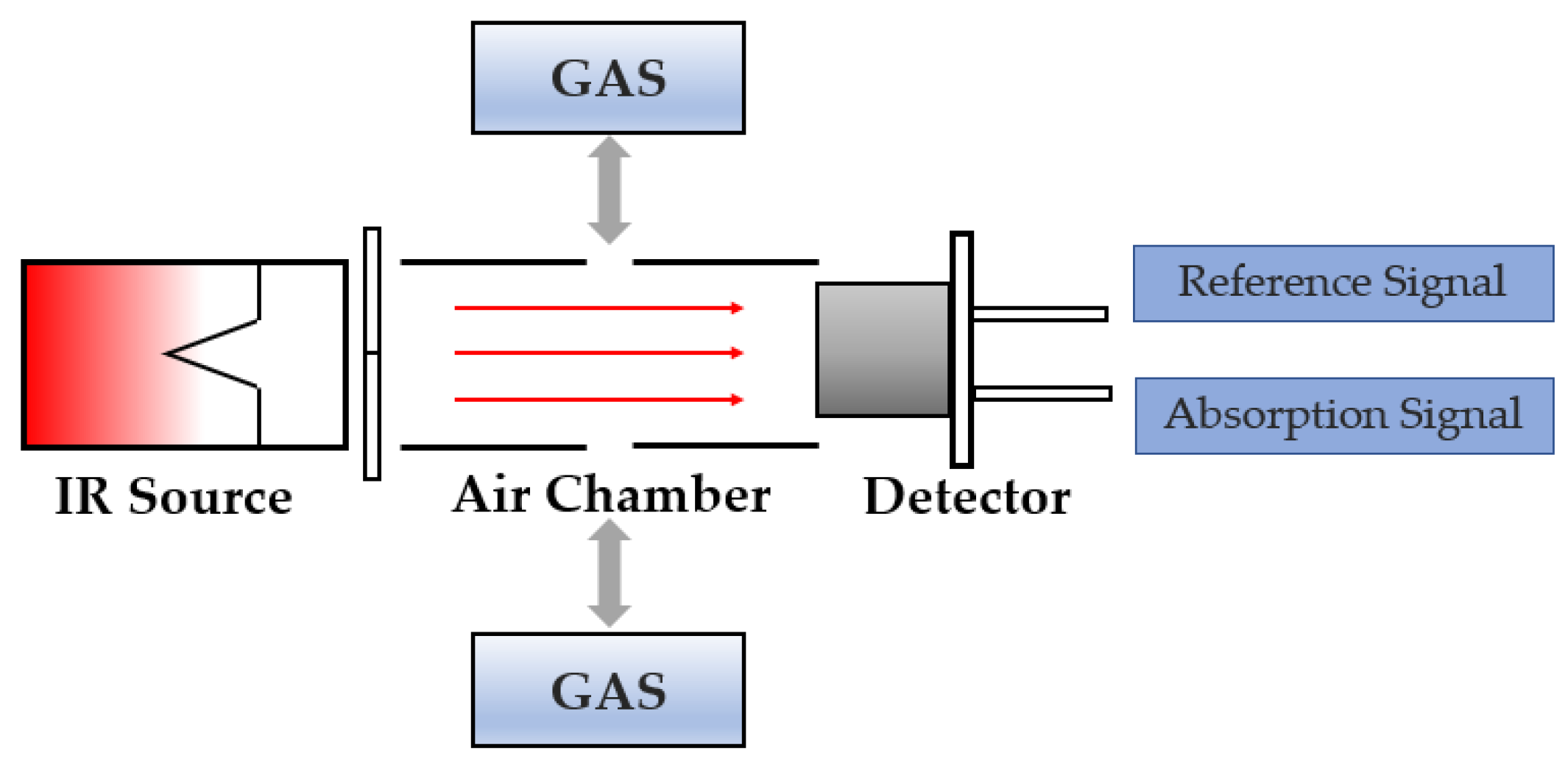
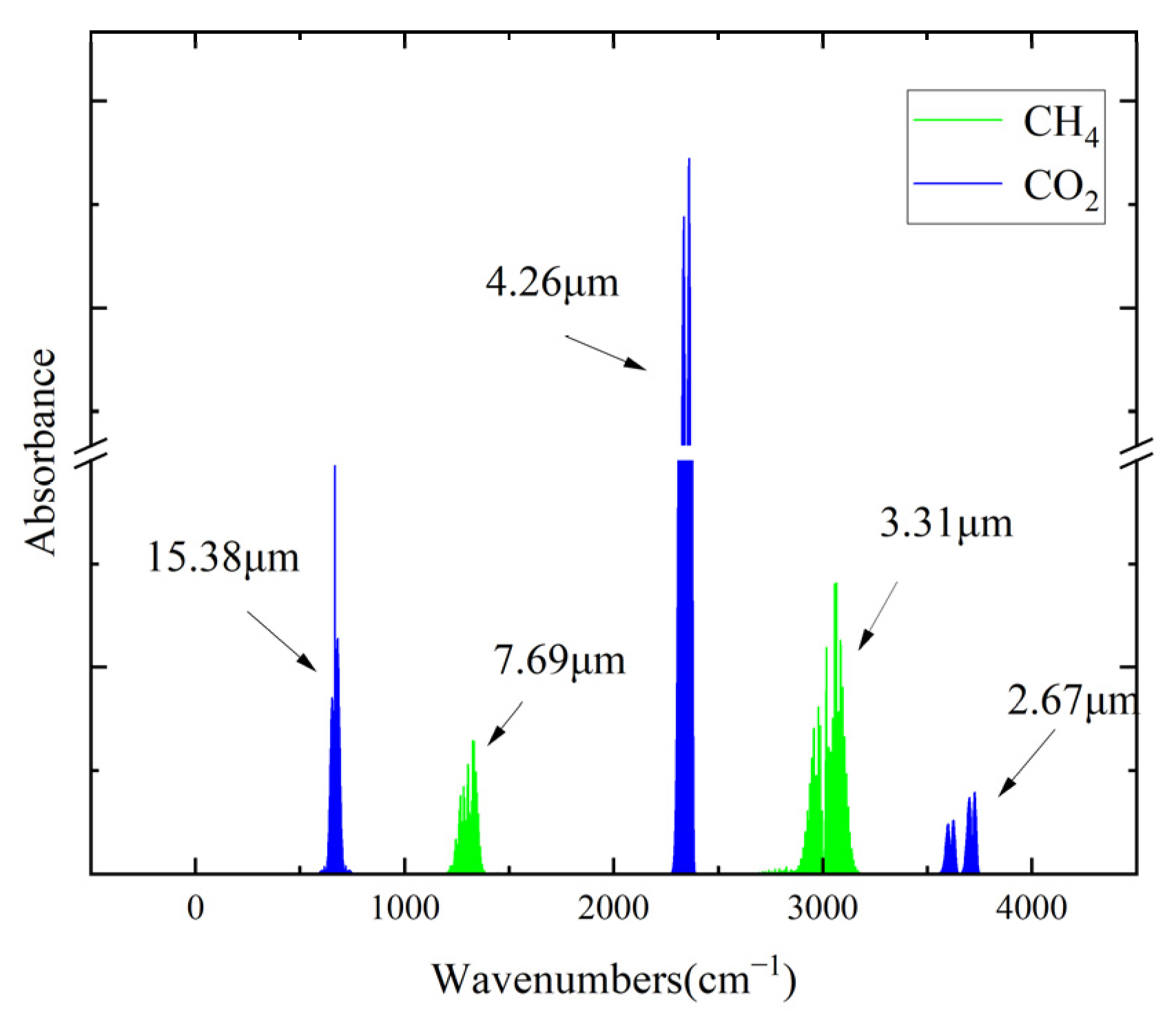
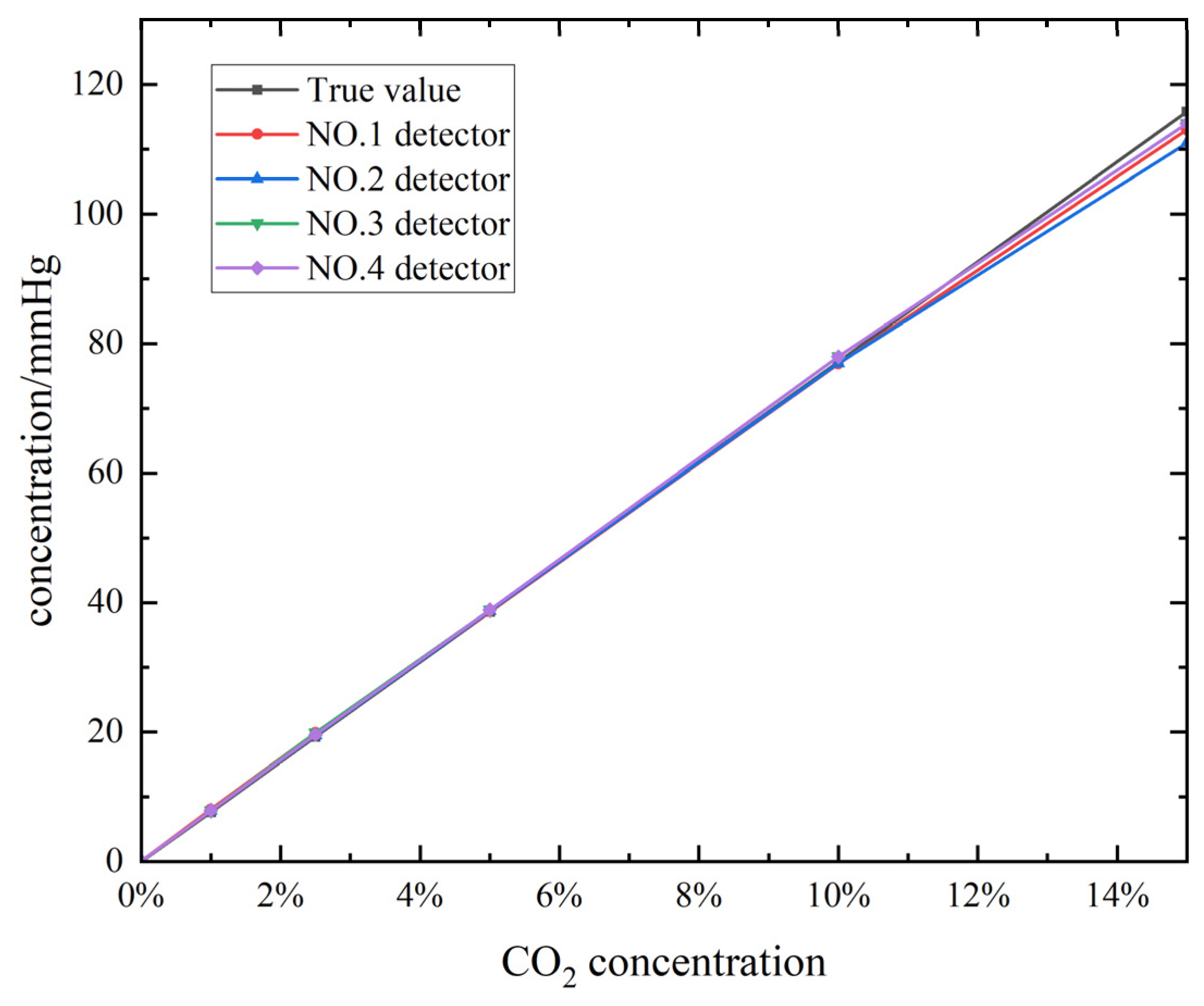
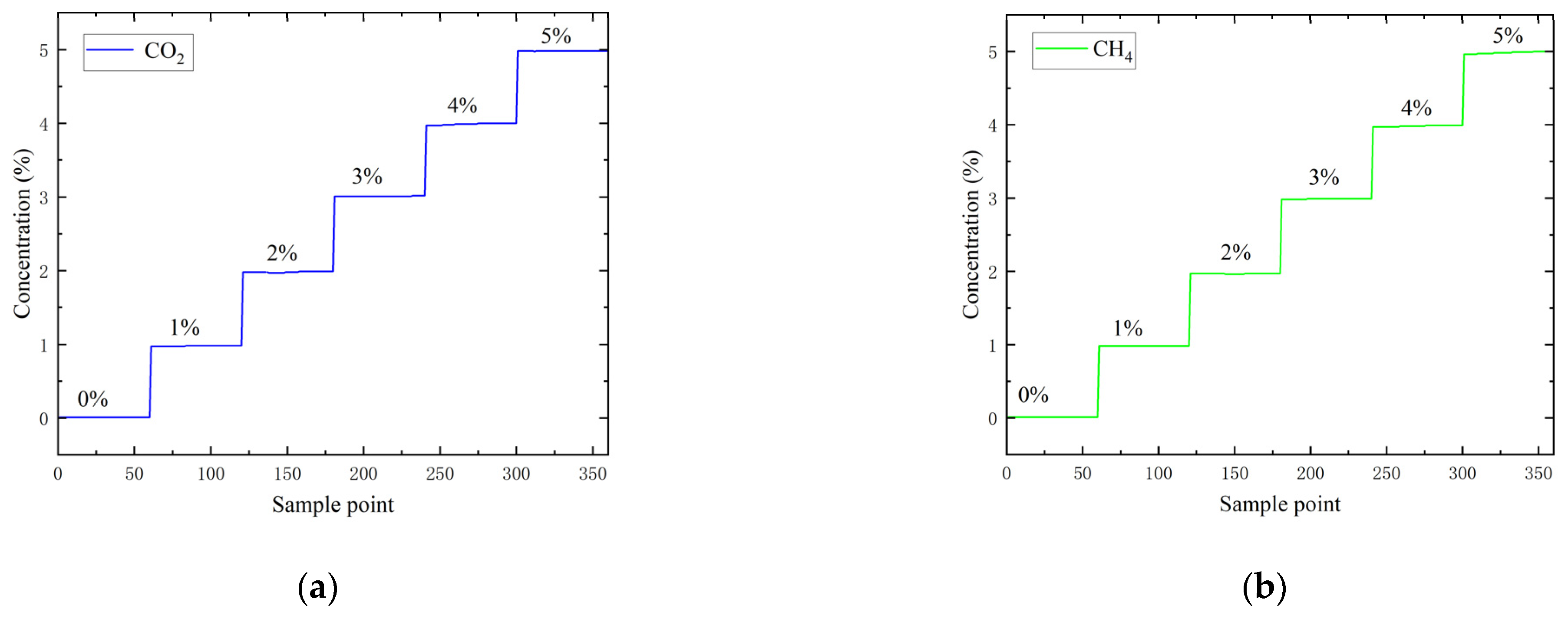
1. Introduction
Lithium-ion batteries (LIBs) have the advantages of high energy density and a long cycle life and are widely used in electric vehicles, hybrid electric vehicles, and power grids [
1,
2]. At present, the cathode materials for automotive lithium-ion batteries are mainly lithium iron phosphate (LiFePO4, LFP) and lithium cobalt manganate (LiCoxNiyMn1-x-yO2, NCM). Generally, lithium-ion batteries exhibit a certain thermal stability within their operating temperature range. In particular, LFP batteries are considered to be relatively safe cathode materials due to the high strength of the P-O bond in the phosphate radical of the cathode material. This bond is not easily broken at high temperatures [
3,
4].
However, in actual use, LIBs still present various forms and causes of failures. The most common on-site fault in the battery system of electric vehicles is overcharging, which is usually caused by failure of the battery management system (BMS) or charger and the inconsistency between batteries [
1,
5]. During overcharging, the positive electrode of a lithium-ion battery becomes excessively de-lithiated, while the negative electrode becomes excessively lithiated, resulting in a series of side reactions. [
6,
7]. Meanwhile, the insufficient thermal stability of the cathode material within the lithium-ion battery induces internal gas production, in addition to the TR risks caused by various gas production processes caused by the internal electricity and heat of the battery. According to the results of previous studies, carbon dioxide (CO
2), methane (CH
4), and other gases are generated in the process of lithium-ion battery failure [
8,
9].
The excessive de-lithiation of lithium leads to the collapse of the cathode structure, accompanied by the generation of heat and the release of oxygen. The release of oxygen accelerates the oxidation and decomposition reactions of the electrolyte inside the lithium-ion battery, resulting in the rapid formation of a large amount of gas inside the battery. Since a lithium-ion battery is a hermetically sealed system, the internal pressure increases sharply in a short time, resulting in a more serious thermal runaway phenomenon, despite the good thermal stability of LFP. The heat released by these side reactions is the main factor that causes a lithium-ion battery to undergo a thermal runaway reaction, leading to fires and explosions [
4]. Furthermore, this process usually involves the premature release of characteristic gases. Therefore, investigation of the gas production process is important for understanding the TR mechanisms of lithium-ion batteries and improving their safety performance.
Zhu et al. [
10] conducted real-time measurements of battery voltage, current, and surface temperature during the overcharging process of a lithium-ion battery with a cathode material of NCM622 and a capacity of 30 Ah. The authors systematically studied the characteristics of an overcharge-induced battery thermal runaway and found that overcharge tests with high capacity rates (C-rates) are more dangerous than those with low C-rates. The peak value of the voltage curve increased linearly with the C-rates, but the temperature increase rate and the maximum surface temperature did not. The authors also found that a sharp drop in voltage always precedes the rapid rise in temperature before thermal runaway; thus, they proposed a safety monitoring signal based on voltage drops to warn users of the imminent risk of thermal runaway for batteries.
Gas production in normally operating lithium-ion batteries is primarily caused by electrolyte decomposition. Kong et al. [
11,
12] found that commercial batteries composed of lithium cobalt oxide (LCO), lithium manganate (LMO), and LFP produced the same gas components, which contained oxides such as CO
2 and CO, hydrocarbons such as C
2H
4 and CH
4, and gases such as H
2 [
13]. The production of these gases was related to the reaction of electrolytes.
Generally, when the temperature reaches high temperatures of 80~120 °C, the negative solid electrolyte interface (SEI) film begins to decompose, and the gas production of the battery becomes significant. When the temperature reaches the melting temperature of the diaphragm (130~170 °C) [
11,
14], the battery will have a large-scale internal short circuit, and the heat will be released instantaneously, resulting in the release of active oxygen from the thermal decomposition of the positive electrode of cobalt-based batteries (such as NCM or NCA batteries), as well as the vaporization and redox reaction of the electrolyte.
The most advanced battery monitoring equipment in automotive battery packs uses Battery Management Systems (BMSs). BMSs are mainly used in electric vehicles for the intelligent management and maintenance of the battery pack and for monitoring the safety of the battery’s status. The main functions of such systems are to ensure that each battery in the battery pack reaches a balanced and consistent state; accurately estimate the state of charge (SOC) of the power battery pack, that is, the remaining power of the battery; ensure that the SOC is maintained within a reasonable range; prevent damage to the battery due to overcharging; in the process of charging and discharging the battery, in real time, collect the terminal voltage and temperature, charging and discharging current, and total voltage of each battery in the battery pack; prevent overcharging or overdischarging of the battery; and simultaneously select problematic batteries to maintain the reliability and efficiency of the entire battery pack operation [
15,
16]. The BMS monitors the working status of the battery pack based on measurements from the temperature sensors, battery voltage, and current; estimates the SOC of the battery pack through algorithms; and maintains the reliability and efficiency of the entire battery pack operation.
New regulations such as GB 38031-2020 and the Electrical Vehicle Safety—Global Technical Regulation (EVS-GTR) dictate that passengers need to be alerted at least five minutes before serious incidents occur [
17,
18]. In order to enhance the safety of batteries and satisfy EVS-GTR20 and GB 38031-2020, gas sensors are added to the battery pack based on BMS to detect the thermal runaway process of automotive batteries, thereby improving the safety of the batteries. BMS mainly detects changes in battery temperature, current and voltage, and impedance. Currently, research on the gas produced during the TR process of automotive batteries has mainly collected and analyzed the gas produced after the TR process. There is less research on monitoring gas production during the TR process. Currently, early thermal runaway warning systems have obvious limitations. (1) The vehicle automotive battery pack is composed of automotive batteries, and when a single battery has a thermal runaway phenomenon, the change is not significant. When BMS detects that the battery has a fault, the battery has often already begun an irreversible thermal runaway process. (2) The current means of improving the detection efficiency and accuracy of BMS systems is achieved through algorithm optimization, which cannot monitor the battery failure process in more dimensions. (3) The battery may fail even though the voltage does not change significantly, resulting in a failure to achieve a timely warning. In the early warnings of current BMS systems, individual batteries in the battery pack already experience irreversible thermal runaway before the alarm, meaning that such systems cannot meet the necessary safety requirements. For battery TR, the main detection gases that are produced include CO
2 and various combustible gases. The most commonly used gas sensors are divided into four categories, electrochemical sensors [
9,
14], semiconductor sensors [
19,
20,
21], Non-Dispersive Infrared Spectroscopy (NDIR) gas sensors [
22], and chemical sensors [
23].
Cai et al. [
24]. proposed a method for the early detection of the battery TR process by detecting CO
2 concentrations and demonstrated through COMSOL simulation that the gas sensor detection method can detect the TR process earlier. Koch et al. [
25] designed a gas sensor system based on evaluating the impact of the TR process and proposed that designing a combination of multiple sensors for different batteries could eliminate the drawbacks of a single sensor and improve the reliability of the entire system. Christiane Essl et al. [
9] tested several commercial gas sensors for four battery fault conditions. The research found that it is possible to use gas sensors for battery fault detection. The report mainly used metal oxide (MOx) gas sensors for detection, as such gas sensors can be used to detect the first venting event before TR in overheating and overcharging experiments. Based on this information, Ze Wang [
26] described the metal oxide gas sensors used for battery thermal runaway.
Current research shows that applying gas sensors to detect the TR process in automotive batteries is more effective than detecting battery voltages and temperatures. Previous research mainly used MOx gas sensors and other types of gas sensors. NDIR gas sensors have the characteristics of low cost, high accuracy, and good stability—offering more accuracy than MOx gas sensors. NDIR gas sensors also have a longer working life and more stable performance than MOX gas sensors, as well as better selectivity for detecting gases. NDIR gas sensors are used in this study in the following ways: (1) to verify the feasibility of NDIR gas sensor TR process detection and (2) to make up for the shortcomings of other gas sensors by applying different sensor principles. We also plan to combine multiple sensors in subsequent research to form a commercially available sensor system for TR process monitoring. In this work, we developed an NDIR gas sensor based on a pyroelectric sensor with high accuracy and temperature stability. The NDIR gas sensor used in this work is based on our own previously developed high-performance pyroelectric infrared detector. This work applies the developed NDIR gas sensing system to monitor and analyze the gas production of lithium-ion batteries in real time. The gas sensing system can detect CO2 and CH4 during an overcharging test on LFP batteries and provide an early warning for the battery TR process.
3. Experiments
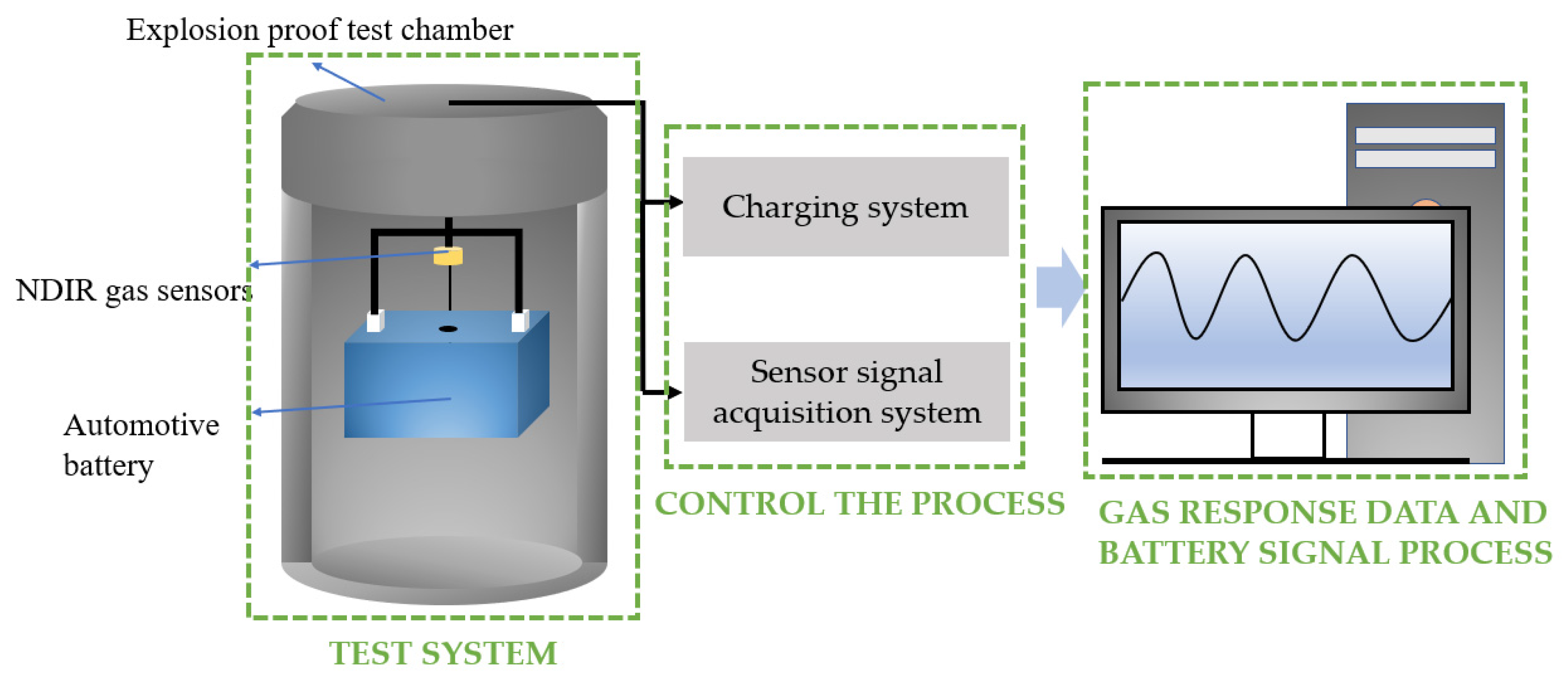
In this experiment, we designed an explosion-proof testing room. The system was installed in an explosion-proof test room due to the possibility of heating and even explosion during the battery overcharge test. Meanwhile, during the failure process of the battery, various types of electrolyte vapors were generated, so the experiment was conducted in a closed environment. Firstly, the chamber was vacuumed and filled with nitrogen to a normal pressure in order to eliminate interference from other gases. The experimental system diagram is shown in
Figure 7. In this study, 50 Ah commercial LIBs with lithium iron phosphate cathodes were used for the overcharging experiment. The main performance parameters of the battery are shown in
Table 1. The gas detector was fixed 12 cm above the battery explosion vent, as shown in
Figure 7. The CO
2 sensor range was 0~20%, and the CH
4 sensor range was 0~5%.
After setting up the test system, the experimental steps taken were as follows. (1) First, the lithium-ion automotive battery was left to stand for 10 min to stabilize the voltage and current and preheat the gas sensor. (2) Then, we used a 50 A constant current to overcharge the automotive battery. When the voltage increased rapidly, a thermal runaway was considered to occur; then, we stopped the overcharging and moved to the static step. (3) The static battery was used to continuously record the voltage change, the gas sensor collected the gas concentration change, and the data acquisition equipment collected the temperature fluctuation at each thermocouple monitoring point on the surface of the automotive battery.
For this experiment, six temperature monitoring points were established by attaching thermocouples. These monitoring points are indicated in
Figure 8, and the position settings are shown in
Table 2. By comparing and analyzing the temperature changes of these six temperature detection points, the corresponding temperature trends of lithium-ion power batteries with overcharging-induced thermal runaway behavior were relatively accurately obtained, allowing us to better analyze the internal mechanisms of battery overcharging thermal runaways.
4. Results and Discussion
In the first experiment, the preliminary feasibility of using NDIR gas sensors to detect gas production in automotive battery failures was verified without opening the automotive battery explosion vent.
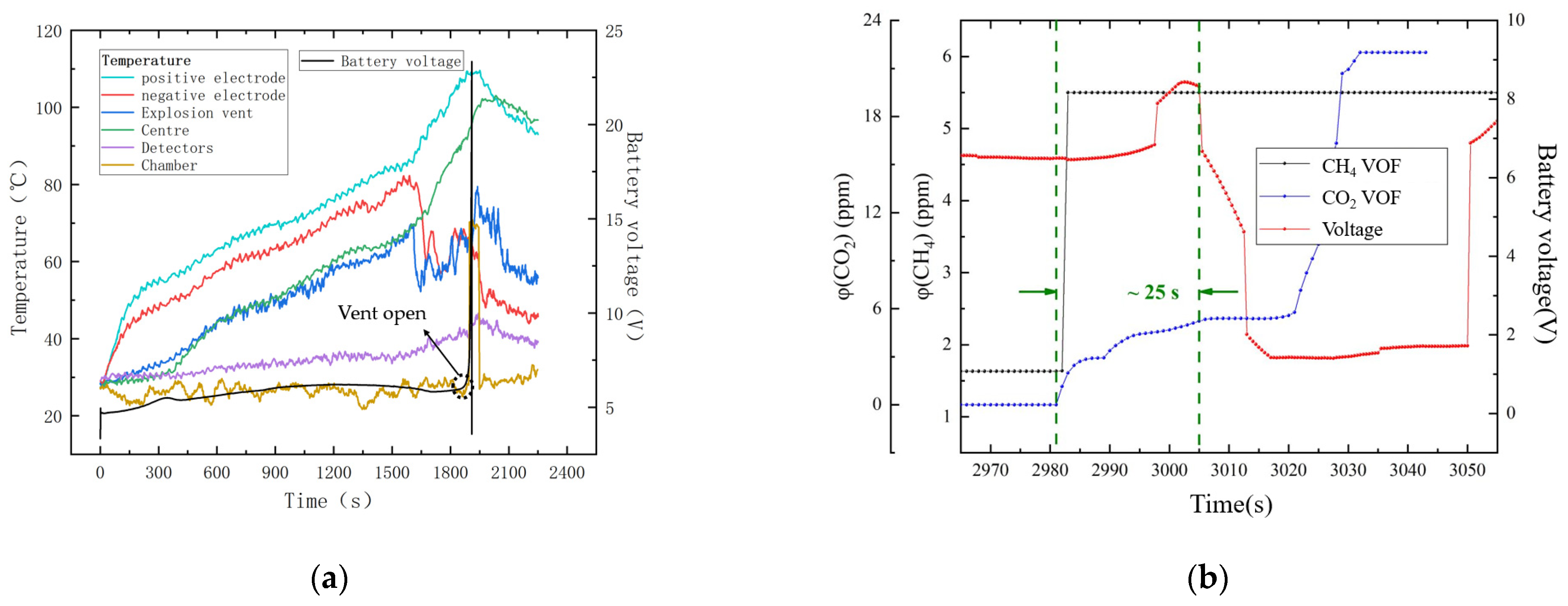
Figure 9a shows the temperature variation at different points during the overcharging experiment. A significant amount of heat was generated during the battery TR, mainly concentrated in the positive electrode and the center of the battery. In
Figure 9a, it can be seen that, as the overcharging reaction of the battery progressed, the overall temperature of the battery gradually increased. At about 1600 s, there was a significant decrease in the temperature of the battery’s negative electrode and the temperature of the battery vent, indicating that a large amount of gas had accumulated inside the battery, reaching the critical value for the vent to open. With the opening of the vent, the release of a large amount of gas carried away some of the heat, causing a decrease in the temperature of the vent. The position of the sensor and the ambient temperature change in the explosion-proof test chamber were within the correction range of the NDIR gas sensor algorithm (less than 80 °C), and the influence of temperature on the concentration change in the detector could be excluded.
Figure 9b shows the changes in gas concentration and battery voltage. In the battery failure experiment, a dramatic change in the positive voltage indicated that the battery had begun to fail. A sharp drop in voltage meant that the explosion vent had opened. As shown in
Figure 9b, the battery voltage dropped sharply after a sharp increase in CO
2 and CH
4 concentrations for 25 s, demonstrating that the developed NDIR gas sensor enabled the early detection of battery failure.
We observed that a large amount of gas accumulated in the battery when the explosion vent of the automotive battery was not opened. When the battery voltage increased sharply, the battery experienced a thermal runaway effect, and the explosion vent was blown away by a large amount of gas. At this time, the sensors were positioned facing the explosion vent, resulting in a sharp increase in gas concentration that quickly exceeded the sensor’s range. As shown in
Figure 9, CH
4 gas reached its full scale after the explosion vent was opened, and the large amount of CO
2 generated as a reaction by-product was oxidized.
Although the application of NDIR gas sensors in the battery TR process was preliminarily verified in the first experiment, the timing of gas signal testing by the sensors was not much earlier than that of the voltage testing in the experiment. At the same time, as a large amount of gas was generated beyond the range of the sensor, it became impossible to accurately measure the ratio of gas production and gas composition during the TR process. After the battery overcharging experiment, the gas sensor that was stimulated by high-temperature gas malfunctioned and could not function properly.
In the second experiment, to comprehensively detect the gas production during automotive battery failure, the vent of the automotive battery was opened, and the experiment was repeated. After opening the vent, the gas generated by the battery during the TR process did not accumulate in the battery, preventing the generation of a large amount of gas from exceeding the range of the gas sensor. At the same time, the gas concentrations generated at different stages of the TR process were also more clearly detected. However, the temperature and voltage changes caused by the overcharging of the lithium-ion automotive battery were uncontrollable, as shown in
Figure 10.
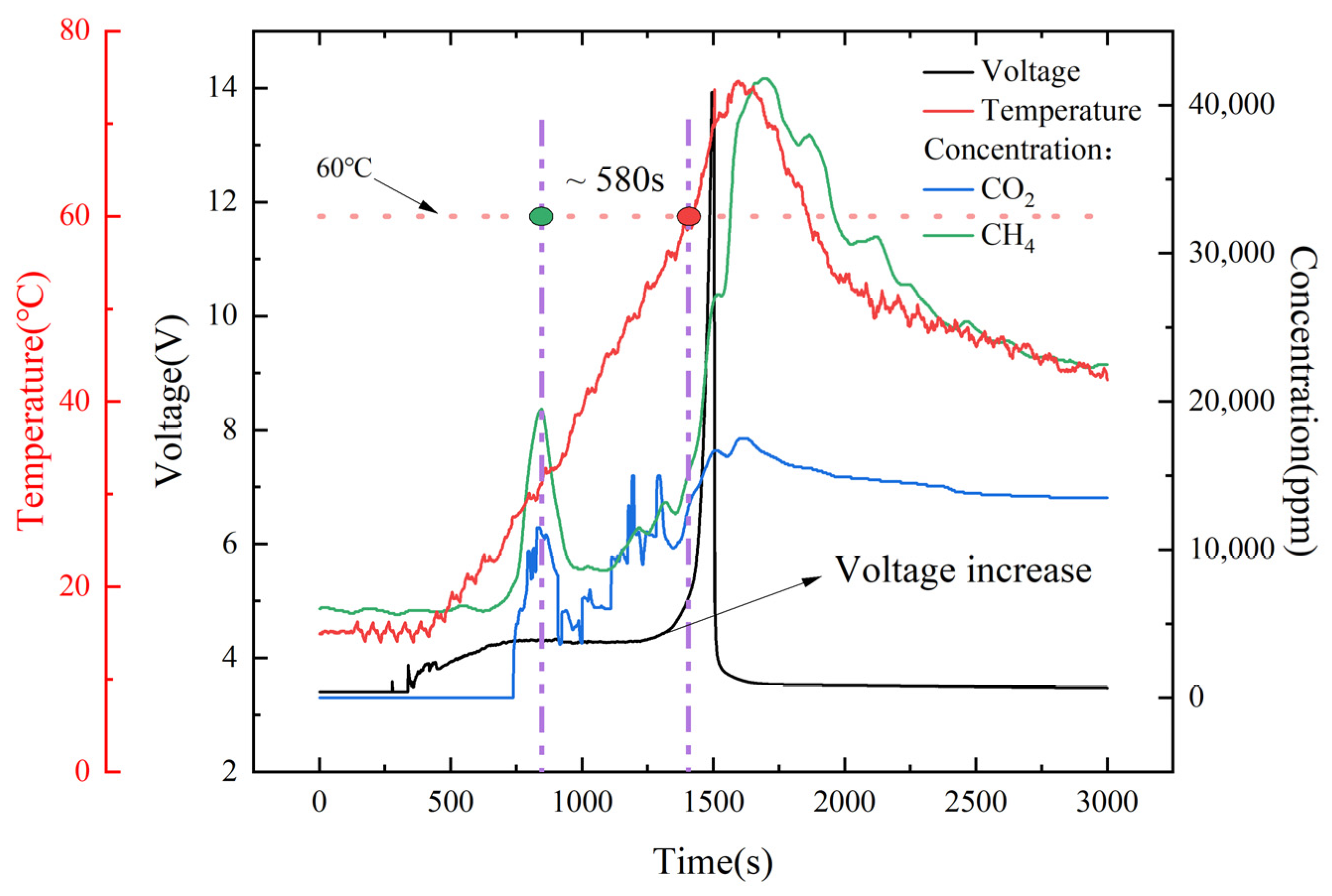
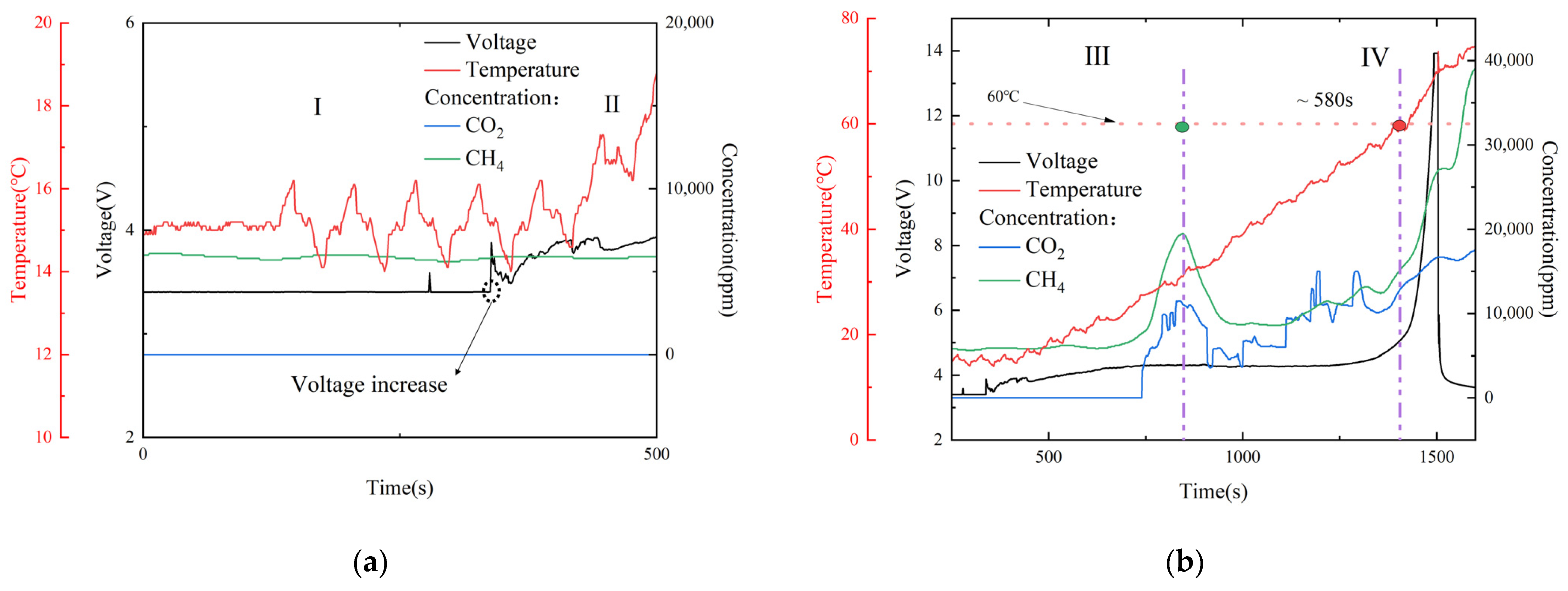
According to the temperature and voltage change characteristics, the voltage curve for the whole process of overcharging and runaway can be divided into four stages.
In the first stage (I), the lithium-ion automotive battery’s initial charging voltage was 3.4 V, and after 338 s of continuous overcharging to the first voltage inflection point, the battery temperature slowly started to rise, with there being no obvious change in the gas sensor, as shown in
Figure 11a.
In the second stage (II), 400 s after the first voltage inflection point, the automotive battery voltage gradually increased to 4.3 V, and the CO
2 and CH
4 gas sensors detected the generation of gas and produced a peak value at the same time, which demonstrated that the automotive battery started to fail at this time, as shown in
Figure 11b. The temperature then increased from 15 to 31.8 °C, with a temperature rise rate of 2.52 °C/min.
In the third stage (III), the automotive battery voltage appeared as a voltage plateau period. The voltage plateau represents the external manifestation of the coexistence of multiple phase states in the internal chemical reaction of the lithium-ion automotive battery, indicating that, at this stage, the lithium ion in the lithium salt was converted to a new relatively stable stage and new substances were beginning to form. The temperature gradually increased from 31.8 to 60 °C, and the temperature increase rate slightly rose to 2.91 °C/min. When the temperature reached 60 °C, the battery management system’s alarm temperature was reached. The concentration of gas produced by the battery also gradually increased. The time from the first peak value collected by the gas sensor to the battery alarm was about 580 s faster than expected, which has a certain significance for battery safety warnings.
In the fourth stage (IV), the temperature of the lithium-ion automotive battery continued to rise, further accelerating the decomposition and exothermic reaction of the positive and negative active materials and the electrolyte. The generation of a large amount of gas in the battery led to a rapid increase in internal resistance, and the voltage increased rapidly to 14 V, causing irreversible thermal loss of control in the automotive battery. The gas production concentration then reached the maximum value.
After the battery experienced thermal runaway and stopped overcharging, the internal energy of the automotive battery began to dissipate, causing the temperature to drop. Additionally, the gas concentration also began to decrease, and the reaction gradually ceased.
In order to meet the new regulations of GB 38031-2020 and EVS-GTR and warn passengers at least five minutes before a serious accident occurs, in this experiment, after opening the venting port, when the gas sensor detected a signal, that signal was compared to the battery temperature reaching the BMS system alarm temperature of 60 °C, which was increased by ~580 s. Compared to Christiane Essl’s research results [
9], this experiment design significantly advanced the warning of battery TR process.
However, the NDIR gas sensor still has some issues: (1) Hydrogen gas is also produced in large quantities during the battery failure process, but NDIR gas sensors cannot selectively detect hydrogen gas because hydrogen gas does not have an infrared characteristic peak. (2) NDIR gas sensors are more expensive than MOx gas sensors. (3) Since NDIR gas sensors are optical sensors, they require a light source and an optical path, creating the demand for a larger volume. To solve these three problems, we could combine multiple sensors to achieve the high precision, low cost, simultaneous detection of multiple gases to improve the reliability of monitoring in future research on monitoring the battery failure process. Going forward, gas sensors could be integrated into the automotive battery to reduce volume. At the same time, the structure of NDIR gas sensors could be further optimized to reduce cost and size.