Comparative Analysis of Acidic and Alkaline Pretreatment Techniques for Bioethanol Production from Perennial Grasses
Abstract
:1. Introduction
2. Lignocellulosic Biomass
2.1. Perennial Grasses
| Grass | Characteristics | Annual Production | Reference |
|---|---|---|---|
| Bermuda grass (Cynodon dactylon) | Grows in tropical and subtropical regions. High carbohydrate content. | 6–27 t/ha | [25] |
| Napier/elephant grass (Pennisetum purpureum schum) | Fodder crop native to tropical and subtropical regions. Fast growing | 46.2 t/ha | [26,27,28] |
| Silver grass (Miscanthus floridulus) | Exhibits excellent resilience to saline conditions and drought. High lignocellulose yield. | 27.0–38.0 t/ha | [29] |
| Switchgrass (Panicum virgatum) | Flourishes in different soil types with minimal fertilizer requirement. Potential for soil carbon sequestration. | 14.8–37.1 t/ha | [21] |
| Fountain grass (Pennisetum alopecuroides) | Can withstand salinity and drought. | 40–50.2 t/ha | [30] |
| Palisade grass (Brachiaria brizantha) | High photosynthesis efficiency | 40 t/ha | [23,31] |
| Giant reed (Arundo donax) | Helps mitigate soil erosion. | 3.00–37.00 t/ha | [32,33,34] |
| King grass (Pennisetum hybridum) | Fast growing, high yield. Grows in a variety of soil conditions | 40–60 t/ha | [35] |
| Cogongrass (Imperata cylindrical) | Globally distributed weed, ecological threat to upland crops, requires minimal water and thrives well in unfertile soil. | 8–20 t/ha | [36] |
2.2. Lignocellulosic Biomass Composition
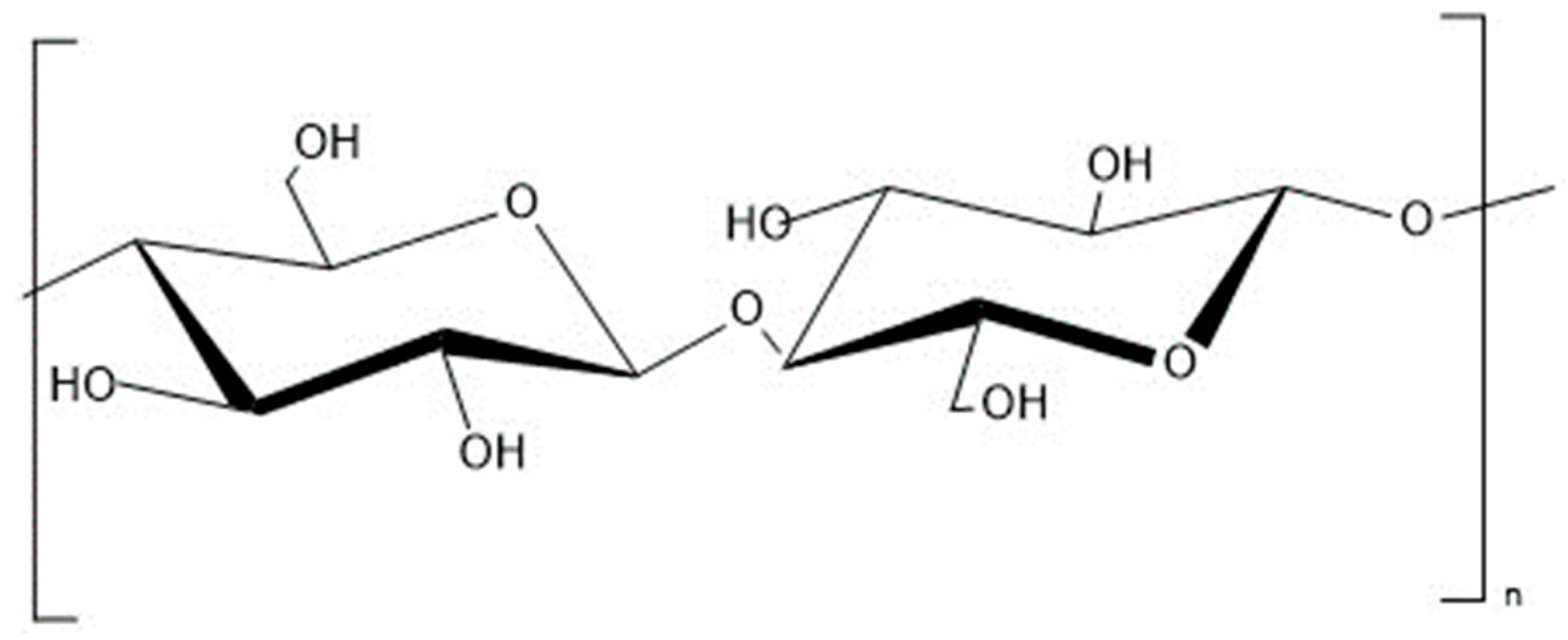
2.2.1. Cellulose
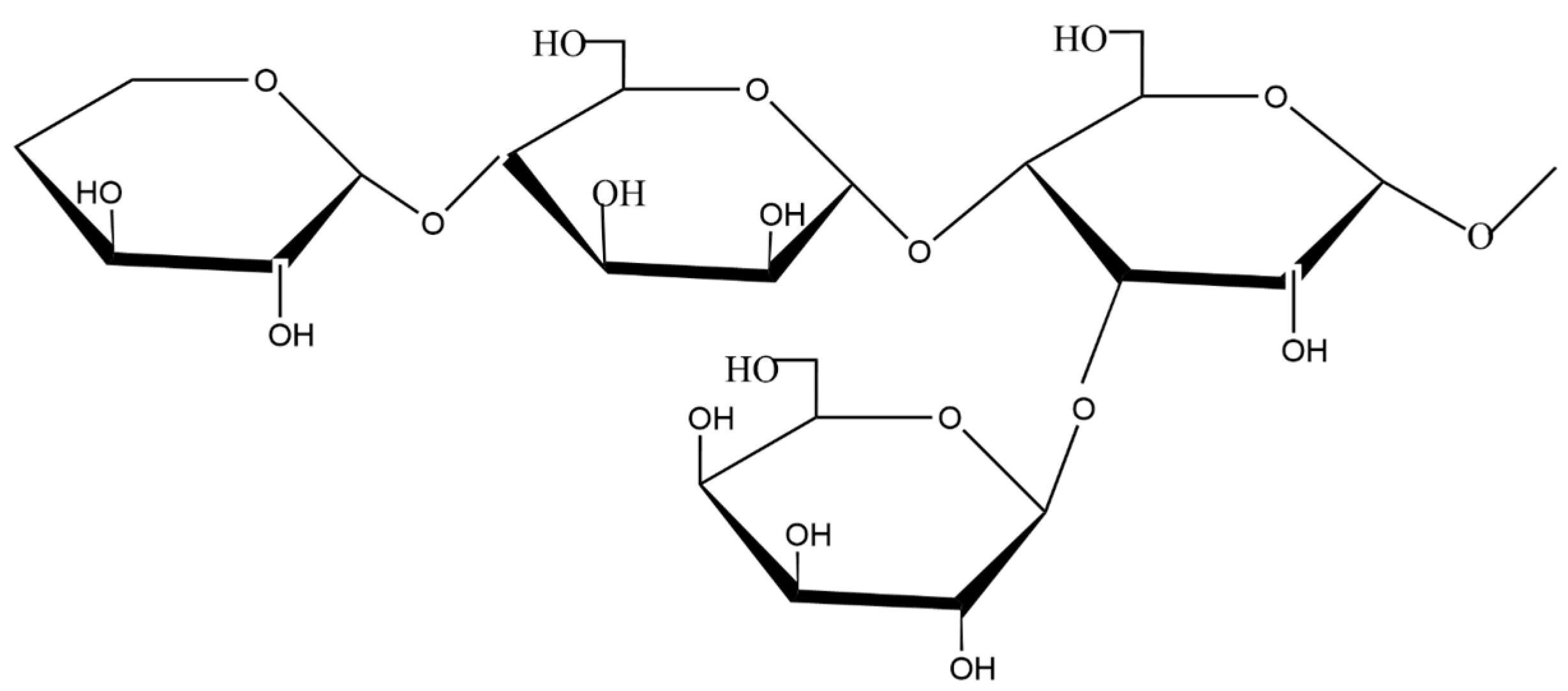
2.2.2. Hemicellulose
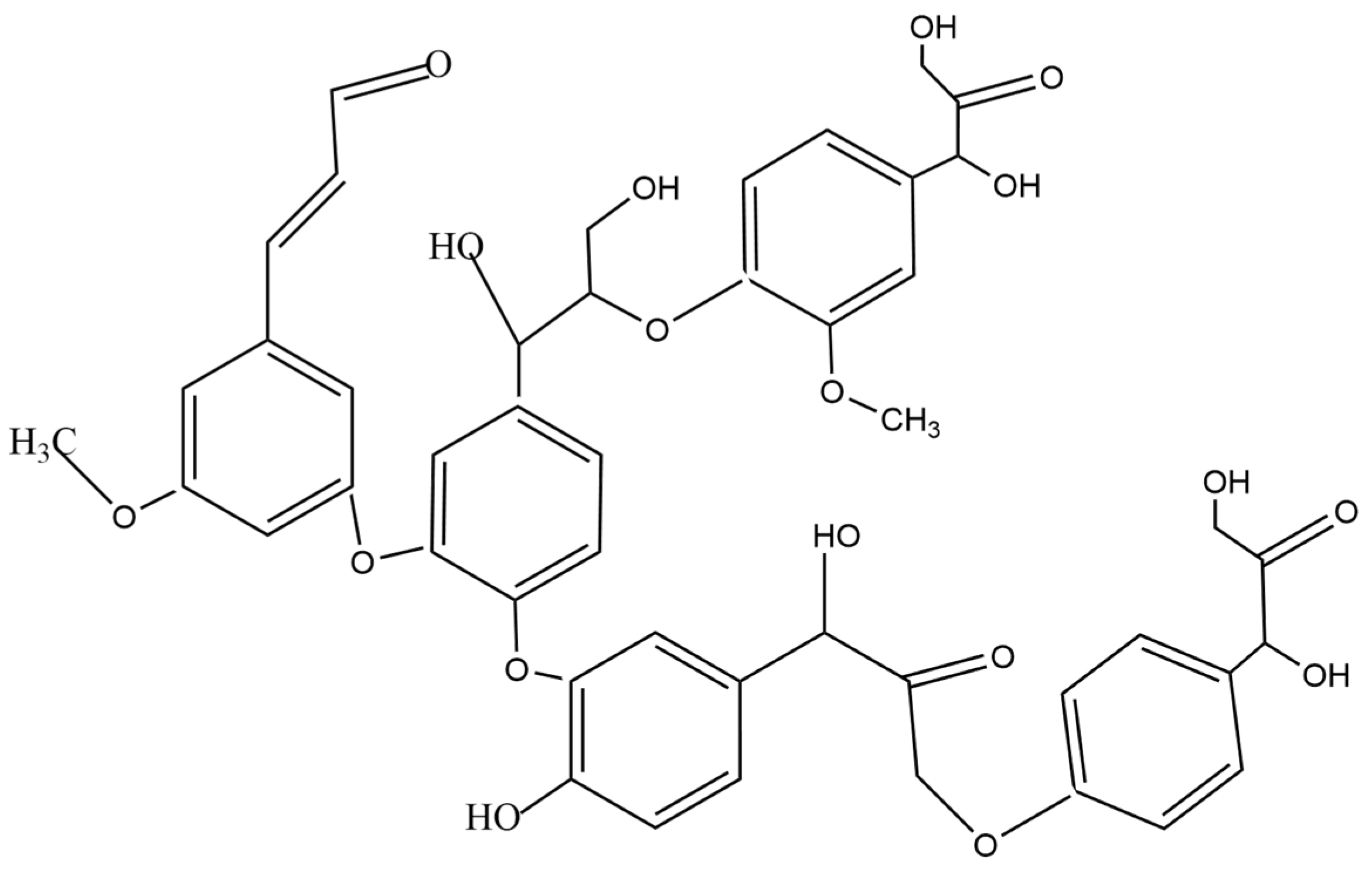
2.2.3. Lignin
2.3. Pretreatment of Biomass
2.3.1. Acidic Pretreatment
2.3.2. Alkaline Pretreatment
3. Enzymatic Hydrolysis and Fermentation
4. Effect of Acidic and Alkaline Pretreatments of Perennial Grasses
4.1. Biomass Composition before and after Pretreatment
4.2. Morphology and Crystallinity before and after Acidic and Alkaline Pretreatment
4.3. Effect of Temperature, Residence Time, and Acid/Alkaline Concentration on the Pretreatment Yield
4.4. Acidic, Alkaline, and Two-Stage Acid–Alkaline/Alkaline–Acid Pretreatment of Perennial Grasses
| Biomass | Pretreatment Efficiency % [w/w] | Enzymatic Hydrolysis and Fermentation | Hydrolysis Efficiency/Total Reducing Sugar Concentration | Bioethanol Yield | Reference |
|---|---|---|---|---|---|
| Napier grass | Alkaline () CR: 62.1 ± 2.7% HR: 67.1 ± 0.2% LR: 63.0 ± 3.2% | Cellulases Cellic CTec2 and HTec2 12 FPU/g. S. cerevisiae SSF | Glucose: 51.60 g/L Xylose: 13.50 g/L Glucose yield: 75.4 ± 2.9% | 0.143 ± 0.006 g/g | [26] |
| Acidic () CR: 47.8% HR: 81.1% LR: 46.2% | Glucose: 29.20 g/L Xylose: 5.10 g/L Glucose yield: 55.4 ± 6.5% | 0.075 ± 0.003 g/g | |||
| Acidic-Alkaline – CR: 67.6% HR: 93.4% LR: 74.1% | Glucose: 56.90 g/L Xylose: 4.20 g/L Glucose yield: 76.4 ± 5.0% | 0.116 ± 0.006 g/g | |||
| Vetiver | Acidic-Alkaline (–) | Cellulase (8 U/g), β-glucosidase (120 U/g). S. cerevisiae SSF | Glucose: 11.08–13.56% Xylose: 44.21–47.84% Arabinose: 15.41–16.34% Galactose: 6.51–7.88% | 0.142–0.151 g/g | [59] |
| Napier grass | Alkaline () CR: 96.5–99% HR: 19.7–82.7% LR: 76.2–88.4% | Accellerase 1500, 30 FPU/g. S. cerevisiae SSF | TRS: 146.90 mg/g | 0.142 g/g (With ) | [12] |
| Acidic () | TRS: 80.00–90.00 mg/g | ||||
| Alkaline (aqueous ) | TRS: 133.60 mg/g | ||||
| Alkaline (Alkaline peroxide) | TRS: 20.00–30.00 mg/g | ||||
| King grass | Alkaline () CR: 55.8–92.8% HR: 22.6–98.4% LR: 53.00–94.00% | Accellerase 1500, 30 FPU/g. S. cerevisiae SSF | TRS: 268.00 mg/g | 0.166 g/g With | [35] |
| Acidic () | TRS: ~120.00 mg/g | ||||
| Alkaline (aqueous ) | TRS: 179.40 mg/g | ||||
| Alkaline (Alkaline peroxide) | TRS: ~50.00 mg/g | ||||
| Switchgrass | Alkaline Aqueous LR: 40.0–50.0% | Cellulase β-glucosidase, 26–77 FPU/mL. S. cerevisiae SSF | Glucose: ~6.40 g/L Xylose: ~7.90 g/L Cellobiose: ~3.80 g/L | 22.00 g/L | [99] |
| Napier grass | Alkaline () LR: 63.79–81.03% | Cellulase 20 FPU/g | Glucose: 18.40 g/L Glucose yield: 45.2–50.6% Xylose yield: 24.0–32.2% | - | [107] |
| Para grass | Acidic () | Commercial Cellulase 20 FPU/g. S. cerevisiae SHF | TRS: 660.00 mg/g | - | [58] |
| Alkaline () | TRS: 696.00 mg/g | 71.8% | |||
| Kans grass | 2-stage – LR: 47.9% | Yeast strain Scheffersomyces stipitis CBS 6054 | TR: 44.30–373.60 mg/g | 0.35 g/g | [17] |
| 2-stage + urea– LR: 51.0% | TRS: 75.20–448.30 mg/g | ||||
| 2-stage Aqueous – LR: 48.0% | TRS: 159.70–580.30 mg/g | ||||
| B. brizantha | Acidic () HR: 73.3% | Commercial cellulase, 20 FPU/g. S. cerevisiae SSF | TRS: 0.19–0.54 g/g Saccharification efficiency: 46.0% | ~6.50 g/L | [23] |
| Bermuda grass | Acidic () LR: 20.0–30.0% | Cellulase, NS50013 (40 FPU/g) and cellobiase NS50010 (70 CBU/g). S. cerevisiae | TRS: 464.80 mg/g TRS yield: 80.00–97.00% | 52.00–83.00% | [110] |
| Bermuda grass | Acidic () | Cellulases (25 FPU/g) and β -glucosidase (75IU/g) | TRS: 203.70–229.30 mg/g Glucose conversion rate; 46.1–83.1% | - | [124] |
| Rye straw | Acidic () | Cellulases (25 FPU/g) and β -glucosidase (75IU/g) | TRS: 125.00–197.10 mg/g Glucose yield: 29.9–52.3% | - | [124] |
| Cogon grass | Alkaline () | Accellerase 150 (0.15-0.25 mL/g-WIS). S. cerevisiae SSF | - | 19.08 g/L (76.2%) | [122] |
| Napier grass | Alkaline () | cellulase Cellic® CTec2 (5–40 FPU/g). S. cerevisiae SHF and SSF | TRS: ~90.00 g/L maximum (SSF) | 30.60 ± 0.40 g/L (SHF) 28.50 ± 2.30 g/L (SSF) | [125] |
| Switchgrass | Acidic () HR: >80.0% | NS 50013 (20 FPU/g) + NS 50010 (10 CBU/g) | TRS: 188.70 mg/g | - | [16] |
| Celli CTec (20 FPU/g) | TRS: 207.50 mg/g | ||||
| Alkaline () LR: >55.0% | NS 50013 (15 FPU/g) + NS 50010 (20 CBU/g) | TRS: 431.40 mg/g | |||
| Cellic CTec (15 FPU/g) + Cellic HTec (25 FXU/g) | TRS: 399.00 mg/g | ||||
| Alkaline ) | NS 50013 (20 FPU/g) + NS 50010 (20 CBU/g) | TRS: 433.00 mg/g | |||
| Cellic CTec (20 FPU/g) + Cellic HTec (25 FXU/g) | TRS: 412.30 mg/g | ||||
| Bermuda grass | Acidic () | NS 50013 (20 FPU/g) + NS 50010 (10 CBU/g) | TRS: 232.10 mg/g | ||
| Celli CTec (20 FPU/g) | TRS: 242.10 mg/g | ||||
| Alkaline () | NS 50013 (15 FPU/g) + NS 50010 (10 CBU/g) | TRS: 396.00 mg/g | |||
| Cellic CTec (10 FPU/g) + Cellic HTec (25 FXU/g) | TRS: 379.50 mg/g | ||||
| Alkaline ( | NS 50013 (20 FPU/g) + NS 50010 (10 CBU/g) | TRS: 426.40 mg/g | |||
| Cellic CTec (15 FPU/g) + Cellic HTec (50 FXU/g) | TRS: 429.70 mg/g | ||||
| Miscanthus floridulus | Alkaline () at RT. HR: 51.5% LR: 73.7% | Accellerase 1000, 10 FPU/g-WIS. S. cerevisiae SSF | 0.071 g/g (48.9%) | [126] | |
| Alkaline () at 90 °C. HR: 85.1% LR: 61.5% | 0.124 g/g (78.4%) |
| Pretreatment | Advantages | Disadvantages | References |
|---|---|---|---|
| Acidic | Short residence times. Mild temperatures and low-pressure operations. Eliminating the need for enzymatic hydrolysis is possible sometimes, as the pretreated feedstock is directly hydrolyzed into fermentable sugars. Achieves high rates of cellulose hydrolysis. | The generation of inhibitory compounds is very high. Requires washing and neutralizing of the acid in the feedstock before enzymatic hydrolysis. High cost to design reactors due to acid-resistant material. Need to recover the acid used. Corrosive. Toxic. | [47,75,112,127] |
| Alkaline | Less degradation of sugars. Low temperature and low-pressure operation. Enhances enzymatic hydrolysis. Low inhibitory compounds generation. Reuse of alkaline solution. | Ideal for low lignin content biomass. Difficult to neutralize. High cost. Extended reaction time. | [38,47,70,112,122,128] |
4.5. Perennial Grasses Pretreatment Comparison with Other LCB
4.6. Inhibitory Compounds Production
4.7. Effect of Enzymatic Hydrolysis and Fermentation on Reducing Sugar Concentrations and Ethanol Yields
4.8. Water Consumption of the Pretreatment Process
| Biomass | Pretreatment | Water usage after Treatment | Reference |
|---|---|---|---|
| Miscanthus | Solid residues washed thoroughly with water until neutral pH. | [146] | |
| Switchgrass | Hot water | Pretreated residue washed with 10 g of distilled water per gram of solids four times; wastewater was discarded. | [147] |
| Bamboo | Glacial acetic acid | Pretreated biomass was washed with tap water; wastewater was discarded. | [148] |
| Hybrid Pennisetum | -valerolactone, tetrahydrofurfuryl alcohol, ethanol, and acetone assisted by | Pretreated biomass was first washed with an equal volume of the organic solvent at least three times then washed to neutral with water; wastewater was discarded. | [149] |
| Hybrid Pennisetum | FeCl3 in ChCl/glycerol | Pretreated biomass was washed with 50% acetone/water (100 mL) until the filtrate was colorless. | [150] |
| Napier grass | and NaOH | Pretreated biomass was washed with tap water until the pH became neutral. | [26] |
| Cogon grass | NaOH | Pretreated biomass washed with 300 mL of water in 3 stages | [36] |
5. Techno-Economic and Environmental Analysis of Bioethanol Production from Perennial Grasses
5.1. Production Costs
5.1.1. Production Cost of Perennial Grasses
5.1.2. Cost of Pretreatment
5.2. Life Cyle Assessemnt
5.2.1. Water Footprint
5.2.2. Carbon Footprint
6. Artificial Intelligence Application in Bioethanol Production from LCB
7. Challenges and Prospects
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raj, T.; Chandrasekhar, K.; Naresh Kumar, A.; Rajesh Banu, J.; Yoon, J.-J.; Kant Bhatia, S.; Yang, Y.-H.; Varjani, S.; Kim, S.-H. Recent Advances in Commercial Biorefineries for Lignocellulosic Ethanol Production: Current Status, Challenges and Future Perspectives. Bioresour. Technol. 2022, 344, 126292. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, D.M.; Sevastyanova, O.; Penna, L.S.; da Silva, B.P.; Lindström, M.E.; Colodette, J.L. Assessment of Chemical Transformations in Eucalyptus, Sugarcane Bagasse and Straw during Hydrothermal, Dilute Acid, and Alkaline Pretreatments. Ind. Crops Prod. 2015, 73, 118–126. [Google Scholar] [CrossRef]
- Papathoti, N.K.; Laemchiab, K.; Megavath, V.S.; Keshav, P.K.; Numparditsub, P.; Le Thanh, T.; Buensanteai, N. Augmented Ethanol Production from Alkali-Assisted Hydrothermal Pretreated Cassava Peel Waste. Energy Sources Part A Recovery Util. Environ. Eff. 2021, 1–11. [Google Scholar] [CrossRef]
- Takkellapati, S.; Li, T.; Gonzalez, M.A. An Overview of Biorefinery Derived Platform Chemicals from a Cellulose and Hemicellulose Biorefinery. Clean Technol. Environ. Policy 2018, 20, 1615–1630. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Xin, R.; Prasongthum, N.; Natewong, P.; Sooknoi, T.; Wang, J.; Reubroycharoen, P.; Fan, X. Catalytic Conversion of Bioethanol to Value-Added Chemicals and Fuels: A Review. Resour. Chem. Mater. 2022, 1, 47–68. [Google Scholar] [CrossRef]
- Swain, M.R.; Singh, A.; Sharma, A.K.; Tuli, D.K. Bioethanol production from rice-and wheat straw: An overview. In Bioethanol Production from Food Crops; Ray, R.C., Ramachandran, S., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 213–231. [Google Scholar] [CrossRef]
- Malode, S.J.; Prabhu, K.K.; Mascarenhas, R.J.; Shetti, N.P.; Aminabhavi, T.M. Recent Advances and Viability in Biofuel Production. Energy Convers. Manag. X 2021, 10, 100070. [Google Scholar] [CrossRef]
- Deshavath, N.; Goud, V.; Dasu, V. Commercialization of 2G Bioethanol as a Transportation Fuel for the Sustainable Energy, Environment, and Economic Growth of India: Theoretical and Empirical Assessment of Bioethanol Potential from Agriculture Crop Residues. Biomass Convers. Biorefin. 2022, 14, 3551–3563. [Google Scholar] [CrossRef]
- Owusu, W.A.; Marfo, S.A. Artificial Intelligence Application in Bioethanol Production. Int. J. Energy Res. 2023, 2023, 7844835. [Google Scholar] [CrossRef]
- Bušić, A.; Marđetko, N.; Kundas, S.; Morzak, G.; Belskaya, H.; Ivančić Šantek, M.; Komes, D.; Novak, S.; Šantek, B. Bioethanol Production from Renewable Raw Materials and Its Separation and Purification: A Review. Food Technol. Biotechnol. 2018, 56, 289–311. [Google Scholar] [CrossRef] [PubMed]
- Jeyakumar, N.; Hoang, A.T.; Nižetić, S.; Balasubramanian, D.; Kamaraj, S.; Lakshmana Pandian, P.; Sirohi, R.; Nguyen, P.Q.P.; Nguyen, X.P. Experimental Investigation on Simultaneous Production of Bioethanol and Biodiesel from Macro-Algae. Fuel 2022, 329, 125362. [Google Scholar] [CrossRef]
- Eliana, C.; Jorge, R.; Juan, P.; Luis, R. Effects of the Pretreatment Method on Enzymatic Hydrolysis and Ethanol Fermentability of the Cellulosic Fraction from Elephant Grass. Fuel 2014, 118, 41–47. [Google Scholar] [CrossRef]
- Santos, C.C.; de Souza, W.; Sant’Anna, C.; Brienzo, M. Elephant Grass Leaves Have Lower Recalcitrance to Acid Pretreatment than Stems, with Higher Potential for Ethanol Production. Ind. Crops Prod. 2018, 111, 193–200. [Google Scholar] [CrossRef]
- Raud, M.; Kikas, T. Perennial Grasses as a Substrate for Bioethanol Production. Environ. Clim. Technol. 2020, 24, 32–40. [Google Scholar] [CrossRef]
- Kamzon, M.A.; Abderafi, S.; Bounahmidi, T. Promising Bioethanol Processes for Developing a Biorefinery in the Moroccan Sugar Industry. Int. J. Hydrogen Energy 2016, 41, 20880–20896. [Google Scholar] [CrossRef]
- Xu, J.; Wang, Z.; Sharma-Shivappa, R.; Cheng, J. Enzymatic Hydrolysis of Switchgrass and Coastal Bermuda Grass Pretreated Using Different Chemical Methods. Bioresources 2011, 6, 2990–3003. [Google Scholar] [CrossRef]
- Chaudhary, G.; Singh, L.K.; Ghosh, S. Alkaline Pretreatment Methods Followed by Acid Hydrolysis of Saccharum Spontaneum for Bioethanol Production. Bioresour. Technol. 2012, 124, 111–118. [Google Scholar] [CrossRef] [PubMed]
- García, J.C.; Alfaro, A.; Loaiza, J.M.; Lozano-Calvo, S.; López, F. Cold Alkaline Extraction of Elephant Grass for Optimal Subsequent Extraction of Hemicelluloses and Energy Production. Biomass Convers. Biorefin. 2022, 1–14. [Google Scholar] [CrossRef]
- Larran, A.; Jozami, E.; Vicario, L.; Feldman, S.R.; Podestá, F.E.; Permingeat, H.R. Evaluation of Biological Pretreatments to Increase the Efficiency of the Saccharification Process Using Spartina Argentinensis as a Biomass Resource. Bioresour. Technol. 2015, 194, 320–325. [Google Scholar] [CrossRef]
- Soares Rodrigues, C.I.; Jackson, J.J.; Montross, M.D. A Molar Basis Comparison of Calcium Hydroxide, Sodium Hydroxide, and Potassium Hydroxide on the Pretreatment of Switchgrass and Miscanthus under High Solids Conditions. Ind. Crops Prod. 2016, 92, 165–173. [Google Scholar] [CrossRef]
- Tye, Y.; Lee, K.T.; Wan Nadiah, W.; Peng, L.C. The World Availability of Non-Wood Lignocellulosic Biomass for the Production of Cellulosic Ethanol and Potential Pretreatments for the Enhancement of Enzymatic Saccharification. Renew. Sust. Energ. Rev. 2016, 60, 155–172. [Google Scholar] [CrossRef]
- Jozami, E.; Mele, F.D.; Piastrellini, R.; Civit, B.M.; Feldman, S.R. Life Cycle Assessment of Bioenergy from Lignocellulosic Herbaceous Biomass: The Case Study of Spartina Argentinensis. Energy 2022, 254, 124215. [Google Scholar] [CrossRef]
- Rodrigues, R.S.; de Almeida, M.N.; Maitan-Alfenas, G.P.; Ventorim, R.Z.; Sartori, S.R.; Visser, E.M.; Guimarães, V.M.; de Rezende, S.T. Brachiaria Brizantha Grass as a Feedstock for Ethanol Production. Braz. Arch. Biol. Technol. 2021, 64, 1–13. [Google Scholar] [CrossRef]
- Himmel, M.E.; Ding, S.-Y.; Johnson, D.K.; Adney, W.S.; Nimlos, M.R.; Brady, J.W.; Foust, T.D. Biomass Recalcitrance: Engineering Plants and Enzymes for Biofuels Production. Science (1979) 2007, 315, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, Z.; Cheng, J.J. Bermuda Grass as Feedstock for Biofuel Production: A Review. Bioresour. Technol. 2011, 102, 7613–7620. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.-H.; Lee, W.-C.; Kuan, W.-C.; Sirisansaneeyakul, S.; Savarajara, A. Evaluation of Different Pretreatments of Napier Grass for Enzymatic Saccharification and Ethanol Production. Energy Sci. Eng. 2018, 6, 683–692. [Google Scholar] [CrossRef]
- da Silva, V.; Daher, R.F.; de Souza, Y.P.; da Silva Menezes, B.R.; Azevedo Santos, E.; Souza Freitas, R.; da Silva Oliveira, E.; Francesconi Stida, W.; Cassaro, S. Assessment of Energy Production in Full-Sibling Families of Elephant Grass by Mixed Models. Renew. Energy 2020, 146, 744–749. [Google Scholar] [CrossRef]
- Collazzo, G.C.; Broetto, C.C.; Perondi, D.; Junges, J.; Dettmer, A.; Dornelles Filho, A.A.; Foletto, E.L.; Godinho, M. A Detailed Non-Isothermal Kinetic Study of Elephant Grass Pyrolysis from Different Models. Appl. Therm. Eng. 2017, 110, 1200–1211. [Google Scholar] [CrossRef]
- Tang, S.; Cao, Y.; Xu, C.; Wu, Y.; Li, L.; Ye, P.; Luo, Y.; Gao, Y.; Liao, Y.; Yan, Q.; et al. One-Step or Two-Step Acid/Alkaline Pretreatments to Improve Enzymatic Hydrolysis and Sugar Recovery from Arundo Donax L. Energies 2020, 13, 948. [Google Scholar] [CrossRef]
- Tang, S.; Xu, C.; Vu, L.T.K.; Liu, S.; Ye, P.; Li, L.; Wu, Y.; Chen, M.; Xiao, Y.; Wu, Y.; et al. Enhanced Enzymatic Hydrolysis of Pennisetum Alopecuroides by Dilute Acid, Alkaline and Ferric Chloride Pretreatments. Molecules 2019, 24, 1751. [Google Scholar] [CrossRef]
- Umami, N.; Widodo, S.; Suhartanto, B.; Suwignyo, B.; Suseno, N.; Noviandi, C. The Effect of Planting Material on Nutrient Quality and Production of Brachiaria Spp. in Yogyakarta, Indonesia. Pak. J. Nutr. 2018, 17, 671–676. [Google Scholar] [CrossRef]
- Ask, M.; Olofsson, K.; Di Felice, T.; Ruohonen, L.; Penttilä, M.; Lidén, G.; Olsson, L. Challenges in Enzymatic Hydrolysis and Fermentation of Pretreated Arundo Donax Revealed by a Comparison between SHF and SSF. Process Biochem. 2012, 47, 1452–1459. [Google Scholar] [CrossRef]
- Lewandowski, I.; Scurlock, J.M.O.; Lindvall, E.; Christou, M. The Development and Current Status of Perennial Rhizomatous Grasses as Energy Crops in the US and Europe. Biomass Bioenergy 2003, 25, 335–361. [Google Scholar] [CrossRef]
- Chandel, A.K.; Singh, O.V. Weedy Lignocellulosic Feedstock and Microbial Metabolic Engineering: Advancing the Generation of “Biofuel”. Appl. Microbiol. Biotechnol. 2011, 89, 1289–1303. [Google Scholar] [CrossRef] [PubMed]
- Cardona, E.; Rios, J.; Peña, J.; Peñuela, M.; Rios, L. King Grass: A Very Promising Material for the Production of Second Generation Ethanol in Tropical Countries. Biomass Bioenergy 2016, 95, 206–213. [Google Scholar] [CrossRef]
- Goshadrou, A. Bioethanol Production from Cogongrass by Sequential Recycling of Black Liquor and Wastewater in a Mild-Alkali Pretreatment. Fuel 2019, 258, 116141. [Google Scholar] [CrossRef]
- Chen, W.-H.; Nižetić, S.; Sirohi, R.; Huang, Z.; Luque, R.; M.Papadopoulos, A.; Sakthivel, R.; Phuong Nguyen, X.; Tuan Hoang, A. Liquid Hot Water as Sustainable Biomass Pretreatment Technique for Bioenergy Production: A Review. Bioresour. Technol. 2022, 344, 126207. [Google Scholar] [CrossRef]
- Zhao, L.; Sun, Z.-F.; Zhang, C.-C.; Nan, J.; Ren, N.-Q.; Lee, D.-J.; Chen, C. Advances in Pretreatment of Lignocellulosic Biomass for Bioenergy Production: Challenges and Perspectives. Bioresour. Technol. 2022, 343, 126123. [Google Scholar] [CrossRef] [PubMed]
- Ilić, N.; Milić, M.; Beluhan, S.; Dimitrijević-Branković, S. Cellulases: From Lignocellulosic Biomass to Improved Production. Energies 2023, 16, 3598. [Google Scholar] [CrossRef]
- Kang, Q.; Tan, T. Exergy and CO2 Analyses as Key Tools for the Evaluation of Bio-Ethanol Production. Sustainability 2016, 8, 76. [Google Scholar] [CrossRef]
- Sebastian, J.; Rouissi, T.; Brar, S.K. Miscanthus Sp. Perennial Lignocellulosic Biomass as Feedstock for Greener Fumaric Acid Bioproduction. Ind. Crops Prod. 2022, 175, 114248. [Google Scholar] [CrossRef]
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic Agriculture Wastes as Biomass Feedstocks for Second-Generation Bioethanol Production: Concepts and Recent Developments. 3 Biotech 2015, 5, 337–353. [Google Scholar] [CrossRef]
- Rajesh Banu, J.; Preethi; Kavitha, S.; Tyagi, V.K.; Gunasekaran, M.; Karthikeyan, O.P.; Kumar, G. Lignocellulosic Biomass Based Biorefinery: A Successful Platform towards Circular Bioeconomy. Fuel 2021, 302, 121086. [Google Scholar] [CrossRef]
- Xu, Z.; Huang, F. Pretreatment Methods for Bioethanol Production. Appl. Biochem. Biotechnol. 2014, 174, 43–62. [Google Scholar] [CrossRef] [PubMed]
- Pendse, D.S.; Deshmukh, M.; Pande, A. Different Pre-Treatments and Kinetic Models for Bioethanol Production from Lignocellulosic Biomass: A Review. Heliyon 2023, 9, 16604. [Google Scholar] [CrossRef] [PubMed]
- Putro, J.N.; Soetaredjo, F.E.; Lin, S.-Y.; Ju, Y.-H.; Ismadji, S. Pretreatment and Conversion of Lignocellulose Biomass into Valuable Chemicals. RSC Adv. 2016, 6, 46834–46852. [Google Scholar] [CrossRef]
- Hassan, S.S.; Williams, G.A.; Jaiswal, A.K. Emerging Technologies for the Pretreatment of Lignocellulosic Biomass. Bioresour. Technol. 2018, 262, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Karimi, K.; Taherzadeh, M.J. A Critical Review of Analytical Methods in Pretreatment of Lignocelluloses: Composition, Imaging, and Crystallinity. Bioresour. Technol. 2016, 200, 1008–1018. [Google Scholar] [CrossRef]
- Sluiter, J.B.; Ruiz, R.O.; Scarlata, C.J.; Sluiter, A.D.; Templeton, D.W. Compositional Analysis of Lignocellulosic Feedstocks. 1. Review and Description of Methods. J. Agric. Food Chem. 2010, 58, 9043–9053. [Google Scholar] [CrossRef]
- Chesson, A. Effects of Sodium Hydroxide on Cereal Straws in Relation to the Enhanced Degradation of Structural Polysaccharides by Rumen Microorganisms. J. Sci. Food Agric. 1981, 32, 745–758. [Google Scholar] [CrossRef]
- Novia, N.; Hasanudin, H.; Hermansyah, H.; Fudholi, A. Kinetics of Lignin Removal from Rice Husk Using Hydrogen Peroxide and Combined Hydrogen Peroxide–Aqueous Ammonia Pretreatments. Fermentation 2022, 8, 157. [Google Scholar] [CrossRef]
- Hermansyah, H.; Cahyadi, H.; Fatma, F.; Miksusanti, M.; Kasmiarti, G.; Panagan, A.T. Delignification of Lignocellulosic Biomass Sugarcane Bagasse by Using Ozone as Initial Step to Produce Bioethanol. Pol. J. Environ. Stud. 2021, 30, 4405–4411. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass, in: Laboratory Analytical Procedure (LAP). Natl. Renew. Energy Lab. 2008, 1617, 1–16. [Google Scholar]
- Sluiter, A.; Sluiter, J.; Wolfrum, E.J. Methods for Biomass Compositional Analysis. In Catalysis for the Conversion of Biomass and Its Derivatives; Max-Planck-Gesellschaft zur Förderung der Wissenschaften: Berlin, Germany, 2013; p. 476. ISBN 978-3-945561-19-5. [Google Scholar]
- Sun, Y.; Cheng, J. Hydrolysis of Lignocellulosic Materials for Ethanol Production: A Review. Bioresour. Technol. 2002, 83, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Ghosh, S. Bioethanol Production from Various Lignocellulosic Feedstocks by a Novel “Fractional Hydrolysis” Technique with Different Inorganic Acids and Co-Culture Fermentation. Fuel 2019, 236, 544–553. [Google Scholar] [CrossRef]
- Raud, M.; Tutt, M.; Olt, J.; Kikas, T. Dependence of the Hydrolysis Efficiency on the Lignin Content in Lignocellulosic Material. Int. J. Hydrogen Energy 2016, 41, 16338–16343. [Google Scholar] [CrossRef]
- Sahoo, D.; Ummalyma, S.B.; Okram, A.K.; Sukumaran, R.K.; George, E.; Pandey, A. Potential of Brachiaria mutica (Para Grass) for Bioethanol Production from Loktak Lake. Bioresour. Technol. 2017, 242, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Neve, S.; Sarkar, D.; Zhang, Z.; Datta, R. Optimized Production of Second-Generation Bioethanol from a Spent C4 Grass: Vetiver (Chrysopogon zizanioides). Energies 2022, 15, 9597. [Google Scholar] [CrossRef]
- Zoghlami, A.; Paës, G. Lignocellulosic Biomass: Understanding Recalcitrance and Predicting Hydrolysis. Front. Chem. 2019, 7, 874. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for Pretreatment of Lignocellulosic Biomass for Efficient Hydrolysis and Biofuel Production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Hallac, B.B.; Ragauskas, A.J. Analyzing Cellulose Degree of Polymerization and Its Relevancy to Cellulosic Ethanol. Biofuels Bioprod. Biorefin. 2011, 5, 215–225. [Google Scholar] [CrossRef]
- Hosseini Koupaie, E.; Dahadha, S.; Bazyar Lakeh, A.A.; Azizi, A.; Elbeshbishy, E. Enzymatic Pretreatment of Lignocellulosic Biomass for Enhanced Biomethane Production—A Review. J. Environ. Manag. 2019, 233, 774–784. [Google Scholar] [CrossRef]
- Galbe, M.; Zacchi, G. Pretreatment: The Key to Efficient Utilization of Lignocellulosic Materials. Biomass Bioenergy 2012, 46, 70–78. [Google Scholar] [CrossRef]
- Eo, M.; Fan, H.; Cho, Y.; Kim, S.; Lee, S. Cellulose Membrane as a Biomaterial: From Hydrolysis to Depolymerization with Electron Beam. Biomater. Res. 2016, 20, 16. [Google Scholar] [CrossRef]
- Sharma, H.K.; Xu, C.; Qin, W. Biological Pretreatment of Lignocellulosic Biomass for Biofuels and Bioproducts: An Overview. Waste Biomass Valorization 2019, 10, 235–251. [Google Scholar] [CrossRef]
- Saha, B. Hemicellulose Bioconversion. J. Ind. Microbiol. Biotechnol. 2003, 30, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Muktham, R.; Bhargava, S.; Bankupalli, S.; Ball, A. A Review on 1st and 2nd Generation Bioethanol Production-Recent Progress. J. Sustain. Bioenergy Syst. 2016, 6, 72–92. [Google Scholar] [CrossRef]
- Zhang, Z.; Donaldson, A.A.; Ma, X. Advancements and Future Directions in Enzyme Technology for Biomass Conversion. Biotechnol. Adv. 2012, 30, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Kumari, D.; Singh, R. Pretreatment of Lignocellulosic Wastes for Biofuel Production: A Critical Review. Renew. Sustain. Energy Rev. 2018, 90, 877–891. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, X.; Li, H.; Lu, J.; Liu, M.; Wang, F. Carbon Modification of Nickel Catalyst for Depolymerization of Oxidized Lignin to Aromatics. ACS Catal. 2018, 8, 1614–1620. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, M.; Roozeboom, K.; Wang, D. Integrated Bioethanol Production to Boost Low-Concentrated Cellulosic Ethanol without Sacrificing Ethanol Yield. Bioresour. Technol. 2018, 250, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Digman, M.F.; Shinners, K.; Muck, R.E.; Dien, B. Pilot-Scale On-Farm Pretreatment of Perennial Grasses with Dilute Acid and Alkali for Fuel Ethanol Production. Trans. ASABE 2010, 53, 1007–1014. [Google Scholar] [CrossRef]
- Lee, J.; Kim, S.; You, S.; Park, Y.-K. Bioenergy Generation from Thermochemical Conversion of Lignocellulosic Biomass-Based Integrated Renewable Energy Systems. Renew. Sustain. Energy Rev. 2023, 178, 113240. [Google Scholar] [CrossRef]
- Soltanian, S.; Aghbashlo, M.; Almasi, F.; Hosseinzadeh-Bandbafha, H.; Nizami, D.A.-S.; Ok, Y.S.; Lam, S.S.; Tabatabaei, M. A Critical Review of the Effects of Pretreatment Methods on the Exergetic Aspects of Lignocellulosic Biofuels. Energy Convers. Manag. 2020, 212, 112792. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, Y.Y.; Kim, T.H. A Review on Alkaline Pretreatment Technology for Bioconversion of Lignocellulosic Biomass. Bioresour. Technol. 2016, 199, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Raj, T.; Vijayaraj, M.; Chopra, A.; Gupta, R.P.; Tuli, D.K.; Kumar, R. Structural Features of Dilute Acid, Steam Exploded, and Alkali Pretreated Mustard Stalk and Their Impact on Enzymatic Hydrolysis. Carbohydr. Polym. 2015, 124, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Sannigrahi, P.; Pu, Y.; Ragauskas, A. Cellulosic Biorefineries—Unleashing Lignin Opportunities. Curr. Opin. Environ. Sustain. 2010, 2, 383–393. [Google Scholar] [CrossRef]
- Ravindran, R.; Jaiswal, A.K. A Comprehensive Review on Pre-Treatment Strategy for Lignocellulosic Food Industry Waste: Challenges and Opportunities. Bioresour. Technol. 2016, 199, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Banu, J.R.; Sugitha, S.; Kavitha, S.; Kannah, R.Y.; Merrylin, J.; Kumar, G. Lignocellulosic Biomass Pretreatment for Enhanced Bioenergy Recovery: Effect of Lignocelluloses Recalcitrance and Enhancement Strategies. Front. Energy Res. 2021, 9, 646057. [Google Scholar] [CrossRef]
- Galbe, M.; Wallberg, O. Pretreatment for Biorefineries: A Review of Common Methods for Efficient Utilisation of Lignocellulosic Materials. Biotechnol. Biofuels 2019, 12, 294. [Google Scholar] [CrossRef]
- Anukam, A.; Berghel, J. Biomass Pretreatment and Characterization: A Review. In Biotechnological Applications of Biomass; Basso, T.P., Basso, T.O., Basso, L.C., Eds.; IntechOpen: Rijeka, Croatia, 2020; pp. 1–17. ISBN 978-1-83881-182-2. [Google Scholar] [CrossRef]
- Kumar, A.K.; Sharma, S. Recent Updates on Different Methods of Pretreatment of Lignocellulosic Feedstocks: A Review. Bioresour. Bioprocess. 2017, 4, 7. [Google Scholar] [CrossRef]
- Vasco-Correa, J.; Zuleta-Correa, A.; Gómez-León, J.; Pérez-Taborda, J.A. Advances in Microbial Pretreatment for Biorefining of Perennial Grasses. Appl. Microbiol. Biotechnol. 2023, 107, 5281–5300. [Google Scholar] [CrossRef]
- Beluhan, S.; Mihajlovski, K.; Šantek, B.; Ivančić Šantek, M. The Production of Bioethanol from Lignocellulosic Biomass: Pretreatment Methods, Fermentation, and Downstream Processing. Energies 2023, 16, 7003. [Google Scholar] [CrossRef]
- Xu, J.; Cheng, J.J.; Sharma-Shivappa, R.R.; Burns, J.C. Lime Pretreatment of Switchgrass at Mild Temperatures for Ethanol Production. Bioresour. Technol. 2010, 101, 2900–2903. [Google Scholar] [CrossRef]
- Rezania, S.; Oryani, B.; Cho, J.; Talaiekhozani, A.; Sabbagh, F.; Hashemi, B.; Rupani, P.F.; Mohammadi, A.A. Different Pretreatment Technologies of Lignocellulosic Biomass for Bioethanol Production: An Overview. Energy 2020, 199, 117457. [Google Scholar] [CrossRef]
- Han, S.H.; Cho, D.H.; Kim, Y.H.; Shin, S.J. Biobutanol Production from 2-Year-Old Willow Biomass by Acid Hydrolysis and Acetone–Butanol–Ethanol Fermentation. Energy 2013, 61, 13–17. [Google Scholar] [CrossRef]
- Alvira, P.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment Technologies for an Efficient Bioethanol Production Process Based on Enzymatic Hydrolysis: A Review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef] [PubMed]
- Solarte-Toro, J.C.; Romero-García, J.M.; Martínez-Patiño, J.C.; Ruiz-Ramos, E.; Castro-Galiano, E.; Cardona-Alzate, C.A. Acid Pretreatment of Lignocellulosic Biomass for Energy Vectors Production: A Review Focused on Operational Conditions and Techno-Economic Assessment for Bioethanol Production. Renew. Sustain. Energy Rev. 2019, 107, 587–601. [Google Scholar] [CrossRef]
- Hoon-Jung, Y.; Heon-Kim, K. Acidic Pretreatment. In Pretreatment of Biomass: Processes and Technologies; Academic Press: Cambridge, MA, USA, 2015; pp. 27–50. [Google Scholar]
- Chiaramonti, D.; Prussi, M.; Ferrero, S.; Oriani, L.; Ottonello, P.; Torre, P.; Cherchi, F. Review of Pretreatment Processes for Lignocellulosic Ethanol Production, and Development of an Innovative Method. Biomass Bioenergy 2012, 46, 25–35. [Google Scholar] [CrossRef]
- Romero-García, J.M.; Niño, L.; Martínez-Patiño, C.; Álvarez, C.; Castro, E.; Negro, M.J. Biorefinery Based on Olive Biomass. State of the Art and Future Trends. Bioresour. Technol. 2014, 159, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Chiranjeevi, T.; Mattam, A.J.; Vishwakarma, K.K.; Uma, A.; Peddy, V.C.R.; Gandham, S.; Ravindra Velankar, H. Assisted Single-Step Acid Pretreatment Process for Enhanced Delignification of Rice Straw for Bioethanol Production. ACS Sustain. Chem. Eng. 2018, 6, 8762–8774. [Google Scholar] [CrossRef]
- Fuertez, J.; Acosta-Pavas, J.; Ruiz, A. Alkaline Delignification of Lignocellulosic Biomass for the Production of Fermentable Sugar Syrups. Dyna 2021, 88, 168–177. [Google Scholar] [CrossRef]
- Wang, Z.; Keshwani, D.R.; Redding, A.P.; Cheng, J.J. Sodium Hydroxide Pretreatment and Enzymatic Hydrolysis of Coastal Bermuda Grass. Bioresour. Technol. 2010, 101, 3583–3585. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Wu, M.; Lin, Y.; Yang, L.; Lin, J.; Cen, P. Artificial Symbiosis for Acetone-Butanol-Ethanol (ABE) Fermentation from Alkali Extracted Deshelled Corn Cobs by Co-Culture of Clostridium Beijerinckii and Clostridium Cellulovorans. Microb. Cell Fact. 2014, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Zdeb, M.; Pawłowska, M.; Skóra, A. Influence of Temperature and Reaction Time on the Efficiency of Alkaline Pretreatment of Hay Biomass. J. Ecol. Eng. 2021, 22, 120–127. [Google Scholar] [CrossRef]
- Isci, A.; Himmelsbach, J.N.; Pometto, A.L.; Raman, D.R.; Anex, R.P. Aqueous Ammonia Soaking of Switchgrass Followed by Simultaneous Saccharification and Fermentation. Appl. Biochem. Biotechnol. 2008, 144, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Digman, M.F.; Shinners, K.J.; Casler, M.D.; Dien, B.S.; Hatfield, R.D.; Jung, H.-J.G.; Muck, R.E.; Weimer, P.J. Optimizing On-Farm Pretreatment of Perennial Grasses for Fuel Ethanol Production. Bioresour. Technol. 2010, 101, 5305–5314. [Google Scholar] [CrossRef] [PubMed]
- Talebnia, F.; Karakashev, D.; Angelidaki, I. Production of Bioethanol from Wheat Straw: An Overview on Pretreatment, Hydrolysis and Fermentation. Bioresour. Technol. 2010, 101, 4744–4753. [Google Scholar] [CrossRef] [PubMed]
- Devi, A.; Singh, A.; Bajar, S.; Pant, D.; Din, Z.U. Ethanol from Lignocellulosic Biomass: An in-Depth Analysis of Pre-Treatment Methods, Fermentation Approaches and Detoxification Processes. J. Environ. Chem. Eng. 2021, 9, 105798. [Google Scholar] [CrossRef]
- Zabed, H.; Sahu, J.N.; Suely, A.; Boyce, A.N.; Faruq, G. Bioethanol Production from Renewable Sources: Current Perspectives and Technological Progress. Renew. Sustain. Energy Rev. 2017, 71, 475–501. [Google Scholar] [CrossRef]
- Miyuranga, K.A.V.; De Silva, S.J.; Arachchige, U.; Jayasinghe, R.A.; Weerasekara, N.A. Comparison of the Properties of Biodiesel-Bioethanol-Diesel Blended Fuel. Asian J. Chem. 2022, 34, 1809–1813. [Google Scholar] [CrossRef]
- Olson, D.G.; McBride, J.E.; Joe Shaw, A.; Lynd, L.R. Recent Progress in Consolidated Bioprocessing. Curr. Opin. Biotechnol. 2012, 23, 396–405. [Google Scholar] [CrossRef]
- Sharma, S.; Tsai, M.L.; Sharma, V.; Sun, P.-P.; Nargotra, P.; Bajaj, B.K.; Chen, C.-W.; Dong, C.-D. Environment Friendly Pretreatment Approaches for the Bioconversion of Lignocellulosic Biomass into Biofuels and Value-Added Products. Environments 2023, 10, 6. [Google Scholar] [CrossRef]
- Haldar, D.; Purkait, M.K. Thermochemical Pretreatment Enhanced Bioconversion of Elephant Grass (Pennisetum purpureum): Insight on the Production of Sugars and Lignin. Biomass Convers. Biorefin. 2022, 12, 1125–1138. [Google Scholar] [CrossRef]
- Xu, J.; Cheng, J.J.; Sharma-Shivappa, R.R.; Burns, J.C. Sodium Hydroxide Pretreatment of Switchgrass for Ethanol Production. Energy Fuels 2010, 24, 2113–2119. [Google Scholar] [CrossRef]
- Phitsuwan, P.; Sakka, K.; Ratanakhanokchai, K. Structural Changes and Enzymatic Response of Napier Grass (Pennisetum purpureum) Stem Induced by Alkaline Pretreatment. Bioresour. Technol. 2016, 218, 247–256. [Google Scholar] [CrossRef]
- Redding, A.; Wang, Z.; Keshwani, D.; Cheng, J. High Temperature Dilute Acid Pretreatment of Coastal Bermuda Grass for Enzymatic Hydrolysis. Bioresour. Technol. 2010, 102, 1415–1424. [Google Scholar] [CrossRef]
- Kaschuk, J.; Santos, D.; Frollini, E.; Canduri, F.; Porto, A. Influence of PH, Temperature, and Sisal Pulp on the Production of Cellulases from Aspergillus Sp. CBMAI 1198 and Hydrolysis of Cellulosic Materials with Different Hemicelluloses Content, Crystallinity, and Average Molar Mass. Biomass Convers. Biorefin. 2020, 10, 483–494. [Google Scholar] [CrossRef]
- Maurya, D.P.; Singla, A.; Negi, S. An Overview of Key Pretreatment Processes for Biological Conversion of Lignocellulosic Biomass to Bioethanol. 3 Biotech 2015, 5, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Chinwatpaiboon, P.; Doolayagovit, I.; Boonsombuti, A.; Savarajara, A.; Luengnaruemitchai, A. Comparison of Acid-, Alkaline-, and Ionic Liquid–Treated Napier Grass as an Immobilization Carrier for Butanol Production by Clostridium Beijerinckii JCM 8026. Biomass Convers. Biorefin. 2020, 10, 1071–1082. [Google Scholar] [CrossRef]
- Reddy, K.O.; Maheswari, C.U.; Reddy, D.J.P.; Rajulu, A.V. Thermal Properties of Napier Grass Fibers. Mater. Lett. 2009, 63, 2390–2392. [Google Scholar] [CrossRef]
- Wang, J.; Xin, D.; Hou, X.; Wu, J.; Fan, X.; Li, K.; Zhang, J. Structural Properties and Hydrolysabilities of Chinese Pennisetum and Hybrid Pennisetum: Effect of Aqueous Ammonia Pretreatment. Bioresour. Technol. 2016, 199, 211–219. [Google Scholar] [CrossRef]
- Martínez-Patiño, J.C.; Ruiz, E.; Romero, I.; Cara, C.; López-Linares, J.C.; Castro, E. Combined Acid/Alkaline-Peroxide Pretreatment of Olive Tree Biomass for Bioethanol Production. Bioresour. Technol. 2017, 239, 326–335. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, S.Z.; Xu, D.H.; Gong, Y.M.; Ma, H.H.; Tang, X.Y. Review of Catalytic Supercritical Water Gasification for Hydrogen Production from Biomass. Renew. Sust. Energ. Rev. 2010, 14, 334–343. [Google Scholar] [CrossRef]
- Gong, J.; Su, J.; Cai, J.; Zou, L.; Chen, Y.; Jiang, Y.; Hu, B. Enhanced Enzymolysis and Bioethanol Yield from Tobacco Stem Waste Based on Mild Synergistic Pretreatment. Front. Energy Res. 2023, 10, 989393. [Google Scholar] [CrossRef]
- Bales, G.L.; Kellogg, D.W.; Miller, D.D. Comparison of Ammonium, Potassium, Sodium, Chloride and Hydroxyl Ions on in Vitro Dry Matter Disappearance of Milo Stalks1. J. Anim. Sci. 1979, 49, 1324–1330. [Google Scholar] [CrossRef]
- Larnaudie, V.; Ferrari, M.D.; Lareo, C. Switchgrass as an Alternative Biomass for Ethanol Production in a Biorefinery: Perspectives on Technology, Economics and Environmental Sustainability. Renew. Sust. Energ. Rev. 2022, 158, 112115. [Google Scholar] [CrossRef]
- Guo, B.; Zhang, Y.; Yu, G.; Lee, W.-H.; Jin, Y.; Morgenroth, E. Two-Stage Acidic–Alkaline Hydrothermal Pretreatment of Lignocellulose for the High Recovery of Cellulose and Hemicellulose Sugars. Appl. Biochem. Biotechnol. 2013, 169, 1069–1087. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lee, W.-C. Simultaneous Saccharification and Fermentation of Alkali-Pretreated Cogongrass for Bioethanol Production. Bioresources 2011, 6, 2744–2756. [Google Scholar] [CrossRef]
- Sahoo, K.; Mani, S.; Das, L.; Bettinger, P. GIS-Based Assessment of Sustainable Crop Residues for Optimal Siting of Biogas Plants. Biomass Bioenergy 2018, 110, 63–74. [Google Scholar] [CrossRef]
- Sun, Y.; Cheng, J. Dilute Acid Pretreatment of Rye Straw and Bermudagrass for Ethanol Production. Bioresour. Technol. 2005, 96, 1599–1606. [Google Scholar] [CrossRef]
- Kongkeitkajorn, M.B.; Yaemdeeka, R.; Chaiyota, I.; Hamsupo, K.; Oraintara, A.; Reungsang, A. Bioethanol from Napier Grass Employing Different Fermentation Strategies to Evaluate a Suitable Operation for Batch Bioethanol Production. Energy Convers. Manag. X 2021, 12, 100143. [Google Scholar] [CrossRef]
- Yeh, R.H.; Lin, Y.S.; Wang, T.H.; Kuan, W.C.; Lee, W.C. Bioethanol Production from Pretreated Miscanthus Floridulus Biomass by Simultaneous Saccharification and Fermentation. Biomass Bioenergy 2016, 94, 110–116. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Karimi, K. Enzymatic-Based Hydrolysis Processes for Ethanol from Lignocellulosic Materials: A Review. Bioresources 2007, 2, 707–738. [Google Scholar] [CrossRef]
- Balat, M.; Balat, H.; Öz, C. Progress in Bioethanol Processing. Prog. Energy. Combust. Sci. 2008, 34, 551–573. [Google Scholar] [CrossRef]
- Guo, G.-L.; Chen, W.-H.; Chen, W.-H.; Men, L.-C.; Hwang, W.-S. Characterization of Dilute Acid Pretreatment of Silvergrass for Ethanol Production. Bioresour. Technol. 2008, 99, 6046–6053. [Google Scholar] [CrossRef]
- Vieira, S.; Barros, M.V.; Sydney, A.C.N.; Piekarski, C.M.; de Francisco, A.C.; Vandenberghe, L.P.d.S.; Sydney, E.B. Sustainability of Sugarcane Lignocellulosic Biomass Pretreatment for the Production of Bioethanol. Bioresour. Technol. 2020, 299, 122635. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.A.; Gomez, L.D.; Steele-King, C.G.; Simister, R.; Bernardinelli, O.D.; Carvalho, M.A.; Rezende, C.A.; Labate, C.A.; deAzevedo, E.R.; McQueen-Mason, S.J.; et al. Evaluating the Composition and Processing Potential of Novel Sources of Brazilian Biomass for Sustainable Biorenewables Production. Biotechnol. Biofuels 2014, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Mishra, C.; Behera, S.S.; Thatoi, H. Application of Pretreatment, Fermentation and Molecular Techniques for Enhancing Bioethanol Production from Grass Biomass—A Review. Renew. Sust. Energ. Rev. 2017, 78, 1007–1032. [Google Scholar] [CrossRef]
- Yang, B.; Dai, Z.; Ding, S.-Y.; Wyman, C.E. Enzymatic Hydrolysis of Cellulosic Biomass. Biofuels 2011, 2, 421–449. [Google Scholar] [CrossRef]
- Pothiraj, C.; Arumugam, R.; Gobinath, M. Sustaining Ethanol Production from Lime Pretreated Water Hyacinth Biomass Using Mono and Co-Cultures of Isolated Fungal Strains with Pichia Stipitis. Bioresour. Bioprocess. 2014, 1, 27. [Google Scholar] [CrossRef]
- Wang, S.; Chen, J.; Yang, G.; Gao, W.; Chen, K. Efficient Conversion of Hubrid Pennisetum to Glucose by Oxygen-Aqueous Alkaline Ionic Liquid Media Pretreatment under Benign Conditions. Bioresour. Technol. 2017, 243, 335–338. [Google Scholar] [CrossRef]
- Gao, F.; Yang, F.; De, Y.; Tao, Y.; Ta, N.; Wang, H.; Sun, Q. Dilute Alkali Pretreatment and Subsequent Enzymatic Hydrolysis of Amur Silvergrass for Ethanol Production. Bioresources 2020, 15, 4823–4834. [Google Scholar] [CrossRef]
- Singh, A.; Bishnoi, N.R. Optimization of Enzymatic Hydrolysis of Pretreated Rice Straw and Ethanol Production. Appl. Microbiol. Biotechnol. 2012, 93, 1785–1793. [Google Scholar] [CrossRef] [PubMed]
- Camesasca, L.; Ramírez, M.; Guigou, M.; Ferrari, M.; Lareo, C. Evaluation of Dilute Acid and Alkaline Pretreatments, Enzymatic Hydrolysis and Fermentation of Napiergrass for Fuel Ethanol Production. Biomass Bioenergy 2015, 74, 193–201. [Google Scholar] [CrossRef]
- Bagudo, B.U.; Dangoggo, S.M.; Usman, J. Bioethanol Production Potential of Andropogon Gayanus. Ann. Biol. Res. 2014, 5, 106–111. Available online: https://api.semanticscholar.org/CorpusID:82882507 (accessed on 18 December 2023).
- Yuan, H.; Tan, L.; Kida, K.; Morimura, S.; Sun, Z.-Y.; Tang, Y.-Q. Potential for Reduced Water Consumption in Biorefining of Lignocellulosic Biomass to Bioethanol and Biogas. J. Biosci. Bioeng. 2021, 131, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Modenbach, A.A.; Nokes, S.E. The Use of High-Solids Loadings in Biomass Pretreatment—A Review. Biotechnol. Bioeng. 2012, 109, 1430–1442. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lee, J.; Wang, D. A Critical Review on Water Overconsumption in Lignocellulosic Biomass Pretreatment for Ethanol Production through Enzymic Hydrolysis and Fermentation. Energy Fuels 2023, 37, 2667–2680. [Google Scholar] [CrossRef]
- Ovejero-Pérez, A.; Ayuso, M.; Rigual, V.; Domínguez, J.C.; García, J.; Alonso, M.V.; Oliet, M.; Rodriguez, F. Technoeconomic Assessment of a Biomass Pretreatment + Ionic Liquid Recovery Process with Aprotic and Choline Derived Ionic Liquids. ACS Sustain. Chem. Eng. 2021, 9, 8467–8476. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Q.; Tan, X.; Qi, W.; Yu, Q.; Zhou, G.; Zhuang, X.; Yuan, Z. High Conversion of Sugarcane Bagasse into Monosaccharides Based on Sodium Hydroxide Pretreatment at Low Water Consumption and Wastewater Generation. Bioresour. Technol. 2016, 218, 1230–1236. [Google Scholar] [CrossRef]
- Fang, C.; Thomsen, M.H.; Brudecki, G.P.; Cybulska, I.; Frankær, C.G.; Bastidas-Oyanedel, J.-R.; Schmidt, J.E. Seawater as Alternative to Freshwater in Pretreatment of Date Palm Residues for Bioethanol Production in Coastal and/or Arid Areas. ChemSusChem 2015, 8, 3823–3831. [Google Scholar] [CrossRef] [PubMed]
- Skiba, E.A.; Ovchinnikova, E.V.; Budaeva, V.V.; Banzaraktsaeva, S.P.; Kovgan, M.A.; Chumachenko, V.A.; Mironova, G.F.; Kortusov, A.N.; Parmon, V.N.; Sakovich, G.V. Miscanthus Bioprocessing Using HNO3-Pretreatment to Improve Productivity and Quality of Bioethanol and Downstream Ethylene. Ind. Crops Prod. 2022, 177, 114448. [Google Scholar] [CrossRef]
- Larnaudie, V.; Ferrari, M.D.; Lareo, C. Enzymatic Hydrolysis of Liquid Hot Water-Pretreated Switchgrass at High Solid Content. Energy Fuels 2019, 33, 4361–4368. [Google Scholar] [CrossRef]
- Song, Y.; Lee, Y.-G.; Cho, E.; Bae, H.-J. Production of Xylose, Xylulose, Xylitol, and Bioethanol from Waste Bamboo Using Hydrogen Peroxicde-Acetic Acid Pretreatment. Fuel 2020, 278, 118247. [Google Scholar] [CrossRef]
- Tan, X.; Zhang, Q.; Wang, W.; Zhuang, X.; Deng, Y.; Yuan, Z. Comparison Study of Organosolv Pretreatment on Hybrid Pennisetum for Enzymatic Saccharification and Lignin Isolation. Fuel 2019, 249, 334–340. [Google Scholar] [CrossRef]
- Wang, Z.-K.; Li, H.; Lin, X.-C.; Tang, L.; Chen, J.-J.; Mo, J.-W.; Yu, R.-S.; Shen, X.-J. Novel Recyclable Deep Eutectic Solvent Boost Biomass Pretreatment for Enzymatic Hydrolysis. Bioresour. Technol. 2020, 307, 123237. [Google Scholar] [CrossRef]
- Aui, A.; Wang, Y.; Wright, M. Evaluating the Economic Feasibility of Cellulosic Ethanol: A Meta-Analysis of Techno-Economic Analysis Studies. Renew. Sust. Energ. Rev. 2021, 145, 111098. [Google Scholar] [CrossRef]
- Larnaudie, V.; Ferrari, M.D.; Lareo, C. Life Cycle Assessment of Ethanol Produced in a Biorefinery from Liquid Hot Water Pretreated Switchgrass. Renew. Energy 2021, 176, 606–616. [Google Scholar] [CrossRef]
- Soldatos, P. Economic Aspects of Bioenergy Production from Perennial Grasses in Marginal Lands of South Europe. Bioenergy Res. 2015, 8, 1562–1573. [Google Scholar] [CrossRef]
- De Laporte, A.V.; Ripplinger, D.G. Economic Viability of Perennial Grass Biomass Feedstock in Northern Climates. Ind. Crops Prod. 2019, 128, 213–220. [Google Scholar] [CrossRef]
- Olba-Zięty, E.; Stolarski, M.J.; Krzyżaniak, M. Economic Evaluation of the Production of Perennial Crops for Energy Purposes—A Review. Energies 2021, 14, 7147. [Google Scholar] [CrossRef]
- Conde-Mejía, C.; Jiménez-Gutiérrez, A.; El-Halwagi, M. A Comparison of Pretreatment Methods for Bioethanol Production from Lignocellulosic Materials. Process Saf. Environ. Prot. 2012, 90, 189–202. [Google Scholar] [CrossRef]
- Sindhu, R.; Binod, P.; Pandey, A. Biological Pretreatment of Lignocellulosic Biomass—An Overview. Bioresour. Technol. 2016, 199, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Hammond, G.P.; Li, B. Environmental and Resource Burdens Associated with World Biofuel Production out to 2050: Footprint Components from Carbon Emissions and Land Use to Waste Arisings and Water Consumption. GCB Bioenergy 2016, 8, 894–908. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhao, F.; Liu, S.; Wang, L.; Qiu, L.; Alexandrov, G.; Jothiprakash, V. Bioenergy Production and Environmental Impacts. Geosci. Lett. 2018, 5, 14. [Google Scholar] [CrossRef]
- VanLoocke, A.; Twine, T.E.; Zeri, M.; Bernacchi, C.J. A Regional Comparison of Water Use Efficiency for Miscanthus, Switchgrass and Maize. Agric. For. Meteorol. 2012, 164, 82–95. [Google Scholar] [CrossRef]
- Chen, Y.; Ale, S.; Rajan, N.; Srinivasan, R. Modeling the Effects of Land Use Change from Cotton (Gossypium hirsutum L.) to Perennial Bioenergy Grasses on Watershed Hydrology and Water Quality under Changing Climate. Agric. Water. Manag. 2017, 192, 198–208. [Google Scholar] [CrossRef]
- Guo, T.; Cibin, R.; Chaubey, I.; Gitau, M.; Arnold, J.G.; Srinivasan, R.; Kiniry, J.R.; Engel, B.A. Evaluation of Bioenergy Crop Growth and the Impacts of Bioenergy Crops on Streamflow, Tile Drain Flow and Nutrient Losses in an Extensively Tile-Drained Watershed Using SWAT. Sci. Total Environ. 2018, 613–614, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Hoekman, S.K.; Broch, A.; Liu, X. (Vivian) Environmental Implications of Higher Ethanol Production and Use in the U.S.: A Literature Review. Part I—Impacts on Water, Soil, and Air Quality. Renew. Sust. Energ. Rev. 2018, 81, 3140–3158. [Google Scholar] [CrossRef]
- Spatari, S.; Bagley, D.M.; MacLean, H.L. Life Cycle Evaluation of Emerging Lignocellulosic Ethanol Conversion Technologies. Bioresour. Technol. 2010, 101, 654–667. [Google Scholar] [CrossRef]
- Smullen, E.; Finnan, J.; Dowling, D.; Mulcahy, P. The Environmental Performance of Pretreatment Technologies for the Bioconversion of Lignocellulosic Biomass to Ethanol. Renew. Energy 2019, 142, 527–534. [Google Scholar] [CrossRef]
- Parajuli, R.; Sperling, K.; Dalgaard, T. Environmental Performance of Miscanthus as a Fuel Alternative for District Heat Production. Biomass Bioenergy 2015, 72, 104–116. [Google Scholar] [CrossRef]
- Bai, Y.; Luo, L.; van der Voet, E. Life Cycle Assessment of Switchgrass-Derived Ethanol as Transport Fuel. Int. J. Life Cycle Assess. 2010, 15, 468–477. [Google Scholar] [CrossRef]
- Iordan, C.-M.; Giroux, B.; Næss, J.S.; Hu, X.; Cavalett, O.; Cherubini, F. Energy Potentials, Negative Emissions, and Spatially Explicit Environmental Impacts of Perennial Grasses on Abandoned Cropland in Europe. Environ. Impact Assess. Rev. 2023, 98, 106942. [Google Scholar] [CrossRef]
- Dunn, J.B.; Mueller, S.; Wang, M.; Han, J. Energy Consumption and Greenhouse Gas Emissions from Enzyme and Yeast Manufacture for Corn and Cellulosic Ethanol Production. Biotechnol. Lett. 2012, 34, 2259–2263. [Google Scholar] [CrossRef]
- MacLean, H.L.; Spatari, S. The Contribution of Enzymes and Process Chemicals to the Life Cycle of Ethanol. Environ. Res. Lett. 2009, 4, 014001. [Google Scholar] [CrossRef]
- Yusof, S.J.H.M.; Roslan, A.M.; Ibrahim, K.N.; Abdullah, S.S.S.; Zakaria, M.R.; Hassan, M.A.; Shirai, Y. Environmental Performance of Bioethanol Production from Oil Palm Frond Petiole Sugars in an Integrated Palm Biomass Biorefinery. IOP Conf. Ser. Mater. Sci. Eng. 2018, 368, 012004. [Google Scholar] [CrossRef]
- Nimmanterdwong, P.; Chalermsinsuwan, B.; Østergård, H.; Piumsomboon, P. Environmental Performance Assessment of Napier Grass for Bioenergy Production. J. Clean. Prod. 2017, 165, 645–655. [Google Scholar] [CrossRef]
- Morandi, F.; Perrin, A.; Østergård, H. Miscanthus as Energy Crop: Environmental Assessment of a Miscanthus Biomass Production Case Study in France. J. Clean. Prod. 2016, 137, 313–321. [Google Scholar] [CrossRef]
- Felix, E.; Tilley, D.R. Integrated Energy, Environmental and Financial Analysis of Ethanol Production from Cellulosic Switchgrass. Energy 2009, 34, 410–436. [Google Scholar] [CrossRef]
- Culaba, A.; Mayol, A.P.; San Juan, J.L.; Vinoya, C.; Concepcion Ii, R.; Bandala, A.; Vicerra, R.; Ubando, A.; Chen, W.-H.; Chang, J.-S. Smart Sustainable Biorefineries for Lignocellulosic Biomass. Bioresour. Technol. 2022, 344, 126215. [Google Scholar] [CrossRef] [PubMed]
- Meena, M.; Shubham, S.; Paritosh, K.; Pareek, N.; Vivekanand, V. Production of Biofuels from Biomass: Predicting the Energy Employing Artificial Intelligence Modelling. Bioresour. Technol. 2021, 340, 125642. [Google Scholar] [CrossRef]
- Cioffi, R.; Travaglioni, M.; Piscitelli, G.; Petrillo, A.; De Felice, F. Artificial Intelligence and Machine Learning Applications in Smart Production: Progress, Trends, and Directions. Sustainability 2020, 12, 492. [Google Scholar] [CrossRef]
- Liao, M.; Yao, Y. Applications of Artificial Intelligence-Based Modeling for Bioenergy Systems: A Review. GCB Bioenergy 2021, 13, 774–802. [Google Scholar] [CrossRef]
- Ahmadian-Moghadam, H.; Elegado, F.B.; Nayve, R. Prediction of Ethanol Concentration in Biofuel Production Using Artificial Neural Networks. Int. J. Model. Optim. 2013, 1, 31–35. [Google Scholar] [CrossRef]
- Grahovac, J.; Jokić, A.; Dodić, J.; Vučurović, D.; Dodić, S. Modelling and Prediction of Bioethanol Production from Intermediates and Byproduct of Sugar Beet Processing Using Neural Networks. Renew. Energy 2016, 85, 953–958. [Google Scholar] [CrossRef]
- Smuga-Kogut, M.; Kogut, T.; Markiewicz, R.; Słowik, A. Use of Machine Learning Methods for Predicting Amount of Bioethanol Obtained from Lignocellulosic Biomass with the Use of Ionic Liquids for Pretreatment. Energies 2021, 14, 243. [Google Scholar] [CrossRef]
- Dodić, J.M.; Vučurović, D.G.; Dodić, S.N.; Grahovac, J.A.; Popov, S.D.; Nedeljković, N.M. Kinetic Modelling of Batch Ethanol Production from Sugar Beet Raw Juice. Appl. Energy 2012, 99, 192–197. [Google Scholar] [CrossRef]
- Li, X.; Dong, Y.; Chang, L.; Chen, L.; Wang, G.; Zhuang, Y.; Yan, X. Dynamic Hybrid Modeling of Fuel Ethanol Fermentation Process by Integrating Biomass Concentration XGBoost Model and Kinetic Parameter Artificial Neural Network Model into Mechanism Model. Renew. Energy 2023, 205, 574–582. [Google Scholar] [CrossRef]
- Hiloidhari, M.; Baruah, D.C.; Singh, A.; Kataki, S.; Medhi, K.; Kumari, S.; Ramachandra, T.V.; Jenkins, B.M.; Thakur, I.S. Emerging Role of Geographical Information System (GIS), Life Cycle Assessment (LCA) and Spatial LCA (GIS-LCA) in Sustainable Bioenergy Planning. Bioresour. Technol. 2017, 242, 218–226. [Google Scholar] [CrossRef]
- Bharti, A.; Paritosh, K.; Mandla, V.R.; Chawade, A.; Vivekanand, V. GIS Application for the Estimation of Bioenergy Potential from Agriculture Residues: An Overview. Energies 2021, 14, 898. [Google Scholar] [CrossRef]
- Wang, Z.; Peng, X.; Xia, A.; Shah, A.A.; Huang, Y.; Zhu, X.; Zhu, X.; Liao, Q. The Role of Machine Learning to Boost the Bioenergy and Biofuels Conversion. Bioresour. Technol. 2022, 343, 126099. [Google Scholar] [CrossRef] [PubMed]
- Negri, V.; Galán-Martín, Á.; Pozo, C.; Fajardy, M.; Reiner, D.M.; Mac Dowell, N.; Guillén-Gosálbez, G. Life Cycle Optimization of BECCS Supply Chains in the European Union. Appl. Energy 2021, 298, 117252. [Google Scholar] [CrossRef]
- Riahi, Y.; Saikouk, T.; Gunasekaran, A.; Badraoui, I. Artificial Intelligence Applications in Supply Chain: A Descriptive Bibliometric Analysis and Future Research Directions. Expert Syst. Appl. 2021, 173, 114702. [Google Scholar] [CrossRef]

| Biomass | Cellulose (%) | Hemicellulose (%) | Lignin (%) | References |
|---|---|---|---|---|
| Switchgrass | 45.00 | 31.40 | 12.00 | [55] |
| Switchgrass | 32.00 | 17.90 | 21.40 | [16] |
| Bermuda grass | 25.60 | 15.90 | 19.30 | [16] |
| Bermuda grass | 25.00 | 35.70 | 6.40 | [55] |
| Napier grass | 41.80 ± 2.20 | 23.20 ± 1.10 | 25.00 ± 0.30 | [26] |
| Kans grass | 44.00 | 21.50 | 28.50 | [56] |
| Miscanthus | 52.10 | 25.80 | 12.60 | [44] |
| Bamboo | 46.50 | 18.80 | 25.70 | [47] |
| Rye | 42.83 | 27.86 | 6.51 | [57] |
| Reed | 49.40 | 31.50 | 8.74 | [57] |
| Amur silver grass | 42.00 | 30.15 | 7.00 | [57] |
| Natural hay | 44.90 | 31.40 | 12.00 | [47] |
| Para grass | 42.00 ± 1.90 | 20.00 ± 0.20 | 19.00 ± 0.20 | [58] |
| Vetiver | 34.48 | 35.07 | 14.34 | [59] |
| Cogongrass | 41.50 | 22.50 | 20.40 | [36] |
| Biomass | Pretreatment | Cellulose [%] | Hemicellulose [%] | Lignin [%] | Reference |
|---|---|---|---|---|---|
| Napier grass | Untreated | 41.80 2.20 | 23.20 1.30 | 25.00 0.30 | [26] |
| Alkaline () | 62.10 2.70 | 16.10 0.80 | 19.50 1.70 | ||
| Acidic () | 47.80 | 8.50 | 26.80 | ||
| Acidic-Alkaline (–) | 67.60 | 4.00 | 17.00 | ||
| Bermuda grass | Untreated | 25.59 | 15.88 | 19.33 | [96] |
| Alkaline () | 25.01–23.11 | 13.60–6.27 | 16.93–2.82 | ||
| B. brizantha | Untreated | 42.15 | 12.70 | 18.80 | [23] |
| Acidic () | 42.85 2.25–54.63 0.63 | 4.07 0.13–12.10 0.10 | - | ||
| Napier grass | Untreated | 22.60 | 20.90 | 19.40 | [12] |
| Alkaline () | 31.20 | 21.90 | 6.80 | ||
| Acidic () | 24.00 | 13.50 | 13.40 | ||
| Alkaline (aqueous ) | 35.50 | 25.40 | 9.20 | ||
| Alkaline () | 39.20 | 10.50 | 11.00 | ||
| King grass | Untreated | 31.60 2.50 | 15.2 0.90 | 15.40 1.00 | [35] |
| Alkaline () | 35.70 0.30 | 21.60 0.90 | 6.80 1.90 | ||
| Acidic () | 43.80 2.30 | 13.10 1.10 | 17.20 1.70 | ||
| Alkaline (aqueous ) | 36.10 0.40 | 23.30 1.30 | 9.90 0.80 | ||
| Alkaline () | 37.90 0.90 | 19.80 0.50 | 14.10 0.60 | ||
| Giant reed (Arundo donax) | Untreated | 33.00 0.70 | 29.70 0.90 | 23.00 1.11 | [29] |
| Acidic () | 55.10 1.30–58.30 0.90 | 12.30 0.70–18.20 1.70 | 18.10 1.30–21.10 1.50 | ||
| Acidic () | 52.90 1.30–62.90 1.20 | 10.60 1.40–15.40 0.90 | 15.80 0.70–20.30 2.10 | ||
| Alkaline () | 48.90 0.50–62.70 0.70 | 16.20 0.40–24.40 0.20 | 13.70 0.8–17.90 0.90 | ||
| Napier grass | Untreated | 36.67 0.17 | 20.23 0.09 | 19.33 0.33 | [107] |
| Alkaline () | 55.83 0.13–72.30 0.17 | 15.45 0.06–20.30 0.12 | 4.33 0.07–7.00 0.23 | ||
| Para grass | Untreated | 42.00 1.90 | 20.00 0.20 | 19.00 0.20 | [58] |
| Alkaline () | 58.13 0.60 | 21.68 0.20 | 10.09 0.10 | ||
| Acidic () | 55.61 0.30 | 9.48 0.05 | 19.01 0.00 |
| Biomass | Pretreatment | Crl before [%] | Crl after [%] | Reference |
|---|---|---|---|---|
| Para grass | Alkaline | 33.40 | 58.60 | [58] |
| Para grass | Acid | 33.40 | 54.30 | [58] |
| Napier grass | Alkaline | 33.00 | 64.02 | [113] |
| Napier grass | Acidic | 33.00 | 49.83 | [113] |
| Chinese Pennisetum | Alkaline | 49.50 | 60.60–63.40 | [115] |
| Hybrid Pennisetum | Alkaline | 43.40 | 56.80–58.60 | [115] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johannes, L.P.; Xuan, T.D. Comparative Analysis of Acidic and Alkaline Pretreatment Techniques for Bioethanol Production from Perennial Grasses. Energies 2024, 17, 1048. https://doi.org/10.3390/en17051048
Johannes LP, Xuan TD. Comparative Analysis of Acidic and Alkaline Pretreatment Techniques for Bioethanol Production from Perennial Grasses. Energies. 2024; 17(5):1048. https://doi.org/10.3390/en17051048
Chicago/Turabian StyleJohannes, Lovisa Panduleni, and Tran Dang Xuan. 2024. "Comparative Analysis of Acidic and Alkaline Pretreatment Techniques for Bioethanol Production from Perennial Grasses" Energies 17, no. 5: 1048. https://doi.org/10.3390/en17051048
APA StyleJohannes, L. P., & Xuan, T. D. (2024). Comparative Analysis of Acidic and Alkaline Pretreatment Techniques for Bioethanol Production from Perennial Grasses. Energies, 17(5), 1048. https://doi.org/10.3390/en17051048









