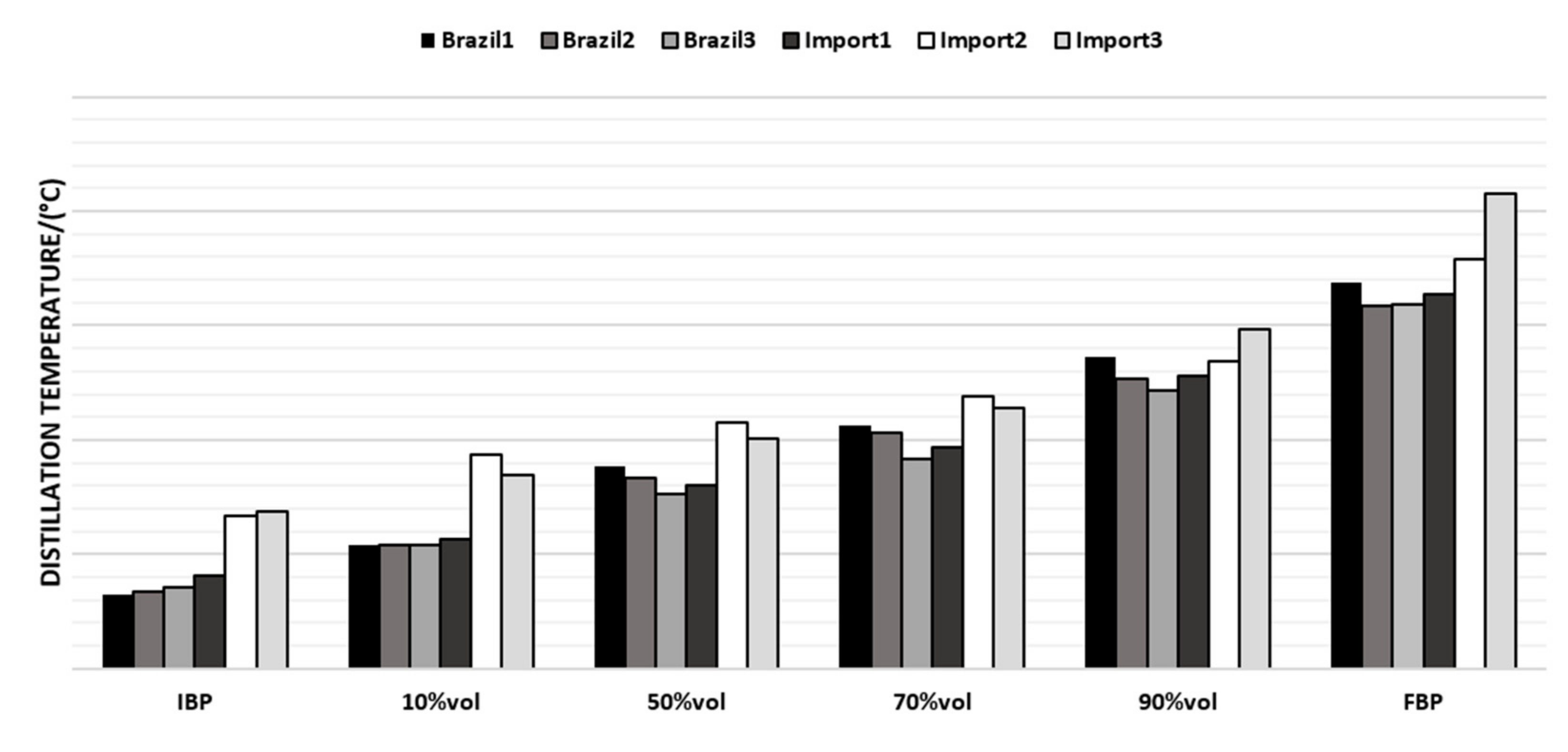
The last parameter evaluated was the distillation curve; the chosen points vary according to the boiling range of the naphtha samples, as reported in Figure 7.
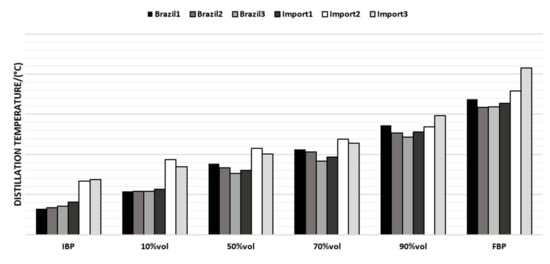
Figure 7.
Average distillation curve values for the naphtha samples.
Regarding the distillation curve, Brazilian naphtha had an initial boiling point (IBP) lower than the imported ones, while the other points had no significant differences. Such an observation may be related to lighter hydrocarbons present in the Brazilian naphtha. The imported naphtha had higher final boiling points and a higher FBP which makes an impact on the presence of heavier molecules, which requires a longer residence time in the coil of the pyrolysis furnace, and can cause coke formation.
In Table 7, it is possible to verify the difference between types of naphtha and their impacts on conversion processes, corroborated with information brought throughout the work.

Table 7.
Comparison between naphtha results.
4. Conclusions
Among the parameters evaluated in the naphtha samples, PIONA composition and total sulfur content showed the greatest variation, confirming the impact of different crude oil origins. Heavier oils have a greater amount of sulfur (sour) and aromatic compounds, while lighter oils have a lower amount of sulfur (sweet) and more paraffinic compounds, which consequently generates lighter fractions. In general, the evaluated Brazilian oils were lighter than the imported ones. High sulfur content in naphtha impairs its conversion processes, so its quality is determined by this parameter and its degree of paraffinity determines whether naphtha will have a better yield when converting into olefins or aromatics. As a suggestion for future work, the use of mathematical modeling to predict the main characteristics of naphtha can be useful to better control and optimize process parameters. Some essential analyses for determining the characteristics of naphtha are laborious and time-consuming; modeling in this process would gain in response time, which would positively impact the conversion process.
Author Contributions
Conceptualization, A.P.S., L.R.-U., W.S.F.-M. and V.O.C.C.; Methodology, A.P.S., J.O.B., R.S.J. and J.L.-P.; Validation, H.M.; Formal analysis, A.P.S., R.S.J. and J.L.-P.; Investigation, V.O.C.C.; Resources, J.O.B., L.R.-U. and W.S.F.-M.; Data curation, J.O.B. and V.O.C.C.; Writing—original draft, A.P.S.; Writing—review & editing, H.M. and J.L.-P.; Supervision, V.O.C.C.; Project administration, A.P.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Brazilian research funding agency CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) grant number 2021.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are grateful to the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq, Brazil), the Coordination for the Improvement of Higher Education Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES, Brazil), and the EAN University and UTRGV for support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Karaba, A.; Zámostny, P.; Belohlav, Z. Substitute Composition of Naphtha Based on Density, SIMDIST, and PIONA for Modeling of Steam Cracking. Chem. Eng. Technol. 2017, 40, 1008–1015. [Google Scholar] [CrossRef]
- BRASKEM. Relatório Anual 2019. Available online: https://www.braskem.com.br/portal/Principal/arquivos/relatorio-anual/2019/BRASKEM_2020_portugues.pdf (accessed on 15 August 2022).
- Da Silva, V.H.; Reboucas, M.V.; Salles, A.R.; Pimentel, M.F.; Pontes, M.J.C.; Pasquini, C. Determination of Naphtha Composition by near Infrared Spectroscopy and Multivariate Regression to Control Steam Cracker Processes. Fuel Process. Technol. 2015, 131, 230–237. [Google Scholar] [CrossRef]
- Speight, J.G. Handbook of Petroleum Product Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2002; Volume 53. [Google Scholar]
- Reza, M.; Jafari, M.; Iranshahi, D. Progress in Catalytic Naphtha Reforming Process: A Review. Appl. Energy 2013, 109, 79–93. [Google Scholar] [CrossRef]
- Speight, J.G. The Chemistry and Technology of Petroleum, 3rd ed.; CRC Press: Boca Raton, FL, USA, 1999; pp. 1–907. [Google Scholar] [CrossRef]
- Podgorski, D.C.; Corilo, Y.E.; Nyadong, L.; Lobodin, V.V.; Bythell, B.J.; Robbins, W.K.; Mckenna, A.M.; Marshall, A.G.; Rodgers, R.P. Heavy Petroleum Composition. 5. Compositional and Structural Continuum of Petroleum Revealed. Energy Fuels 2013, 27, 1268–1276. [Google Scholar] [CrossRef]
- Antos, G.; Aitani, A. Catalytic Naphtha Reforming, 2nd ed.; Marcel Dekker: New York, NY, USA, 2004. [Google Scholar]
- Mei, H.; Yupeng, D.; Zhenlei, W.; Feng, Q. Naphtha Characterization Based on a Molecular Type Homologous Series Vector Representation. J. Tsinghua Univ. 2016, 56, 723–727. [Google Scholar]
- Weber, B.M.; Walsh, P.; Harynuk, J.J. Determination of Hydrocarbon Group-Type of Diesel Fuels by Gas Chromatography with Vacuum Ultraviolet Detection. Anal. Chem. 2016, 88, 5809–5817. [Google Scholar] [CrossRef]
- IHS. Light and Heavy Naphtha: International Market Analysis. Available online: https://cdn.ihs.com/www/pdf/Naptha-Service-Prospectus.pdf (accessed on 15 August 2022).
- Farah, M.A. Petróleo e Seus Derivados; LTC: Rio de Janeiro, Brazil, 2013. [Google Scholar]
- Barazandeh, K.; Dehghani, O.; Hamidi, M.; Aryafard, E.; Rahimpour, M.R. Investigation of coil outlet temperature effect on the performance of naphtha cracking furnace. Chem. Eng. Res. Des. 2015, 94, 307–316. [Google Scholar] [CrossRef]
- Haghighi, S.S.; Rahimpour, M.R.; Raeissi, S.; Dehghani, O. Investigation of Ethylene Production in Naphtha Thermal Cracking Plant in Presence of Steam and Carbon Dioxide. Chem. Eng. J. 2013, 228, 1158–1167. [Google Scholar] [CrossRef]
- Zakria, M.H.B.; Mohd Nawawi, M.G.; Abdul Rahman, M.R. Propylene Yield from Naphtha Pyrolysis Cracking Using Surface Response Analysis. Polyolefins J. 2022, 9, 15–24. [Google Scholar] [CrossRef]
- Kim, J.; Joo, C.; Kim, M.; An, N.; Cho, H.; Moon, I.; Kim, J. Multi-Objective Robust Optimization of Profit for a Naphtha Cracking Furnace Considering Uncertainties in the Feed Composition. Expert Syst. Appl. 2023, 216, 119464. [Google Scholar] [CrossRef]
- Leite, L.F. Olefinas Leves: Tecnologia, Mercado e Aspectos Econômicos; Interciência: Rio de Janeiro, Brazil, 2013; Volume 4, pp. 21–59. [Google Scholar]
- Hu, S.; Towler, G.; Zhu, X.X. Combine Molecular Modeling with Optimization to Stretch Refinery Operation. Ind. Eng. Chem. Res. 2002, 41, 825–841. [Google Scholar] [CrossRef]
- Rodríguez, M.A.; Ancheyta, J. Detailed Description of Kinetic and Reactor Modeling for Naphtha Catalytic Reforming. Fuel 2011, 90, 3492–3508. [Google Scholar] [CrossRef]
- Sadighi, S.; Mohaddecy, R.S. Predictive Modeling for an Industrial Naphtha Reforming Plant Using Artificial Neural Network with Recurrent Layers. Int. J. Technol. 2013, 4, 102. [Google Scholar] [CrossRef]
- Pulido, J.; Martinez Arias, E.; Wolf Maciel, M.; Maciel Filho, R. Heat Integrated Reactive Distillation Column (r-hidic): Implementing a New Technology Distillation. Chem. Eng. Trans. 2011, 24, 1303–1308. [Google Scholar] [CrossRef]
- Van Geem, K.M.; Hudebine, D.; Franc, M.; Verstraete, J.J.; Marin, G.B. Molecular Reconstruction of Naphtha Steam Cracking Feedstocks Based on Commercial Indices. Comput. Chem. Eng. 2007, 31, 1020–1034. [Google Scholar] [CrossRef]
- ASTM D5134; Standard Test Method for Detailed Analysis of Petroleum Naphthas through N-Nonane by Capillary Gas Chromatography. ASTM International: West Conshohocken, PA, USA, 2014. [CrossRef]
- Kostecki, P.; Morrison, R.; Dragun, J. Hydrocarbons. In Encyclopedia of Soils in the Environment; No. 1999; Elsevier: Amsterdam, The Netherland, 2005. [Google Scholar]
- ASTM D4052; Standard Test Method for Density, Relative Density, and API Gravity of Liquids by Digital Density Meter. ASTM International: West Conshohocken, PA, USA, 2018. [CrossRef]
- Gomez-Prado, J.; Zhang, N.; Theodoropoulos, C. Characterisation of Heavy Petroleum Fractions Using Modified Molecular-Type Homologous Series (MTHS) Representation. Energy 2008, 33, 974–987. [Google Scholar] [CrossRef]
- Mei, H.; Cheng, H.; Wang, Z.; Li, J. Molecular Characterization of Petroleum Fractions Using State Space Representation and Its Application for Predicting Naphtha Pyrolysis Product Distributions. Chem. Eng. Sci. 2017, 164, 81–89. [Google Scholar] [CrossRef]
- Meirelles, L.B.; da Silva, S.M.C.; Rajagopal, K. Caracterização da Nafta Petroquímica para a produção de Aromáticos. In Anais do XX Congresso Brasileiro de Engenharia Química - COBEQ 2014 [= Blucher Chemical Engineering Proceedings, v.1, n.2]; Blucher: São Paulo, Brazil, 2015; pp. 13879–13885. [Google Scholar] [CrossRef]
- ASTM D4294; Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive. ASTM International: West Conshohocken, PA, USA, 2012. [CrossRef]
- ASTM D86; Standard Test Method for Distillation of Petroleum Products and Liquid Fuels at Atmospheric Pressure. ASTM International: West Conshohocken, PA, USA, 2018. [CrossRef]
- Neto, A.A.D.N.; Gurgel, A.; Refino de Petróleo e Petroquímica. UFRN 2014, 3–17. Available online: http://www.nupeg.ufrn.br/downloads/deq0370/curso_refino_ufrn-final_1.pdf (accessed on 15 November 2022).
- Szklo, A.S.; Uller, V.C.; Bonfá, M.H. Fundamentos do Refino do Petróleo—Tecnologia e Economia; Interciência: Rio de Janeiro, Brazil, 2012. [Google Scholar]
- Ziarifar, E.; Fakhrhoseini, S.M.; Ghiassi, H. Optimization of Coil Outlet Temperature for Producing Maximum Products in an Olefin Furnace. Pet. Sci. Technol. 2013, 31, 596–602. [Google Scholar] [CrossRef]
- Ziarifar, E.; Fakhrhoseini, S.M.; Ghiassi, H.; Masoumi, M.E.; Sadrameli, S.M.; Towfighi, J.; Niaei, A.; Kostecki, P.; Morrison, R.; Dragun, J. Simulation, Optimization and Control of a Thermal Cracking Furnace. Energy 2006, 31, 516–527. [Google Scholar] [CrossRef]
- Thomas, J.E. Fundamentos de Engenharia de Petróleo, Segunda; Editora Interciência: Rio de Janeiro, Brazil, 2004. [Google Scholar]
- Filho, R.S., Jr. Corrosão do aço Carbono Devido a Elevação da Concentração de CS 2 (Dissulfeto de Carbono). Intercorr 2016, 2, 1–13. Available online: https://abraco.org.br/src/uploads/intercorr/2016/INTERCORR2016_158.pdf (accessed on 15 February 2023).
- Salem, A.B.S.H.; Hamid, H.S. Removal of Sulfur Compounds from Naphtha Solutions Using Solid Adsorbents. Chem. Eng. Technol. 1997, 20, 342–347. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).