1. Introduction
Combustion engines emit complex mixtures of volatile organic compounds (VOCs) and polycyclic aromatic hydrocarbons (PAHs). The European Commission Directive [
1] states that a volatile organic compound is any organic compound with an initial boiling point less than or equal to 250 °C measured at a standard pressure of 1010.3 kPa. The most numerous groups of VOCs include aliphatic hydrocarbons, cyclic hydrocarbons, aromatic hydrocarbons, halogenated hydrocarbons, nitro hydrocarbons, alcohols, and phenols. It is estimated that VOCs constitute 60% of all substances polluting the atmosphere, while as many as 73% of them are on the list of carcinogenic compounds. They can therefore cause a number of short- and long-term effects, ranging from the irritation of the mucous membranes of the nose, eyes, and throat, headache, nausea, and dizziness to the development of cancer and damage to the nervous system. Emissions of these compounds in the range of 200–300 µg/m
3 are considered potentially harmful, while the range of 300–500 µg/m
3 is deemed harmful to human health. According to reports [
2,
3] by the National (Poland) Center for Emission Balancing and Management, the main anthropogenic source of VOCs with the second largest share (after the use of solvents and other products of this type) includes the transport sector. Polycyclic aromatic hydrocarbons, on the other hand, are compounds consisting of two or more aromatic rings. They are characterized by various structural forms with different relative positions of benzene rings in the molecule. These compounds have strong genotoxic, mutagenic, and carcinogenic properties [
4] and are always present in a mixture, never individually. It is estimated that in India, 37% of CO (carbon oxide) emissions and 17.5% of volatile organic compound (VOC) emissions from transport are generated by two-wheelers. The cited data demonstrate the scale of the problem of emissions from two-wheeled vehicles, although the scale varies depending on the location.
Currently, the interdisciplinarity of scientific research is important, which enables a multi-aspect approach to a selected issue. Therefore, the identification of toxic compounds from motor vehicles in real operating conditions, in addition to tests using PEMS (Portable Emission Measurement System) equipment, can be additionally undertaken with a qualitative chemical analysis [
5]. Due to the fact that some semi-volatile organic compounds emitted from motor vehicles (i.e., PAHs—polycyclic aromatic hydrocarbons) play a significant role in the formation of photochemical smog and secondary organic aerosol, there is a need to understand the sources and fate of these compounds in the atmosphere. Analyses of this type require researchers to optimize sample collection and determine an appropriate analytical method. A literature review showed that scientists use a number of available methods of collecting research material and chromatographic methods, i.e., GC–MS [
6,
7,
8,
9,
10,
11,
12,
13,
14], LC–MS [
15], TD–GC–MS [
16] with sampling into sorbent tubes [
14], feeding whole air or raw exhaust gases into Tedlar bags [
9,
17,
18,
19,
20,
21,
22,
23], microextraction into the solid phase [
24], or the flame ionization method [
25,
26,
27]. In the work of other research centers [
22,
23], volatile organic compounds were collected from two-wheeled vehicles into a set of Tedlar bags. Taking into account the current state of knowledge presented in the following chapters, theoretical and laboratory work should be developed in the field to generate a more detailed assessment of exhaust emissions. Therefore, the article presents a test procedure for a two-wheeled vehicle, based on GC–MS analysis, in traffic conditions representative of the tested vehicle. The methodology used, including the selection of specific operating conditions, was a novelty in the field of this type of work.
3. Chromatographic Analysis of the Chemical Composition of Exhaust Gas Samples
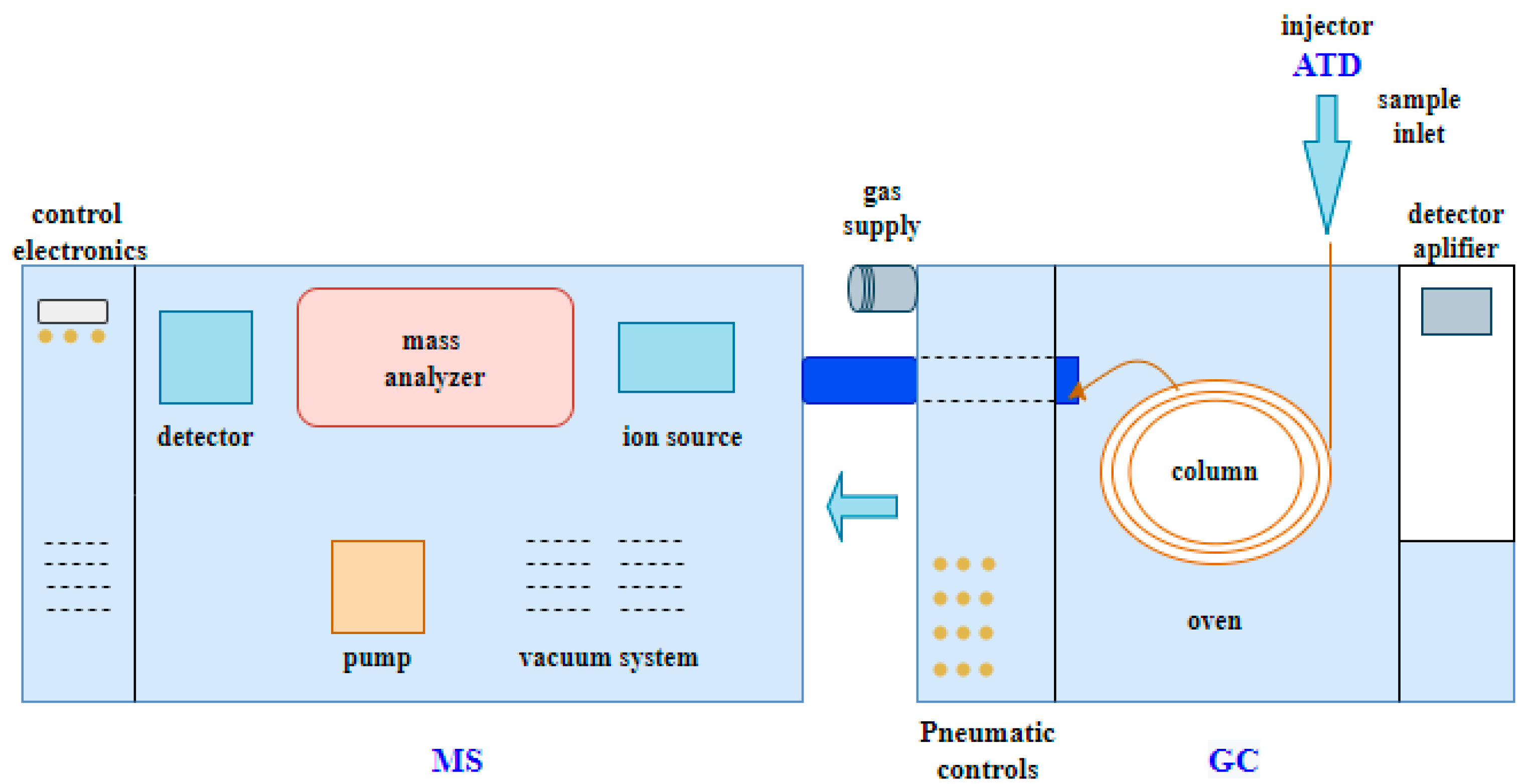
The use of the gas chromatography–thermal desorption–mass spectrometry method provides valuable information, primarily about volatile substances produced at particular operating points of a gasoline-powered engine. The identification of exhaust compounds (qualitative analysis) was carried out by locating chromatographic peaks of individual substances and determining their retention time (Rt). A chromatogram was obtained for each analyzed sample (
Figure 7). Additional confirmation was provided by the analysis of mass spectra (MS), which were compared with the commercial library of NIST (National Institute of Standards and Technology). This enabled the final identification of compounds based on mass-to-charge ratios (
m/
z) and the relative amounts of molecular and fragment ions resulting from electronic ionization. Mass spectrometry is a method that allows the determination of the molecular weight of a tested compound and the mass of fragments resulting from its decay during ionization in a mass spectrometer. Thus, mass spectra were compiled for the detected compounds with approximate retention times of Rt = 1.66 and 1.67.
In the mass spectrum of 3,3-dimethyl-1,2-epoxybutane, the most visible molecular ion had a mass-to-charge ratio
m/
z = 83, while for 4-methyl-1-heptene, the ion was measured to have a ratio
m/
z = 42 (
Figure 8a,b). The relative intensity was calculated in relation to the ion with the highest measured intensity, which was taken as the main ion and assigned an intensity of 100%. The intensities of the remaining peaks were then presented as percentages relative to the main peak.
Table 4 shows the chemicals identified for all samples that were detected using the measurement method described in
Section 2. The tabular list of chemical components contained in the gas samples obtained from an urban two-wheeled vehicle was additionally supplemented with a literature review. Studies and scientific articles that discussed each specific substance were analyzed. Their presence was recorded in chemical tests of samples of pollutants from motor vehicles, which were described in the cited works.
The obtained results indicated the presence of volatile organic compounds in the tested samples, including many aromatic and cyclic hydrocarbons (cycloalkanes). Qualitative analysis did not reveal significant differences in the four samples obtained (
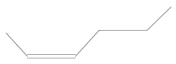
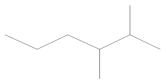
Table 5). Gasoline is a fuel that does not occur in the natural environment but is produced in primary or destructive refining processes, which means that it consists of up to several hundred substances with various physical and chemical properties. However, the main components of gasoline include aliphatic hydrocarbons with carbon atoms ranging from 5 to 12 and trace amounts of unsaturated and aromatic hydrocarbons. Therefore, alkenes and alkanes were observed in all samples, regardless of the engine operating point, as they are the main components of petroleum gases, i.e., butene, octane, heptane, hexene, and 3,3-dimethyl-1,2-epoxybutane, formed as a result of the oxidation of 4-heptanal and 2,3-dimethylhexane. These are primarily branched alkanes with long carbon chains and retention times of 1.66–1.95.
Gasoline components that were a product of crude oil distillation—4-methyl-1-heptene, 2,4-dimethylpentane, and 1,4-epoxycyclohexane—were also found in each of the analyzed samples. Moreover, toxicologically important compounds in the composition of gasoline were noted, defined as aromatic hydrocarbons from the BTX group (benzene, toluene, and xylenes),their derivatives (cumene, phenylethane, and dimethylbenzene), and benzoic acid, resulting from the oxidation of toluene.
The presence of these components in the exhaust gases may indicate incomplete combustion and complex reactions in the engine combustion chamber. The qualitative assessment of exhaust gas samples showed that the two-wheeled vehicle was characterized by greater qualitative diversity depending on the engine operating point in the case of aromatic hydrocarbon derivatives of the BTX group.
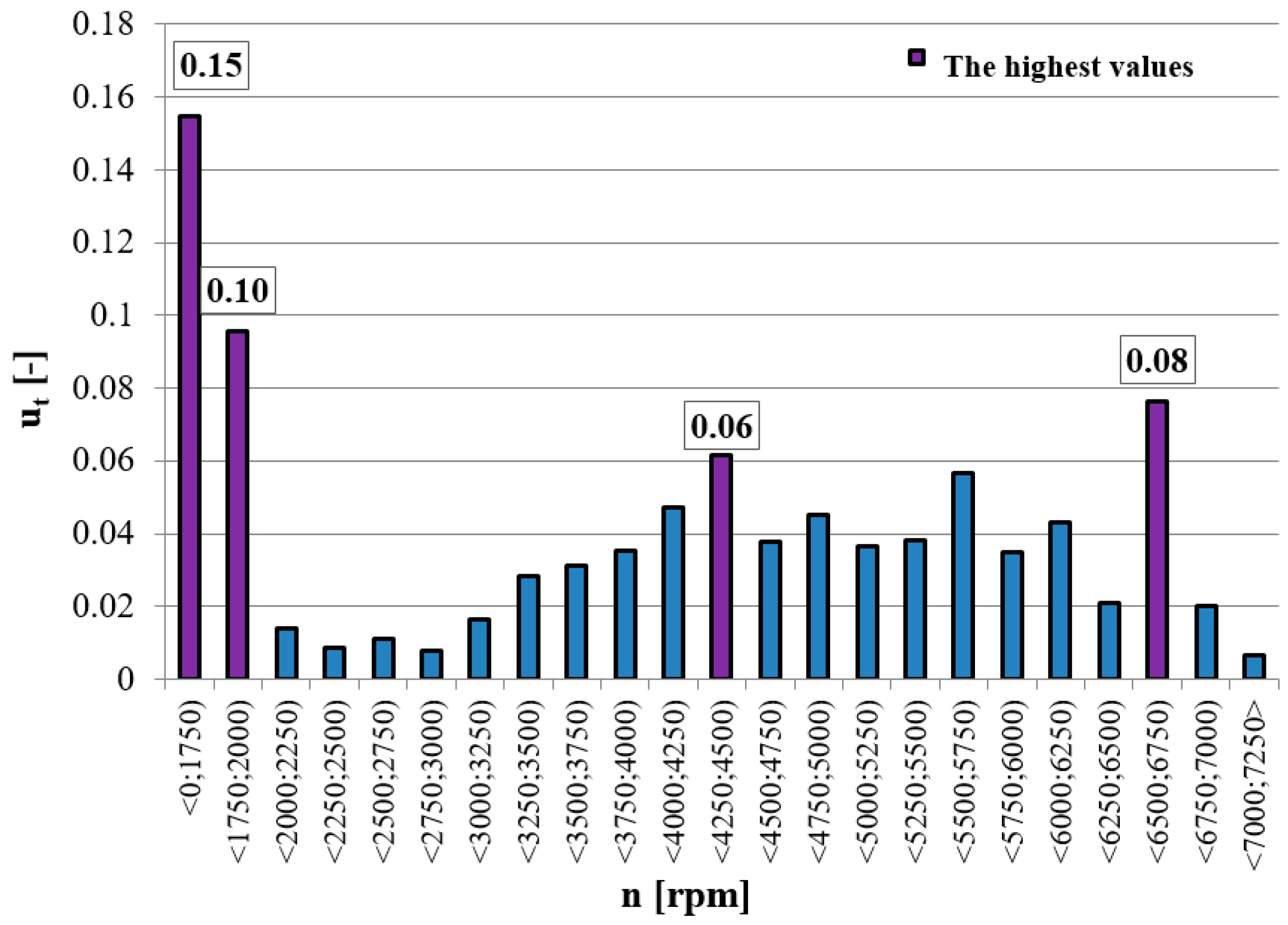
Ethylbenzene (phenylethane) with a retention time of 4.77 was not recorded in sample no. 4 (crankshaft rotational speed in the range of 6500–6750 rpm). The high temperature reached at this operating point resulted in a complete combustion of this compound. Increasing the speed and, consequently, a more effective combustion process also resulted in the lack of the presence of cumene (belonging to the group of aromatic hydrocarbons) in the last two analyzed samples. A more efficient combustion process resulted in the oxidation of aromatic hydrocarbons or their complete combustion, thus removing them from the emissions. The compound 1,5-Heptadien-3-in also appeared only in sample no. 1. Too low a temperature in the combustion chamber and cold walls and piston head were the causes of the formation of this compound. This is referred to as the wall effect—where the flame is dimmed near the cold walls of the combustion chamber.
In order to confirm the validity of the above considerations regarding the chemical composition of a gaseous sample from an urban two-wheeled vehicle, a gasoline sample was also analyzed to determine its detailed composition. The gas chromatography–mass spectrometry method was also used in this case. The technique utilizes three primary components: the separation power of high-resolution capillary gas chromatography, a mass spectrometer with controlled ion source and ion fragmentation coefficients, and unique data processing and reporting software. The method covers the range of hydrocarbons from C4 to C12 in gasoline.
There are a number of items in the literature regarding the gas chromatographic analysis of gasoline and related petroleum products. Retention index databases and computer software for detailed gasoline analysis are available from references [
48,
49,
50,
51,
52,
53,
54]. Gas chromatographic classification of liquid petroleum products is a well-established technique. The primary parameters used to distinguish different classes of products include boiling point range, aliphatic and aromatic hydrocarbon content, and the relative concentration of major and minor components.
The analysis showed that the composition of gasoline is mainly light aliphatic hydrocarbons (butane, pentane, methylpentane, hexane, methylhexane, and heptane) and aromatic compounds (toluene, xylenes, and trimethylbenzenes), with smaller amounts of olefinic hydrocarbons (pentanes, hexenes, heptenes, octenes, and nonenes;
Table 6). It is worth noting that conventional combustion engine solutions use gasoline and diesel oil, which are derivatives of crude oil. The approximate elemental structure of an average crude oil sample is assumed to be 84% carbon, 14% hydrogen, 1–3% sulfur, and less than 1% nitrogen, oxygen atoms, metals, and salts. There is a wide range of hydrocarbon compounds in crude oil consisting of alkanes, alkenes, naphthenes, and aromatics. These are very small molecular structures, such as propane (C
3H
8) and butane (C
4H
10), but can also consist of mixtures of different structures with very large molecules, such as heavy oils and asphalt. Therefore, crude oil must be distilled to be used for automotive applications.
As a result of the thermal distillation of crude oil, petroleum products are obtained, such as petroleum gases, aviation fuel, kerosene, gasoline, diesel oil, heavy fuels, machine oils, and asphalt. Generally, crude oil distillation produces on average 30% gasoline, 20–40% diesel oil, 20% heavy fuel oil, and heavy oils from 10 to 20% [
55]. The lack of derivatives of many compounds in the tested samples from the urban two-wheeler was due to the combustion reaction taking place in the engine and the detection limit of the concentration of some emitted substances. Additionally, the analysis time, which differed between the analysis of a liquid substance (gasoline) and a gaseous substance, could also have played a significant role.
Of the identified volatile toxic compounds found in a sample from an urban two-wheeler, almost half were aromatic compounds that are hazardous to health, including benzene, toluene, xylenes, and ethylbenzene. Moreover, most of them take part in the process of creating photochemical smog. VOCs can cause serious health effects such as drowsiness, headaches and dizziness, irritation of mucous membranes, and permanent damage to the liver or nervous system. Some of them are characterized by highly toxic, carcinogenic, neurotoxic, or mutagenic properties. They enter the body through the skin, respiratory, and digestive systems. Benzene and its derivatives are considered to be the most toxic compounds detected in exhaust gas samples from urban two-wheeled vehicles. Benzene is a simple aromatic hydrocarbon that causes both acute and chronic poisoning. It usually occurs through the inhalation of vapors through the respiratory system, but absorption through the skin and food intake are also possible. It may also lead to genetic defects [
55].
Toluene, although much less toxic than benzene, is considered a compound harmful to the respiratory, circulatory, reproductive, nervous, and immune systems, as well as the kidneys and the liver. In the case of toluene, the nervous system is particularly vulnerable, as toluene has a high affinity for tissues rich in lipids, in which it is soluble. Xylenes are compounds from the BTX group, which, due to the lowest volatility and higher boiling point, are considered safer than benzene and toluene. However, its unpleasant-smelling vapors are extremely easily absorbed by the respiratory tract, mucous membranes, and skin.
Table 7 presents the types of hazards and the chemical compounds assigned to them that were recorded in exhaust gas samples from two-wheeled vehicles [
56].
Most of the substances detected in the tested exhaust gases, depending on the method of exposure, may cause more or less serious health effects. For example, contact with liquid heptane causes redness, rash, and dry skin and is irritating to the eyes, but short-term exposure to heptane vapors may cause dizziness, headache, vomiting, a feeling of intoxication, problems with motor coordination, and loss of consciousness. Moreover, after entering water, heptane settles on solids in the water, including animals. For this reason, it is defined as a compound that is highly toxic to aquatic organisms and causes negative and long-lasting effects in the aquatic environment. Hexene, heptanal, 1,5-Hexadiene, and octane found in exhaust gases also have similar properties. The main way octane enters organisms is through the inhalation of its vapors [
56]. This aliphatic hydrocarbon mainly affects the central nervous system; however, in case of aspiration, there are also symptoms related to the respiratory system, i.e., cough, irritation of the upper respiratory tract, and in extreme cases it may lead to hemorrhagic pneumonia and pleurisy [
57,
58].