Livestock Agriculture Greenhouse Gases for Electricity Production: Recent Developments and Future Perspectives
Abstract
:1. Introduction
- (i)
- To investigate the technologies employed to capture greenhouse gases from livestock agriculture.
- (ii)
- To investigate the technological innovations employed to transform greenhouse gases from livestock agriculture into energy.
- (iii)
- To identify future directions and emerging solutions that transform greenhouse gases from livestock agriculture.
2. Materials and Methods
2.1. Stage I: Identifying the Research Question
What innovations are currently being employed to capture and use greenhouse gases produced within livestock farms for energy production, and what future directions are expected?
2.2. Stage II: Development of a Search Strategy
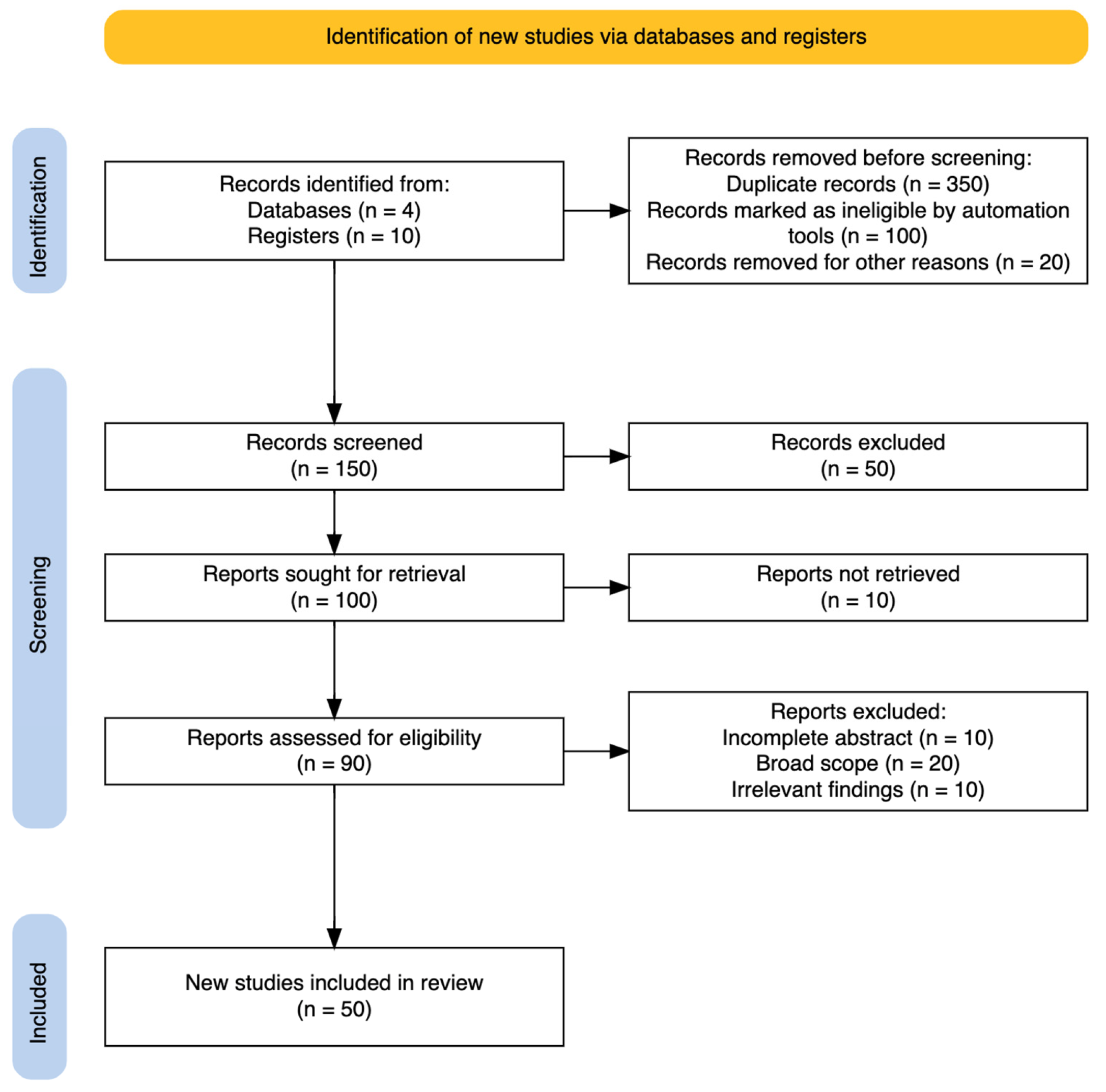
2.3. Stage III: Study Selection
2.4. Stage IV: Reporting the Findings
3. Findings
3.1. Advances in Measuring GHG Emissions from Livestock Agriculture
3.1.1. Animal-Based Techniques
Direct Gas Exchange Measurement Techniques
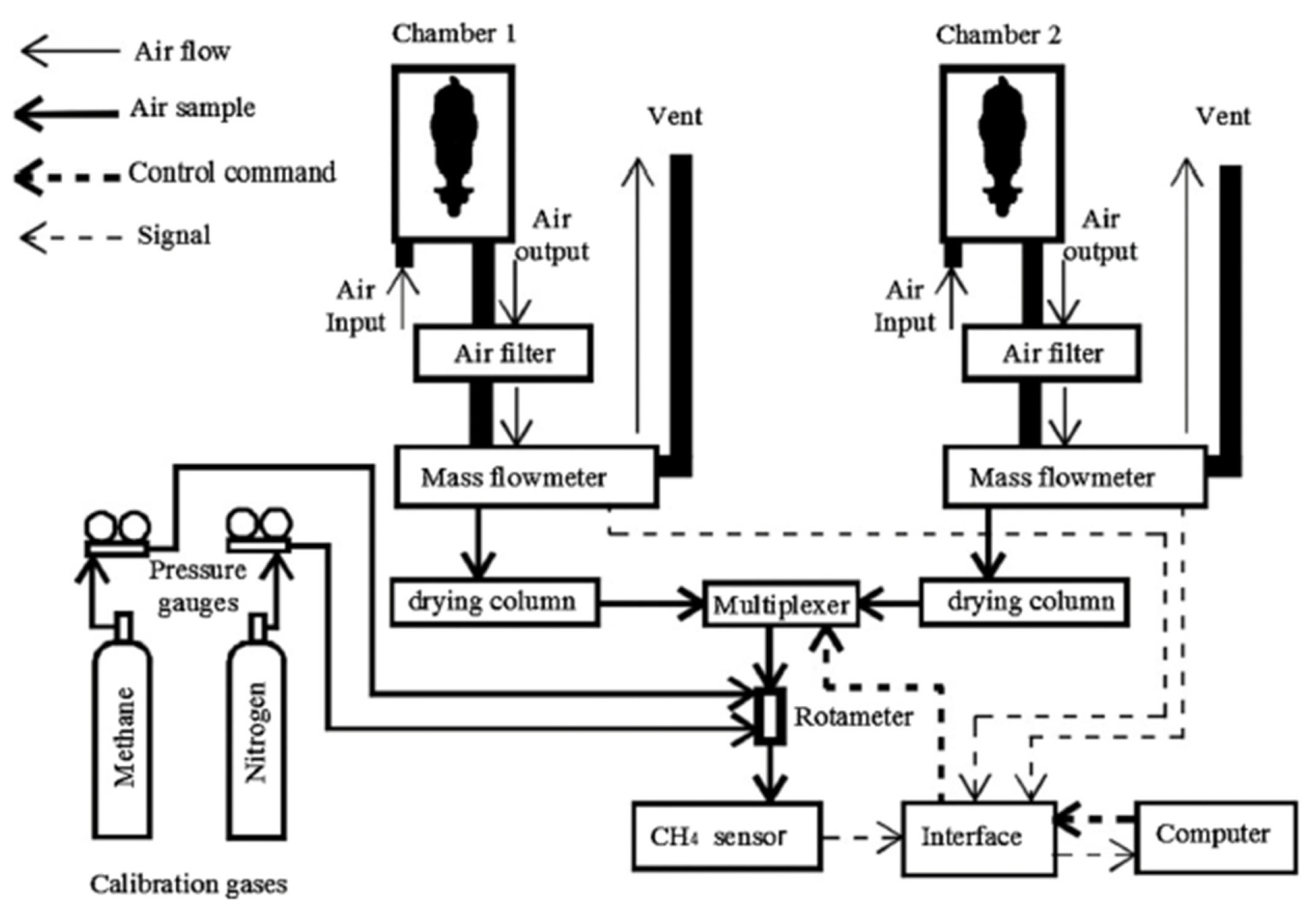
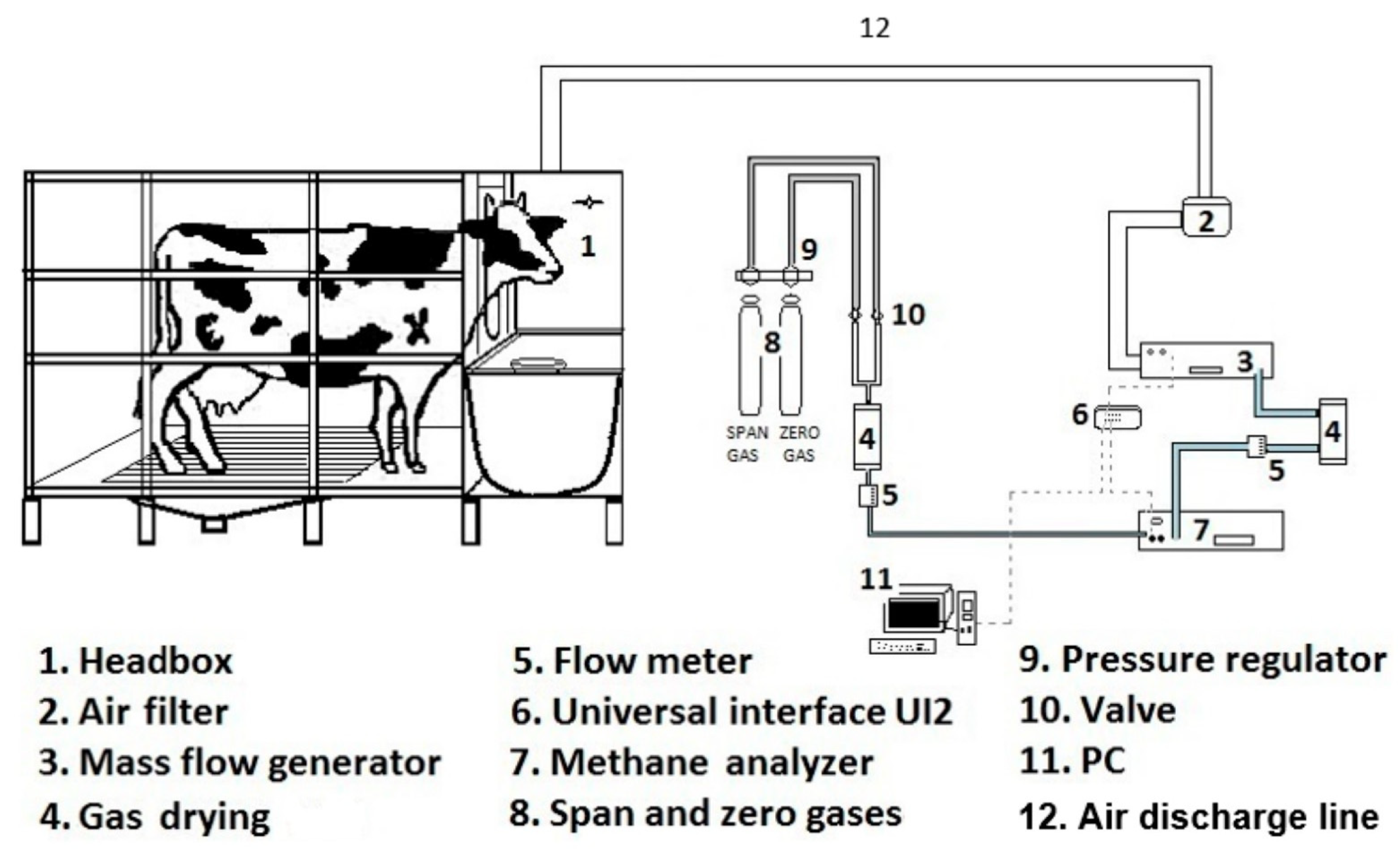
Respiration Chambers
Spot Sampling
Tracer Techniques
Laser-Based Techniques
3.1.2. Facility-Based Techniques
Manure Storage
3.1.3. Emerging Techniques to Measure CH4 from Ruminants
Blood CH4 Concentration Tracer
Infrared (IR) Thermography
Intraruminal Telemetry
Eddy Covariance (EC) Technique
Carbon Dioxide as a Tracer Gas
3.2. Technologies to Transform GHG Emissions in Livestock Agriculture into Energy
3.3. Emerging Technologies and Future Directions to Transform GHG into Energy
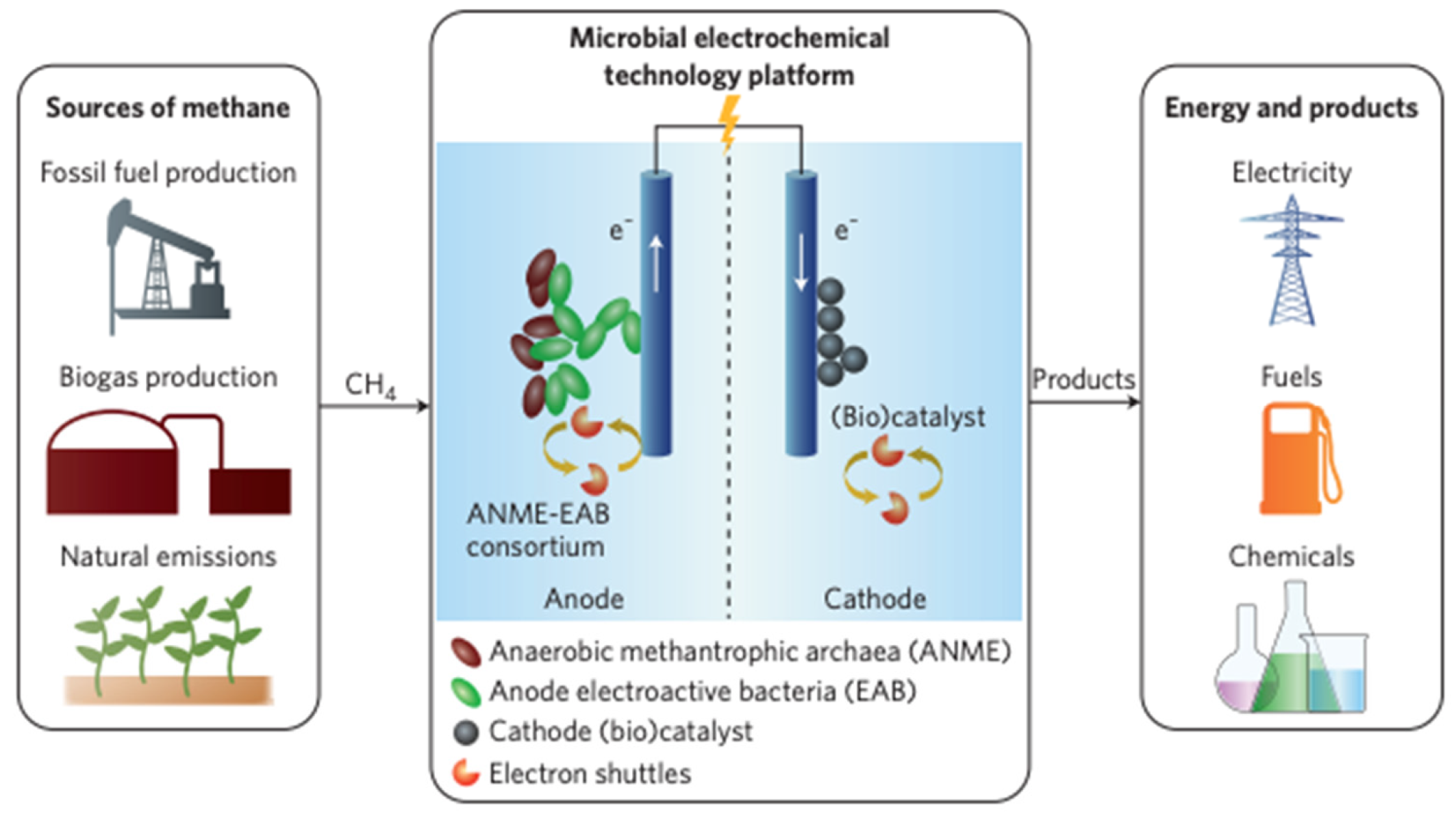
3.3.1. Microbial Fuel Cells
3.3.2. Biogas Dry Reforming
3.3.3. Biogas Steam Methane Reforming
3.3.4. Other Techniques
4. Discussion
4.1. Addressing the Research Objectives
4.2. Economic and Life Cycle Assessment
5. Conclusions
5.1. Study Conclusions
5.2. Recommendations
5.3. Future Directions and Perspectives
5.4. Further Research Needs
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A. Literature Matrix
| Number | Authors | Title | Methodology | Findings | Recommendation |
| 1 | [25] P. C. Garnsworthy et al. | Comparison of Methods to Measure Methane for Use in Genetic Evaluation of Dairy Cattle | Quantitative comparison of respiration chambers, SF6, breath sampling during milking and feeding, greenfeed and laser methane detector | The respiration chamber indirect calorimetry technique is the gold standard method used in the measurement of enteric methane from ruminants where the animal is confined in a chamber for 2 to 7 days. Breath sampling generated high throughput. | Use of respiration chambers to measure methane from ruminants |
| 2 | [26] J. C. Ku-Vera et al. | Determination of methane yield in cattle fed tropical grasses as measured in open-circuit respiration chambers | 66 individual determinations of dry matter intake (DMI) and 42 determinations of organic matter intake (OMI) for enteric methane production | An average intake of 8.22 kg DM and 7.8 kg OM was also recorded for the heifers with the open-circuit gas exchange measurement, revealing an average CH4 of 88 g per heifer on a daily basis | Open-circuit respiration chambers were reliable in measuring CH4 from cattle fed tropical grasses. |
| 3 | [27] Blümmel et al. | Comparison of methane produced by straw fed sheep in open-circuit respiration with methane predicted by fermentation characteristics measured by an in vitro gas procedure | Comparison of feed intake, digestibility, and methane production by open-circuit respiration measurements in 15 sheep fed untreated, sodium hydroxide (NaOH) treated and anhydrous ammonia (NH3) treated wheat, barley, and oat straws | Daily methane production from the open-circuit respiration chambers compared well with that from the in vitro fermentation features | Adopt open-circuit respiration chambers to measure methane from animals compared to in vitro techniques. |
| 4 | [28] M. G. G. Chagunda and T. Yan | Do methane measurements from a laser detector and an indirect open-circuit respiration calorimetric chamber agree sufficiently? | Pearson correlation and analysis of agreement based on the Bland and Altman methodology to test the laser methane detector (LMD) and the indirect open-circuit respiration calorimetric chamber | The two methods compared well against each other given the close inverse regression estimates and high correlation coefficients in the different techniques. The LMD generated higher numerical methane measurements as compared to the open-circuit respiration chambers | Both LMD and open-circuit chambers generate effective measurements of methane from ruminants |
| 5 | [29] N. W. Tomkins, S. M. McGinn, D. A. Turner, and E. Charmley | Comparison of open-circuit respiration chambers with a micrometeorological method for determining methane emissions from beef cattle grazing a tropical pasture | Quantify CH4 emissions from beef cattle at herd scale and from individual animals using open circuit respiration chambers and an open-path laser | Using the micrometeorological method led to comparable methane quantities to those from the open-circuit chamber method—29.7 ± 3.70 g/kg dry matter (DM) vs. 30.1 ± 2.19 g/kg, respectively | Open-circuit chambers generated comparable results to micrometeorological methods |
| 6 | [30] B. Suybeng et al. | Response to Climate Change: Evaluation of Methane Emissions in Northern Australian Beef Cattle on a High-Quality Diet Supplemented with Desmanthus Using Open-Circuit Respiration Chambers and GreenFeed Emission Monitoring Systems | Experiment 1: sixteen yearling Brangus steers fed a basal diet of Rhodes grass (Chloris gayana) hay in four treatments; the three Desmanthus cultivars and lucerne (Medicago sativa) at 30% dry matter intake (DMI) Experiment 2—GEM utilized forty-eight animals allocated to four treatments including a basal diet of Rhodes grass hay plus the three Desmanthus cultivars in equal proportions at 0%, 15%, 30% and 45% DMI. | There were no differences between the levels of methane measured using the different techniques | Open-circuit respiration chambers and GreenFeed emission monitoring systems generated comparable results regarding methane measurement |
| 7 | [31] A. Jonker et al., | Genetic parameters of methane emissions determined using portable accumulation chambers in lambs’ and ewes’ grazing pasture and genetic correlations with emissions determined in respiration chambers | Quantitative measurement of CH4 and carbon dioxide (CO2) emissions using several 1-h portable accumulation chamber (PAC) measurements from lambs and again as ewes while grazing ryegrass-based pastures | The methane/methane + carbon dioxide ratio was comparable to the PAC and respiration chamber techniques | Open-circuit respiration chambers and PAC Systems generated comparable results regarding methane measurement |
| 8 | [32] G. F. Difford et al. | Ranking cows’ methane emissions under commercial conditions with sniffers versus respiration chambers | 20 lactating dairy cows (10 Holstein and 10 Jerseys were recorded using sniffers installed in milking robots for three weeks of lactation and subsequently in respiration chambers (RC) where they were each recorded on three occasions within the RC | High correlation between methane produced from sniffers and respiration chambers: 0.77 ± 0.18 vs. 0.75 ± 0.20, respectively | Sniffers both on the farm and in respiration chambers had a potential for the measurement of methane emissions from large-scale dairy cattle |
| 9 | [33] D. Sorg et al. | Comparison of a laser methane detector with the GreenFeed and two breath analyzers for on-farm measurements of methane emissions from dairy cows | Data obtained with a handheld laser methane detector (LMD) and the GreenFeed system (GF), as well as data obtained with LMD and Fourier Transformed Infrared (FTIR) and Non-dispersive Infrared (NDIR) breath analyzers (sniffers) installed in the feed bin of automatic milking systems. | The different spot sampling devices were able to measure methane emissions on a daily basis, as a strong repeated measures correlation of LMD and GF was reported at 0.66 | Different spot sampling methods ranked the cows similarly, which implied that comparable results were generated |
| 10 | [34] O. A. Castelán Ortega et al. | Construction and Operation of a Respiration Chamber of the Head-Box Type for Methane Measurement from Cattle | Six assays were conducted to determine the pure CH4 recovery rate of the whole system in order to validate it and comply with the standards of chamber operation | Methane yields obtained from the cows and heifers: 19.7 ± 3.4 g and 17.1 ± 3.4 g CH4 kg−1 of dry matter were comparable to those from the literature | Head box chambers generated comparable methane measurements to the literature |
| 11 | [35] J. Rey et al. | Comparison Between Non-Invasive Methane Measurement Techniques in Cattle | Tests were conducted between the non-dispersive infrared methane analyzer (NDIR) method and the hand-held laser methane detector (LMD). Methane (CH4) was measured simultaneously with the two devices totaling 164 paired measurements. | There was higher methane concentration with the NDIR sniffer (0.42) as compared to the LMD (0.23) | The LMD and NDIR were not interchangeable, as they captured methane levels differently when used in livestock farms |
| 12 | [36] P. J. Moate et al. | Measurement of Enteric Methane Emissions by the SF6 Technique Is Not Affected by Ambient Weather Conditions | Six different cohorts of dairy cows (40 per cohort) were kept outdoors and fed a common diet during spring in 3 consecutive years. Methane production from individual cows was measured daily over the last 5 days of each 32 day period. An automated weather station measured air temperature, wind speed, relative humidity, and rainfall every 10 min. | Use of the SF6 technique was feasible in outdoor settings where there were varied conditions of humidity, rainfall, temperature, and wind speed. | Using the SF6 technique facilitated the measurement of methane emissions in different outdoor weather conditions |
| 13 | [37] M. Doreau, M. Arbre, Y. Rochette, C. Lascoux, M. Eugène, and C. Martin | Comparison of three methods for estimating enteric methane and carbon dioxide emission in nonlactating cows | Comparison undertaken in eight dry cows receiving a diet made of 70% hay and 30% concentrates given in limited and constant amounts, in a 15-wk experiment. Two periods in free stalls for SF6 and GF and in chambers for OC were used; in addition, SF6 was determined in chambers for one period. | Correlation coefficients for CH4 emission and CH4 yield were high and significant for OC and SF6, while they were not significant between OC and GF, and GF and SF6 | OC and SF6 generated comparable methane emission measurements |
| 14 | [38] I. C. de F. Maciel et al., | Could the breed composition improve performance and change the enteric methane emissions from beef cattle in a tropical intensive production system? | Steers (n = 8) from each breed composition were randomly selected in each phase to measure CH4 production using a sulfur hexafluoride (SF6) tracer technique and DM intake (DMI) using titanium dioxide. | The NEL had less methane intensity in grazing and average daily gain as compared to the AN. The results were also indicative that breed composition did not generate an impact on the methane yield based on dry matter intake (DMI) | Crossbreeding was an effective strategy to reduce methane levels emitted per kg of meat produced |
| 15 | [39] T. K. Flesch et al. | Methane emissions from cattle grazing under diverse conditions: An examination of field configurations appropriate for line-averaging sensors | Experimental design using an inverse dispersion method (IDM) to measure enteric methane (CH4) emissions, and its application to 15 rather distinct cattle trials in three types of feeding situations: summer grazing, winter swath grazing, and winter feeding | There was good agreement between the IDM designs across the 15 trials and based on the consistent forage types | Open path laser techniques were feasible in scenarios where narrow paddock IDM were used, and they generated advantages such as non-interference with the animals in their natural environments |
| 16 | [40] W. Bekele, A. Guinguina, A. Zegeye, A. Simachew, and M. Ramin, | Contemporary Methods of Measuring and Estimating Methane Emission from Ruminants | Quantitative comparison of six categories of methods for measuring and estimating CH4 emissions from ruminants | IR thermography was a non-invasive and inexpensive technique that is adopted to detect methane emissions from ruminants by assessing changes in temperature in the rumen | Use of IR thermography technique to measure methane emissions from rumen in ruminants |
| 16 | [41] K. Kang et al. | Application of a hand-held laser methane detector for measuring enteric methane emissions from cattle in intensive farming | Experiment 1 was conducted with four Hanwoo steers (584 ± 57.4 kg body weight [BW]) individually housed in metabolic cages. In experiment 2, 30 Hanwoo growing steers (343 ± 24.6 kg BW), blocked by BW, were randomly divided into three groups. Three different diets were provided to each group: high FC ratio (35:65) with low-energy concentrate (HFC-LEC), high FC ratio with high-energy concentrate (HFC-HEC), and low FC ratio (25:75) with high-energy concentrate (LFC-HEC). | the LMD method was effective in measuring methane emissions from cattle | the LMD method was effective in measuring methane emissions from cattle |
| 17 | [42] R. Roessler and E. Schlecht | Application of the laser methane detector for measurements in freely grazing goats: impact on animals’ behavior and methane emissions | LMD to assess the CH4 concentration in air exhaled by four pasture-fed female Thuringian Forest goats when they were either expressing their natural grazing behavior or when they were manually restrained at three times of the day over five consecutive days. | Methane emissions did not vary under restraint (6.5 ppm-m) and free-roaming (6.6 ppm-m). However, higher CH4 concentrations were established in the exhaled air from the ruminants during the afternoons as compared to morning sessions | LMD was effective in measuring methane emissions from animals |
| 18 | [48] A. B. Meireles et al. | Use of infrared thermography in an animal model as a complementary tool for monitoring the inflammatory process: a preliminary study | CFA-induced paw edema on rats (n = 5) was performed and discrepancies between animals treated or not with anti-inflammatory drugs such as triamcinolone acetonide and diclofenac sodium were analyzed. | Using infrared thermographic cameras to measure the changes in temperature on different surfaces indicated methane emissions | |
| 19 | [49] C. McSweeney | Measuring methane in the rumen under different production systems as a predictor of methane emissions | Real time data from the capsule in the rumen can be relayed via an ear tag to a remote personal computer using the public G3 network communication system. The power supply to the device enables data logging for approximately a month when the sampling rate is set at 20–30-min intervals. | Use of IR sensors and wireless networks to measure the concentration of different gases including methane, carbon dioxide and hydrogen in the rumen by utilizing intraruminal devices | Intraruminal telemetry facilitated measurement of methane gas within the rumen |
| 20 | [50] P. Prajapati and E. A. Santos | Estimating Herd-Scale Methane Emissions from Cattle in a Feedlot Using Eddy Covariance Measurements and the Carbon Dioxide Tracer Method | A closed-path EC system was used to measure CH4 and CO2 fluxes from a feedlot in Kansas. The EC flux measurements were scaled from landscape to animal scale using footprint analyses. | There were consistent methane emissions in the CO2 tracer method and the EC techniques with only 3% deviations being reported during dry and cold months | Use of eddy covariance was important to measure methane emissions from cattle |
| 21 | [51] P. Dumortier et al. | Beef cattle methane emission estimation using the eddy covariance technique in combination with geolocation | Methane emissions of a grazing herd of Belgian Blue cattle were estimated per individual on the field by combining eddy covariance measurements with the geolocation of the cattle and a footprint model | The results from the method revealed an estimate of methane emissions of 220 ± 35 g CH4 LU−1 day−1 which indicated methane emissions per livestock units per day | Eddy covariance technique in combination with geolocation was effective for methane measurement from beef cattle |
| 22 | [52] P. C. Stoy et al. | Methane efflux from an American bison herd | Measured methane and carbon dioxide fluxes from a bison herd on an enclosed pasture during daytime periods in winter using eddy covariance. | In the absence of the bison, methane emissions were negligible in the study area but were greater than zero when the bison were incorporated 0.048 ± 0.082 µmol m−2 s−1 for the mean and standard deviations | Adopt eddy covariance to measure methane emissions from bison herd |
| 23 | [75] P. Huhtanen, A. R. Bayat, P. Lund, A. L. F. Hellwing, and M. R. Weisbjerg | Short communication: Variation in feed efficiency hampers use of carbon dioxide as a tracer gas in measuring methane emissions in on-farm conditions | Data (307 cow-period observations) from two locations using the same setup for measuring CH4 and CO2 in respiration chambers were compiled, and observed production of CH4 and CO2 was compared with the equivalent predicted production using two different approaches. | An overestimation of methane emissions by 17% for the low-efficiency cows which indicated that the method favored low-efficient cows | Disparity in results identified in the use of carbon dioxide as a tracer gas in measuring methane emissions in on-farm conditions |
| 24 | [43] T. L. I. Vergote, S. Bodé, A. E. J. De Dobbelaere, J. Buysse, E. Meers, and E. I. P. Volcke | Monitoring methane and nitrous oxide emissions from digestate storage following manure mono-digestion | Quantified methane (CH4) and nitrous oxide (N2O) emissions from farm-scale mono-digested dairy manure by continuous monitoring in an on-site digestate storage for three months, in autumn | Generated results showed that the methane emission generated ranged from 4.6 to 14 g m−3 d−1 per day, while the nitrous oxide emission varied from 0.004 to 0.13 g m−3 d−1. The results further indicated that total emissions of the greenhouse gases ranged between 170 and 478 g [CO2, eq.] m−3 d−1, where only 10% was attributed to N2O | Use of an n-line gas phase analyzer to quantify the CH4 and N2O emissions from stored manure |
| 25 | [44] N. T. Vechi, N. S. Jensen, and C. Scheutz | Methane emissions from five Danish pig farms: Mitigation strategies and inventory-estimated emissions | Methane emissions were quantified by using the tracer gas dispersion method. Farms were measured between five and eight times throughout a whole year. One of the farms housed sows and weaners (P1) and the others focused on fattening pigs (P2–P5). The farms had different manure treatment practices including biogasification (P3), acidification (P4–P5), and no manure treatment (liquid slurry) (P1–P2). | Results showed that farms that had no manure treatment and where pigs were fattened had the highest methane emission rates, while the lowest rates of methane emission were identified in farms that had acidification manure treatment. | Manure storage generates methane emissions that can be measured using the tracer gas dispersion method. |
| 26 | [45] A. Cárdenas et al. | Methane emissions from the storage of liquid dairy manure: influences of season, temperature, and storage duration | Manure from the summer and winter season was stored under controlled conditions in barrels at ambient temperature to simulate manure storage conditions. Methane emissions from the manure samples from the winter season were measured in two time periods: 0 to 69 and 0 to 139 days. For the summer storage period, the experiments covered four time periods: from 0 to 70, 0 to 138, 0 to 209, and 0 to 279 continuous days, with probing every 10 weeks. | Storing the livestock manure in a liquid form (slurry) in conditions of higher temperature and over a long duration of time maximized the total methane emissions that could be generated. | Methane emissions were generated from stored slurry that was maintained in conditions of high temperature. |
| 27 | [46] J. E. Hilgert et al. | Methane Emissions from Livestock Slurry: Effects of Storage Temperature and Changes in Chemical Composition | Dairy and fattening pig manure samples were stored at five different temperatures (5–25 °C) for 90 days in a laboratory-scale experiment to measure the methane production. The chemical composition of the slurry samples was analyzed, and the Biochemical Methane Potential (BMP) tests were performed before and after storage. | Results showed that temperatures between 20 °C and 25 °C accounted for the highest methane emissions at a biochemical methane potential (BMP) of 69.3% and 50.3%, respectively | Temperature had an impact on methane emissions from livestock slurry |
| 28 | [47] C. Ma, L. B. Guldberg, M. J. Hansen, L. Feng, and S. O. Petersen | Frequent Export of Pig Slurry for Outside Storage Reduced Methane But Not Ammonia Emissions in Cold and Warm Seasons | The study examined CH4 and NH3 emissions from liquid pig manure (pig slurry) removed from the in-house slurry collection pits at three different frequencies, i.e., three times per week (T2.3), once per week (T7), or once after 40 days (T40, reference). The slurry from treatments T2.3 and T7 was transferred for outside storage weekly over four weeks, and slurry from treatment T40 once after 40 days, in connection with summer and winter production cycles with growing-finishing pigs. The slurry was stored in pilot-scale storage tanks with solid cover and continuous ventilation. | The frequent export of slurry to the external storage led to more ammonia emissions and lower methane emissions | To generate more methane emissions, it was important to export the slurry to external storage sites |
| 29 | [53] M. J. B. Kabeyi and O. A. Olanrewaju | Technologies for biogas to electricity conversion | Quantitative comparison of prime movers and different technologies that could be used to generate biogas | Prime movers useful for biogas power generation included steam and gas turbines, diesel engines, otto cycle engines and Stirling engines | Performance of biogas prime movers could be enhanced where enriched biogas or biomethane could be used in place of raw biogas |
| 30 | [54] A. Yatim, A. Luthfi, and R. Chemilo | Burner Design for biogas-fueled Stirling engine for electric power generation | Experiment involving design of a Stirling engine for biogas-fueled power generation system | The system generated 5 kW capacity fueled by 165 kg/day solid waste from a local farm with a biodigester of 20 m3. The burner provided simultaneous air preheater for lower fuel consumption leading to 37% lower consumption of fuel. | Use of Stirling engine provided flexible flue usage for power generation |
| 31 | [55] S. Abanades et al. | A conceptual review of sustainable electrical power generation from biogas | Conceptual examination of biogas-based electrical power-production systems | Use of upgraded biogas technologies increased the lower heating values of biogas by removing the pollutants. An economic analysis of the biogas-fueled systems was also undertaken | The use of upgraded biogas technologies improved electrical power production. |
| 32 | [56] U. U. R. Zia et al., | Technological Assessment of Bio Energy Production through Livestock Waste in Azad Jammu and Kashmir (AJK) | Quantitative assessment of the production capacity of biogas technologies | More than four million tons of biomass could be treated via technologies such as bio-methane engines, gas turbines, and steam turbines to generate over 300 TWh of energy. | The use of different types of bio-methane technologies including gas turbines, steam turbines and engines led to heat and electricity generation |
| 33 | [57] H. Barzegaravval, S. E. Hosseini, M. A. Wahid, and A. Saat | Effects of fuel composition on the economic performance of biogas-based power generation systems | An experiment to study the effects of fuel composition on exergetic and economic performance of biogas-based gas turbine systems with preheaters | Changing methane content from 0.95 to 0.6 increased the cost rate of the plant by 1%. The cost of the generated electricity varied from 0.05 $kWh to 0.18 $kWh | It is important to increase methane content in biogas-based turbine systems in order to reduce total cost rates of electricity |
| 34 | [58] R. Yamasaki, T. Maeda, and T. K. Wood | Electron carriers increase electricity production in methane microbial fuel cells that reverse methanogenesis | The reversal of methanogenesis in microbial fuel cells to produce electricity from methane through combining archaeal strains with Geobacter and methane acclimated sludge | Adding more electron carriers and changing the order of strains of the consortium led to increased power density and current density. MFCs were able to convert methane to electricity and were limited by electron carriers. | Power density and current density were comparable for any microbial fuel cells that used non-gaseous substrates or Shewanella |
| 35 | [59] M. J. McAnulty et al. | Electricity from methane by reversing methanogenesis | Experiment where a synthetic consortium was constructed that comprised of archaeal strain with Geobacter and methane acclimated sludge | The MFC operated at high Coulombic efficiency | The use of MFC is recommended to convert methane to electricity to avoid transportation |
| 36 | [60] Z. J. Ren | Microbial fuel cells: Running on gas | Experiment to create a microbial electrochemical technology platform comprised of a consortium of microorganisms | The MFC efficiently converted methane directly to current | use of MFC is recommended to convert methane to electricity |
| 37 | [61] J. Ding et al. | Decoupling of DAMO archaea from DAMO bacteria in a methane-driven microbial fuel cell | Experiment to investigate decoupling of denitrifying anaerobic methane oxidation archaea and DAMO bacteria in a MFC where methane was used as fuel | The DAMO fuel cell worked successfully but demonstrated weak electrogenic capability with around 25 mV production. After 45 days’ enrichment, the sequencing and fluorescence in situ hybridization results showed the DAMO archaea percentage had increased from 26.96% (inoculum) to 65.77% (electrode biofilm), while the DAMO bacteria percentage decreased from 24.39% to 2.07%. | The MFC may be used as a potential device to separate DAMO archaea from DAMO bacteria. |
| 38 | [62] S. Chen and A. L. Smith | Methane-driven microbial fuel cells recover energy and mitigate dissolved methane emissions from anaerobic effluents | Experiment using microbial fuel cells, single chamber MFCs, and dual-chamber MFCs to recover energy and mitigate methane emissions from anaerobic effluents | Generated results showed that up to 85% dissolved methane removal was achieved, resulting in the generation of 0.55 ± 0.06 V | Use of the MFC generated electricity from methane and reduced total emissions |
| 39 | [63] J. Myung, P. E. Saikaly, and B. E. Logan | A two-staged system to generate electricity in microbial fuel cells using methane | MFC experiment which involved a two-step strategy where methane was converted to methanol and electricity generated using the methanol | The methanol-fed MFC produced a maximum power density of 426 ± 17 mW/m2. It was also shown that the methanol-rich medium produced from the first step can be directly supplied to the MFCs, removing the need for the purification of methanol. | The analysis demonstrated that MFCs based on methanol could generate high power |
| 40 | [64] A. Nishimura, T. Takada, S. Ohata, and M. L. Kolhe | Biogas Dry Reforming for Hydrogen through Membrane Reactor Utilizing Negative Pressure | Quantitative study involving a membrane reactor to promote biogas dry reforming | Concentrations of hydrogen and carbon monoxide increase with increasing reaction temperature. The hydrogen concentration at the outlet chamber reduced with decreasing pressure sweep. The highest concentration of hydrogen was obtained in the molar ration of CH4:CO2 = 1:1 | To increase the conversion of methane to hydrogen gas, it was important to ensure a ratio of 1:1 for methane to carbon dioxide concentration. |
| 41 | [65] M. Chaghouri et al. | Impact of impurities on biogas valorization through dry reforming of methane reaction | Use of gas chromatography to examine biogas composition | Methane and carbon dioxide represented 60% of the composition of biogas, which led to promising results using dry reforming. Using a toluene catalyst led to a progressive increase in catalytic activity and higher carbon deposition. The addition of water decreased carbon dioxide conversion and the formation of carbon, thereby increasing the hydrogen/CO values closer to 1 | Incorporate toluene catalysts to increase the conversion of methane to hydrogen from biogas |
| 42 | [66] A. Cabello et al. | Production of hydrogen by chemical looping reforming of methane and biogas using a reactive and durable Cu-based oxygen carrier | Use of autothermal chemical looping reforming (CLRa) to evaluate the suitability of Cu-based oxygen carriers in a continuous pilot plant to produce blue and green hydrogen | The operation of 950 °C in fuel and air reactors resulted in the conversion of methane and hydrogen at 96% and 2.60 mol of hydrogen per mole of methane. The Cu-based oxygen carrier maintained mechanical integrity and chemical stability under harsh operating conditions. | The use of Cu-based oxygen carriers was considered a promising alternative to Ni-based materials to produce green and blue hydrogen through the CLRa process |
| 43 | [67] A. G. Georgiadis et al., | Biogas dry reforming over Ni/LnOx-type catalysts (Ln = La, Ce, Sm or Pr) | Use of the sol-gel citrate method to prepare Ni/LnO-type catalysts for the dry reforming of biogas | LNO was observed to possess higher catalytic activity in comparison to other materials. The use of Sm (Ni/Sm2O3) was considered an alternative strategy to restrict catalyst deactivation | The use of Ni/LnO catalysts facilitated the dry reforming of biogas |
| 44 | [68] P. Durán et al. | Pure hydrogen from biogas: Intensified methane dry reforming in a two-zone fluidized bed reactor using permselective membranes | Quantitative experiment to generate stable pure hydrogen by using a fluidized bed reactor coupled with permselective Pd/Ag membranes in the dry reforming of biogas | Hydrogen over-yields compared with conventional fluidized bed reactors in the range +200% to 100% were obtained for the interval of temperatures of 475 °C to 575 °C, while the stable operation by continuous catalyst regeneration was maintained. | Use of the bed reactor increased methane conversion and selectivity to hydrogen expressed as hydrogen to CO ratio |
| 45 | [69] R. Chein and Z. Yang | Experimental Study on Dry Reforming of Biogas for Syngas Production over Ni-Based Catalysts | Experiment to produce syngas from the dry reforming of biogas using catalysts such as Ni/Al2O3, Pt/Al2O3, and Pt-Ni/Al2O3 | The bimetallic Pt-Ni catalysts exhibit the best activity and thermal stability among the three types of catalysts due to better carbon deposition resistance. Adding H2O to the dry biogas reforming process leads to the steam reforming of methane as the dominant reaction which results in higher hydrogen and CO yields with biogas containing lower amounts of CO2. | Incorporating Ni-based catalysts in dry biogas reforming leads to the best conversion of methane to hydrogen |
| 46 | [70] C.-M. Vo et al., | Toward syngas production from simulated biogas dry reforming: Promotional effect of calcium on cobalt-based catalysts performance | Experiment to upgrade simulated biogas with equal mole ratio of methane and carbon dioxide through dry reforming over calcium promoted on cobalt-based catalysts | At low calcium dosages in a range of 0.1–0.2 wt%, the average Co3O4 crystalline size decreased from 8.15 nm to 6.01–7.43 nm, suggesting well-dispersed cobalt on the surface. In addition, the reducibility and basicity of catalysts were also enhanced with a sufficient addition of promoters. The optimal catalyst, 0.2Ca-10Co/Al2O3, exhibited the best performance, with roughly 84% and 89% of CH4 and CO2 conversions, respectively | The use of earth-abundant catalysts is important to enhance the dry reforming of simulated biogas |
| 47 | [71] A. Iulianelli et al. | Sustainable H2 generation via steam reforming of biogas in membrane reactors: H2S effects on membrane performance and catalytic activity | Experiment to steam reform synthetic biogas stream which contains 200 ppm of H2S, carried out in a non-commercial supported Pd–Au/Al2O3 membrane reactor (7–8 μm selective layer thickness) at 823 K and 150 kPa over a non-commercial Rh(1%)/MgAl2O4/Al2O3 catalyst | The developed system was able to recover 80% of total hydrogen produced during the reaction, which shows a good resistance to H2S contamination, which was confirmed by the stable conversion of methane for more than 400 h under operation. | Using the Pd–Au/Al2O3 membrane reactor over a non-commercial Rh(1%)/MgAl2O4/Al2O3 catalyst generated higher hydrogen recovery due to lower H2S contamination |
| 48 | [72] J. G. F. Madeira et al. | Hydrogen production from swine manure biogas via steam reforming of methane (SRM) and water gas shift (WGS): An ecological, technical, and economic analysis | The utilization of steam reforming and water gas shift processes to produce hydrogen from swine manure biogas. | The ecological efficiency, pollution indicator and energy efficiency of the process were 93.73%, 19.15%, and 79.06%, respectively, showing the viability from an ecological standpoint. An 8-year payback with a hydrogen production cost of $0.14 kWh, a production scenario of 8760 per year, and exergetic efficiency of 76%. | The production of hydrogen using this approach is economical and provides a high exegetic yield. |
| 49 | [73] M.-J. Park et al. | System optimization for effective hydrogen production via anaerobic digestion and biogas steam reforming | Experiment to test the stability of an optimized anaerobic digestion reactor over 60 days | Conditions of the reaction such as reaction temperature and steam/methane ratio had an impact on biogas steam reforming reactions. | Steam reforming biogas from anaerobic digesters was achieved for 25 h without any significant fluctuation or deactivation |
| 50 | [74] N. Schiaroli et al. | Biogas to Syngas through the Combined Steam/Dry Reforming Process: An Environmental Impact Assessment | An experiment to produce syngas from clean biogas using a combination of steam/dry reforming. | Clean biogas-to-syngas could be generated by using the reforming processes and had the potential to reduce anthropogenic impacts on the environment | The use of combined steam and dry reforming technology was effective in producing syngas from clean biogas. |
Appendix B. Literature CASP Appraisal
| Study | Was the Aim Stated Clearly? | Was the Quantitative Methodology Used? | Was the Research Design Suitable in This Study? | Was the Sampling of Participants Suitable in This Study? | Was the Data Collected in a Way That Addressed the Research Problem? | Is There a Positive Relationship between the Researcher and the Participants? | Have Ethical Issues Been Implemented? | Was the Data Well Analyzed? | Are the Findings Well Stated? |
| [25] P. C. Garnsworthy et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [26] J. C. Ku-Vera et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [27] Blümmel et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [28] M. G. G. Chagunda and T. Yan | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [29] N. W. Tomkins et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [30] B. Suybeng et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [31] A. Jonker et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [32] G. F. Difford et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [33] D. Sorg et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [34] O. A. Castelán Ortega et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [35] J. Rey et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [36] P. J. Moate et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [37] M. Doreau et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [38] I. C. de F. Maciel et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [39] T. K. Flesch et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [40] W. Bekele, et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [41] K. Kang et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [42] R. Roessler and E. Schlecht | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [48] A. B. Meireles et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [49] C. McSweeney | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [50] P. Prajapati and E. A. Santos | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [51] P. Dumortier et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [52] P. C. Stoy et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [75] P. Huhtanen, A. R. et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [43] T. L. I. Vergote et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [44] N. T. Vechi, N. S. Jensen, and C. Scheutz | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [45] A. Cárdenas et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [46] J. E. Hilgert et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [47] C. Ma, L. B. Guldberg, M. J. Hansen, L. Feng, and S. O. Petersen | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [53] M. J. B. Kabeyi and O. A. Olanrewaju | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [54] A. Yatim, A. Luthfi, and R. Chemilo | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [55] S. Abanades et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [56] U. U. R. Zia et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [57] H. Barzegaravval, S. E. Hosseini, M. A. Wahid, and A. Saat | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [58] R. Yamasaki, T. Maeda, and T. K. Wood | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [59] M. J. McAnulty et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [60] Z. J. Ren | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [61] J. Ding et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [62] S. Chen and A. L. Smith | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [63] J. Myung, P. E. Saikaly, and B. E. Logan | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [64] A. Nishimura, T. Takada, S. Ohata, and M. L. Kolhe | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [65] M. Chaghouri et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [66] A. Cabello et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [67] A. G. Georgiadis et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [68] P. Durán et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [69] R. Chein and Z. Yang | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [70] C.-M. Vo et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [71] A. Iulianelli et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [72] J. G. F. Madeira et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [73] M.-J. Park et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [74] N. Schiaroli et al. | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
References
- Ritchie, H.; Roser, M. Meat and Dairy Production. In Our World in Data. 2017. Available online: https://ourworldindata.org/meat-production (accessed on 21 January 2023).
- Liu, Z.; Ahmad, M.; Li, G.; Yang, Y.; Liu, Y.; Gao, M.; Luo, Q. Decoupling of Greenhouse Gas Emissions from Livestock Industrial Development: Evidence from China Agricultural Green Development Modern Zone. Front. Environ. Sci. 2022, 10, 1563. [Google Scholar] [CrossRef]
- Zhuang, M.; Lu, X.; Caro, D.; Gao, J.; Zhang, J.; Cullen, B.; Li, Q. Emissions of Non-CO2 Greenhouse Gases from Livestock in China during 2000–2015: Magnitude, Trends and Spatiotemporal Patterns. J. Environ. Manag. 2019, 242, 40–45. [Google Scholar] [CrossRef]
- Llonch, P.; Haskell, M.J.; Dewhurst, R.J.; Turner, S.P. Current available strategies to mitigate greenhouse gas emissions in livestock systems: An animal welfare perspective. Animal 2016, 11, 274–284. [Google Scholar] [CrossRef]
- Tubiello, F.N.; Salvatore, M.; Rossi, S.; Ferrara, A.; Fitton, N.; Smith, P. The FAOSTAT database of greenhouse gas emissions from agriculture. Environ. Res. Lett. 2013, 8, 015009. [Google Scholar] [CrossRef]
- Gerber, P.J.; Henderson, B.; Harinder, P.S. Makkar, and Food and Agriculture Organization of the United Nations. In Mitigation of Greenhouse Gas Emissions in Livestock Production: A Review of Technical Options for Non-CO2 Emissions; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013. [Google Scholar]
- The World Bank. Moving Towards Sustainability: The Livestock Sector and the World Bank. 2019. Available online: https://www.worldbank.org/en/topic/agriculture/brief/moving-towards-sustainability-the-livestock-sector-and-the-world-bank (accessed on 21 January 2023).
- Shine, P.; Upton, J.; Sefeedpari, P.; Murphy, M.D. Energy Consumption on Dairy Farms: A Review of Monitoring, Prediction Modelling, and Analyses. Energies 2020, 13, 1288. [Google Scholar] [CrossRef]
- Paris, B.; Vandorou, F.; Tyris, D.; Balafoutis, A.T.; Vaiopoulos, K.; Kyriakarakos, G.; Manolakos, D.; Papadakis, G. Energy Use in the EU Livestock Sector: A Review Recommending Energy Efficiency Measures and Renewable Energy Sources Adoption. Appl. Sci. 2022, 12, 2142. [Google Scholar] [CrossRef]
- Frorip, J.; Kokin, E.; Praks, J.; Poikalainen, V.; Ruus, A.; Veermäe, I.; Lepasalu, L.; Schäfer, W.; Mikkola, H.; Ahokas, J. Energy Consumption in Animal Production—Case Farm Study. Agron. Res. 2012, 10, 39–48. [Google Scholar]
- Upton, J.; Murphy, M.; Shalloo, L.; Koerkamp, P.G.; De Boer, I. A Mechanistic Model for Electricity Consumption on Dairy Farms: Definition, Validation, and Demonstration. J. Dairy Sci. 2014, 97, 4973–4984. [Google Scholar] [CrossRef]
- Guan, Y.; Yan, J.; Shan, Y.; Zhou, Y.; Hang, Y.; Li, R.; Liu, Y.; Liu, B.; Nie, Q.; Bruckner, B.; et al. Burden of the Global Energy Price Crisis on Households. Nat. Energy 2023, 8, 304–316. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, L.; Song, S.; Yu, S. Impacts of Energy Price on Agricultural Production, Energy Consumption, and Carbon Emission in China: A Price Endogenous Partial Equilibrium Model Analysis. Sustainability 2022, 14, 3002. [Google Scholar] [CrossRef]
- Shirzad, M.; Panahi, H.K.S.; Dashti, B.B.; Rajaeifar, M.A.; Aghbashlo, M.; Tabatabaei, M. A Comprehensive Review on Electricity Generation and GHG Emission Reduction Potentials through Anaerobic Digestion of Agricultural and Livestock/Slaughterhouse Wastes in Iran. Renew. Sustain. Energy Rev. 2019, 111, 571–594. [Google Scholar] [CrossRef]
- Grossi, G.; Goglio, P.; Vitali, A.; Williams, A.G. Livestock and Climate change: Impact of Livestock on Climate and Mitigation Strategies. Anim. Front. 2018, 9, 69–76. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock: A Global Assessment of Emissions and Mitigation Opportunities; FAO: Rome, Italy, 2013. [Google Scholar]
- Kraus, S.; Breier, M.; Dasí-Rodríguez, S. The Art of Crafting a Systematic Literature Review in Entrepreneurship Research. Int. Entrep. Manag. J. 2020, 16, 1023–1042. [Google Scholar] [CrossRef]
- Aromataris, E.; Riitano, D. Constructing a Search Strategy and Searching for Evidence. Am. J. Nurs. 2014, 114, 49–56. [Google Scholar] [CrossRef]
- Bramer, W.M.; De Jonge, G.B.; Rethlefsen, M.L.; Mast, F.; Kleijnen, J. A Systematic Approach to Searching: An Efficient and Complete Method to Develop Literature Searches. J. Med. Libr. Assoc. 2018, 106, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Ogie, R.; O’Brien, S.; Federici, F. Towards Using Agent-Based Modelling for Collaborative Translation of Crisis Information: A Systematic Literature Review to Identify the Underlying Attributes, Behaviors, Interactions, and Environment of Agents. Int. J. Disaster Risk Reduct. 2022, 68, 102717. [Google Scholar] [CrossRef]
- Long, H.A.; French, D.P.; Brooks, J.M. Optimizing the Value of the Critical Appraisal Skills Program (CASP) Tool for Quality Appraisal in Qualitative Evidence Synthesis. Res. Methods Med. Health Sci. 2020, 1, 31–42. [Google Scholar] [CrossRef]
- Patel, J.J.; Hill, A.; Lee, Z.-Y.; Heyland, D.K.M.; Stoppe, C. Critical Appraisal of a Systematic Review: A Concise Review. Crit. Care Med. 2022; publish ahead of print. [Google Scholar] [CrossRef]
- Graham, M.W.; Butterbach-Bahl, K.; du Toit, C.J.L.; Korir, D.; Leitner, S.; Merbold, L.; Mwape, A.; Ndung’u, P.W.; Pelster, D.E.; Rufino, M.C.; et al. Research Progress on Greenhouse Gas Emissions from Livestock in Sub-Saharan Africa Falls Short of National Inventory Ambitions. Front. Soil Sci. 2022, 2, 927452. [Google Scholar] [CrossRef]
- Tedeschi, L.O.; Abdalla, A.L.; Álvarez, C.; Anuga, S.W.; Arango, J.; Beauchemin, K.A.; Becquet, P.; Berndt, A.; Burns, R.; De Camillis, C.; et al. Quantification of Methane Emitted by Ruminants: A Review of Methods. J. Anim. Sci. 2022, 100, skac197. [Google Scholar] [CrossRef]
- Garnsworthy, P.C.; Difford, G.F.; Bell, M.J.; Bayat, A.R.; Huhtanen, P.; Kuhla, B.; Lassen, J.; Peiren, N.; Pszczola, M.; Sorg, D.; et al. Comparison of Methods to Measure Methane for Use in Genetic Evaluation of Dairy Cattle. Animals 2019, 9, 837. [Google Scholar] [CrossRef]
- Ku-Vera, J.C.; Valencia-Salazar, S.S.; Piñeiro-Vázquez, A.T.; Molina-Botero, I.C.; Arroyave-Jaramillo, J.; Montoya-Flores, M.D.; Lazos-Balbuena, F.J.; Canul-Solís, J.R.; Arceo-Castillo, J.I.; Ramírez-Cancino, L.; et al. Determination of Methane Yield in Cattle Fed Tropical Grasses as Measured in Open-Circuit Respiration Chambers. Agric. For. Meteorol. 2018, 258, 3–7. [Google Scholar] [CrossRef]
- Blümmel, M.; Givens, D.; Moss, A. Comparison of methane produced by straw fed sheep in open-circuit respiration with methane predicted by fermentation characteristics measured by an in vitro gas procedure. Anim. Feed Sci. Technol. 2005, 123–124, 379–390. [Google Scholar] [CrossRef]
- Chagunda, M.; Yan, T. Do methane measurements from a laser detector and an indirect open-circuit respiration calorimetric chamber agree sufficiently closely? Anim. Feed Sci. Technol. 2011, 165, 8–14. [Google Scholar] [CrossRef]
- Tomkins, N.; McGinn, S.; Turner, D.; Charmley, E. Comparison of open-circuit respiration chambers with a micrometeorological method for determining methane emissions from beef cattle grazing a tropical pasture. Anim. Feed Sci. Technol. 2011, 166–167, 240–247. [Google Scholar] [CrossRef]
- Suybeng, B.; Mwangi, F.W.; McSweeney, C.S.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Malau-Aduli, A.E.O. Response to Climate Change: Evaluation of Methane Emissions in Northern Australian Beef Cattle on a High-Quality Diet Supplemented with Desmanthus Using Open-Circuit Respiration Chambers and GreenFeed Emission Monitoring Systems. Biology 2021, 10, 943. [Google Scholar] [CrossRef]
- Jonker, A.; Hickey, S.M.; Rowe, S.J.; Janssen, P.H.; Shackell, G.H.; Elmes, S.; Bain, W.E.; Wing, J.; Greer, G.J.; Bryson, B.; et al. Genetic parameters of methane emissions determined using portable accumulation chambers in lambs and ewes grazing pasture and genetic correlations with emissions determined in respiration chambers1. J. Anim. Sci. 2018, 96, 3031–3042. [Google Scholar] [CrossRef] [PubMed]
- Difford, G.F.; Olijhoek, D.W.; Hellwing, A.L.F.; Lund, P.; Bjerring, M.A.; de Haas, Y.; Lassen, J.; Løvendahl, P. Ranking cows’ methane emissions under commercial conditions with sniffers versus respiration chambers. Acta Agric. Scand. Sect. A Anim. Sci. 2018, 68, 25–32. [Google Scholar] [CrossRef]
- Sorg, D.; Difford, G.F.; Mühlbach, S.; Kuhla, B.; Swalve, H.H.; Lassen, J.; Strabel, T.; Pszczola, M. Comparison of a laser methane detector with the GreenFeed and two breath analyzers for on-farm measurements of methane emissions from dairy cows. Comput. Electron. Agric. 2018, 153, 285–294. [Google Scholar] [CrossRef]
- Ortega, O.A.C.; Beltrán, P.E.P.; Pineda, G.S.H.; Benaouda, M.; Ronquillo, M.G.; Molina, L.T.; Vera, J.C.K.; Pérez, H.D.M.; Carrillo, M.F.V. Construction and Operation of a Respiration Chamber of the Head-Box Type for Methane Measurement from Cattle. Animals 2020, 10, 227. [Google Scholar] [CrossRef]
- Rey, J.; Atxaerandio, R.; Ruiz, R.; Ugarte, E.; González-Recio, O.; Garcia-Rodriguez, A.; Goiri, I. Comparison between Non-Invasive Methane Measurement Techniques in Cattle. Animals 2019, 9, 563. [Google Scholar] [CrossRef]
- Moate, P.J.; Pryce, J.E.; Marett, L.C.; Garner, J.B.; Deighton, M.H.; Ribaux, B.E.; Hannah, M.C.; Wales, W.J.; Williams, S.R.O. Measurement of Enteric Methane Emissions by the SF6 Technique Is Not Affected by Ambient Weather Conditions. Animals 2021, 11, 528. [Google Scholar] [CrossRef]
- Doreau, M.; Arbre, M.; Rochette, Y.; Lascoux, C.; Eugène, M.; Martin, C. Comparison of 3 methods for estimating enteric methane and carbon dioxide emission in nonlactating cows. J. Anim. Sci. 2018, 96, 1559–1569. [Google Scholar] [CrossRef] [PubMed]
- Maciel, I.C.D.F.; Barbosa, F.A.; Tomich, T.R.; Ribeiro, L.G.P.; Alvarenga, R.C.; Lopes, L.; Malacco, V.M.R.; Rowntree, J.E.; Thompson, L.R.; Lana, A. Could the breed composition improve performance and change the enteric methane emissions from beef cattle in a tropical intensive production system? PLoS ONE 2019, 14, e0220247. [Google Scholar] [CrossRef] [PubMed]
- Flesch, T.K.; Basarab, J.A.; Baron, V.S.; Wilson, J.D.; Hu, N.; Tomkins, N.W.; Ohama, A.J. Methane emissions from cattle grazing under diverse conditions: An examination of field configurations appropriate for line-averaging sensors. Agric. For. Meteorol. 2018, 258, 8–17. [Google Scholar] [CrossRef]
- Bekele, W.; Guinguina, A.; Zegeye, A.; Simachew, A.; Ramin, M. Contemporary Methods of Measuring and Estimating Methane Emission from Ruminants. Methane 2022, 1, 82–95. [Google Scholar] [CrossRef]
- Kang, K.; Cho, H.; Jeong, S.; Jeon, S.; Lee, M.; Lee, S.; Baek, Y.; Oh, J.; Seo, S. Application of a hand-held laser methane detector for measuring enteric methane emissions from cattle in intensive farming. J. Anim. Sci. 2022, 100, skac211. [Google Scholar] [CrossRef]
- Roessler, R.; Schlecht, E. Application of the laser methane detector for measurements in freely grazing goats: Impact on animals’ behavior and methane emissions. Animal 2021, 15, 100070. [Google Scholar] [CrossRef] [PubMed]
- Vergote, T.L.; Bodé, S.; De Dobbelaere, A.E.; Buysse, J.; Meers, E.; Volcke, E.I. Monitoring methane and nitrous oxide emissions from digestate storage following manure mono-digestion. Biosyst. Eng. 2020, 196, 159–171. [Google Scholar] [CrossRef]
- Vechi, N.T.; Jensen, N.S.; Scheutz, C. Methane emissions from five Danish pig farms: Mitigation strategies and inventory estimated emissions. J. Environ. Manag. 2022, 317, 115319. [Google Scholar] [CrossRef]
- Cárdenas, A.; Ammon, C.; Schumacher, B.; Stinner, W.; Herrmann, C.; Schneider, M.; Weinrich, S.; Fischer, P.; Amon, T.; Amon, B. Methane emissions from the storage of liquid dairy manure: Influences of season, temperature and storage duration. Waste Manag. 2021, 121, 393–402. [Google Scholar] [CrossRef]
- Hilgert, J.E.; Amon, B.; Amon, T.; Belik, V.; Dragoni, F.; Ammon, C.; Cárdenas, A.; Petersen, S.O.; Herrmann, C. Methane Emissions from Livestock Slurry: Effects of Storage Temperature and Changes in Chemical Composition. Sustainability 2022, 14, 9934. [Google Scholar] [CrossRef]
- Ma, C.; Guldberg, L.B.; Hansen, M.J.; Feng, L.; Petersen, S.O. Frequent Export of Pig Slurry for Outside Storage Reduced Methane but not Ammonia Emissions in Cold and Warm Seasons. SSRN Electron. J. 2023. [Google Scholar] [CrossRef]
- Meireles, A.B.; Cruz, T.M.; Moreira, I.C.B.; de Almeida, V.G.; Avelar-Freitas, B.; Ottoni, M.H.F.; Araújo, C.P.; de Melo, G.E.B.A.; Gonçalves, P.F.; Pereira, W.D.F. Use of infrared thermography in an animal model as a complementary tool for monitoring the inflammatory process: A preliminary study. Res. Sq. 2021. [Google Scholar] [CrossRef]
- McSweeney, C. Measuring Methane in the Rumen under Different Production Systems as a Predictor of Methane Emissions. 2015. Available online: https://www.mla.com.au/contentassets/92d46123c2a640268f7a978c1d50c787/b.cch.6210_final_report.pdf (accessed on 21 January 2023).
- Prajapati, P.; Santos, E.A. Estimating Herd-Scale Methane Emissions from Cattle in a Feedlot Using Eddy Covariance Measurements and the Carbon Dioxide Tracer Method. J. Environ. Qual. 2019, 48, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Dumortier, P.; de la Motte, L.G.; Andriamandroso, A.; Aubinet, M.; Beckers, Y.; Bindelle, J.; De Cock, N.; Lebeau, F.; Heinesch, B. Beef cattle methane emission estimation using the eddy covariance technique in combination with geolocation. Agric. For. Meteorol. 2021, 297, 108249. [Google Scholar] [CrossRef]
- Stoy, P.C.; Cook, A.A.; Dore, J.E.; Kljun, N.; Kleindl, W.; Brookshire, E.N.J.; Gerken, T. Methane efflux from an American bison herd. Biogeosciences 2021, 18, 961–975. [Google Scholar] [CrossRef]
- Kabeyi, M.J.B.; Olanrewaju, O.A. Technologies for biogas to electricity conversion. Energy Rep. 2022, 8, 774–786. [Google Scholar] [CrossRef]
- Yatim, A.; Luthfi, A.; Chemilo, R. Burner Design for biogas-fueled Stirling engine for electric power generation. E3S Web Conf. 2018, 67, 02028. [Google Scholar] [CrossRef]
- Abanades, S.; Abbaspour, H.; Ahmadi, A.; Das, B.; Ehyaei, M.A.; Esmaeilion, F.; Assad, M.E.H.; Hajilounezhad, T.; Hmida, A.; Rosen, M.A.; et al. A conceptual review of sustainable electrical power generation from biogas. Energy Sci. Eng. 2021, 10, 630–655. [Google Scholar] [CrossRef]
- Zia, U.U.R.; Rashid, T.U.; Awan, W.N.; Bin Ahmed, T.; Siddique, M.A.; Habib, M.; Asid, R.M. Technological Assessment of Bio Energy Production through Livestock Waste in Azad Jammu and Kashmir (AJK). In Proceedings of the 2019 International Conference on Electrical, Communication, and Computer Engineering (ICECCE), Swat, Pakistan, 24–25 July 2019. [Google Scholar] [CrossRef]
- Barzegaravval, H.; Hosseini, S.E.; Wahid, M.A.; Saat, A. Effects of fuel composition on the economic performance of biogas-based power generation systems. Appl. Therm. Eng. 2018, 128, 1543–1554. [Google Scholar] [CrossRef]
- Yamasaki, R.; Maeda, T.; Wood, T.K. Electron carriers increase electricity production in methane microbial fuel cells that reverse methanogenesis. Biotechnol. Biofuels 2018, 11, 211. [Google Scholar] [CrossRef]
- McAnulty, M.J.; Poosarla, V.G.; Kim, K.-Y.; Jasso-Chávez, R.; Logan, B.E.; Wood, T.K. Electricity from methane by reversing methanogenesis. Nat. Commun. 2017, 8, 15419. [Google Scholar] [CrossRef]
- Ren, Z.J. Microbial fuel cells: Running on gas. Nat. Energy 2017, 2, 17093. [Google Scholar] [CrossRef]
- Ding, J.; Lu, Y.-Z.; Fu, L.; Ding, Z.-W.; Mu, Y.; Cheng, S.H.; Zeng, R.J. Decoupling of DAMO archaea from DAMO bacteria in a methane-driven microbial fuel cell. Water Res. 2017, 110, 112–119. [Google Scholar] [CrossRef]
- Chen, S.; Smith, A.L. Methane-driven microbial fuel cells recover energy and mitigate dissolved methane emissions from anaerobic effluents. Environ. Sci. Water Res. Technol. 2018, 4, 67–79. [Google Scholar] [CrossRef]
- Myung, J.; Saikaly, P.E.; Logan, B.E. A two-staged system to generate electricity in microbial fuel cells using methane. Chem. Eng. J. 2018, 352, 262–267. [Google Scholar] [CrossRef]
- ANishimura, A.; Takada, T.; Ohata, S.; Kolhe, M.L. Biogas Dry Reforming for Hydrogen through Membrane Reactor Utilizing Negative Pressure. Fuels 2021, 2, 194–209. [Google Scholar] [CrossRef]
- Chaghouri, M.; Hany, S.; Cazier, F.; Tidahy, H.L.; Gennequin, C.; Abi-Aad, E. Impact of impurities on biogas valorization through dry reforming of methane reaction. Int. J. Hydrog. Energy 2022, 47, 40415–40429. [Google Scholar] [CrossRef]
- Georgiadis, A.G.; Siakavelas, G.I.; Tsiotsias, A.I.; Charisiou, N.D.; Ehrhardt, B.; Wang, W.; Sebastian, V.; Hinder, S.J.; Baker, M.A.; Mascotto, S.; et al. Biogas dry reforming over Ni/LnOx-type catalysts (Ln = La, Ce, Sm or Pr). Int. J. Hydrog. Energy 2023, in press. [Google Scholar] [CrossRef]
- Durán, P.; Sanz-Martínez, A.; Soler, J.; Menéndez, M.; Herguido, J. Pure hydrogen from biogas: Intensified methane dry reforming in a two-zone fluidized bed reactor using permselective membranes. Chem. Eng. J. 2019, 370, 772–781. [Google Scholar] [CrossRef]
- Chein, R.; Yang, Z. Experimental Study on Dry Reforming of Biogas for Syngas Production over Ni-Based Catalysts. ACS Omega 2019, 4, 20911–20922. [Google Scholar] [CrossRef]
- Vo, C.-M.; Cao, A.N.T.; Qazaq, A.S.; Pham, C.Q.; Nguyen, D.L.T.; Alsaiari, M.; Vu, T.V.; Sharma, A.; Phuong, P.T.; Van, T.T.; et al. Toward syngas production from simulated biogas dry reforming: Promotional effect of calcium on cobalt-based catalysts performance. Fuel 2022, 326, 125106. [Google Scholar] [CrossRef]
- Iulianelli, A.; Manisco, M.; Bion, N.; Le Valant, A.; Epron, F.; Colpan, C.; Esposito, E.; Jansen, J.; Gensini, M.; Caravella, A. Sustainable H2 generation via steam reforming of biogas in membrane reactors: H2S effects on membrane performance and catalytic activity. Int. J. Hydrog. Energy 2021, 46, 29183–29197. [Google Scholar] [CrossRef]
- Madeira, J.G.F.; Oliveira, E.M.; Springer, M.V.; Cabral, H.L.; Barbeito, D.F.D.C.; Souza, A.P.G.; Moura, D.A.d.S.; Delgado, A.R.S. Hydrogen production from swine manure biogas via steam reforming of methane (SRM) and water gas shift (WGS): A ecological, technical, and economic analysis. Int. J. Hydrog. Energy 2021, 46, 8961–8971. [Google Scholar] [CrossRef]
- Park, M.-J.; Kim, J.-H.; Lee, Y.-H.; Kim, H.-M.; Jeong, D.-W. System optimization for effective hydrogen production via anaerobic digestion and biogas steam reforming. Int. J. Hydrog. Energy 2020, 45, 30188–30200. [Google Scholar] [CrossRef]
- Schiaroli, N.; Volanti, M.; Crimaldi, A.; Passarini, F.; Vaccari, A.; Fornasari, G.; Copelli, S.; Florit, F.; Lucarelli, C. Biogas to Syngas through the Combined Steam/Dry Reforming Process: An Environmental Impact Assessment. Energy Fuels 2021, 35, 4224–4236. [Google Scholar] [CrossRef]
- Cabello, A.; Mendiara, T.; Abad, A.; Izquierdo, M.; García-Labiano, F. Production of hydrogen by chemical looping reforming of methane and biogas using a reactive and durable Cu-based oxygen carrier. Fuel 2022, 322, 124250. [Google Scholar] [CrossRef]
- Huhtanen, P.; Bayat, A.; Lund, P.; Hellwing, A.; Weisbjerg, M. Short communication: Variation in feed efficiency hampers use of carbon dioxide as a tracer gas in measuring methane emissions in on-farm conditions. J. Dairy Sci. 2020, 103, 9090–9095. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maraveas, C.; Simeonaki, E.; Loukatos, D.; Arvanitis, K.G.; Bartzanas, T.; Kotzabasaki, M.I. Livestock Agriculture Greenhouse Gases for Electricity Production: Recent Developments and Future Perspectives. Energies 2023, 16, 3867. https://doi.org/10.3390/en16093867
Maraveas C, Simeonaki E, Loukatos D, Arvanitis KG, Bartzanas T, Kotzabasaki MI. Livestock Agriculture Greenhouse Gases for Electricity Production: Recent Developments and Future Perspectives. Energies. 2023; 16(9):3867. https://doi.org/10.3390/en16093867
Chicago/Turabian StyleMaraveas, Chrysanthos, Eleni Simeonaki, Dimitrios Loukatos, Konstantinos G. Arvanitis, Thomas Bartzanas, and Marianna I. Kotzabasaki. 2023. "Livestock Agriculture Greenhouse Gases for Electricity Production: Recent Developments and Future Perspectives" Energies 16, no. 9: 3867. https://doi.org/10.3390/en16093867
APA StyleMaraveas, C., Simeonaki, E., Loukatos, D., Arvanitis, K. G., Bartzanas, T., & Kotzabasaki, M. I. (2023). Livestock Agriculture Greenhouse Gases for Electricity Production: Recent Developments and Future Perspectives. Energies, 16(9), 3867. https://doi.org/10.3390/en16093867







