Ceramic Mineral Waste-Forms for Nuclear Waste Immobilization
Abstract
1. Introduction
- (1)
- The ability of the structure to include various cations in different combinations and ratios.
- (2)
- Known high stability of structure to the action of the destructive factors of the environment during their prolonged exposure (“mineral-like” compounds preferred while “the nature suggests”) such as high temperatures, thermal “stresses”, radiation levels, the corrosive action of water and other chemical solutions. Criteria for the resistance of materials to such effects are justified by the features of the crystal structure of materials including small interatomic distances, and the possibility of their controlled variation in the desired direction when cations and/or anions of given sizes are included in the crystallographic positions. Most of the crystalline matrices discussed in the present work meet these criteria in full or in part. Herewith the classification criteria for crystalline ceramics were based on considering first simple and then more complex structures, e.g., starting with oxides (from simple oxides to complex oxides) and moving to salt compositions (from simple salt to complex ones).
2. Theoretical Aspects of Substitution
- (1)
- Sufficient lattice vacancies exist in the structure or,
- (2)
- A variable valence cation such as Fe or Ti is present in a neighboring lattice site balancing the charge.
3. Synthesis of Ceramic Waste-forms
4. Crystalline Ceramic Phase:
4.1. Simple Oxides
4.2. Complex Oxides
- (1)
- [8]A3[6]B2[TiO4]3, e.g., [8](Ca,Gd, actinides)[6]Fe2[4]Fe3O12.
- (2)
- A3B2(XO4)3; distorted cubic structure; BO6 octahedra and XO4 tetrahedra establish a framework structure alternately sharing corners; A and B sites can host actinides, REs, Y, Mg, Ca, Fe2+, Mn2+ and X = Cr3+, Fe3+, Al3+, Ga3+, Si4+, Ge4+ and V5+. Structure: Cubic, Sp. gr. Ia3d, z = 8. Ceramics were prepared by cold pressing and sintering and using Spark Plasma Sintering with high relative density up to 98–99% of theoretical.
4.3. Framework Silicates
- (1)
- Sodalite Na8Cl2Al6Si6O24, also written as (Na,K)6[Al6Si6O24]·(2NaCl) to demonstrate that 2Cl and associated Na atoms are in a cage structure defined by the aluminosilicate tetrahedra of six adjoining NaAlSiO4, is a naturally occurring feldspathoid mineral. It incorporates alkali, alkaline earths, rare earth elements, halide fission products and trace quantities of U and Pu. Sodalite was and it is being investigated as a durable host for the waste generated from electro-refining operations deployed for the reprocessing of metal fuel. Supercalcines which are high temperature, silicate-based “natural mineral” assemblages proposed for HLW waste stabilization in the United States in 1973–1985, contained sodalites as minor phases retaining Cs, Sr and Mo, e.g., Na6[Al6Si6O24](NaMoO4)2. Sodalite structures are known to retain B, Ge, I, Br and Re in the cage-like structures. Structure of Sodalite showing (a) two-dimensional projection of the (b) three-dimensional structure and (c) the four fold ionic coordination of the Na site to the Cl-ion and three framework oxygen bonds. Structure: Cubic, Sp. gr. , z = 1; may include: Na, K, Mg, Ca, Mn, Fe, Al, Si, Ti, Cl, SO4 and CO3. Ceramics were prepared by cold pressing and sintering; by HIP.
- (2)
- Nosean, (Na,K)6[Al6Si6O24](Na2SO4)), silica “stuffed derivative” sodalite cage type structure host mineral for sulfate or sulfide species.
- (3)
- Hauyne, (Na)6[Al6Si6O24]((Ca,Na)SO4)1-2 sodalite family; can accommodate either Na2SO4 or CaSO4.
- (4)
- Helvite (Mn4[Be3Si3O12]S: Be (beryllium) can be substituted in place of Al and S2 in the cage structure along with Fe, Mn and Zn.
- (5)
- Danalite (Fe4[Be3Si3O12]S).
- (6)
- Genthelvite (Zn4[Be3Si3O12]S).
- (7)
- Lazurite, (Ca,Na)6[Al6Si6O24]((Ca,Na)S,SO4,Cl)x; can accommodate either SO4 or S2, Ca or Na and Cl.
4.4. Phosphates
4.5. Tungstate, Molybdates
5. Summary of Crystalline Ceramic Waste-forms
6. Conclusions
- Ceramic waste-forms for nuclear waste immobilization are investigated in different countries with a focus on improving environmental safety during storage, transport and disposal.
- Inorganic compounds of oxide and salt character, having structural analogs with natural minerals, are being studied as most perspective materials for the immobilization of radioactive waste.
- Approaches based on crystallochemistry principles are used when choosing the most favorable structural forms. They are based on the materials science concept “composition-structure-method of synthesis-property” accounting for the real task to be achieved. The basic principle is the isomorphism of cations and anions in compounds when choosing a real structure. Possible isomorphic substitutions in both cationic and anionic structural sites were considered in the works analyzed.
- Crystalline ceramic waste-forms are intended to increase the environmental safety barrier when isolating radioactive materials (containing both actinides and fission products) from the biosphere. Among the methods of obtaining ceramic waste-forms, special attention in recent years is paid to sintering methods which ensure the formation of ceramics that, first, are almost non-porous e.g., have a relative density of up to 99.0–99.9% of theoretical, and, second, can be obtained within a small processing time e.g., within a few minutes (i.e., 2–3 min). These requirements are met by high-speed electric pulse sintering processes e.g., so-called Spark Plasma Sintering (SPS), although hot pressing enables the synthesis of very dense ceramics as well.
Author Contributions
Funding
Conflicts of Interest
References
- Hatch, L.P. Ultimate Disposal of Radioactive Wastes. Am. Sci. 1953, 41, 410–421. [Google Scholar]
- McCarthy, G.J. Quartz-Matrix Isolation of Radioactive Wastes. J. Mater. Sci. 1973, 8, 1358–1359. [Google Scholar] [CrossRef]
- McCarthy, G.J.; Davidson, M.T. Ceramic Nuclear Waste Forms: I. Crystal Chemistry and Phase Formation. Bull. Am. Ceram. Soc. 1975, 54, 782–786. [Google Scholar]
- Roy, R. Rationale Molecular Engineering of Ceramic Materials. J. Am. Ceram. Soc. 1977, 60, 350–363. [Google Scholar] [CrossRef]
- Jantzen, C.M.; Lee, W.E.; Ojovan, M.I. Radioactive waste (RAW) conditioning, immobilization, and encapsulation processes and technologies: overview and advances. In Radioactive Waste Management and Contaminated Site Clean-Up. Processes, Technologies and International Experience; Lee, W.E., Ojovan, M.I., Jantzen, C.M., Eds.; Woodhead Published Limited: Oxford, UK; Cambridge, UK; Philadelphia, PA, USA; New Delhi, India, 2013; Chapter 6; pp. 171–272. [Google Scholar]
- Schoebel, R.O. Stabilization of High Level Waste in Ceramic Form. Bull. Am. Ceram. Soc. 1975, 54, 459. [Google Scholar]
- Ringwood, A.E.; Oversby, V.M.; Kesson, S.E. SYNROC: Leaching Peformance and Process Technology. In Proceedings of the Seminar on Chemistry and Process Engineering for High Level Liquid Waste Solidification, Jülichw, Germany, 1–5 June 1981; pp. 495–506. [Google Scholar]
- Ringwood, A.E. Safe Disposal of High Level Nuclear Reactor Wastes: A New Strategy; Australian Nuclear University Press: Canberra, Australia, 1978; pp. 1–64. [Google Scholar]
- Ringwood, A.E.; Kesson, S.E.; Ware, N.G.; Hibberson, W.O.; Major, A. The SYNROC Process: A Geochemical Approach to Nuclear Waste Immobilization. Geochem. J. 1979, 13, 141–165. [Google Scholar] [CrossRef]
- Vance, E.R.; Moricca, S.A.; Stewart, M.W.A. Progress at ANSTO on a synroc plant for intermediate-level waste from reactor production of 99Mo. Adv. Sci. Technol. 2014, 94, 111–114. [Google Scholar] [CrossRef]
- McCarthy, G.J.; Pepin, J.G.; Pfoertsch, D.E.; Clarke, D.R. Crystal Chemistry of the Synthetic Minerals in Current Supercalcine-Ceramics; U.S. DOE Report CONF-790420; Battelle Pacific Northwest Labs.: Richland, WA, USA, 1979; pp. 315–320. [Google Scholar]
- Lutze, W.; Ewing, R.C. Radioactive Waste Forms for the Future; North-Holland Publishers: Amsterdam, The Netherlands, 1988; p. 778. [Google Scholar]
- Jantzen, C.M.; Flintoff, J.; Morgan, P.E.D.; Harker, A.B.; Clarke, D.R. Ceramic Nuclear Waste Forms. In Proceedings of the Seminar on Chemistry and Process Engineering for High-Level Liquid Waste Solidification, Jülichw, Germany, 1–5 June 1981; Volume 2, pp. 693–706. [Google Scholar]
- Raison, P.E.; Haire, R.G.; Sato, T.; Ogawa, T. Fundamental and Technological Aspects of Actinide Oxide Pyrochlores: Relevance for Immobilization Matrices; MRS Online Proceedings Library Archive: Warrendale, PA, USA, 1999; p. 556. [Google Scholar]
- Burakov, B.E.; Ojovan, M.I.; Lee, W.E. Crystalline Materials for Actinide Immobilization; Imperial College Press: London, UK, 2010. [Google Scholar]
- Dacheux, N.; Clavier, N.; Podor, R. Monazite as a promising long-term radioactive waste matrix: Benefits of high-structural flexibility and chemical durability. Am. Miner. 2013, 98, 833–847. [Google Scholar] [CrossRef]
- Clavier, N.; Podor, R.; Dacheux, N. Crystal chemistry of the monazite structure. J. Eur. Ceram. Soc. 2011, 31, 941–976. [Google Scholar] [CrossRef]
- Orlova, A.I.; Lizin, A.A.; Tomilin, S.V.; Lukinykh, A.N. On the Possibility of Realization of Monazite and Langbeinite Structural Types in Complex Americium and Plutonium Phosphates. Synthesis and X-ray Diffraction Studies. Radiochemistry 2011, 53, 63–68. [Google Scholar] [CrossRef]
- Alamo, J.; Roy, R. Revision of Crystalline Phases in the System ZrO2-P2O5. Commun. Am. Ceram. Soc. 1984, 67, C80–C82. [Google Scholar] [CrossRef]
- Hawkins, H.T.; Spearing, D.R.; Veirs, D.K.; Danis, J.A.; Smith, D.M.; Tait, C.D.; Runde, W.H. Synthesis and Characterization of Uranium(IV)-Bearing Members of the [NZP] Structural Family. Chem. Mater. 1999, 11, 2851–2857. [Google Scholar] [CrossRef]
- Orlova, A.I. Isomorphism in Crystalline Phosphates of the NaZr2(PO4)3 structural type and Radiochemical Problems. Radiochemistry 2002, 44, 423–445. [Google Scholar] [CrossRef]
- Volkov, Y.F.; Tomlin, S.V.; Orlova, A.I.; Lizin, A.A.; Spirjakov, V.I.; Lukinikh, A.N. Actinide Phosphates AIM2IV(PO4)3 (MIV = U, Np, Pu; AI = Na, K, Rb) with rombohedral structure. Radiochemistry 2003, 46, 319–328. [Google Scholar] [CrossRef]
- Gregg, D.J.; Karatchevtseva, I.; Triani, G.; Lumpkin, G.R.; Vance, E.R. The thermophysical properties of calcium and barium zirconium phosphate. J. Nucl. Mater. 2013, 441, 203–210. [Google Scholar] [CrossRef]
- Gregg, D.J.; Karatchevtseva, I.; Thorogood, G.J.; Davis, J.; Bell, B.; Jackson, M.; Dayal, P.; Ionescu, M.; Triani, G.; Short, K.; et al. Ion Beam Irradiation Effects in Strontium Zirconium Phosphate with NZP-structure type. J. Nucl. Mater. 2014, 446, 224–231. [Google Scholar] [CrossRef]
- Orlova, A.I.; Koryttseva, A.K.; Loginova, E.E. A Family of Phosphates of Langbeinite Structure. Crystal-Chemical Aspect of Radioactive Waste Immobilization. Radiochemistry 2011, 53, 51–62. [Google Scholar] [CrossRef]
- Orlova, A.I. Chemistry and structural chemistry of anhydrous tri- and tetravalent actinide orthophosphates. In Structural Chemistry of Inorganic Actinide Compounds; Chapter 8; Krivovichev, S.V., Burns, P.C., Tananaev, I.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 315–340. [Google Scholar]
- Huittinen, N.; Scheinost, A.C.; Ji, Y.; Kowalski, P.M.; Arinicheva, Y.; Wilden, A.; Neumeier, S.; Stumpf, T. A spectroscopic and computational study of Cm3+ incorporation in lanthanide phosphate rhabdophane (LnPO4·0.67H2O) and monazite (LnPO4). Inorg. Chem. 2018, 57, 6252–6265. [Google Scholar] [CrossRef]
- Clarke, D.R. Preferential Dissolution of an Intergranular Amorphous Phase in a Nuclear Waste Ceramic. J. Am. Ceram. Soc. 1981, 64, 89–90. [Google Scholar] [CrossRef]
- Cooper, J.A.; Cousens, D.R.; Hanna, J.A.; Lewis, R.A.; Myhra, S.; Segall, R.L.; Smart, R.S.C.; Turner, P.S.; White, T.J. Intergranular Films and Pore Surfaces in Synroc C: Structure, Composition, and Dissolution characteristics. J. Am. Ceram. Soc. 1986, 69, 347–352. [Google Scholar] [CrossRef]
- Buykx, W.J.; Hawkins, K.; Levins, D.M.; Mitamura, H.; Smart, R.S.C.; Stevens, G.T.; Watson, K.G.; Weedon, D.; White, T.J. Titanate Ceramics for the Immobilization of SodiumBearing High-Level Nuclear Waste. J. Am. Ceram. Soc. 1988, 71, 768–788. [Google Scholar] [CrossRef]
- Dickson, F.J.; Mitamura, H.; White, T.J. Radiophase Development in Hot-Pressed Alkoxide-Derived Titanate Ceramics for Nuclear Waste Stabilization. J. Am. Ceram. Soc. 1989, 72, 1055–1059. [Google Scholar] [CrossRef]
- Buykx, W.J.; Levins, D.M.; Smart, R.S.C.; Smith, K.L.; Stevens, G.T.; Watson, K.G.; Weedon, D.; White, T.J. Interdependence of Phase Chemistry, Microstructure, and Oxygen Fugacity in Titanate Nuclear Waste Ceramics. J. Am. Ceram. Soc. 1990, 73, 1201–1207. [Google Scholar] [CrossRef]
- Buykx, W.J.; Levins, D.M.; St Smart, R.C.; Smith, K.L.; Stevens, G.T.; Watson, K.G.; White, T.J. Processing Impurities as Phase Assemblage Modifiers in Titanate Nuclear Waste Ceramics. J. Am. Ceram. Soc. 1990, 73, 217–225. [Google Scholar] [CrossRef]
- Mitamura, H.; Matsumoto, S.; Hart, K.P.; Miyazaki, T.; Vance, E.R.; Tamura, Y.; Togashi, Y.; White, T.J. Aging Effects on Curium-Dopped Titantate Ceramics Containing Sodium-Bearing High-Level Nuclear Waste. J. Am. Ceram. Soc. 1992, 75, 392–400. [Google Scholar] [CrossRef]
- Zhang, Z.; Carter, M.L. An X-Ray Photoelectron Spectroscopy Investigation of Highly Soluble Grain-Boundary Impurity Films in Hollandite. J. Am. Ceram. Soc. 2010, 93, 894–899. [Google Scholar] [CrossRef]
- Harker, A.B.; Clarke, D.R.; Jantzen, C.M.; Morgan, P.E.D. The Effect of Interfacial Material on Tailored Ceramic Nuclear Waste Form Dissolution, Surfaces and Interfaces in Ceramic and Ceramic-Metal Systems; Pask, J., Evans, A., Eds.; Plenum Press: New York, NY, USA, 1981; Volume 14, pp. 207–216. [Google Scholar]
- Roy, R. Hydroxylated Ceramic Waste Forms and the Absurdity of Leach Tests. In Proceedings of the International Seminar on Chemistry and Process Engineering for High-Level Liquid Waste Solidification, Julich, India, 1–5 June 1981; pp. 576–602. [Google Scholar]
- Mason, J.B.; Oliver, T.W.; Carson, M.P.; Hill, G.M. Studsvik Processing Facility Pyrolysis/Steam Reforming Technology for Volume and Weight Reduction and Stabilization of LLRW and Mixed Wastes; American Institute of Chemical Engineers: New York, NY, USA, 1999. [Google Scholar]
- Mason, J.B.; McKibbin, J.; Ryan, K.; Schmoker, D. Steam Reforming Technology for Denitration and Immobilization of DOE Tank Wastes; THOR Treatment Technologies, LLC.: Richland, WA, USA, 2003. [Google Scholar]
- Jantzen, C.M.; Lorier, T.H.; Marra, J.C.; Pareizs, J.M. Durability Testing of Fluidized Bed Steam Reforming (FBSR) Waste Forms WM06 Paper № 6373; WWM Symposia: Phoenix, AZ, USA, 2006. [Google Scholar]
- Jantzen, C.M.; Lorier, T.H.; Pareizs, J.M.; Marra, J.C. Fluidized Bed Steam Reformed (FBSR) Mineral Waste Forms: Characterization and Durability Testing; MRS Online Proceedings Library Archive: Warrendale, PA, USA, 2007; pp. 379–386. [Google Scholar]
- Jantzen, C.M. Mineralization of Radioactive Wastes By Fluidized Bed Steam Reforming (FBSR): Comparisons to Vitreous Waste Forms and Pertinent Durability Testing; U.S. DOE Report WSRC-STI-2008-00268; Westinghouse Savannah River Company, Savannah River Site: Aiken, SC, USA, 2008. [Google Scholar]
- Jantzen, C.M.; Crawford, C.L.; Burket, P.B.; Daniel, W.G.; Cozzi, A.D.; Bannochie, C.J. Radioactive Demonstrations of Fluidized Bed Steam Reforming (FBSR) as a Supplementary Treatment for Hanford’s Low Activity Waste (LAW) and Secondary Wastes (SW); Waste Management WM11, Paper № 11593; WM Symposia: Phoenix, AZ, USA, 2011. [Google Scholar]
- Jantzen, C.M.; Crawford, C.L.; Burket, P.R.; Bannochie, C.J.; Daniel, W.G.; Nash, C.A.; Cozzi, A.D.; Herman, C.C. Radioactive Demonstrations of Fluidized Bed Steam Reforming (FBSR) with Actual Hanford Low Activity Wastes: Verifying FBSR as a Supplemental Treatment; WM12 Paper № 12317; WM Symposia: Phoenix, AZ, USA, 2012. [Google Scholar]
- Jantzen, C.M.; Glasser, F.P. Crystallochemical Stabilization of Radwaste Elements in Portland Cement Clinker. In Proceedings of the International Symposium on Ceramics in Nuclear Waste Management; CONF-790420, Cincinnati, OH, USA, 30 April–2 May 1979; pp. 342–348. [Google Scholar]
- Jantzen, C.M.; Glasser, F.P. Stabilization of Nuclear Waste Constituents in Portland Cement. Am. Ceram. Soc. Bull. 1979, 58, 459–466. [Google Scholar]
- Bragg, L.; Claringbull, G.F.; Taylor, W.H. Crystal Structures of Minerals; G. Bell & Sons: London, UK, 1965; p. 409. [Google Scholar]
- Shannon, R.D.; Prewitt, C.T. Effective Ionic Radii in Oxides and Fluorides. Acta Cryst. 1969, B25, 925–946. [Google Scholar] [CrossRef]
- Shannon, R.D.; Prewitt, C.T. Revised Values of Effective Ionic Radii. Acta Cryst. 1970, B26, 1046–1048. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides. Acta Cryst. 1976, A32, 751–767. [Google Scholar] [CrossRef]
- Felsche, J. Rare Earth Silicates with the Apatite Structure. J. Solid State Chem. 1972, 5, 266–275. [Google Scholar] [CrossRef]
- Jantzen, C.M.; Glasser, F.P.; Lachowsli, E.E. Radioactive Waste-Portland Cement Systems: I. Radionuclide Distribution. J. Am. Ceram. Soc. 1984, 67, 668–673. [Google Scholar] [CrossRef]
- Jantzen, C.M.; Glasser, F.P.; Lachowsli, E.E. Radioactive Waste-Portland Cement Systems: II. Leaching Characteristics. J. Am. Ceram. Soc. 1984, 67, 674–678. [Google Scholar] [CrossRef]
- Ringwood, A.E.; Kesson, S.E. Immobilization of High-Level Wastes in Synroc Titanate Ceramic. In Proceedings of the International Symposium on Ceramics in Nuclear WaMc Management, CONF-790420, Cincinnati, OH, USA, 30 April–2 May 1979; pp. 174–178. [Google Scholar]
- Cooper, J.A.; Cousens, D.R.; Lewis, R.A.; Myhra, S.; Segall, R.L.; Smart, R.S.C.; Turner, P.S.; White, T.J. Microstructural Characterization of Synroc C and E by Electron Microscopy. J. Am. Ceram. Soc. 1985, 68, 64–70. [Google Scholar] [CrossRef]
- Aubin, V.; Caurant, D.; Gourier, D.; Baffier, N.; Advocat, F.; Bart, F.; Leturcq, G.; Costantini, J.M. Synthesis, Characterization and Study of the Radiation Effects on Hollandite Ceramics Developed for Cesium Immobilization. Mater. Res. Soc. Symp. Proc. 2004, 807, 315–320. [Google Scholar] [CrossRef]
- Hart, K.P.; Vance, E.R.; Day, R.A.; Begg, B.D.; Angel, P.J. Immobilization of Separated Tc and Cs/Sr in SYNROC. Mater. Res. Soc. Symp. Proc. 1996, 412, 281–287. [Google Scholar] [CrossRef]
- Buerger, M.J.; Klein, G.E.; Hamburger, G.E. Structure of Nepheline. Geol. Soc. Am. Bull. 1946, 57, 1182–1183. [Google Scholar]
- Buerger, M.J.; Klein, G.E.; Hamburger, G.E. The structure of nepheline. Am. Mineral. 1947, 32, 197. [Google Scholar]
- Simmons, W.B.; Peacor, D.R. Refinement of the crystal structure of a volcanic nepheline. Am. Mineral. 1972, 57, 1711–1719. [Google Scholar]
- Rossi, G.; Oberti, R.; Smith, D.C. The crystal structure of a K-poor Ca-rich silicate with the nepheline framework, and crystal-chemical relationships in the compositional space (K,Na,Ca,_)8(Al,Si)16O32. Eur. J. Mineral. 1989, 1, 59–70. [Google Scholar] [CrossRef]
- Tait, K.T.; Sokolova, E.V.; Hawthorne, F.C.; Khomyakov, A.P. The crystal chemistry of nepheline. Can. Mineral. 2003, 41, 61–70. [Google Scholar] [CrossRef]
- Hassan, I.; Antao, S.M.; Hersi, A.A.M. Single-crystal XRD, TEM, and thermal studies of the satellite reflections in nepheline. Can. Mineral. 2003, 41, 759–783. [Google Scholar] [CrossRef]
- Gatta, G.D.; Angel, R.J. Elastic behavior and pressure-induced structural evolution of nepheline: Implications for the nature of the modulated superstructure. Am. Mineral. 2007, 92, 1446–1455. [Google Scholar] [CrossRef]
- Angel, R.J.; Gatta, G.C.; Ballaran, T.B.; Carpenter, M.A. The mechanism of coupling in the modulated structure of nepheline. Can. Mineral. 2008, 46, 1465–1476. [Google Scholar] [CrossRef]
- Antao, S.M.; Nepheline, H.I. Structure of three samples from the Bancroft area, Ontario,obtained using synchrotron high-resolution powder x-ray diffraction. Can. Mineral. 2010, 48, 69–80. [Google Scholar] [CrossRef]
- Chapman, K.W.; Chupas, P.J.; Nenoff, T.M. Radioactive Iodine Capture in Silver-Containing Mordenites through Nanoscale Silver Iodide Formation. J. Am. Chem. Soc. 2010, 132, 8897–8899. [Google Scholar] [CrossRef]
- Matyas, J.; Fryxell, G.; Busche, B.; Wallace, K.; Fifield, L. Functionalised silica aerogels: Advanced materials to capture and immobilise radioactive iodine. In Ceramic Engineering and Science Proceedings; American Ceramic Society, Inc.: Columbus, OH, USA, 2011; pp. 23–32. [Google Scholar]
- Riley, B.J.; Chun, J.; Ryan, J.V.; Matyáš, J.; Li, X.S.; Matson, D.W.; Sundaram, S.K.; Strachan, D.M.; Vienna, J.D. Chalcogen-based aerogels as a multifunctional platform for remediation of radioactive iodine. RSC Adv. 2011, 1, 1704–1715. [Google Scholar] [CrossRef]
- Yang, J.H.; Cho, Y.-J.; Shin, J.M.; Yim, M.-S. Bismuth-embedded SBA-15 mesoporous silica for radioactive iodine capture and stable storage. J. Nucl. Mater. 2015, 465, 556–564. [Google Scholar] [CrossRef]
- Subrahmanyam, K.S.; Sarma, D.; Malliakas, C.D.; Polychronopoulou, K.; Riley, B.J.; Pierce, D.A.; Chun, J.; Kanatzidis, M.G. Chalcogenide Aerogels as Sorbents for Radioactive Iodine. Chem. Mater. 2015, 27, 2619–2626. [Google Scholar] [CrossRef]
- Matyas, J.; Canfield, N.; Silaiman, S.; Zumhoff, M. Silica-based waste form for immobilization of iodine from reprocessing plant off-gas streams. J. Nucl. Mater. 2016, 476, 255–261. [Google Scholar] [CrossRef]
- Vienna, J.D.; Kroll, J.O.; Hrma, P.R.; Lang, J.B.; Crum, J.V. Submixture Model to Predict Nepheline Precipitation in Waste Glasses. Int. J. Appl. Glass Sci. 2017, 8, 143–157. [Google Scholar] [CrossRef]
- Asmussen, R.M.; Matyáš, J.; Qafoku, N.P.; Kruger, A.A. Silver-functionalized silica aerogels and their application in the removal of iodine from aqueous environments. J. Hazard. Mater. 2018. [Google Scholar] [CrossRef]
- Asmussen, R.M.; Ryan, J.V.; Matyas, J.; Crum, J.V.; Reiser, J.T.; Avalos, N.; McElroy, E.M.; Lawter, A.R.; Canfield, N.C. Investigating the Durability of Iodine Waste Forms in Dilute Conditions. Materials 2019, 12, 686. [Google Scholar] [CrossRef]
- Burghatz, M.; Matzke, H.; Leger, C.; Vambenepe, G.; Rome, M. Inert Matrices for the Transmuation of Actinides; Fabrication, Thermal Properties and Radiation Stability of Ceramic Materials. J. Nucl. Mater. 1998, 271, 544–548. [Google Scholar]
- Sickafus, K.E.; Hanrahan, R.J.; McClellan, K.J.; Mitchell, J.N.; Wetteland, C.J.; Butt, D.P.; Chodak, P.; Ramsey, K.B.; Blair, T.H.; Chidester, K.; et al. Burn and Bury Option for Plutonium. Bull. Am. Ceram. Soc. 1999, 78, 69–74. [Google Scholar]
- Gong, W.L.; Lutze, W.; Ewing, R.C. Zirconia Ceramics for Excess Weapons Plutonium Waste. J. Nucl. Mater. 2000, 277, 239–249. [Google Scholar] [CrossRef]
- Zacate, M.O.; Minervini, L.; Bradfield, D.J.; Grimes, R.W.; Sickafus, K.E. Defect cluster formation in M2O3-doped cubic ZrO2. Solid State Ion. 2000, 128, 243–254. [Google Scholar] [CrossRef]
- Sickafus, K.E.; Minervini, L.; Grimes, R.W.; Valdez, J.A.; Ishimaru, M.; Li, F.; McClellan, K.J.; Hartmann, T. Radiation tolerance of complex oxides. Science 2000, 259, 748–751. [Google Scholar] [CrossRef]
- Burakov, B.; Anderson, E.; Yagovkina, M.; Zamoryanskaya, M.; Nikolaeva, E. Behavior of 238Pu-Doped Ceramics Based on Cubic Zirconia and Pyrochlore under Radiation Damage. J. Nucl. Sci. A Technol. 2002, 3, 733–736. [Google Scholar] [CrossRef]
- Poinssot, C.; Ferry, C.; Kelm, M.; Grambow, B.; Martinez-Esparza, A.; Johnson, L.; Andriambololona, Z.; Bruno, J.; Cachoir, C.; Cavedon, J.M.; et al. Final Report of the European Project: Spent Fuel Stability under Repository Conditions; 2005; Available online: https://inis.iaea.org/collection/NCLCollectionStore/_Public/37/038/37038431.pdf?r=1&r=1 (accessed on 19 August 2019).
- Rendtorff, N.M.; Grasso, S.; Hu, C.; Suarez, G.; Aglietti, E.F.; Sakka, Y. Zircon–zirconia (ZrSiO4–ZrO2) dense ceramic composites by spark plasma sintering. J. Eur. Ceram. Soc. 2012, 32, 787–793. [Google Scholar] [CrossRef]
- Truphémus, T.; Belin, R.C.; Richaud, J.-C.; Reynaud, M.; Martinez, M.-A.; Félines, I.; Arredondo, A.; Miard, A.; Dubois, T.; Adenot, F.; et al. Structural studies of the phase separation in the UO2–PuO2–Pu2O3 ternary system. J. Nucl. Mater. Vol. 2013, 432, 378–387. [Google Scholar] [CrossRef]
- Burakov, B.E.; Yagovkina, M.A. A study of accelerated radiation damage effects in PuO2 and gadolinia- stabilized cubic zirconia, Zr0.79Gd0.14Pu0.07O1.93, doped with 238Pu. J. Nucl. Mater. 2015, 467, 534–536. [Google Scholar] [CrossRef]
- Diaz-Guillen, J.A.; Dura, O.J.; Diaz-Guillen, M.R.; Bauer, E.; Lopez de la Torre, M.A.; Fuentes, A.F. Thermophysical properties of Gd2Zr2O7powders prepared by mechanical milling: Effect of homovalent Gd3+ substitution. J. Alloys Compd. 2015, 649, 1145–1150. [Google Scholar] [CrossRef]
- Orlova, A.I.; Chuvildeev, V.N. Chemistry, Crystal Chemistry and SPS technology for elaboration of perspective materials for nuclear wastes and minor actinides consolidation. J. Nucl. Energy Sci. Rower Gener. Technol. 2017, 6, 36. [Google Scholar]
- Zhang, L.; Shelyug, A.; Navrotsky, A. Thermochemistry of UO2-ThO2 and UO2-ZrO2 Fluorite Solid Solutions. J. Chem. Thermodyn. 2017, 114, 48–54. [Google Scholar] [CrossRef]
- Orlova, A.I.; Chuvildeev, V.N.; Nokhrin, A.V.; Boldin, M.S.; Potanina, E.A.; Mikhailov, D.A.; Golovkina, L.S.; Malanina, N.A.; Tokarev, M. Next Generation Ceramic Materials for Consolidation of radioactive alpha-wastes using the Innovative Technology Spark Plasma Sintering for their preparation. In Proceedings of the 3rd World Congress on Materials Science & Engineering, Barcelona, Spain, 24–26 August 2017. [Google Scholar]
- Zubekhina, B.Y.; Burakov, B.E. Plutonium leaching from polycrystalline and monocrystalline PuO2. Radiochim. Acta 2018, 106, 119–123. [Google Scholar] [CrossRef]
- Shelyug, A.; Palomares, R.I.; Lang, M.; Navrotsky, A. Energetics of defect production in fluorite-structured CeO2 induced by highly ionizing radiation. Phys. Rev. Mater. 2018, 2, 093607. [Google Scholar] [CrossRef]
- Zhang, L.; Dzik, E.; Sigmon, G.; Szymanowski, J.; Navrotsky, A.; Burns, P. Experimental Thermochemistry of Neptunium Oxides: Np2O5 and NpO2. J. Nucl. Mater. 2018, 501, 398–404. [Google Scholar] [CrossRef]
- Yavo, N.; Sharma, G.; Kimmel, G.; Lubomirsky, I.; Navrotsky, A.; Yeheskel, O. Energetics of Bulk Lutetium Doped Ce1-xLuxO2-x/2 Compounds. J. Am. Ceram. Soc. 2018, 101, 3520–3526. [Google Scholar] [CrossRef]
- Chakoumakos, B.C. Systematics of the Pyrochlore Structure Type. Ideal A2B2X6Y. J. Solid State Chem. 1984, 53, 120–129. [Google Scholar] [CrossRef]
- Chakoumakos, B.C.; Ewing, R.C. Crystal Chemical Constraints on the Formation of Actinide Pyrochlores. In Materials Research Society Symposium Proceedings. In Scientific Basis for Nuclear Waste Management; Jantzen, C.M., Stone, J.A., Ewing, R.C., Eds.; Materials Research Society: Pittsburgh, PA, USA, 1985; pp. 641–646. [Google Scholar]
- Castro, A.; Rasines, I.; Turrillas, X.M. Synthesis, X-ray diffraction study, and ionic conductivity of new AB2O6 pyrochlores. J. Solid State Chem. 1989, 80, 227–234. [Google Scholar] [CrossRef]
- Sobolev, I.A.; Stefanovsky, S.V.; Omelianenko, B.I.; Ioudintsev, S.V.; Vance, E.R.; Jostsons, A. Comparative Study of Synroc-C Ceramics Produced by Hotpressing and Inductive Melting. Mater. Res. Soc. Symp. Proc. 1997, 465, 371–378. [Google Scholar] [CrossRef]
- Yudintsev, S.V.; Yudintseva, T.S. Nonstoichiometry of pyrochlore Ca(U,Pu)Ti2O7 and problem of brannerite (U,Pu)Ti2O6 in ceramic for actinide immobilization. In Proceedings of the 8th International Conference on Radioactive Waste Management and Environmental Remediation, Bruges, Belgium, 30 September–4 October 2001; ASME: New York, NY, USA, 2011; Volume 2, pp. 547–552. [Google Scholar]
- Kar, T.; Choudhary, R.N.P. Structural, dielectric and electrical conducting properties of CsBʹBʺO6 (Bʹ = Nb, Ta; Bʺ = W, Mo) ceramics. Mater. Sci. Eng. 2002, B90, 224–233. [Google Scholar] [CrossRef]
- Ewing, R.C.; Weber, W.J.; Lian, J. Pyrochlore (A2B2O7): A Nuclear Waste Form for the Immobilization of Plutonium and “Minor” Actinides, (Focus Review). J. Appl. Phys. 2004, 95, 5949–5971. [Google Scholar] [CrossRef]
- Whittle, K.R.; Lumpkin, G.R.; Ashbrook, S.E. Neutron diffraction and MAS NMR of Cesium Tungstate defect pyrochlores. J. Solid State Chem. 2006, 179, 512–521. [Google Scholar] [CrossRef]
- Thorogood, G.J.; Saines, P.J.; Kennedy, B.J.; Withers, R.L.; Elcombe, M.M. Diffuse scattering in the cesium pyrochlore CsTi0.5W1.5O6. Mater. Res. Bull. 2008, 43, 787–795. [Google Scholar] [CrossRef]
- Fukuda, K.; Akatsuka, K.; Ebina, Y.; Ma, R.; Takada, K.; Nakai, I.; Sasaki, T. Exfoliated Nanosheet Crystallite of Cesium Tungstate with 2D Pyrochlore. Structure: Synthesis, Characterization, and Photochromic Properties. ACS Nano 2008, 2, 1689–1695. [Google Scholar] [CrossRef]
- Lukinykh, A.N.; Tomilin, S.V.; Lizin, A.A.; Yudintsev, S.V. Radiation and Chemical durability of Actinide Crystalline Matrices, In Book of Abstracts. In Proceedings of the III International Pyroprocessing Research Conference, Dimitrovgrad, Russia, 29 November–3 December 2010; Volume 42. [Google Scholar]
- Laverov, N.P.; Yudintsev, S.V.; Livshits, T.S.; Stefanovsky, S.V.; Lukinykh, A.N.; Ewing, R.C. Synthetic Minerals with the Pyrochlore and Garnet Structures for Immobilization of Actinide-Containing Wastes. Geochem. Int. 2010, 48, 1–14. [Google Scholar] [CrossRef]
- Laverov, N.P.; Urusov, V.S.; Krivovichev, S.V.; Pakhomova, A.S.; Stefanovsky, S.V.; Yudintsev, S.V. Modular Nature of the Polysomatic Pyrochlore-Murataite Series. Geol. Ore Depos. 2011, 53, 273–294. [Google Scholar] [CrossRef]
- Hartmann, T.; Alaniz, A.; Poineau, F.; Weck, P.F.; Valdes, J.A.; Tang, M.; Jarvinen, G.D.; Czerwinski, K.R.; Sickafus, K.E. Structure studies on lanthanide technetium pyrochlores as prospective host phases to immobilize 99 technetium and fission lanthanides from effuents of reprocessed used nuclear fuels. J. Nucl. Mater. 2011, 411, 60–71. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Urusov, V.S.; Yudintsev, S.V.; Stefanovsky, S.V.; Karimova, O.V.; Organova, N.N. Crystal Structure of Murataite Mu-5, a Member of the Murataite-Pyrochlore Polysomatic Series. In Minerals as Advanced Materials II; Springer: Berlin/Heidelberg, Germany, 2012; pp. 293–304. [Google Scholar]
- Gregg, D.J.; Zhang, Y.; Middleburgh, S.C.; Conradson, S.D.; Lumpkin, G.R.; Triani, G.; Vance, E.R. The incorporation of plutonium in lanthanum zirconate pyrochlores. J. Nucl. Mater. 2013, 443, 444–451. [Google Scholar] [CrossRef]
- Nash, K.L.; Lumetta, G.J.; Vienna, J.D. Irradiated nuclear Fuel management: resource versus waste. In Radioactive Waste Management and Contaminated Siite Clean-up. Processes, Technologies and International Experience; Lee, W.E., Ojovan, M.I., Jansen, M.C., Eds.; Woodhead Publishing Limited: Oxford, UK; Cambridge, UK; Philadelphia, PA, USA; New Delhi, India, 2013; Chapter 5; pp. 145–170. [Google Scholar]
- Yudintsev, S.V.; Stefanovsky, S.V.; Nikonov, B.S. A Pyrochlore based matrix for isolation of the REE-actinide fraction of wastes from spent nuclear fuel reprocessing. Dokl. Earth Sci. 2014, 454, 54–58. [Google Scholar] [CrossRef]
- Jafar, M.; Sengupta, P.; Achary, S.N.; Tuagi, A.K. Phase evolution and microstructural studies in CaZrTi2O7 (zirconolite)–Sm2Ti2O7 (pyroclore) system. J. Eur. Ceram. Soc. 2014, 34, 4373–4381. [Google Scholar] [CrossRef]
- Jafar, M.; Sengupta, P.; Achary, S.N.; Tuagi, A.K. Phase Evolution and Microstructural Studies in CaZrTi2O7 —Nd2Ti2O7 System. J. Am. Ceram. Soc. 2014, 97, 609–616. [Google Scholar] [CrossRef]
- Hollmann, D.; Merka, O.; Schwertmann, L.; Marschall, R.; Wark, M.; Brückner, A. Active Sites for Light Driven Proton Reduction in Y2Ti2O7 and CsTaWO6 Pyrochlore Catalysts Detected by In Situ EPR. Top. Catal. 2015, 58, 769–775. [Google Scholar] [CrossRef]
- Kim, J.; Shih, P.-C.; Tsao, K.-C.; Pan, Y.-T.; Yin, X.; Sun, C.-J.; Yang, H. High-Performance Pyrochlore-Type Yttrium Ruthenate. Electrocatalyst for Oxygen Evolution Reaction in Acidic Media. J. Am. Chem. Soc. 2017, 139, 12076–12083. [Google Scholar] [CrossRef]
- McMaster, S.A.; Ram, R.; Faris, N.; Pownceby, M.I. Radionuclide disposal using the pyrochlore supergroup of minerals as a host matrix-A review. J Hazard Mater. 2018, 360, 257–269. [Google Scholar] [CrossRef]
- Kim, J.; Shih, P.-C.; Qin, Y.; Al-Bardan, Z.; Sun, C.-J.; Yang, H. A Porous Pyrochlore Y2[Ru1.6Y0.4]O7- δ Electrocatalyst for Enhanced Performance towards the Oxygen Evolution Reaction in Acidic Media. Angew. Chem. Int. Ed. 2018, 57, 13877–13881. [Google Scholar] [CrossRef]
- Morgan, P.E.D.; Ryerson, F.J. A new “Cubic” Crystal Compound. J. Mater. Sci. Lett. 1982, 1, 351–352. [Google Scholar] [CrossRef]
- Sobolev, I.A.; Stefanovsky, S.V.; Ioudintsev, S.V.; Nikonov, B.S.; Omelianenko, B.I.; Mokhov, A.V. Study of Melted Synroc Doped with Simulated High-level Waste. Mater. Res. Soc. Symp. Proc. 1997, 465, 363–370. [Google Scholar] [CrossRef]
- Stefanovsky, S.V.; Yudintsev, S.V.; Nikonov, B.S.; Mokhov, A.V.; Perevalov, S.A.; Stefanovsky, O.I.; Ptashkin, A.G. Phase Compositions and Leach Resistance of Actinide-Bearing Murataite Ceramics. Mater. Res. Soc. Symp. Proc. 2006, 893, 0893-JJ05-23. [Google Scholar] [CrossRef]
- Lian, I.; Yudintsev, S.V.; Stefanovsky, S.V.; Kirjanova, O.I.; Ewing, R.C. Ion Induced Amorphization of Murataite. Mater. Res. Soc. Symp. Proc. 2002, 713, 455–460. [Google Scholar] [CrossRef]
- Urusov, V.S.; Organova, N.I.; Karimova, O.V.; Yudintsev, S.V.; Stefanovsky, S.V. Synthetic “Murataits” as Modular Members of Pyrochlore-Murataite Polysomatic Series. Dokl. Earth Sci. 2005, 401, 319–325. [Google Scholar]
- Stefanovsky, S.V.; Yudintsev, S.V.; Nikonov, B.S.; Stefanovsky, O.I. Rare Earth-Bearing Murataite Ceramics. Mater. Res. Soc. Symp. Proc. 2007, 985, 175–180. [Google Scholar] [CrossRef]
- Stefanovsky, S.V.; Ptashkin, A.G.; Knyazev, O.A.; Dimitriev, S.A.; Yudintsev, S.V.; Nikonov, B.S. Inductive Cold Crucible Melting of Actinide-bearning Murataite-based Ceramics. J. Alloys Compd. 2007, 444, 438–442. [Google Scholar] [CrossRef]
- Stefanovsky, S.V.; Yudintsev, S.V.; Perevalov, S.A.; Startseva, I.V.; Varlakova, G.A. Leach Resistance of Murataite-based Ceramics Containing Actinides. J. Alloys Compd. 2007, 444, 618–620. [Google Scholar] [CrossRef]
- Stefanovsky, S.V.; Ptashkin, A.G.; Knyazev, O.A.; Zen’kovskaja, M.S.; Stefanovsky, O.I.; Yudintsev, S.V.; Nikonov, B.S.; Lapina, M.I. Melted Murataite Ceramics Containing Simulated Actinide/Rere Earth Fraction of High Level Waste. In Proceedings of the WM2008 Conference, Phoenix, AZ, USA, 24–28 February 2008. [Google Scholar]
- Krivovichev, S.V.; Yudintsev, S.V.; Stefanovsky, S.V.; Organova, N.I.; Karimova, O.V.; Urusov, V.S. Murataite–Pyrochlore Series: A Family of Complex Oxides with Nanoscale Pyrochlore Clusters. Angew. Chem. Int. Ed. 2010, 49, 9982–9984. [Google Scholar] [CrossRef]
- Laverov, N.P.; Yudintsev, S.V.; Stefanovskii, S.V.; Omel’yanenko, B.I.; Nikonov, B.S. Murataite Matrices for Actinide Wastes. Radiochemistry 2011, 53, 229–243. [Google Scholar] [CrossRef]
- Pakhomova, A.S.; Krivovichev, S.V.; Yudintsev, S.V.; Stefanovsky, S.V. Synthetic Murataite-3c, a complex form for long-term immobilization of nuclear waste: crystal structure and its comparison with natural analogue. Z. Kristallogr 2013, 228, 151–156. [Google Scholar] [CrossRef]
- Poglyad, S.S.; Pryzhevskaya, E.A.; Lizin, A.A.; Tomilin, S.V.; Murasova, O.V. On possibility of the murataite fusion temperature lowering for radioactive waste immobilization. J. Phys. Conf. Ser. 2018, 1133, 012019. [Google Scholar] [CrossRef]
- Lizin, A.A.; Tomilin, S.V.; Poglyad, S.S.; Pryzhevskaya, E.A.; Yudintsev, S.V.; Stefanovsky, S.V. Murataite: a matrix for immobilizing waste generated in radiochemical reprocessing of spent nuclear fuel. J. Radioanal. Nucl. Chem. 2018, 318, 2363–2372. [Google Scholar] [CrossRef]
- Clinard, F., Jr.; Hobbs, L.W.; Land, C.C.; Peterson, D.E.; Rohr, D.L.; Roof, R.B. Alpha Decay Self-irradiation Damage in 238Pu-substituted Zirconolite. J. Nucl. Mater. 1982, 105, 248–256. [Google Scholar]
- Clinard, F., Jr.; Peterson, D.E.; Rohr, D.L.; Hobbs, L.W. Self-irradiation Effects in 238Pu-substituted Zirconolite: I. Temperature Dependence of Damage. J. Nucl. Mater. 1984, 126, 245–254. [Google Scholar]
- Boult, K.A.; Dalton, J.T.; Evans, J.P.; Hall, A.R.; Inns, A.J.; Marples, J.A.C.; Paige, E.L. The Preparation of fully-active Synroc and its radiation stability–Final Report October 1988; UKAEA: Harwell, UK, 1988.
- Vance, E.R.; Ball, C.J.; Day, R.A.; Smith, K.L.; Blackford, M.G.; Begg, B.D.; Angel, P.J. Actinide and Rare Earth Incorporation in Zirconolite. J. Alloys Compd. 1994, 213–214, 406–409. [Google Scholar] [CrossRef]
- Vance, E.R.; Angel, P.J.; Begg, B.D.; Day, R.A. Zirconolite-Rich Titanate Ceramics for High-Level Actinide Wastes. In Scientific Basis for Nuclear Waste Management XVII; van Konynenburg, R., Barkatt, A.A., Eds.; Materials Research Society: Pittsburgh, PA, USA, 1994; pp. 293–298. [Google Scholar]
- Zhang, Y.; Stewart, M.W.A.; Li, H.; Carter, M.L.; Vance, E.R.; Moricca, S. Zirconolite-rich Titanate Ceramics for Immobilization of Actinides–Waste form/HIP Can Interactions and Chemical Durability. J. Nucl. Mater. 2009, 395, 69–74. [Google Scholar] [CrossRef]
- Whittle, K.R.; Hyatt, N.C.; Smith, K.L.; Margiolaki, I.; Berry, F.J.; Knight, K.E.; Lumpkin, G.R. Combined neutron and X-ray diffraction determination of disorder in doped zirconolite-2M. Am. Mineral. 2012, 97, 291–298. [Google Scholar] [CrossRef]
- Bohre, A.; Avasthi, K.; Shrivastava, O.P. Synthesis, Characterization, and Crystal Structure Refinement of Lanthanum and Yttrium Substituted Polycrystalline 2M Type Zirconolite Phases: Ca1-xMexZrTi2O7 (Me = Y, La and x = 0.2). J. Powder Technol. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Lumpkin, G.R.; Gao, Y.; Giere, R.; Williams, C.T.; Mariano, A.N.; Geisler, T. The role of Th-U minerals in assessing the performance of nuclear waste forms. Mineral. Mag. 2014, 78, 1071–1095. [Google Scholar] [CrossRef]
- Deschanels, K.; Seydonx-Guillaume, A.M.; Morgin, V.; Mesbah, A.; Tribet, M.; Moloney, M.P.; Serruys, Y.; Peuget, S. Swelling induced by alpha decay in monazite and zirconolite ceramics: A XRD and TEM comparative study. J. Nucl. Mater. 2014, 448, 184–194. [Google Scholar] [CrossRef]
- Zhang, K.; Wen, G.; Zhang, H.; Teng, Y. Self-propagating high-temperature synthesis of CeO2 incorporated zirconolite-rich waste forms and the aqueous durability. J. Eur. Cream. Soc. 2015, 35, 3085–3093. [Google Scholar] [CrossRef]
- Zhang, K.; Wen, G.; Jia, Z.; Teng, Y.; Zhang, H. Self-Propagating High-Temperature Synthesis of Zirconolite Using CuO and MoO3 as the Oxidants. Int. J. Appl. Ceram. Technol. 2015, 12, E111–E120. [Google Scholar] [CrossRef]
- Wen, G.; Zhang, K.; Yin, D.; Zhang, H. Solid-state reaction synthesis and aqueous durability of Ce-doped zirconolite-rich ceramics. J. Nucl. Mater. 2015, 466, 113–119. [Google Scholar] [CrossRef]
- Popa, K.; Cologna, M.; Martel, L.; Staicu, D.; Cambriani, A.; Ernstberger, M.; Raison, P.E.; Somers, J. CaTh(PO4)2 cheralite as a candidate ceramic nuclear waste form: Spark plasma sintering and physicochemical characterization. J. Eur. Ceram. 2016, 36, 4115–4121. [Google Scholar] [CrossRef]
- Wen, J.; Sun, C.; Dholabhai, P.P.; Xia, Y.; Tang, M.; Chen, D.; Yang, D.Y.; Li, Y.H.; Uberuaga, B.P.; Wang, Y.Q. Temperature dependence of the radiation tolerance of nanocrystalline pyrochlores A2Ti2O7 (A = Gd, Ho and Lu). Acta Mater. 2016, 110, 175–184. [Google Scholar] [CrossRef]
- Clark, B.M.; Sundaram, S.K.; Misture, S.T. Polymorphic Transitions in Cerium-Substituted Zirconolite (CaZrTi2O7). Sci. Rep. 2017, 7, 5920. [Google Scholar] [CrossRef]
- Sun, S.K.; Stennett, M.C.; Corkhill, C.L. Reactive spark plasma synthesis of CaZrTi2O7 zirconolite ceramics for plutonium disposition. J. Nucl. Mater. 2018, 500, 11–14. [Google Scholar] [CrossRef]
- McCaughherty, S.; Crosvenor, A.P. Low-temperature synthesis of CaZrTi2O7 zirconolite-type materialss using ceramic, coprecipitation, and soil-gel methods. J. Mater. Chem. C 2019, 7, 177–187. [Google Scholar] [CrossRef]
- Zhang, K.; Yin, D.; Xu, K.; Zhang, H. Self-Propagating Synthesis and Characterization Studies of Gd-Bearing Hf-Zirconolite Ceramic Waste Forms. Materials 2019, 12, 178. [Google Scholar] [CrossRef]
- Boult, A.; Dalton, J.T.; Evans, J.P.; Hall, A.R.; Inn, A.J.; Marples, J.A.C.; Paige, E.L. The Preparation of Fully-active Synroc and its Radiation Stability. Rep. Aere-R. 1987, 13, 318. [Google Scholar]
- Vance, E.R.; Carter, M.L.; Zhang, Z.; Finnie, K.S.; Thomson, S.J.; Begg, B.D. Uranium valences in perovskite, CaTiO3. In Environmental Issues and Waste Management Technologies in the Ceramic & Nuclear Industries IX; The American Ceramic Society: Westeville, OH, USA, 2004; Volume 155, pp. 3–10. [Google Scholar]
- Bozadjiev, L.; Georgiev, G.; Parashkevov, D. Synthesis of perovskites and perovskite based technical stones. In National Conference “GEOSCIENSES 2006”; Bulgarian Geophysical Society: Sofia, Bulgaria, 2006; pp. 127–129. [Google Scholar]
- Bowles, J.F.W.; Howie, R.A.; Vaughan, D.J.; Zussman, J. Rock-Forming Minerals, V5A: Non-Silicates: Oxides, Hydroxides and Sulphides; The Geological Society: London, UK, 2011; p. 920. [Google Scholar]
- Stewart, M.W.A.; Vance, E.R.; Moricca, S.A.; Brew, D.R.; Cheung, C.; Eddowes, T.; Bermudez, W. Immobilisation of Higher Activity Wastes from Nuclear Reactor Production of 99Mo. Sci. Technol. Nucl. Install. 2013, 2013, 1–16. [Google Scholar] [CrossRef]
- Ghosh, B.; Dutta, A.; Shannigrahi, S.; Sinha, T.P. Combined XPS and first principles study of double-perovskite Ca2GdTaO6. J. Mater. Sci. 2014, 49, 819–826. [Google Scholar] [CrossRef]
- Bohre, A.; Avasthi, K.; Shrivastava, O.P. Structure Refinement of Polycrystalline Orthorombic Calcium Titanate Substituted by Lanthanum: Ca1-xLnxTiO3 (x = 0.1–0.4). Crystallogr. Rep. 2014, 59, 944–948. [Google Scholar] [CrossRef]
- Livshits, T.S.; Zhang, J.; Yudintsev, S.V.; Stefanovsky, S.V. New titanate matrices for immobilization of REE-actinide high level waste. J. Radioanal. Nucl. Chem. 2014, 304, 47–52. [Google Scholar] [CrossRef]
- Mahadik, P.S.; Sengupta, P.; Halder, R.; Abraham, G.; Dey, G.K. Perovskite-Ni composite: A potential route for management of radioactive metallic waste. J. Hazard. Mater. 2015, 287, 207–216. [Google Scholar] [CrossRef]
- Carter, M.L.; Vance, E.R.; Mitchell, D.R.G.; Hanna, J.V.; Zhang, Z.; Loi, E. Fabrication, Characterization, and Leach Testing of Hollandite, (Ba,Cs)(Al,Ti)2Ti6O16. J. Mater. Res. 2002, 17, 2578–2589. [Google Scholar] [CrossRef]
- Carter, M.L.; Vance, E.R.; Li, H. Hollandite-rich Ceramic Melts for the Immobilization of Cs. Mater. Res. Soc. Proc. 2004, 807, 249–254. [Google Scholar] [CrossRef]
- Whittle, K.R.; Ashbrook, S.; Redfem, S.; Lumpkin, G.R. Structural Studies of Hollandite-Based Radioactive Waste Forms Structural Studies of Hollandite-Based Radioactive Waste Forms. Mater. Res. Soc. Symp. Proc. 2004, 87, 1–7. [Google Scholar]
- Nishiyama, N.; Rapp, R.P.; Irifune, T.; Sanehira, T.; Yamazaki, D.; Funakoshi, K. Stability and P-V-T equation of state KAlSi3O8-hollandite determined by in situ X-Ray observations and implications for dynamics of subducted continental crust material. Phys. Chem. Miner. 2005, 32, 627–637. [Google Scholar] [CrossRef]
- Addelouas, A.; Utsunomiya, S.; Suzuki, T.; Grambow, B.; Advocat, T.; Bart, F.; Ewing, R.C. Effect of ionizing radiation on the hollandite structure-type: Ba0.85Cs0.26Al1.35Fe0.77Ti5.90O16. Am. Mineral. 2008, 93, 241–247. [Google Scholar] [CrossRef]
- Shluk, L.; Niewa, R. Crystal Structure and Magnetic Properties of the Novel Hollandite Ba1.3Co1.3Ti6.7O16. Z. Naturforsch. 2011, 66b, 1097–1100. [Google Scholar] [CrossRef]
- Shabalin, B.; Titov, Y.; Zlobenko, B.; Bugera, S. Ferric Titanous Hollandite Analogues – Matrices for Immobilization of Cs-Containing Radioactive Waste: Synthesis. Mineral. J. 2013, 35, 12–18. [Google Scholar]
- Chen, T.-Y.; Maddrell, E.R.; Hyatt, N.C.; Hriljac, J.A. A potential wasteform for Cs immobilization: synthesis, structure determination, and aqueous durability of Cs2TiNb6O18. Inorg. Chem. 2016, 55, 12686–12695. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Xu, Y.; Shuller-Nickles, L.; Amoroso, J.; Frenkel, A.I.; Li, Y.; Gong, W.; Lilova, K.; Navrotsky, A.; Brinkman, K.S. Compositional control of radionuclide retention in hollandite-based ceramic waste forms for Cs immobilization. J. Am. Ceram. Soc. 2018, 102, 4314–4324. [Google Scholar] [CrossRef]
- Grote, R.; Zhao, M.; Shuller-Nickles, L.; Amoroso, J.; Gong, W.; Lilova, K.; Navrotsky, A.; Tang, M.; Brinkman, K.S. Compositional control of tunnel features in hollandite-based ceramics: Structure and stability of (Ba,Cs)1.33(Zn,Ti)8O16. J. Mater. Sci. 2018, 54, 1112–1125. [Google Scholar] [CrossRef]
- Geller, S.; Miller, C.E. Silicate garnet yttrium-iron garnet solid solution. Am. Mineral. 1959, 44, 1115–1120. [Google Scholar]
- Ito, J.; Frondel, C. Synthesis zirconium and titanium garnets. Am. Mineral. 1967, 52, 773–781. [Google Scholar]
- Rickwood, P.C. On recasting analyses of garnet into end-member molecules. Contrib. Mineral. Petrol. 1968, 18, 175–198. [Google Scholar] [CrossRef]
- Novak, G.A.; Gibbs, G.V. The crystal chemistry of the silicate garnets. Am. Mineral. 1971, 56, 791–825. [Google Scholar]
- Dowty, E. Crystal chemistry of titanian and zirconian garnet: I. Review and spectral studies. Am. Mineral. 1971, 56, 1983–2009. [Google Scholar]
- Kanke, Y.; Navrotsky, A. A calorimetric study of the lanthanide aluminum oxides and the lantha-nide gallium oxides: Stability of the perovskites and the garnets. J. Solid State Chem. 1998, 141, 424–436. [Google Scholar] [CrossRef]
- Burakov, B.E.; Anderson, E.B.; Zamoryanskaya, M.V.; Petrova, M.A. Synthesis and Study of 239Pu-Doped Gadolinium-Aluminum Garnet. In Material Research Society Symposium Proceedings Scientific Basis for Nuclear Waste Management XXIII; Materials Research Society: Warendalle, PA, USA, 2000; Volume 608, pp. 419–422. [Google Scholar]
- Yudintsev, S.V. Incorporation of U, Th, Zr, and Gd into the Garnet-structured Host. In Proceedings of the 8th International Conference on Radioactive Waste Management and Environmental Remediation, The American Society of Mechanical Engineers, Bruges, Belgium, 30 September–4 October 2001. [Google Scholar]
- Yudintsev, S.V.; Lapina, M.I.; Ptashkin, A.G. Accommodation of uranium into the garnet structure. MRS Symp. Proc. 2002, 713, 477–480. [Google Scholar] [CrossRef]
- Utsunomiya, S.; Wang, L.M.; Yudintsev, S.; Ewing, R.C. Ion irradiation-induced amorphization and nano-crystal formation in garnets. J. Nucl. Mater. 2002, 303, 177–187. [Google Scholar] [CrossRef]
- Yudintsev, S.V. A Structural-chemical Approach to Selecting Crystalline Matrices for Actinide Immobilization. Geol. Ore Depos. 2003, 45, 151–165. [Google Scholar]
- Utsunomiya, S.; Yudintsev, S.V.; Ewing, R.C. Radiation effects in ferrate garnet. J. Nucl. Mater. 2005, 336, 251–260. [Google Scholar] [CrossRef]
- Maslakov, K.I.; Teterin, U.A.; Vukchevich, L.; Udintseva, T.C.; Udintsev, S.V.; Ivanov, K.E.; Lapina, M.I. Issledovanie obraztsov keramiki (Ca2.5Th0.5)Zr2Fe3O12, (Ca1.5GdTh0.5)(ZrFe)Fe3O12 i (Ca2.5Ce0.5)Zr2Fe3O12 so strukturoi granata. Radiohimiya 2007, 49, 31–37. [Google Scholar]
- Suarez, M.; Fernandez, A.; Menendez, J.L.; Torrecillas, R. Transparent Yttrium Aluminium Garnet Obtained by Spark Plasma Sintering of Lyophilized Gels. J. Nanomater. 2009, 2009, 1–5. [Google Scholar] [CrossRef]
- Tomilin, S.V.; Lizin, A.A.; Lukinykh, A.N.; Livshits, T.S. Radiation Resistance and Chemical of Yttrium Aluminium Garnet. Radiochemistry 2011, 53, 186–190. [Google Scholar] [CrossRef]
- Golovkina, L.S.; Orlova, A.I.; Nokhrin, A.V.; Boldin, M.S.; Sakharov, N.V. Ceramics based on Yttrium Aluminium Garnet Containing Nd and Sm obtained by Spark Plasma Sintering. Adv. Ceram. Sci. Eng. 2013, 2, 261–265. [Google Scholar]
- Hanc, E.; Zając, W.; Lu, L. On fabrication procedures of Li-ion conducting garnets. J. Solid State Chem. 2017, 248, 51–60. [Google Scholar] [CrossRef]
- Yamada, H.; Ito, T.; Basappa, R.H.; Bekarevich, R.; Mitsuishi, K. Influence of strain on local structure and lithium ionic conduction in garnet-type solid electrolyte. J. Power Sources 2017, 368, 97–106. [Google Scholar] [CrossRef]
- Selvi, M.M.; Chakraborty, D.; Venkateswaran, C. Magnetodielectric coupling in multiferroic holmium iron garnets. J. Magn. Magn. Mater. 2017, 423, 39–45. [Google Scholar] [CrossRef]
- Golovkina, L.; Orlova, A.; Boldin, M.; Sakharov, N.; Chuvil’deev, V.; Nokhrin, A.; Konings, R.; Staicu, D. Development of composite ceramic materials with improved thermal conductivity and plasticity based on garnet-type oxides. J. Nucl. Mater. 2017, 489, 158–163. [Google Scholar] [CrossRef]
- Golovkina, L.S.; Nokhrin, A.V.; Boldin, M.S.; Lantsev, E.A.; Orlova, A.I.; Chuvil’deev, V.N.; Murashov, A.A.; Sakharov, N.V. Preparation of Fine-Grained Y2.5Nd0.5Al5O12 + MgO composite ceramics for Inert Matrix Fuels by Spark Plasma Sintering. Inorg. Mater. 2018, 54, 1291–1298. [Google Scholar] [CrossRef]
- Golovkina, L.S.; Orlova, A.I.; Nokhrin, A.V.; Boldin, M.S.; Lantsev, E.A.; Chuvil’deev, V.N.; Sakharov, N.V.; Shotin, S.V.; Zelenov, A.Y. Spark Plasma Sintering of fine-grained ceramic-metal composites YAG: Nd-(W,Mo) based on garnet-type oxide Y2.5Nd0.5Al5O12 for inert matrix fuel. J. Nucl. Mater. 2018, 511, 109–121. [Google Scholar] [CrossRef]
- Golovkina, L.S.; Orlova, A.I.; Nokhrin, A.V.; Boldin, M.S.; Chuvil’deev, V.N.; Sakharov, N.V.; Belkin, O.A.; Shotin, S.V.; Zelenov, A.Y. Spark Plasma Sintering of fine-grain ceramic-metal composites based on garnet-structure oxide Y2.5Nd0.5Al5O12 for inert matrix fuel. Mater. Chem. Phys. 2018, 214, 516–526. [Google Scholar] [CrossRef]
- Golovkina, L.S.; Orlova, A.I.; Chuvil’deev, V.N.; Boldin, M.S.; Lantcev, E.A.; Nokhrin, A.V.; Sakharov, N.V.; Zelenov, A.Y. Spark Plasma Sintering of high-density fine-grained Y2.5Nd0.5Al5O12 + SiC composite ceramics. Mater. Res. Bull. 2018, 103, 211–215. [Google Scholar] [CrossRef]
- Golovkina, L.; Orlova, A.; Boldin, M.; Lantsev, E.; Sakharov, N.; Zelenov, A.; Chuvil’deev, V. Composite Ceramics Based on Garnet-type Oxide Y2.5Nd0.5Al5O12 and Silicon Carbide. Mater. Res. Bull. 2018, 4, 518–524. [Google Scholar]
- Gong, W.L.; Ewing, R.C.; Wang, L.M.; Xie, H.S. Crichtonite Structure Type (AM21O38 and A2M19O36) as a Host Phase in Crystalline Waste Form Ceramics. Scientific Basis for Nuclear Waste Management XVIII. T. Murakami and R.C. Ewing, Eds. Proc. Mater. Res. Soc. 1995, 353, 807–815. [Google Scholar] [CrossRef]
- Grey, I.E.; Lloyd, D.J.; White, J.S., Jr. The Structure of Crichtonite and its Relationship to Senaite. Am. Mineral. 1976, 61, 1203–1212. [Google Scholar]
- Wulser, P.-A.; Meisser, N.; Brugger, J.; Schenk, K.; Ansermet, S.; Bonin, M.; Bussy, F. Cleusonite, (Pb,Sr)(U4+,U6+)(Fe2+,Zn)2(Ti,Fe2+,Fe3+)18(O,OH)38, a new mineral species of the crichtonite group from the western Swiss Alps. Eur. J. Mineral. 2005, 17, 933–942. [Google Scholar] [CrossRef]
- Mills, S.J.; Bindi, L.; Cadoni, M.; Kampf, A.R.; Ciriotti, M.E.; Ferraris, G. Paseroite, PbMn2+(Mn2+,Fe2+)2(V5+,Ti,Fe3+,□)18O38, a new member of the crichtonite group. Eur. J. Mineral. 2012, 24, 1061–1067. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Aksenov, S.M.; Chukanov, N.V.; Menezes, L.A.D. Crystal Structure of Almeidaite, a New Mineral of the Crichtonite Group. Dokl. Chem. 2014, 455, 53–57. [Google Scholar] [CrossRef]
- Biagioni, C.; Orlandi, P.; Pasero, M.; Nestola, F.; Bindi, L. Mapiquiroite, (Sr,Pb)(U,Y)Fe2(Ti,Fe3+)18O38, a new member of the crichtonite group from the Apuan Alps, Tuscany, Italy. Eur. J. Mineral. 2014, 26, 427–437. [Google Scholar] [CrossRef]
- Menezes Filho, L.A.D.; Chukanov, N.V.; Rastsvetaeva, R.K.; Aksenov, S.M.; Pekov, I.V.; Chaves, M.L.S.C.; Richards, R.P.; Atencio, D.; Brandao, P.R.G.; Scholz, R.; et al. Almeidaite, Pb(Mn,Y)Zn2(Ti,Fe3+)18O36(O,OH)2, a new crichtonite-group mineral, from Novo Horizonte, Bahia. Braz. Mineral. Mag. 2015, 79, 269–283. [Google Scholar] [CrossRef]
- Rezvukhin, D.I.; Malkovets, V.G.; Sharygin, I.S.; Kuzmin, D.V.; Gibsher, A.A.; Litasov, K.D.; Pokhilenko, A.P.; Sobolev, N.V. Inclusions of Crichtonite Group Minerals in Pyropes from the Internatsionalnaya Kimberlite Pipe. Dokl. Akad. Nauk. 2016, 466, 714–717. [Google Scholar] [CrossRef]
- Ishiguro, T.; Tanaka, K.; Marumo, F.; Ismail, M.G.M.U.; Hirano, S. Somiya, Freudenbergite. Acta Cryst. 1978, B34, 255–256. [Google Scholar] [CrossRef]
- Vance, E.R.; Angel, P.J.; Cassidy, D.J.; Stewart, M.W.A.; Blackford, M.G.; McGlinn, P.A. Freudenbergite: A Possible Synroc Phase for Sodium-Bearing High-Level Waste. J. Am. Ceram. Soc. 1994, 77, 1576–1580. [Google Scholar] [CrossRef]
- Ren, X.; Komarneni, S.; Roy, D.M. Novel CsAl2PO6 of pollucite structure: synthesis and characterization. Mater. Res. Bull. 1990, 25, 665–670. [Google Scholar] [CrossRef]
- Komarneni, S.; Menon, V.C.; Li, Q.H.; Roy, R.; Ainger, F. Microwave-hydrothermal processing of BiFeO3 and CsAl2PO6. J. Am. Ceram. Soc. 1996, 75, 1409–1412. [Google Scholar] [CrossRef]
- Aloy, A.S.; Kol’tsova, T.I.; Trofimenko, A.V.; Tutov, A.G. New compound with pollucite structure forming in process of synthesis and crystallization of cesium-alumphosphate glasses. Radiochemistry 2000, 42, 273–274. [Google Scholar]
- Hirst, J.P.; Claridge, J.B.; Rosseinsky, M.J.; Bishop, P. High temperature synthesis of a noncentrosymmetric site-ordered cobalt aluminophosphate related to the pollucite structure, The Royal Society of Chemistry. Chem. Commun. 2003, 6, 684–685. [Google Scholar] [CrossRef]
- Orlova, A.I. Crystal Chemical View on Elaboration of Ecology Safe Materials for Immobilization of Alkaline elements of Radwaste. In III International Pyroprocessing Research Conference; NIIAR: Dimitrovgrad, Russia, 2010; pp. 27–28. [Google Scholar]
- Loginova, E.E.; Orlova, A.I.; Mikhailov, D.A.; Troshin, A.N.; Borovikova, E.Y.; Samoilov, S.G.; Kazantsev, G.N.; Kazakova, A.Y.; Demarin, V.T. Phosphorus-Containing Compounds of Pollucite Structure and Radiochemical Problems. Radiochemistry 2011, 53, 593–603. [Google Scholar] [CrossRef]
- Orlova, A.I.; Volgutov, V.Y.; Mikhailov, D.A.; Troshin, A.N.; Golovkina, L.S.; Skuratov, V.A.; Kirilkin, N.S.; Chuvil’diev, V.N.; Nokhrin, A.V.; Boldin, M.S.; et al. Mineral like compounds with NZP and Pollucite structures: synthesis of high density ceramic and radiation testing. In European Congress on Advanced Materials and Processes; EUROMAT 2011; FEMS: Montpellier, France, 2011. [Google Scholar]
- Yanase, I.; Saito, Y.; Kobayashi, H. Synthesis and thermal expansion of (V, P, Nb)-replaced pollucite. Ceram. Int. 2012, 38, 811–815. [Google Scholar] [CrossRef]
- Orlova, A.I.; Troshin, A.N.; Mikhailov, D.A.; Chuvil’deev, V.N.; Boldin, M.S.; Sakharov, N.V.; Nokhrin, A.V.; Skuratov, V.A.; Kirilkin, N.S. Phosphorus-Containing Cesium Compounds of Pollucite Structure. Preparation of High-Density Ceramic and Its Radiation Tests. Radiochemistry 2014, 56, 98–104. [Google Scholar] [CrossRef]
- Klapshin, Y.P. Chemical and Phase Transformations during the Synthesis of Cs[MgR0.5P1.5O6] (R = B, Al, Fe) Complex Oxides from Metal Chlorides. Russ. J. Inorg. Chem. 2018, 63, 1156–1163. [Google Scholar] [CrossRef]
- Klapshin, Y.P. Chemical and Phase Transformations during the Synthesis of Cs[MgR0.5P1.5O6] (R = B, Al, Fe) Complex Oxides from Metal Nitrates. Russ. J. Inorg. Chem. 2018, 63, 1381–1388. [Google Scholar] [CrossRef]
- Townes, W.D.; Fang, J.H.; Perrotta, A.J. The Crystal Structure and Refinement of Ferromagnetic Barium Ferrite, BaFe12O19. Z. Kristallogr. 1967, 125, 437–449. [Google Scholar] [CrossRef]
- Morgan, P.E.D.; Clarke, D.R.; Jantzen, C.M.; Harker, A.B. High Alumina Tailored Nuclear Waste Ceramics. J. Am. Ceram. Soc. 1981, 64, 249–258. [Google Scholar] [CrossRef]
- Harker, A.B.; Jantzen, C.M.; Clarke, D.R.; Morgan, P.E.D. Tailored Ceramic Nuclear Waste Forms: Preparation and Characterization. In Science Basis for Nuclear Waste Management, III; Moore, J.G., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 139–146. [Google Scholar]
- Morgan, P.E.D.; Cirlin, E.H. The Magnetoplumbite Crystal Structure as a Radwaste Host. J. Am. Ceram. Soc. 1982, 65, C114–C115. [Google Scholar] [CrossRef]
- Jantzen, C.M.; Clarke, D.R.; Morgan, P.E.D.; Harker, A.B. Leaching of Polyphase Nuclear Waste Ceramics: Microstructural and Phase Characterization. J. Am. Ceram. Soc. 1982, 65, 292–300. [Google Scholar] [CrossRef]
- Bansal, N.P.; Zhu, D. Thermal properties of oxides with magnetoplumbite structure for advanced thermal barrier coatings. Surf. Coat. Technol. 2008, 202, 2698–2703. [Google Scholar] [CrossRef]
- Men, D.; Patel, M.K.; Usov, I.O.; Pivin, J.C.; Porter, J.R.; Mecartney, M.L. Radiation Damage of LaMgAl11O19 and CeMgAl11O19 Magnetoplumbite. J. Am. Ceram. Soc. 2013, 96, 3325–3332. [Google Scholar]
- Rakshit, S.K.; Parida, S.C.; Lilova, K.; Navrotsky, A. Thermodynamic studies of CaLaFe11O19(s). J. Solid State Chem. 2013, 2, 68–74. [Google Scholar] [CrossRef]
- Angle, J.P.; Nelson, A.T.; Men, D.; Mecartney, M.L. Thermal measurements and computational simulations of three-phase (CeO2–MgAl2O4–CeMgAl11O19) and four-phase (3Y-TZP–Al2O3– MgAl2O4–LaPO4) composites as surrogate inert matrix nuclear fuel. J. Nucl. Mater. 2014, 454, 69–76. [Google Scholar] [CrossRef]
- Ewing, R.C.; Lutze, W.; Weber, W.J. Zircon: A host-phase for the disposal of weapons plutonium. J. Mater. Res. 1995, 10, 243–246. [Google Scholar] [CrossRef]
- Shi, Y.; Huang, X.; Yan, D. Fabrication of Hot-Pressed Zircon Ceramics: Mechanical Properties and Microstructure. Ceram. Int. 1997, 23, 457–462. [Google Scholar] [CrossRef]
- Meldrum, A.; Boatner, L.A.; Ewing, R.C. A comparison of radiation effects in crystalline ABO4-type phosphates and silicates. Mineral. Mag. 2000, 64, 185–194. [Google Scholar] [CrossRef]
- Burakov, B.E.; Anderson, E.B.; Zamoryanskaya, M.V.; Yagovkina, M.A.; Strykanova, E.E.; Nikolaeva, E.V. Synthesis and Study of 239Pu-Doped Ceramics Based on Zircon, (Zr,Pu)SiO4, and Hafnon, (Hf,Pu)SiO4. In Material Research Society Symposium Proceedings Scientific Basis for Nuclear Waste Management XXIV; Materials Research Society: Warrendale, PA, USA, 2001; Volume 663, pp. 307–313. [Google Scholar]
- Hanchar, J.M.; Burakov, B.E.; Anderson, E.B.; Zamoryanskaya, M.V. Investigation of Single Crystal Zircon, (Zr,Pu)SiO4, Doped with 238Pu. In Scientific Basis for Nuclear Waste Management XXVI, Materials Research Society Symposium Proceedings; Finch, R.J., Bullen, D.B., Eds.; Materials Research Society: Warrendale, PA, USA, 2003; Volume 757, pp. 215–225. [Google Scholar]
- Geisler-Wierwille, T.; Burakov, B.E.; Zirlin, V.; Nikolaeva, L.; Pöml, P. A Raman spectroscopic study of high-uranium zircon from the Chernobyl “Lava”. Eur. J. Mineral. 2005, 17, 883–894. [Google Scholar] [CrossRef]
- Burakov, B.E.; Domracheva, Y.V.; Zamoryanskaya, M.V.; Petrova, M.A.; Garbuzov, V.M.; Kitsay, A.A.; Zirlin, V.A. Development and synthesis of durable self-glowing crystals doped with plutonium. J. Nucl. Mater. 2009, 385, 134–136. [Google Scholar] [CrossRef]
- Rendtorff, N.M.; Grasso, S.; Hu, C.; Suarez, G.; Aglietti, E.F.; Sakka, Y. Dense zircon (ZrSiO4) ceramics by high energy ball milling and spark plasma sintering. Ceram. Int. 2012, 38, 1793–1799. [Google Scholar] [CrossRef]
- IAEA. Radiation Protection and NORM Residue Management in the Zircon and Zirconia Industries; Safety Reports Series 51; IAEA: Vienna, Austria, 2007; 149p. [Google Scholar]
- Pöml, P.; Burakov, B.; Geisler, T.; Walker, C.T.; Grange, M.L.; Nemchin, A.A.; Berndt, J.; Fonseca, R.O.C.; Bottomley, P.D.W.; Hasnaoui, R. Micro-analytical uranium isotope and chemical investigations of zircon crystals from the Chernobyl “lava” and their nuclear fuel inclusions. J. Nucl. Mater. 2013, 439, 51–56. [Google Scholar] [CrossRef]
- Ojovan, M.I.; Burakov, B.E.; Lee, W.E. Radiation-induced microcrystal shape change as a mechanism of wasteform degradation. J. Nucl. Mater. 2018, 501, 162–171. [Google Scholar] [CrossRef]
- Hayden, L.A.; Watson, E.B.; Wark, D.A. A thermobarometer for sphene (titanite). Contrib. Mineral. Petrol. 2008, 155, 529–540. [Google Scholar] [CrossRef]
- Park, T.J.; Li, S.; Navrotsky, A. Thermochemistry of glass forming Y-substituted Sr-analogues of titanite (SrTiSiO5). J. Mater. Res. 2009, 24, 3380–3386. [Google Scholar] [CrossRef]
- Scanu, T.; Guglielmi, J.; Colomban, P. Ion exchange and hot corrosion of ceramic composites matrices: A vibrational and microstructural study. Solid State Ion. 1994, 70–71, 109–120. [Google Scholar] [CrossRef]
- Weber, W.J. Radiation-induced Swelling and Amorphization in Ca2Nd8(SiO4)6O2. Radiat. Eff. Defects Solids 1983, 77, 295–308. [Google Scholar] [CrossRef]
- Fahey, J.A.; Weber, W.J.; Rotella, F.J. An X-ray and Neutron Powder Diffraction Study of the Ca2+xNd8−x(SiO4)6O2−0.5x System. J. Solid State Chem. 1985, 60, 145–158. [Google Scholar] [CrossRef]
- Weber, W.J. Alpha-decay-induced Amorphization in Complex Silicate Structures. J. Am. Ceram. Soc. 1993, 76, 1729–1738. [Google Scholar] [CrossRef]
- Utsunomiya, S.; Yudintsev, S.; Wang, L.M.; Ewing, R.C. Ion-beam and Electron-beam Irradiation of Synthetic Britholite. J. Nucl. Mater. 2003, 322, 180–188. [Google Scholar] [CrossRef]
- Leo’n-Reina, L.; Losilla, E.R.; Martı´nez-Lara, M.; Bruque, S.; Llobet, A.; Sheptyakov, D.V.; Aranda, M.A.G. Interstitial Oxygen in Oxygen-stoichiometric Apatites. J. Mater. Chem. 2005, 15, 2489–2498. [Google Scholar] [CrossRef]
- Terra, O.; Dacheux, N.; Audubert, F.; Podor, R. Immobilization of tetravalent actinides in phosphate ceramics. J. Nucl. Mater. 2006, 352, 224–232. [Google Scholar] [CrossRef]
- Malavasi, L.; Fisher, C.A.J.; Islam, M.S. Oxide-ion and proton conducting electrolyte materials for clean energy applications: Structural and mechanistic features. Chem. Soc. Rev. 2010, 39, 4370–4387. [Google Scholar] [CrossRef]
- Knyazev, A.V.; Bulanov, E.N.; Korshunov, A.O.; Krasheninnikova, O.V. Synthesis and thermal expansion of some lanthanide-containing apatites. Inorg. Mater. 2013, 49, 1133–1137. [Google Scholar] [CrossRef]
- Bulanov, E.N.; Knyazev, A.V. High-temperature in situ XRD investigations in apatites. Structural interpretation of thermal deformations. In Apatite: Synthesis, Structural Characterization and Biomedical Applications; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2014; Chapter 7; pp. 173–200. [Google Scholar]
- Bulanov, E.N.; Wang, J.; Knyazev, A.V.; White, T.; Manyakina, M.; Baikie, T.; Lapshin, A.N.; Dong, Z. Structure and thermal expansion of calcium-thorium apatite, [Ca4]F[Ca2Th4]T [(SiO4)6]O2. Inorg. Chem. 2015, 54, 11356–11361. [Google Scholar] [CrossRef]
- Knyazev, A.V.; Bulanov, E.N.; Smirnova, N.N.; Korokin, V.Z.; Shushunov, A.N.; Blokhina, A.G.; Xu, Z. Thermodynamic and thermophysics properties of synthetic britholite SrPr4(SiO4)3O. J. Chem. Thermodyn. 2017, 108, 38–44. [Google Scholar] [CrossRef]
- Cronstedt, A.F. Observation and Description of an Unknown Kind of Rock to be Named Zeolites. Kong Vetenskaps Acad. Handl. Stockh. 1756, 12, 120–123. (In Swedish) [Google Scholar]
- Smith, J.V. Structural Classification of Zeolites. Mineral. Soc. Am. Spec. Pap. 1963, 1, 281. [Google Scholar]
- Breck, D.A. Zeolite Molecular Sieves: Structure, Chemistry and Use; Wiley-Interscience: New York, NY, USA, 1974. [Google Scholar]
- Smith, J.V. Origin and Structure of Zeolites. In Zeolite Chemistry and Catalysis; A.C.S. Series; Rabo, J.A., Ed.; American Chemical Society: Washington, DC, USA, 1976; Volume 171, pp. 1–79. [Google Scholar]
- Barrer, R.M. Hydrothermal Chemistry of Zeolites; Academic Press: New York, NY, USA, 1982. [Google Scholar]
- Harjula, R.; Lehto, J. Effect of sodium and potassium ions on cesium absorption from nuclear power plant waste solutions on synthetic zeolites. Nucl. Chem. Waste Manag. 1986, 6, 133–137. [Google Scholar] [CrossRef]
- Higgins, F.M.; de Leeuw, N.H.; Parker, S.C. Modelling the Effect of Water on Cation Exchange in Zeolite A. J. Mater. Chem. 2002, 12, 124–131. [Google Scholar]
- Lima, E.; Ibarra, A.; Bosch, P.; Bulbulian, S. Vitrification of CsA and CsX zeolites. Stud. Surf. Sci. Catal. 2004, 154, 1907–1911. [Google Scholar]
- Tsukada, T.; Takahashi, K. Absorption Characteristics of Fission Product Elements on Zeolite. Nucl. Technol. 2008, 162, 229–243. [Google Scholar] [CrossRef]
- Kaminski, M.D.; Mertz, C.J.; Ferrandon, M.; Dietz, N.L.; Sandi, G. Physical properties of an alumino-silicate waste form for cesium and strontium. J. Nucl. Mater. 2009, 392, 510–518. [Google Scholar] [CrossRef]
- Cappelletti, P.; Rapisardo, G.; De Gennaro, B.; Colella, A.; Langella, A.; Fabio, S.; Lee, D.; De Gennaro, M. Immobilization of Cs and Sr in aluminosilicate matrices derived from natural zeolites. J. Nucl. Mater. 2011, 414, 451–457. [Google Scholar] [CrossRef]
- Gatta, G.D.; Merlini, M.; Lotti, P.; Lausi, A.; Rieder, M. Microporous and Mesoporous Materials Phase stability and thermo-elastic behavior of CsAlSiO4 (ABW): A potential nuclear waste disposal material. Microporous Mesoporous Mater. 2012, 163, 147–152. [Google Scholar] [CrossRef]
- Yamagishi, I.; Nagaishi, R.; Kato, C.; Morita, K.; Terada, A.; Kamiji, Y. Characterization and storage of radioactive zeolite waste. J. Nucl. Sci. Technol. 2014, 51, 1044–1053. [Google Scholar] [CrossRef]
- Gallis, F.S.; Ermanoski, I.; Greathouse, J.A.; Chapman, K.W.; Nenoff, T.M. Iodine Gas Adsorption in Nanoporous Materials: A Combined Experiment–Modeling Study. Ind. Eng. Chem. Res. 2017, 56, 2331–2338. [Google Scholar] [CrossRef]
- Lee, H.Y.; Kim, H.S.; Jeong, H.-K.; Park, M.; Chung, D.-Y.; Lee, E.-H.; Lim, W.T. Selective Removal of Radioactive Cesium from Nuclear Waste by Zeolites: On the Origin of Cesium Selectivity Revealed by Systematic Crystallographic Studies. J. Phys. Chem. C 2017, 121, 10594–10608. [Google Scholar] [CrossRef]
- Papynov, E.K. Spark plasma sintering of ceramic and glass-ceramic matrices for cesium radionuclides immobilization. In Glas-Ceramics: Properties, ApplIcations and Technolology; Narag, K., Ed.; Nova Science Publisher, Inc.: New York, NY, USA, 2018; pp. 109–153. [Google Scholar]
- Papynov, E.K.; Shichalin, O.O.; Mayorov, V.Y.; Kuryavyi, V.G.; Kaidalova, T.A.; Teplukhina, L.V.; Portnyagin, A.S.; Slobodyuk, A.B.; Belov, A.A.; Tananaev, I.G.; et al. SPS technique for ionizing radiation source fabrication based on dense cesium-containing core. J. Hazard. Mater. 2019, 369, 25–30. [Google Scholar] [CrossRef]
- Strachan, D.M.; Schulz, W.W. Characterization of Pollucite as a Material for Long-Term Storage of Cesium-137. Am. Ceram. Soc. Bull. 1979, 58, 865–871. [Google Scholar]
- Gallagher, S.A.; McCarthy, G.J. Preparation and X-ray Characterization of Pollucite (CsAlSi2O6). Inorg. Nucl. Chem. 1981, 43, 1773–1777. [Google Scholar] [CrossRef]
- Komameni, S.; White, W.B. Stability of Pollucite in Hydrothermal Fluids. Sci. Basis Nucl. Waste Manag. 1981, 3, 387–396. [Google Scholar]
- Yanagisawa, K.; Nishioka, M.; Yamasaki, N. Immobilization of Cesium into Pollucite Structure by Hydrothermal Hot-Pressing. J. Nucl. Sci. Technol. 1987, 24, 51–60. [Google Scholar] [CrossRef]
- Mielearski, M. Preparation of 137Cs Pollucite Source Core. Isotopenpraxis 1989, 25, 404–408. [Google Scholar] [CrossRef]
- Mimura, H.; Shibata, M.; Akiba, K. Surface Alteration of Pollucite under Hydrothermal Conditions. J. Nucl. Sci. Technol. 1990, 27, 835–843. [Google Scholar] [CrossRef][Green Version]
- Anthony, R.G.; Phillip, C.V.; Dosch, R.G. Selective Adsorption and Ion Exchange of Metal Cations and Anions with Silico-Titanates and Layered Titanates. Waste Manag. 1993, 13, 503–512. [Google Scholar] [CrossRef]
- Balmer, M.L.; Bunker, B.C. Inorganic Ion Exchange Evaluation and Design-Silicotitanate Ion Exchange Waste Conversion; PNL-10460; Pacific Northwest Laboratory: Richland, WA, USA, 1995.
- Su, Y.; Balmer, M.L.; Bunker, B.C. Evaluation of Cesium Silicotitanates as an Alternate Waste Form. Mater. Res. Soc. Symp. Proc. 1996, 465, 457–464. [Google Scholar] [CrossRef]
- McCready, D.E.; Balmer, M.L.; Keefer, K.D. Experimental and Calculated X-ray Diffraction Data for Cesium Titanium Silicate, CsTiSi2O6,5: A New Zeolite. Powder Diffr. 1997, 12, 40–46. [Google Scholar] [CrossRef]
- Su, Y.; Balmer, M.L.; Wang, L.; Bunker, B.C.; Nyman, M.; Nenoff, T.; Navrotsky, A. Evaluation of Thermally Converted Silicotitanate Waste Forms. Mater. Res. Soc. Symp. Proc. 1999, 556, 77–84. [Google Scholar] [CrossRef]
- Xu, H.; Navrotsky, A.; Nyman, M.D.; Nenoff, T.M. Thermochemistry of Microporous Silicotitanate Phases in the Na2O-Cs2O-SiO2-TiO2-H2O System. J. Mater. Res. 2000, 15, 815–823. [Google Scholar] [CrossRef]
- Xu, H.; Navrotsky, A.; Balmer, M.L.; Su, Y.; Bitten, E.R. Energetics of Substituted Pollucites along the CsAlSi2O6-CsTiSi2O6,5 Join: A High-Temperature Calorimetric Strudy. J. Am. Ceram. Soc. 2001, 84, 555–560. [Google Scholar] [CrossRef]
- Bubnova, R.S.; Stepanov, N.K.; Levin, A.A.; Filatov, S.K.; Paufler, P.; Meyer, D.C. Crystal structure and thermal behavior of boropollucite CsBSi2O6. Solid State Sci. 2004, 6, 629–637. [Google Scholar] [CrossRef]
- Richerson, D.W.; Hummel, F.A. Synthesis and Thermal Expansion of Polycrystalline Cesium Minerals. J. Am. Ceram. Soc. 2006, 55, 269–273. [Google Scholar] [CrossRef]
- Rehspringer, J.-L.; Balencie, J.; Vilminot, S.; Burger, D.; Boos, A.; Estournes, C. Confining caesium in expanded natural Perlite. J. Eur. Ceram. Soc. 2007, 27, 619–622. [Google Scholar] [CrossRef][Green Version]
- Garino, T.G.; Nenoff, T.M.; Park, T.J.; Navrotsky, A. The Crystallization of Ba-Substituted CsTiSi2O6.5 Pollucite using CsTiSi2O6.5 Seed Crystals. J. Am. Ceram. Soc. 2009, 92, 2144–2146. [Google Scholar] [CrossRef]
- Park, T.-J.; Garino, T.J.; Nenoff, T.M.; Rademacher, D.; Navrotsky, A. The Effect of Vacancy and Ba-Substitution on the Stability of the CsTiSi2O6.5 Pollucite. J. Am. Ceram. Soc. 2011, 94, 3053–3059. [Google Scholar] [CrossRef]
- He, P.; Jia, D. Low-temperature sintered pollucite ceramic from geopolymer precursor using synthetic metakaolin. J. Mater. Sci. 2013, 48, 1812–1818. [Google Scholar] [CrossRef]
- Garino, T.J.; Rademacher, D.X.; Rodriguez, M.; Nenoff, T.M. The Synthesis of Ba and Fe Substituted CsAlSi2O6 Pollucites. J. Am. Ceram. Soc. 2013, 96, 2966–2972. [Google Scholar]
- Jing, Z.; Hao, W.; He, X.; Fan, J.; Zhang, Y.; Miao, J.; Jin, F. A novel hydrothermal method to convert incineration ash into pollucite for the immobilization of a simulant radioactive cesium. J. Hazard. Mater. 2016, 306, 220–229. [Google Scholar] [CrossRef]
- Fan, J.; Jing, Z.; Zhang, Y.; Miao, J.; Chen, Y.; Jin, F. Mild hydrothermal synthesis of pollucite from soil for immobilization of Cs in situ and its characterization. Chem. Eng. J. 2016, 304, 344–350. [Google Scholar] [CrossRef]
- Vance, E.R.; Gregg, D.J.; Griffiths, G.J.; Gaugliardo, P.R.; Grant, C. Incorporation of Ba in Al and Fe pollucite. J. Nuc. Mater. 2016, 478, 256–260. [Google Scholar] [CrossRef]
- Henderson, C.M.B.; Charnock, J.M.; Bell, J.M.; van der Laan, G.C.M.B. X-ray absorption study of 3d transition-metals and Mg in glasses and analogue crystalline materials in AFe3+Si2O6 and A2X2+Si5O12, where A = K, Rb, or Cs and X = Mg, Mn, Fe, Co, Ni, Cu, or Zn. J. Non-Cryst. Solids 2016, 451, 23–48. [Google Scholar] [CrossRef]
- Omerasevic, M.; Matovic, L.; Ruzic, J.; Golubovic, Z.; Jovanovic, U.; Mentus, S.; Dondur, V. Safe trapping of cesium into pollucite structure by hot-pressing method. J. Nuc. Mater. 2016, 474, 35–44. [Google Scholar] [CrossRef]
- Hamada, S.; Kishimura, H.; Matsumoto, H.; Takahashi, K.; Aruga, A. Effect of shock compression on luminescence properties of CsAlSi2O6:Eu2+ for white-light-emitting diodes. Opt. Mater. 2016, 62, 192–198. [Google Scholar] [CrossRef]
- Orlova, A.I.; Chuvildeev, V.N.; Mikhailov, D.A.; Boldin, M.S.; Belkin, O.A.; Nokhrin, A.V.; Sakharov, N.V.; Skuratov, V.A.; Kirilkin, S. Preparation of ceramic materials with mineral-like structures by mean of SPS technology for the purpose of radwaste consolidation and radiation and hydrolytic investigations. J. Nucl. Energy Sci. Power Gener. Technol. 2017, 6, 33. [Google Scholar]
- Berry, L.G.; Mason, B. Mineralogy Concepts, Descriptions, Determinations; W.H. Freeman & Co.: San Francisco, CA, USA, 1959. [Google Scholar]
- Deer, W.A.; Howie, R.A.; Zussman, J. Rock-Forming Minerals, Vol IV; John Wiley & Sons, Inc.: New York, NY, USA, 1963. [Google Scholar]
- Klingenberg, R.; Felsche, J. Interstitial Cristobalite-type Compounds (Na2O)≤0.33Na[AlSiO4]). J. Solid State Chem. 1986, 61, 40–46. [Google Scholar] [CrossRef]
- Kim, J.G.; Lee, J.H.; Kim, I.T.; Kim, E.H. Fabrication of a Glass-Bonded Zeolite Waste Form for Waste LiCl Salt. J. Ind. Eng. Chem. 2007, 13, 292–298. [Google Scholar]
- Brookins, D.G. Geochemical Aspects of Radioactive Waste Disposal; Springer: Berlin, Germany, 1984. [Google Scholar]
- Fleet, M.E. Structures of Sodium Alumino-Germanate Sodalites. Acta Cryst. 1989, C45, 843–847. [Google Scholar]
- McFarlane, H.F.; Goff, K.M.; Felicione, F.S.; Dwight, C.C.; Barber, D.B. Hot Demonstrations of Nuclear-Waste Processing Technologies. JOM 1997, 49, 14–21. [Google Scholar] [CrossRef]
- Deer, W.A.; Howie, R.A.; Zussman, J. Rock-Forming Minerals. Vol. 2A. Single-Chain Silicates; Geological Society: London, UK, 1997. [Google Scholar]
- Nakazawa, T.; Kato, H.; Okada, K.; Ueta, S.; Mihara, M. Iodine Immobilization by Sodalite Waste Form. Mater. Res. Soc. Symp. Proc. 2001, 663, 51–57. [Google Scholar] [CrossRef]
- Olson, A.L.; Soelberg, N.R.; Marshal, D.W.; Anderson, G.L. Fluidized Bed Steam Reforming of INEEL SBW Using THOR Mineralizing Technology; INEEL/EXT-04-02564; Idaho National Laboratory: Idaho Falls, ID, USA, 2004. [Google Scholar]
- Deer, W.A.; Howie, R.A.; Wise, W.S.; Zussman, J. Rock-Forming Minerals, Vol. 4B, Framework Silicates: Silica Minerals, Feldspathoids and the Zeolites; The Geological Society: London, UK, 2004. [Google Scholar]
- Mattigod, S.V.; McGrail, B.P.; McCready, D.E.; Wang, L.; Parker, K.E.; Young, J.S. Synthesis and Structure of Perrhenate Sodalite. J. Microporous Mesopourous Mater. 2006, 91, 139–144. [Google Scholar] [CrossRef]
- Angelis, G.D.; Capone, M.; Mannielo, A.; Mariani, M.; Maceratu, E.; Conti, C. Different methods for conditioning Chlorite Salt waste from Pyroprocesses. In Book of Abstracts “III International Pyroprocessing Research Conference; NIIAR: Dimitrovgrad, Russia, 2010; p. 48. [Google Scholar]
- Simpson, M.F.; Allensworth, J.R.; Phongikaroon, S.; Williams, A.N.; Dunzik-Gourgar, M.L.; Ferguson, C. Immobilization of Salt from Zone Freezing Process in Zeolite-A. In III International Pyroprocessing Research Conference; NIIAR: Dimitrovgrad, Russia, 2010; pp. 30–31. [Google Scholar]
- Akimkhan, A.M. Structural and Ion-Exchange Properties of Natural Zeolite. Ion Exch. Technol. 2012, 10, 261–283. [Google Scholar]
- Tendeloo, L.V.; Blochouse, B.; Dom, D.; Vancluysen, J.; Snellings, R.; Martens, J.A.; Kirschhock, C.E.A.; Maes, A.; Breynaert, E. Cation Exchange Properties of Zeolites in Hyper Alkaline Aqueous Media. Environ. Sci. Technol. 2015, 49, 1729–1737. [Google Scholar] [CrossRef]
- Olszewska, W.; Miśkiewicz, A.; Zakrzewska-Kołtuniewicz, G.; Lankof, L.; Pająk, L. Multibarrier system preventing migration of radionuclides from radioactive waste repository. Nukleonika 2015, 60, 557–563. [Google Scholar] [CrossRef]
- Kim, H.S.; Park, J.S.; Lim, W.T. Site Competition of Ca2+ and Cs+ Ions in the Framework of Zeolite Y (Si/Al = 1.56) and Their Crystallographic Studies. J. Mineral. Soc. Korea 2018, 31, 235–248. [Google Scholar] [CrossRef]
- Ovhal1, S.; Butler, I.S.; Xu, S. The Potential of Zeolites to Block the Uptake of Radioactive Strontium-90 in Organisms. Contemp. Chem. 2018, 1, 1–13. [Google Scholar]
- Dyer, A.; Hriljac, J.; Evans, N.; Stokes, I.; Rand, P.; Kellet, S.; Harjula, R.; Moller, T.; Maher, Z.; Heatlie-Branson, R.; et al. The use of columns of the zeolite clinoptilolite in the remediation of aqueous nuclear waste streams. J. Radioanal. Nucl. Chem. 2018, 318, 2473–2491. [Google Scholar] [CrossRef]
- Grundy, H.D.; Hassan, I. The crystal structure of a carbonate-rich cancrinite. Can. Mineral. 1982, 20, 239–251. [Google Scholar]
- Zhao, H.; Deno, Y.; Harsh, J.B.; Flury, M.; Boyle, J.S. Alteration of Kaolinite to Cancrinite and Sodalite by simulated Hanford tank waste and its inpact on cesium retention. Clay Mater. 2004, 52, 1–13. [Google Scholar]
- Mon, J.; Deng, Y.; Flury, M.; Harsh, J.B. Cesium incorporation and diffusion in cancrinite, sodalite, zeolite, and allophone. Microporous Microporous Mater. 2005, 86, 277–286. [Google Scholar] [CrossRef]
- Hassan, I.; Antao, S.M.; Parise, J.B. Cancrinite: Crystal structure, phase transitions, and dehydration behavior with temperature. Am. Mineral. 2006, 91, 1117–1124. [Google Scholar] [CrossRef]
- Deng, Y.; Flury, M.; Harsh, J.B.; Felmy, A.R.; Qafoku, O. Cancrinite and sodalite formation in the presence of cesium, potassium, magnesium, calcium and strontium in Hanford tank waste stimulant. Appl. Geochem. 2006, 21, 2049–2063. [Google Scholar] [CrossRef]
- Dickson, J.O.; Harsh, J.B.; Flury, M.; Lukens, W.W.; Pierce, E.M. Immobilization and Exchange of Perrhenate in Sodalite and Cancrinite. Microporous Mesoporous Mater. 2015, 214, 115–120. [Google Scholar] [CrossRef][Green Version]
- Zheng, Z.; Anthony, R.G.; Miller, J.E. Modeling Multicomponent Ion Exchange Equilibrium Utilizing Hydrous Crystalline Silicotitanates by a Multiple Interactive Ion Exchange Site Model. Ind. Eng. Chem. Res. 1997, 36, 2427–2434. [Google Scholar] [CrossRef]
- Miller, J.E.; Brown, N.E. Development and Properties of Crystalline Silicotitanate (CST) Ion Exchangers for Radioactive Waste Applications; SAND97-0771; Sandia National Laboratories: Albuquerque, NM, USA, 1997.
- Andrews, M.K.; Harbour, J. Glass Formulation Requirements for Hanford Coupled Operations Using Crystalline Silicotitanates (CST); WSRC-RP-97-0265; Westinghouse Savannah River Company, Savannah River Site: Aike, SC, USA, 1997. [Google Scholar]
- Yu, B.; Chen, J.; Song, C. Crystalline Silicotitanate: A New Type of Ion Exchange for Cs Removal from Liquid Waste. J. Mater. Sci. Technol. 2002, 18, 206–210. [Google Scholar]
- Tripathi, A.; Medvedev, D.G.; Nyman, M.; Clearfield, A. Selectivity for Cs and Sr in Nb-Substituted Titanosilicate with Sitinakite Topology. J. Solid State Chem. 2003, 175, 72–83. [Google Scholar] [CrossRef]
- Keppler, H. Ion Exchange Reactions Between Dehydroxylated Micas and Salt Melts and the Crystal Chemistry of the Interlayer Cation in Micas. Am. Minerial. 1990, 75, 529–538. [Google Scholar]
- Fleet, M.E. Rock-Forming Minerals: Sheet Silicates: Micas, V. 3A; The Geological Society: Bath, Englad, 2003. [Google Scholar]
- Jantzen, C.M.; Williams, M.R.; Bibler, N.E.; Crawford, C.L.; Jurgensen, A.R. Fluidized Bed Steam Reformed (FBSR) Mineral Waste Forms: Application to Cs-137/Sr-90 Wastes for the Global Nuclear Energy Partnership (GNEP); U.S. DOE Report WSRC-MS; Savannah River National Laboratory: Jackson, MS, USA, 2008.
- Jantzen, C.M.; Williams, M.R. Fluidized Bed Steam Reforming (FBSR) Mineralization for High Organic and Nitrate Waste Streams for the Global Nuclear Energy Partnership (GNEP); Waste Management 08, Paper #8314 (2008); WM Symposia: Phoenix, AZ, USA, 2008; p. 8314. [Google Scholar]
- Neeway, J.J.; Qafoku, N.P.; Peterson, R.A.; Brown, C.F. Characterization and Leaching Tests of the Fluidized Bed Steam Reforming (FBSR) Waste Form for LAW Immobilizatio; Waste Management 2013, Paper 13400; WM Symposia: Phoenix, AZ, USA, 2013. [Google Scholar]
- Kumar, A.; Singh, Y.P.; Pradhan, G.; Dhawan, N. Utilization of Mica for Potassium Recovery. In Proceedings. Mater. Today 2018, 5, 17030–17034. [Google Scholar]
- Boatner, L.A.; Sales, B.C. Monazite. In Radiation Waste Forms for the Future; Lutze, W., Ewing, R.C., Eds.; North-Holland Press: Amsterdam, The Netherlands, 1988; pp. 495–564. [Google Scholar]
- Ewing, R.C.; Weber, W.J.; Lutze, W. Crystalline Ceramics: Waste Forms for the Disposal of Weapons Plutonium. In Disposal of Ex-weapons Plutonium as Waste; NATIO ASI Series; Merz, E.R., Walter, C.E., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1996; pp. 65–83. [Google Scholar]
- Merz, E.R.; Walter, C.E. Disposal of Ex-weapons Plutonium as Waste, NATIO ASI Series; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1996; pp. 65–83. [Google Scholar]
- Van Emden, B.; Thornber, M.R.; Graham, J.; Lincoln, F.J. The incorporation of actinides in monazite and xenotime from placer deposits in Western Australia. Can. Mineral. 1997, 35, 95–104. [Google Scholar]
- Chang, L.L.Y.; Howie, R.A.; Zussman, J. Rock-Forming Minerals, V.5B Non-Silicates. In Sulphates, Carbonates, Phosphates, Halides, 2nd ed.; The Geological Society: London, UK, 1998; Volume 383, ICBN 978-1897799901. [Google Scholar]
- Genet, M.; Dacheux, N.; Thomas, A.C.; Chassigneux, B.; Pichot, E.; Brandel, V. Thorium phosphate-diphosphate as a ceramic for the immobilization of tetravalent uranium, neptunium and plutonium. Waste Manag. 1999, 99, 38. [Google Scholar]
- Clavier, N.; Dacheux, N.; Podor, R.; Le Coustumer, P. Study of Actinides Incorporation in Thorium Phosphate-Diphosphate/Monazite Based Ceramics. Mater. Res. Soc. Symp. Proc. 2004, 802, DD3.6.1–DD3.6.6. [Google Scholar] [CrossRef]
- Burakov, B.E.; Yagovkina, M.A.; Garbuzov, V.M.; Kitsay, A.A.; Zirlin, V.A. SelfIrradiation of Monazite Ceramics: Contrasting Behavior of PuPO4 and (La,Pu)PO4 Doped with Pu-238. In Scientific Basis for Nuclear Waste Management XXVIII; Hanchar, J.M., Stroes-Gascoyne, S., Browning, L., Eds.; Materials Research Society Symposium Proceedings: San Francisco, CA, USA, 2004; Volume 824, pp. 219–224. [Google Scholar]
- Montel, J.M.; Glorieux, B.; Seydoux-Guilaume, A.M.; Wirth, R. Synthesis and Sintering of a Monazite-brabantite Solid Solution Ceramic for Nuclear Waste Storage. J. Phys. Chem. Solids 2006, 67, 2489–2500. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Vance, E.R. Plutonium in Monazite and Brabanite: Diffuse Reflectance Spectroscopy Study. J. Nucl. Mater. 2008, 375, 311–314. [Google Scholar] [CrossRef]
- Glorieux, B.; Montel, J.M.; Matecki, M. Synthesis and Sintering of a Monazite-brabantite Solid Solution Ceramics Using Metaphosphate. J. Eur. Ceram. Soc. 2009, 29, 1679–1686. [Google Scholar] [CrossRef]
- Brandt, F.; Neumeier, S.; Schuppik, T.; Arinicheva, Y.; Bukaemskiy, A.; Modolo, G.; Bosbach, D. Conditioning of minor actinides in lanthanum monazite ceramics: A surrogate with Europium. Prog. Nucl. Energy 2014, 72, 140–143. [Google Scholar] [CrossRef]
- Arinicheva, Y.; Bukaemskiy, A.; Neumeier, S.; Modolo, G.; Bosbach, D. Studies on thermal and mechanical properties of monazite-type ceramics for the conditioning of minor actinides. Prog. Nucl. Energy 2014, 72, 144–148. [Google Scholar] [CrossRef]
- Mezentseva, L.P.; Kruchinina, I.Y.; Osipov, A.V.; Kuchaeva, S.K.; Ugolkov, V.L.; Popova, V.F.; Pugachev, K.E. Nanopowders of Orthophosphate LaPO4–YPO4–H2O System and Ceramics Based on Them. Glass Phys. Chem. 2014, 40, 356–361. [Google Scholar] [CrossRef]
- Ma, J.; Teng, Y.; Huang, Y.; Wu, L.; Zhang, K.; Zhao, X. Effects of sintering process, pH and temperature on chemical durability of Ce0.5Pr0.5PO4 ceramics. J. Nucl. Mater. 2015, 465, 550–555. [Google Scholar] [CrossRef]
- Zhao, X.; Teng, Y.; Yang, H.; Huang, Y.; Ma, J. Comparison of microstructure and chemical durability of Ce0.9Gd0.1PO4 ceramics prepared by hot-press and pressureless sintering. Ceram. Int. 2015, 41, 11062–11068. [Google Scholar] [CrossRef]
- Teng, Y.; Zeng, P.; Huang, Y.; Wu, L.; Wang, X. Hot-pressing of monazite Ce0.5Pr0.5PO4 ceramic and its chemical durability. J. Nucl. Mater. 2015, 465, 482–487. [Google Scholar] [CrossRef]
- Zhao, X.; Teng, Y.; Wu, L.; Huang, Y.; Ma, J.; Wang, G. Chemical durability and leaching mechanism of Ce0.5Eu0.5PO4 ceramics: Effects of temperature and pH values. J. Nucl. Mater. 2015, 466, 187–193. [Google Scholar] [CrossRef]
- Teng, Y.; Wang, X.; Huang, Y.; Wu, L.; Zeng, P. Hot-pressure sintering, microstructure and chemical durability of Ce0.5Eu0.5PO4 monazite ceramics. Ceram. Int. 2015, 41, 10057–10062. [Google Scholar] [CrossRef]
- Meng, C.; Ding, X.; Zhao, J.; Ren, C.; Fu, H.; Yang, H. Phase evolution and microstructural studies of Gd1−xYbxPO4 (0≤x≤1) ceramics for radioactive waste storage. J. Eur. Ceram. Soc. 2016, 36, 773–779. [Google Scholar] [CrossRef]
- Potanina, E.; Golovkina, L.; Orlova, A.; Nokhrin, A.; Boldin, M.; Sakharov, N. Lanthanide (Nd, Gd) compounds with garnet and monazite structures. Powders synthesis by “wet” chemistry to sintering ceramics by Spark Plasma Sintering. J. Nucl. Mater. 2016, 473, 93–98. [Google Scholar] [CrossRef]
- Ji, Y.; Kowalski, P.M.; Neumeier, S.; Deissmann, G.; Kulriya, P.K.; Gale, J.D. Atomistic modeling and experimental studies of radiation damage in monazite-type LaPO4 ceramics. Nucl. Instrum. Methods Phys. Res. 2017, 393, 54–58. [Google Scholar] [CrossRef]
- Babelot, C.; Bukaemskiy, A.; Neumeier, S.; Modolo, G.; Bosbach, D. Crystallization processes, compressibility, sinterability and mechanical properties of La-monazite-type ceramics. J. Eur. Ceram. Soc. 2017, 37, 1681–1688. [Google Scholar] [CrossRef]
- Guo, L.; Yan, Z.; Li, Z.; Yu, J.; Wang, Q.; Li, M.; Ye, F. GdPO4 as a novel candidate for thermal barrier coating applications at elevated temperatures. Surf. Coat. Technol. 2018, 349, 400–406. [Google Scholar] [CrossRef]
- Arinicheva, Y.; Gausse, C.; Neumeier, S.; Brandt, F.; Rozov, K.; Szenknect, S.; Dacheux, N.; Bosbach, D.; Deissmann, G. Influence of temperature on the dissolution kinetics of synthetic LaPO4-monazite in acidic media between 50 and 130 °C. J. Nucl. Mater. 2018, 509, 488–495. [Google Scholar] [CrossRef]
- Arinicheva, Y.; Clavier, N.; Neumeier, S.; Podor, R.; Bukaemskiy, A.; Klinkenberg, M.; Roth, G.; Dacheux, N.; Bosbach, D. Effect of powder morphology on sintering kinetics, microstructure and mechanical properties of monazite ceramics. J. Eur. Ceram. Soc. 2018, 38, 227–234. [Google Scholar] [CrossRef]
- Zhao, X.; Li, Y.; Teng, Y.; Wu, L.; Bi, P.; Wang, L.; Wang, S. The structure, sintering process, and chemical durability of Ce0.5Gd0.5PO4 ceramics. Ceram. Int. 2018, 44, 19718–19724. [Google Scholar] [CrossRef]
- Zhao, X.; Li, Y.; Teng, Y.; Wu, L.; Bi, P.; Yang, X.; Wan, L. The effect of Ce content on structure and stability of Gd1-xCexPO4: Theory and experiment. J. Eur. Ceram. Soc. 2019, 39, 1555–1563. [Google Scholar] [CrossRef]
- Milligan, W.O.; Mullica, D.F.; Beall, G.W.; Boatner, L.A. Structural investigation of YPO4, ScPO4, and LuPO4. Inorg. Chim. Acta 1982, 60, 39–43. [Google Scholar] [CrossRef]
- Boatner, L. Synthesis, Structure, and Properties of Monazite, Pretulite, and Xenotime. Rev. Mineral. Geochem. 2002, 48, 87–121. [Google Scholar] [CrossRef]
- Hetherington, C.J.; Hasrlov, D.E.; Budzyn, B. Experimental metasomatism of monazite and xenotime: Mineral stability, REE mobility and fluid composition. Miner. Petrol. 2010, 99, 165–184. [Google Scholar] [CrossRef]
- Ji, Y.; Beridze, G.; Bosbach, D.; Kowalski, P.M. Heat capacities of xenotime-type ceramics: An accurate ab initio prediction. J. Nucl. Mater. 2017, 494, 172–181. [Google Scholar] [CrossRef]
- Kondrat’eva, O.N.; Nikiforova, G.E.; Tyurin, A.V.; Ryumin, M.A.; Gurevich, V.M.; Kritskaya, A.P.; Gavrichev, K.S. Calorimetric study of ytterbium orthovanadate YbVO4 polycrystalline ceramics. Ceram. Int. 2018, 44, 18103–18107. [Google Scholar] [CrossRef]
- Weber, W.J.; Turcotte, R.P.; Bunnell, L.R.; Roberts, F.P.; Westsik, J.H., Jr. Radiation Effects in Vitreous and Devitrified Simulated Waste Glass (Contains Apatite). In Proceedings of the International Symposium on Ceramics in Nuclear Waste Management, Cincinnati, OH, USA, 28 April–3 May 1979; pp. 294–299. [Google Scholar]
- Weber, W.J. Radiation Damage in Rare-earth Silicate with the Apatite Structure. J. Am. Ceram. Soc. 1982, 65, 544–548. [Google Scholar] [CrossRef]
- Bros, R.; Carpens, J.; Sere, V.; Beltritti, A. Occurrence of Plutonium and Fissiogenic REE in Hydrothermal Apatites from the Nuclear Reactor 16 at Oklo (Gabon). Radiochim. Acta 1996, 74, 277–282. [Google Scholar] [CrossRef]
- Weber, W.J.; Ewing, R.C.; Meldrum, A. The Kinetics of Alpha-decay-induced Amorphization in Zircon and Apatite Containing Weapons-grade Plutonium or Other Actinides. J. Nucl. Mater. 1997, 250, 147–155. [Google Scholar] [CrossRef]
- Audubert, F.J.; Lacout, J.L.; Tetard, F. Elaboration of an Iodine-Bearing Apatite Iodine Diffusion into a Pb3(VO4)2 Matrix. Solid State Ion. 1997, 95, 113–119. [Google Scholar] [CrossRef]
- Boyer, L.; Carpena, J.; Lacout, J.L. Synthesis of Phosphate-Silicate Apatites at Atmospheric Pressure. Solid State Ion. 1997, 95, 121–129. [Google Scholar] [CrossRef]
- Carpena, J.; Donazzon, B.; Ceraulo, E.; Prene, S. Composite Apatitic Cement as a Material to Retain Cesium and Iodine. Comptes Rendus de L Academie Des Sciences Serie II Fascicule C—Chimie 2001, 4, 301–308. [Google Scholar]
- Park, H.S.; Kim, I.T.; Kim, H.Y.; Lee, K.S.; Ryu, S.K.; Kim, J.H. Application of Apatite Waste Form for the Treatment of Water-soluble Wastes Containing Radioactive Elements. Part 1: Investigation on the Possibility. J. Ind. Eng. Chem. 2002, 8, 318–327. [Google Scholar]
- Elliott, J.C.; Wilson, R.M.; Dowker, S.E.P. Apatite Structures. Adv. X-ray Anal. 2002, 45, 172–181. [Google Scholar]
- Kim, J.Y.; Dong, Z.L.; White, T.J. Model Apatite Systems for the Stabilization of Toxic Metals: II, Ccation and Metalloid Substitutions in Chlorapatites. J. Am. Ceram. Soc. 2005, 88, 1253–1260. [Google Scholar] [CrossRef]
- Carpena, J.; Lacout, J.L. Calcium Phosphate Nuclear Materials: Apatitic Ceramics for Separated Wastes. Actual. Chim. 2005, 66–71. [Google Scholar]
- Jothinathan, E.; Vammeesel, K.; Vleugels, J. Apatite type lanthanum silicate and composite anode half cells. Solid State Ion. 2011, 192, 419–423. [Google Scholar] [CrossRef]
- Knyazev, A.V.; Chernorukov, N.G.; Bulanov, E.N. Apatite-structured compounds: Synthesis and high-temperature investigation. Mater. Chem. Phys. 2012, 132, 773–781. [Google Scholar] [CrossRef]
- Wang, J. Incorporation of iodine into apatite structure: a crystal chemistry approach using Artificial Neural Network. Front. Earth Sci. 2015, 3, 1–11. [Google Scholar] [CrossRef]
- Kirkland, C.L.; Yakymchuk, C.; Szilas, K.; Evans, N.; Hollis, J.; McDonald, B.; Gardiner, N.J. Apatite: A U-Pb thermochronometer or geochronomete. Lithos 2018, 318–319, 143–157. [Google Scholar] [CrossRef]
- Hong, H.Y.-P. Crystal structures and crystal chemistry in the system Na1+xZr2SixP3-xO12. Mater. Res. Bull. 1976, 11, 173–182. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Hong, H.Y.-P.; Kafalas, J.A. Fast Na+-ion transport in skeleton structures. Mater. Res. Bull. 1976, 11, 203–220. [Google Scholar] [CrossRef]
- Boilot, J.P.; Salanié, J.P.; Desplanches, G.; Le Potier, D. Phase transformation in Na1+xSixZr2P3−xO12 compounds. Mater. Res. Bull. 1979, 14, 1469–1477. [Google Scholar] [CrossRef]
- de la Rochère, M.; d’Yvoire, F.; Collin, G.; Comès, R.; Boilot, J.P. NASICON type materials—Na3M2(PO4)3 (M = Sc, Cr, Fe): Na+-Na+ correlations and phase transitions. Solid State Ion. 1983, 9–10 Pt 2, 825–828. [Google Scholar] [CrossRef]
- Manthiram, A.; Goodenough, J.B. Lithium Insertion into Fe2(MO4)3 Frameworks: Comparison of M = W with M = Mo. J. Solid State Chem. 1987, 71, 349–360. [Google Scholar] [CrossRef]
- Roy, R.; Vance, E.R.; Alamo, J. [NZP], a new radiophase for ceramic nuclear waste forms. Mater. Res. Bull. 1982, 17, 585–589. [Google Scholar] [CrossRef]
- Alamo, R.; Roy, R. Crystal chemistry of the NaZr2(PO4)3, NZP or CTP, structure family. J. Mater. Sci. 1986, 21, 444–450. [Google Scholar] [CrossRef]
- Scheetz, B.E.; Roy, R. Novel Waste Forms. In Radioactive Waste Forms for the Future; Lutze, W., Ewing, R.C., Eds.; North-Holland: Amsterdam, The Netherlands, 1988; pp. 596–599. [Google Scholar]
- Orlova, A.I.; Zyryanov, V.N.; Kotel’nikov, A.R.; Demarin, V.T.; Rakitina, E.V. Ceramic phosphate matrices for high level waste. Behaviour in hydrothermal conditions. Radiokhimiya 1993, 35, 120–126. [Google Scholar]
- Orlova, A.I.; Volkov, Y.F.; Melkaya, R.F.; Masterova, L.Y.; Kulikov, I.A.; Alferov, V.A. Synthesis and Radiation Stability of NZP Phosphates Containg F-elements. Radiochemistry 1994, 36, 322–325. [Google Scholar]
- Scheetz, B.E.; Agrawal, D.K.; Breval, E.; Roy, R. Sodium Zirconium-phosphate (NZP) as a Host Structure for Nuclear Waste Immobilization—A Review. Waste Manag. 1994, 14, 489–505. [Google Scholar] [CrossRef]
- Hawkins, H.T.; Scheetz, B.E.; Guthrie, G.D., Jr. Preparation of Monophasic [NZP] Radiophases: Potential Host Matrices for the Immobilization of Reprocessed Commercial High-Level Wastes. In Scientific Basis for Nuclear Waste Management. XX; Gray, W.J., Triay, I.R., Eds.; Material Research Society: Pittsburgh, PA, USA, 1997; pp. 387–394. [Google Scholar]
- Zyryanov, V.N.; Vance, E.R. Comparison of Sodium Zirconium Phosphate-Structured HLW forms and Synroc for High-Level Nuclear Waste Immobilization. In Scientific Basis for Nuclear Waste Management. XX; Gray, W.J., Triay, I.R., Eds.; Material Research Society: Pittsburgh, PA, USA, 1997; pp. 409–416. [Google Scholar]
- Miyajima, Y.; Miyoshi, T.; Tamaki, J.; Matsuoka, M.; Yamamoto, Y.; Masquelier, C.; Tabuchi, M.; Saito, Y.; Kageyama, H. Solubility range and ionic conductivity of large trivalent ion doped Na1+xMxZr2-xP3O12 (M: In, Yb, Er, Y, Dy, Tb, Gd) solid electrolytes. Solid State Ion. 1999, 124, 201–211. [Google Scholar] [CrossRef]
- Orlova, A.I.; Charlamova, A.A.; Volkov, Y.F. Investigation of Plutonium, Americium and Curium Phosphates as a Basis for Inclusion into Kosnarite-type Ceramic Waste Worms. In Review of Excess Weapons Plutonium Disposition, LLNL Contract Work in Russia, Proceedings of the 3rd Annual Meeting for Coordination and Review of LLNL Contract Work, St. Petersburg, Russia, 14–18 January 2002; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2002; pp. 407–418. [Google Scholar]
- Orlova, A.I.; Orlova, V.A.; Buchirin, A.V.; Beskrovnyi, A.I.; Kurazhkovskaya, V.S. Cesium and Its Analogs, Rubidium and Potassium, in Rhombohedral [NaZr2(PO4)3 Type] and Cubic (Langbeinite Type) Phosphates: 1. Crystal-Chemical Studies. Radiochemistry 2005, 47, 225–234. [Google Scholar] [CrossRef]
- Bykov, D.M.; Orlova, A.I.; Tomilin, S.V.; Lizin, A.A.; Lukinykh, A.N. Americium and Plutonium on Trigonal Phosphates (NZP Type) AM1/3Zr2(PO4)3 and Pu1/4Zr2(PO4)3. Radiochemistry 2006, 48, 234–239. [Google Scholar] [CrossRef]
- Bykov, D.M.; Konings, R.J.M.; Orlova, A.I. High Temperature Investigations of the rare earth NZP phosphates R1/3Zr2(PO4)3 (R = La, Nd, Eu, Lu) by drop calorimetry. J. Alloy. Compd. 2007, 439, 376–379. [Google Scholar] [CrossRef]
- Nalk, A.H.; Deb, S.B.; Chalke, A.B.; Saxena, M.K.; Ramakumar, K.L.; Venugopal, V.; Dharwadkar, S.R. Microwave-assisted low temperature synthesis of sodium zirconium phosphate (NZP) and the leachability of some selected fission products incorporated in its structure—A case study of leachability of cesium. J. Chem. Sci. 2010, 122, 71–82. [Google Scholar]
- Orlova, A.I.; Lizin, A.A.; Tomilin, S.V.; Lukinykh, A.N.; Kanunov, A.E.; Chuvil’deev, V.N.; Boldin, M.S.; Sakharov, N.V.; Nokhrin, A.V. Actinide Phosphates with NaZr2(PO4)3 Structure. High-Speed Production of Dense Ceramics. In Book of Abstracts: The 49-th Conference on Hot Laboratories and Remote Handling “HOTLAB 2012”; CEA: Marcoule, France, 2012. [Google Scholar]
- Orlova, A.I.; Volgutov, V.Y.; Mikhailov, D.A.; Bykov, D.M.; Skuratov, V.A.; Chuvil’deev, V.N.; Nokhrin, A.V.; Boldin, M.S.; Sakharov, N.V. Phosphate Ca1/4Sr1/4Zr2(PO4)3 of the NaZr2(PO4)3 type: Synthesis of a dense ceramic material and its radiation testing. J. Nucl. Mater. 2014, 441, 232–239. [Google Scholar] [CrossRef]
- Bohre, A.; Shrivastava, O.P.; Awasthi, K. Crystal Chemistry of Immobilization of Tetravalent Ce and Se in Ceramic Matrix of Sodium Zirconium Phosphate. Phys. Chem. Res. 2014, 2, 21–29. [Google Scholar]
- Pet’kov, V.; Asabina, E.; Loshkarev, V.; Sukhanov, M. Systematic investigation of the strontium zirconium phosphate ceramic form for nuclear waste immobilization. J. Nucl. Mater. 2016, 471, 122–128. [Google Scholar] [CrossRef]
- Pet’kov, V.I.; Dmitrienko, A.S.; Sukhanov, M.V.; Koval’skii, A.M.; Borovikova, E.Y. Synthesis, phase formation, and thermal expansion of sulfate phosphates with the NaZr2(PO4)3 structure. Russ. J. Inorg. Chem. 2016, 61, 623–629. [Google Scholar] [CrossRef]
- Glukhova, I.O.; Asabina, E.A.; Pet’kov, V.I.; Borovikova, E.Y.; Koval’skii, A.M. Phase Formation, Structure, and Thermal Expansion of Phosphates M0.5(1+x)FexTi2–x(PO4)3 (M = Mn, Zn). Russ. J. Inorg. Chem. 2016, 61, 681–687. [Google Scholar] [CrossRef]
- Liu, T.; Wang, B.; Gu, X.; Wang, L.; Ling, M.; Liu, G.; Wang, D.; Zhang, S. All-climate sodium ion batteries based on the NASICON electrode materials. Nano Energy 2016, 30, 756–761. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Park, S.; Seo, I.; Kim, Y. Na ion-conducting ceramic as solid electrolyte for rechargeable seawater batteries. Electrochim. Acta 2016, 191, 1–7. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Z.J. Luminescence properties and energy transfer studies of color tunable Tb3+-doped RE1/3Zr2(PO4)3 (RE = Y, La, Gd and Lu). J. Alloys Compd. 2016, 685, 841–847. [Google Scholar] [CrossRef]
- Ribero, D.; Seymour, K.C.; Kriven, W.M.; White, M.A. Synthesis of NaTi2(PO4)3 by the inorganic–organic steric entrapment method and its thermal expansion behavior. J. Am. Ceram. Soc. 2016, 99, 3586–3593. [Google Scholar] [CrossRef]
- Ananthanarayanan, A.; Ambashta, R.D.; Sudarsan, V.; Ajithkumar, T.; Sen, D.; Mazumder, S.; Wattal, P.K. Structure and short time degradation studies of sodium zirconium phosphate ceramics loaded with simulated fast breeder (FBR) waste. J. Nucl. Mater. 2017, 487, 5–12. [Google Scholar] [CrossRef]
- Kanunov, A.; Glorieux, B.; Orlova, A.; Borovikova, E.; Zavedeeva, G. Synthesis, structure and luminescence properties of phosphates A1-3xEuxZr2(PO4)3 (A-alkali metal). Bull. Mater. Sci. 2017, 40, 7–16. [Google Scholar] [CrossRef]
- Orlova, A. Next Generation Ceramic Materials for Consolidation of radioactive alpha-wastes using the Innovative Technology Spark Plasma Sintering for their preparation. In Book of Abstracts: 3rd World Congress on Materials Science, Engineering, Oil, Gas and Petrochemistry; StatNano: Dunbai, UAE, 2018. [Google Scholar]
- Kanunov, A.E.; Orlova, A.I. Phosphors Based on Phosphates of NaZr2(PO4)3 and Langbeinite Structural Families. Rev. J. Chem. 2018, 8, 1–33. [Google Scholar] [CrossRef]
- Hallopeau, L.; Bregiroux, D.; Rousse, G.; Portehault, D.; Stevens, P.; Toussaint, G.; Laberty-Robert, C. Microwave-assisted reactive sintering and lithium ion conductivity of Li1.3Al0.3Ti1.7(PO4)3 solid electrolyte. J. Power Sources 2018, 378, 48–52. [Google Scholar] [CrossRef]
- Wang, H.; Okubo, K.; Inada, M.; Hasegawa, G.; Enomoto, N.; Hayashi, K. Low temperature-densified NASICON-based ceramics promoted by Na2O-Nb2O5-P2O5 glass additive and spark plasma sintering. Solid State Ion. 2018, 322, 54–60. [Google Scholar] [CrossRef]
- Savinkh, D.O.; Khainakov, S.A.; Orlova, A.I.; Garcia-Granda, S. New Phosphate-Sulfates with NZP Structure. Russ. J. Inorg. Chem. 2018, 63, 714–724. [Google Scholar] [CrossRef]
- Savinkh, D.O.; Khainakov, S.A.; Orlova, A.I.; Garcia-Granda, S. Preparation and Thermal Expansion of Calcium Iron Zirconium Phosphates with the NaZr2(PO4)3 Structure. Inorg. Mater. 2018, 54, 591–595. [Google Scholar] [CrossRef]
- Orlova, A.; Khainakov, S.; Alexandrov, A.; Garcia-Granda, S.; Savinykh, D. Crystallographic studies of NaZr2(PO4)3 phosphates at high temperatures. In Book of Abstracts: 31-st European Crystallographic Meeting “ECM31”; European Crystallographic Association: Oviedo, Spain, 2018. [Google Scholar]
- Savinkh, D.O.; Khainakov, S.A.; Boldin, M.S.; Orlova, A.I.; Aleksandrov, A.A.; Lantsev, E.A.; Sakharov, N.V.; Murashov, A.A.; Garcia-Granda, S.; Nokhrin, A.V.; et al. Preparation of NZP-Type Ca0.75+0.5xZr1.5Fe0.5(PO4)3-x(SiO4)x Powders and Ceramic, Thermal Expansion Behavior. Inorg. Mater. 2018, 54, 1267–1273. [Google Scholar] [CrossRef]
- Orlova, A.I.; Loginova, E.E.; Logacheva, A.A.; Demarin, V.T.; Shmidt, O.V.; Nikolaev, A.Y. A Crystal-Chemical Approach in the Development of Phosphate Materials as Environmentally Safe Chemical Forms of Utilization of Spent Cs-Containing Ferrocyanide Sorbents. Radiochemistry 2010, 52, 462–468. [Google Scholar] [CrossRef]
- Lizin, A.A.; Tomilin, S.V.; Gnevashov, O.E.; Lukinykh, A.N.; Orlova, A.I. Orthophosphates of Langbeinite Structure for Immobilization of Alkali Metal Cations of Salt Wastes from Pyrochemical Processes. Radiochemistry 2012, 54, 542–548. [Google Scholar] [CrossRef]
- Martynov, K.V.; Nekrasov, A.N.; Kotel’nikov, A.R.; Tananaev, I.G. Synthesis and study of the chemical stability and strength of zirconium phosphates with the structure of langbeinite with imitators of high-level radioactive waste (HLRW). Glass Phys. Chem. 2017, 43, 75–82. [Google Scholar] [CrossRef]
- Mold, P.; Bull, R.K.; Durrani, S.A. Constancy of 244Pu distribution in chondritic whitlockite. Nuclear Tracks 1981, 5, 27–31. [Google Scholar] [CrossRef]
- Nakamura, S.; Otsuka, R.; Aoki, H.; Akao, M.; Miura, N.; Yamamoto, T. Thermal expansion of hydroxyapatite-β-tricalcium phosphate ceramics. Thermochim. Acta 1990, 165, 57–72. [Google Scholar] [CrossRef]
- Belik, A.A.; Morozov, V.A.; Grechkin, S.V.; Khasanov, S.S.; Lazoryak, B.I. Crystal Structures of Double Vanadates, Ca9R(VO4)7. III. R = Nd, Sm, Gd, or Ce. Crystallogr. Rep. 2000, 45, 798–803. [Google Scholar]
- Orlova, A.I.; Orlova, M.P.; Solov’eva, E.N.; Loginova, E.E.; Demarin, V.T.; Kazantsev, G.N.; Samojlov, S.G.; Stefanovsky, S.V. Lanthanides in Phosphates with the structure of whitlockite mineral (analog of β-Ca3(PO4)2). Radiochemistry 2006, 48, 561–567. [Google Scholar] [CrossRef]
- Orlova, M.; Glorieux, B.; Orlova, A.; Montel, J.M.; Kazantsev, G.; Samoilov, S. Phosphates with structure of mineral whitlockite (beta-Ca3(PO4)3. In Book of Abstracts: Engineering Conf. International ECI “Alternative Materials for Radioactive Waste Stabilization and Nuclear Materials Containment”; ECI: Brooklyn, NY, USA, 2007. [Google Scholar]
- Orlova, A.I.; Orlova, M.P.; Loginova, E.E.; Lizin, A.A.; Tomilin, S.V.; Lukinykh, A.N.; Khainakov, S.A.; Garcia-Granda, S.; Demarin, V.T.; Oleneva, T.A.; et al. Thorium, Plutonium, Lanthanides and Some 1- and 2-Valent Elements in the New Orthophosphates with the Structure of Mineral Whitlockite. Chemistry, Structure, Stability. In Abstracts Booklet “Plutoniun Future—“The Science”, Proceedings of the Topical Conference on Plutonium and Actinides, Dijon, France, 7–11 July 2008; Elsevier: Amsterdam, Netherlands, 2008; pp. 313–314. [Google Scholar]
- Orlova, A.I.; Khainakov, S.A.; Loginova, E.E.; Oleneva, T.A.; Garcia Granda, S.; Kurazhkovskaya, V.S. Calcium Thorium Phosphate (whitlockite type Mineral). Synthesis and Structure Refinement. Crystallogr. Rep. 2009, 54, 591–597. [Google Scholar] [CrossRef]
- Benhamou, R.A.; Bessiere, A.; Wallez, G.; Viana, B.; Elaatmani, M.; Daoud, M.; Zegzouti, A. New insight in the structure-luminescence relationship of Ca9Eu(PO4)7. J. Solid State Chem. 2009, 182, 2319–2325. [Google Scholar] [CrossRef]
- Orlova, A.I.; Orlova, M.P.; Loginova, E.E.; Khainakov, S.; Garcia-Granda, S.; Lizin, A.A.; Tomilin, S.V.; Lukinikh, A.N.; Kurazshkovskay, V.S. Nature “experience” and experimental data on immobilization of actinides into calcium phosphate with whitelochite mineral structure. In Book of Abstracts: 6th Russian Conference on Radiochemistry “Radiochemistry-2009”; Lomonosov Moscow State University: Moscow, Russia, 2009; p. 294. [Google Scholar]
- Orlova, A.I.; Malanina, N.V.; Chuvil’deev, V.N.; Boldin, M.S.; Sakharov, N.V.; Nokhrin, A.V. Praseodymium and neodymium phosphates Ca9Ln(PO4)7 of whitlockite structure. Preparation of a ceramic with a high relative density. Radiochemistry 2014, 56, 380–384. [Google Scholar] [CrossRef]
- Adcock, C.T.; Tschauner, O.; Hausrath, E.M.; Udry, A.; Luo, S.N.; Cai, Y.; Ren, M.; Lanzirotti, A.; Newville, M.; Kunz, M.; et al. Shock-transformation of whitlockite to merrillite and the implications for meteoritic phosphate. Nat. Commun. 2017, 8, 14667. [Google Scholar] [CrossRef]
- Carrasco, I.; Piccinelli, F.; Bettinelli, M. Optical Spectroscopy of Ca9Tb1-xEux(PO4)7 (x = 0, 0.1, 1): Weak Donor Energy Migration in the Whitlockite Structure. J. Phys. Chem. C 2017, 121, 16943–16950. [Google Scholar] [CrossRef]
- Bénard, P.; Brandel, V.; Dacheux, N.; Jaulmes, S.; Launay, S.; Lindecker, C.; Genet, M.; Louër, D.; Quarton, M. Th4(PO4)4P2O7, a New Thorium Phosphate: Synthesis, Characterization, and Structure Determination. Chem. Mater. 1996, 8, 181–188. [Google Scholar] [CrossRef]
- Dacheux, N.; Podor, R.; Chassigneux, B.; Brandel, V.; Genet, M. Actinides Iimmobilization in New Matrices Bbased on Solid Solutions: Th4-xMxIV(PO4)(4)P2O7, (M-IV = U-238, Pu-239). J. Alloy. Compd. 1998, 271, 236–239. [Google Scholar] [CrossRef]
- Dacheux, N.; Podor, R.; Brandel, V.; Genet, M. Investigations of systems ThO2-MO2-P2O5 (M = U, Ce, Zr, Pu). Solid Solutions of Thorium-Uranium(IV) and ThoriumPlutonium(IV) Phosphate-diphosphates. J. Nucl. Mater. 1998, 252, 179–186. [Google Scholar] [CrossRef]
- Pichot, E.; Dacheux, N.; Brandel, V.; Genet, M. Investigation of Cs-137(+), Sr-85(2+) and Am-241(3+) Ion Exchange on Thorium Phosphate Hydrogenphosphate and their Immobilization in the Thorium Phosphate Diphosphate. New J. Chem. 2000, 24, 1017–1023. [Google Scholar] [CrossRef]
- Clavier, N.; Dacheux, N.; Martinez, P.; Du Fou de Kerdaniel, E.; Aranda, L.; Podor, R. Sintering of β-Thorium−Uranium(IV) Phosphate−Diphosphate Solid Solutions from Low-Temperature Precursors. Chem. Mater. 2004, 16, 3357–3366. [Google Scholar] [CrossRef]
- Clavier, N.; Dacheux, N.; Podor, R. Synthesis, characterization, sintering, and leaching of β-TUPD/monazite radwaste matrices. Inorg. Chem. 2006, 45, 22. [Google Scholar] [CrossRef]
- Clavier, N.; Du Fou de Kerdaniel, E.; Dacheux, N.; Le Coustumer, P.; Drot, R.; Ravaux, J.; Simoni, E. Behavior of thorium–uranium (IV) phosphate–diphosphate sintered samples during leaching tests. Part II. Saturation processes. J. Nucl. Mater. 2006, 349, 304–316. [Google Scholar] [CrossRef]
- Morozov, V.A.; Mironov, A.V.; Lazoryak, B.I.; Khaikina, E.G.; Basovich, O.M.; Rossell, M.D.; Van Tendeloo, G. Ag1/8Pr5/8MoO4: An incommensurately modulated scheelite-type structure. J. Solid State Chem. 2006, 179, 1183–1191. [Google Scholar] [CrossRef]
- Maček Kržmanc, M.; Logar, M.; Budič, B.; Suvorov, D. Dielectric and Microstructural Study of the SrWO4, BaWO4, and CaWO4 Scheelite Ceramics. J. Am. Ceram. Soc. 2011, 94, 2464–2472. [Google Scholar] [CrossRef]
- Cheng, J.; He, J. Electrical properties of scheelite structure ceramic electrolytes for solid oxide fuel cells. Mater. Lett. 2017, 209, 525–527. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, Q.; Zhao, C.; Chang, A. Comparison of structure and electrical properties of vacuum-sintered and conventional-sintered Ca1-xYxCeNbWO8 NTC ceramics. J. Alloy. Compd. 2017, 698, 1–6. [Google Scholar] [CrossRef]
- Xiao, M.; Sun, H.; Zhou, Z.; Zhang, P. Bond ionicity, lattice energy, bond energy, and microwave dielectric properties of Ca1-xSrxWO4 ceramics. Ceram. Int. 2018, 44, 20686–20691. [Google Scholar] [CrossRef]
- Potanina, E.A.; Orlova, A.I.; Nokhrin, A.V.; Boldin, M.S.; Sakharov, N.V.; Belkin, O.A.; Chuvil’deev, V.N.; Tokarev, M.G.; Shotin, S.V.; Zelenov, A.Y. Characterization of Nax(Ca/Sr)1-2xNdxWO4 complex tungstates fine-grained ceramics obtained by Spark Plasma Sintering. Ceram. Int. 2018, 44, 4033–4044. [Google Scholar] [CrossRef]
- Potanina, E.A.; Orlova, A.I.; Mikhailov, D.A.; Nokhrin, A.V.; Chuvil’deev, V.N.; Boldin, M.S.; Sakharov, N.V.; Lantcev, E.A.; Tokarev, M.G.; Murashov, A.A. Spark Plasma Sintering of fine-grained SrWO4 and NaNd(WO4)2 tungstates ceramics with the scheelite structure for nuclear waste immobilization. J. Alloys Compd. 2019, 774, 182–190. [Google Scholar] [CrossRef]
- Pang, L.-X.; Zhou, D.; Yue, Z.-X. Temperature independent low firing [Ca0.25(Nd1-xBix)0.5]MoO4 (0.2 ≤ x ≤ 0.8) microwave dielectric ceramics. J. Alloys Compd. 2019, 781, 385–388. [Google Scholar] [CrossRef]
- Hanusa, J. Raman scattering and ifra-red spectra of tungstates KLn(WO4)2-family (Ln: La – Lu). J. Mol. Struct. 1984, 114, 471–474. [Google Scholar] [CrossRef]
- Pages, M.; Freundlich, W. Phases of scheelite structure in the neptunium molybdate and sodium or litium molybdate systems. J. Inorg. Nucl. Chem. 1972, 34, 2797–2801. [Google Scholar] [CrossRef]
- Lee, M.R.; Mahe, P. Molybdates et tungstates d`uranium IV et de sodium. C. R. Acad. Sci. Paris. 1974, 279, 1137–1170. [Google Scholar]
- Tabuteau, A.; Pages, M. Identification and crystal chemistry of double molybdates of alkalimetals (K, Rb, Cs) and transuranium elements (Np, Pu, Am). J. Inorg. Nucl. Chem. 1980, 42, 401–403. [Google Scholar] [CrossRef]
- Tabuteau, A.; Pages, M.; Freundlich, W. Sur les phases de structure sheelite dans les systems molybdate deplutonium-molybdate de litium du sodium. Mater. Res. Bull. 1972, 7, 691–697. [Google Scholar] [CrossRef]
- Müller-Buschbaum, H.; Gallinat, S. Synthese und Röntgenstrukturanalyse von KCuGd2Mo4O16 und CuTb2Mo4O16. Z. Naturforsch. 1995, 50, 1794–1798. [Google Scholar] [CrossRef]
- 454. Basovich, M.; Khaikina, E.G.; Vasil’ev, E.V.; Frolov, A.M. Phase formation in the Li2MoO4—Rb2MoO4—Ln2(MoO4)3 systems and the poperties of LiRbLn2(MoO4)4. Zh. Neorg. Khim. 1995, 40, 2047-251. [Google Scholar]
- Basovich, O.M.; Khaikina, E.G.; Solodovnikov, S.F.; Tsyrenova, G.D. Phase formation in the systems Li2MoO4—K2MoO4—Ln2(MoO4)3 (Ln = La, Nd, Dy, Er) and properties of triple molybdates LiKLn2(MoO4)4. J. Solid State Chem. 2005, 178, 1580–1588. [Google Scholar] [CrossRef]
- Szillat, H.; Müller-Buschbaum, H. Synthese und Kristallstructur von KCuHoMo4O16. Z. Nat. 1994, 49, 350–354. [Google Scholar]
- Wang, Y.; Wu, W.; Fu, X.; Liu, M.; Cao, J.; Shao, C.; Chen, S. Metastable scheelite CdWO4:Eu3+ nanophosphors: Solvothermal synthesis, phase transitions and their polymorph-dependent luminescence properties. Dyes Pigment. 2017, 147, 283–290. [Google Scholar] [CrossRef]
- Donald, I.W. Waste Immobilisation in Glass and Ceramic Based Hosts; Wiley: Chichester, UK, 2010; 507p. [Google Scholar]
- Kinoshita, H. Development of ceramic matrices for high level radioactive waste. In Handbook of Advanced Radioactive Waste Conditioning Technologies; Ojovan, M., Ed.; Woodhead: Cambridge, UK, 2011; Chapter 10; p. 293. [Google Scholar]
- Ojovan, M.I.; Lee, W.E.; Kalmykov, S.N. An Introduction to Nuclear Waste Immobilisation, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2019; p. 497. [Google Scholar]
- Zhang, Y.; Wei, T.; Zhang, Z.; Kong, L.; Dayal, P.; Gregg, D.J. Uranium brannerite with Tb(III)/Dy(III) ions: Phase formation, structures, and crystallizations in glass. J. Am. Ceram. Soc. 2019, 1–11. [Google Scholar] [CrossRef]

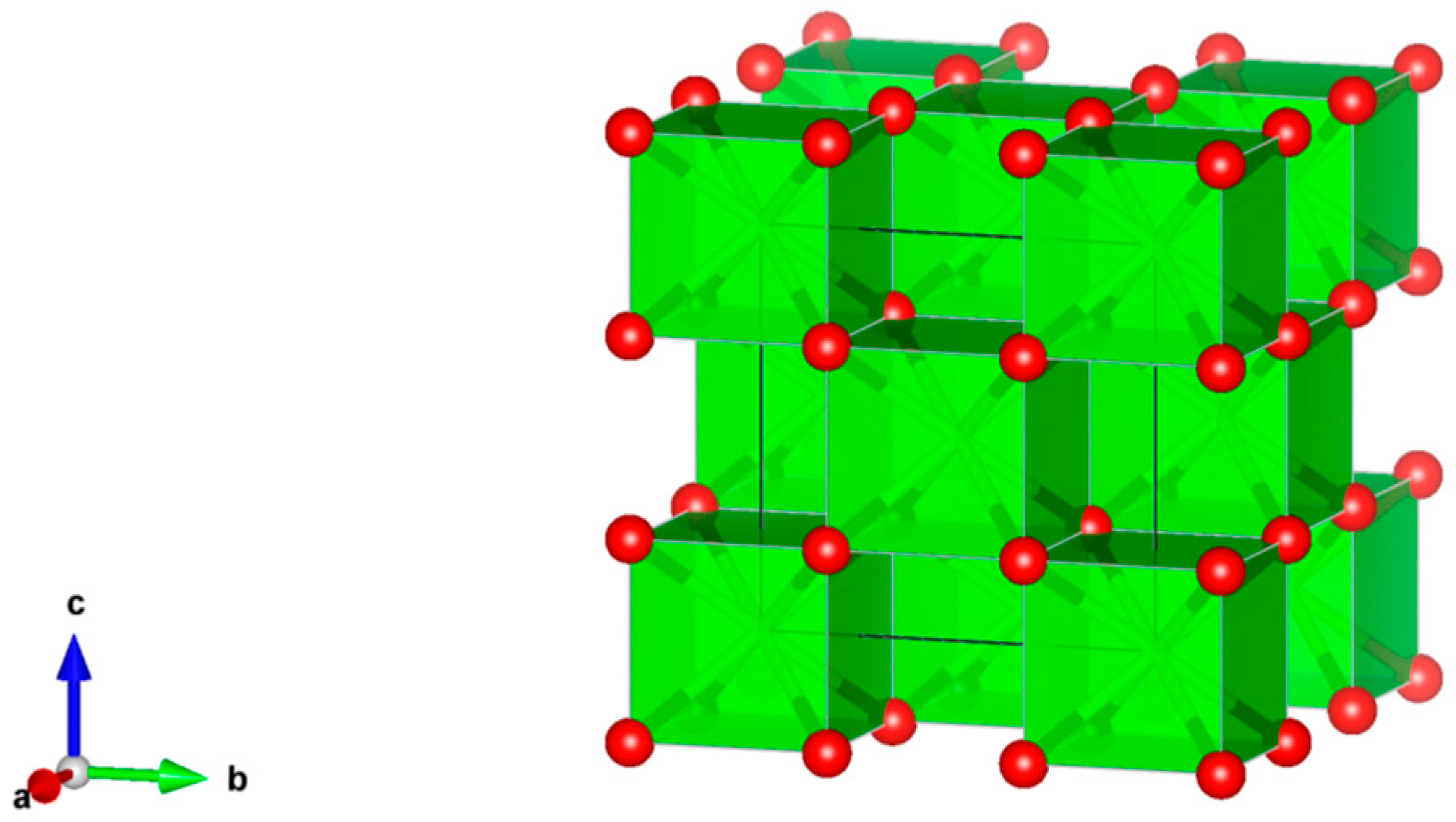
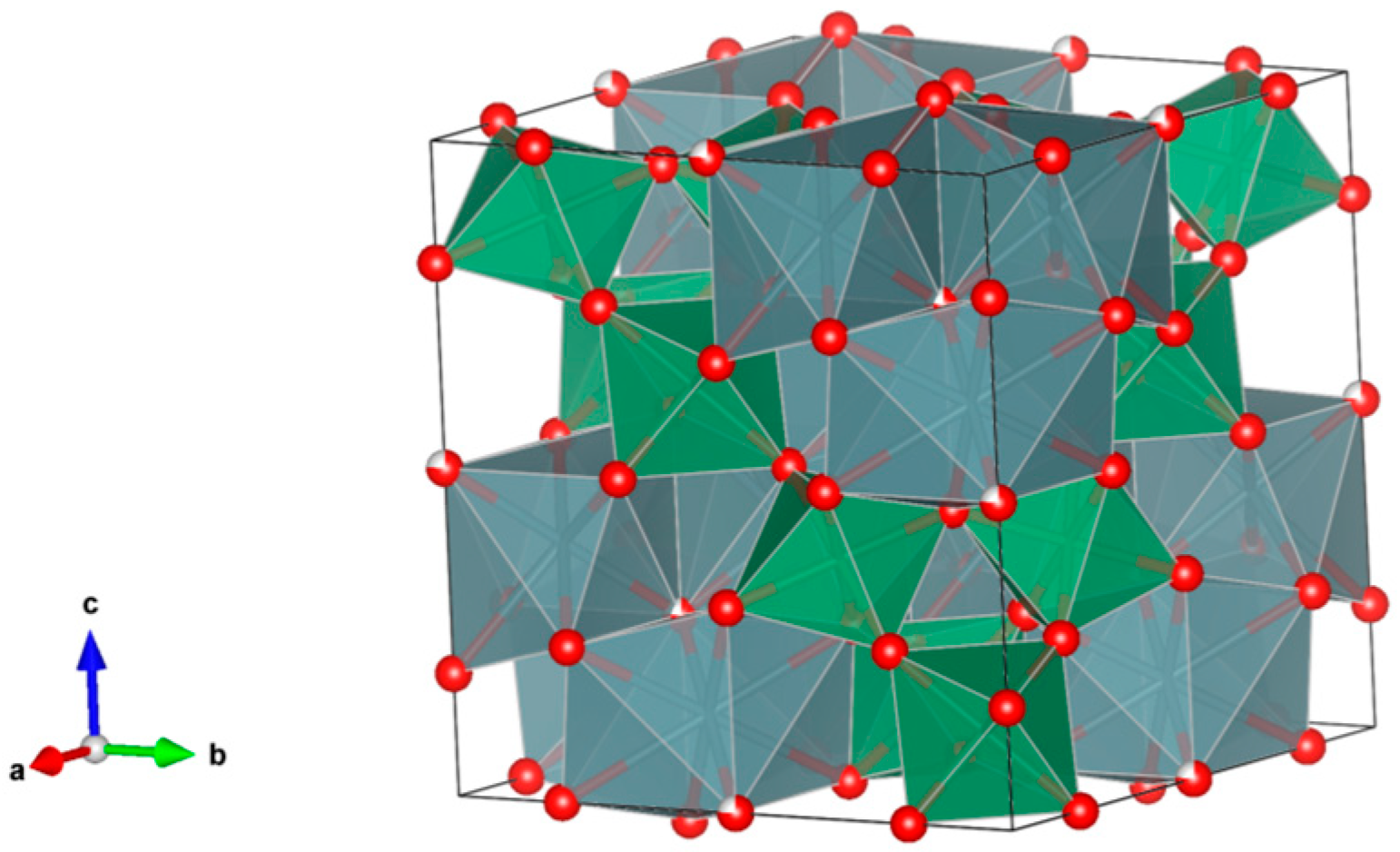

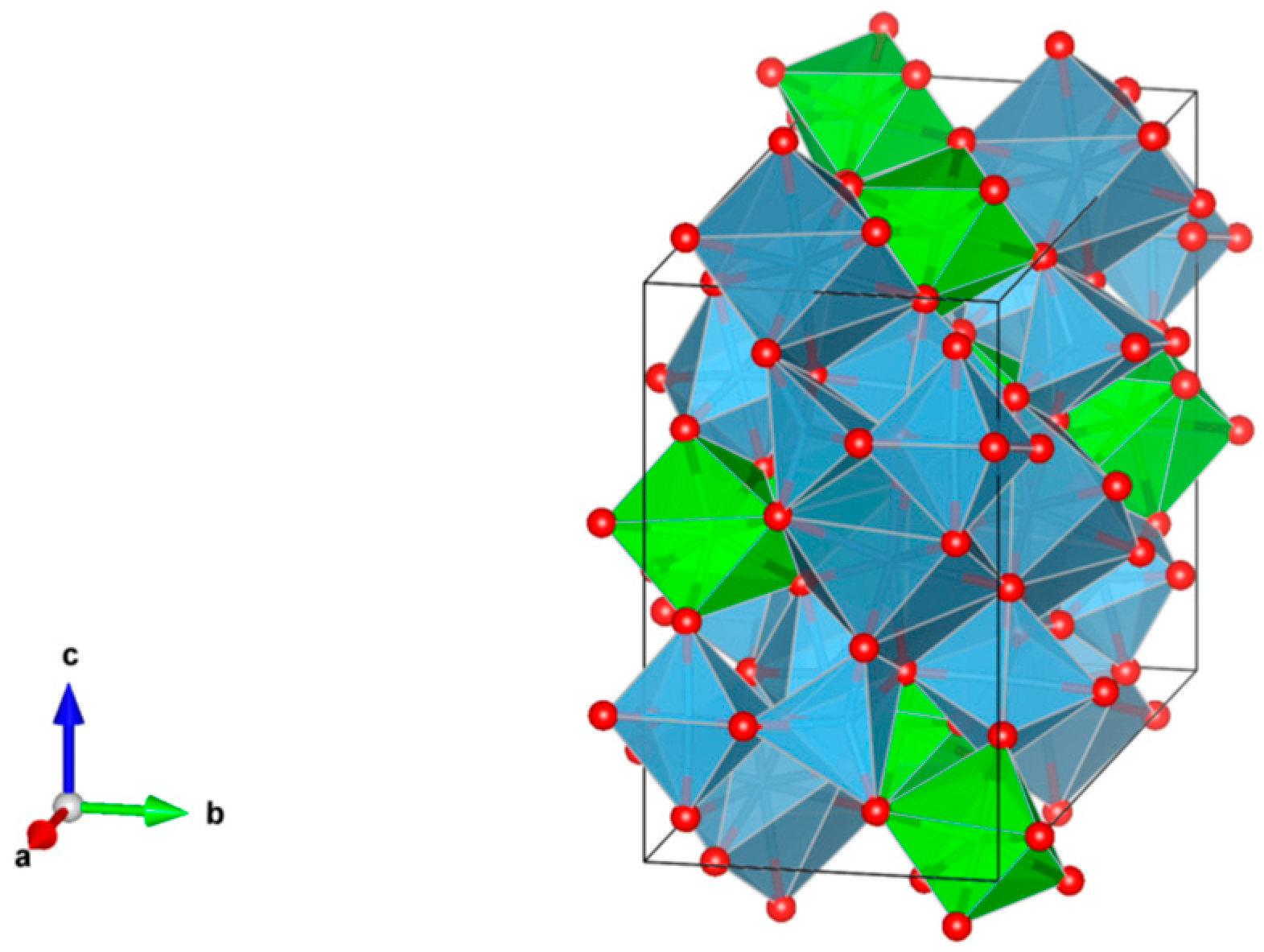
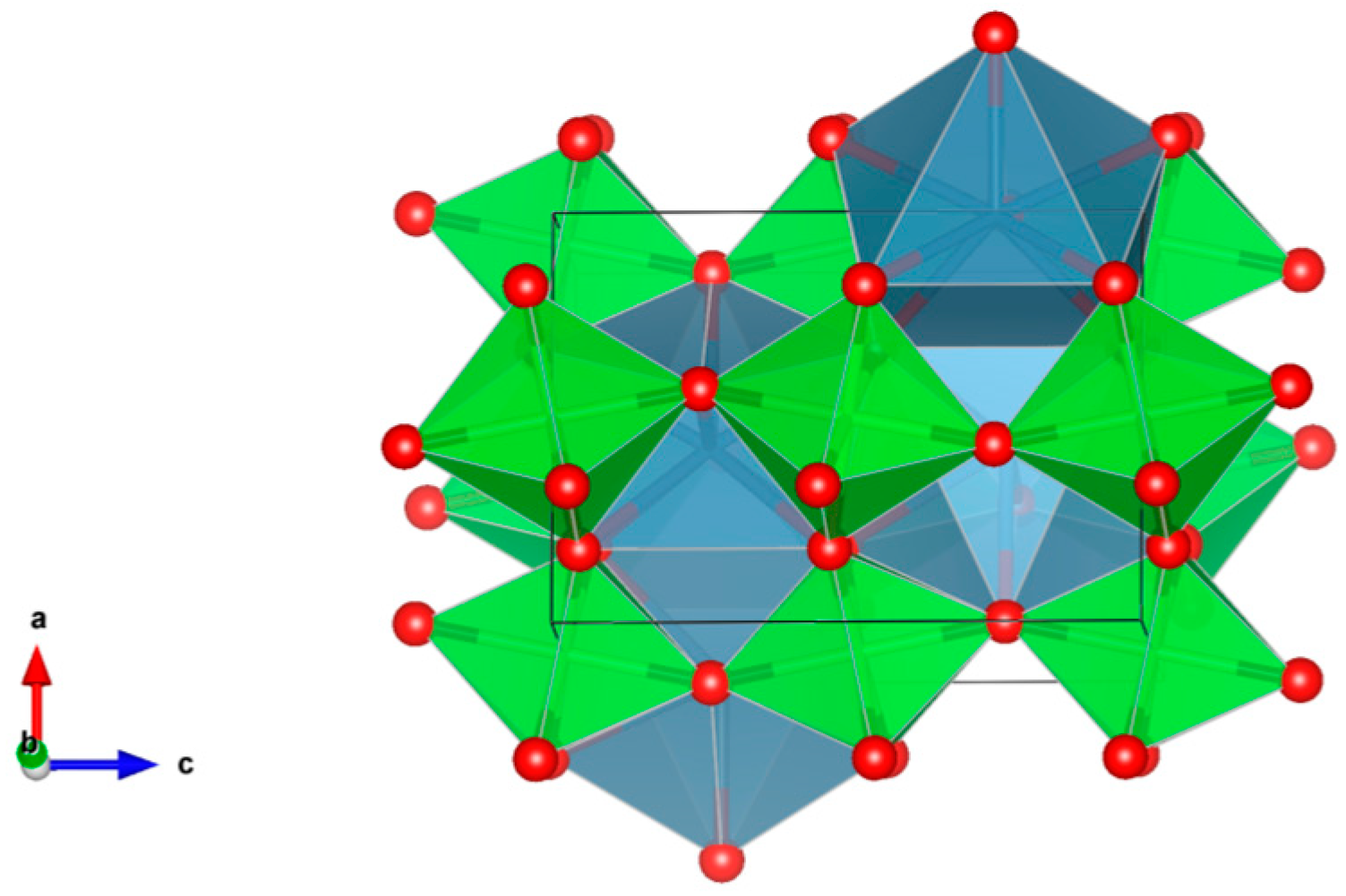
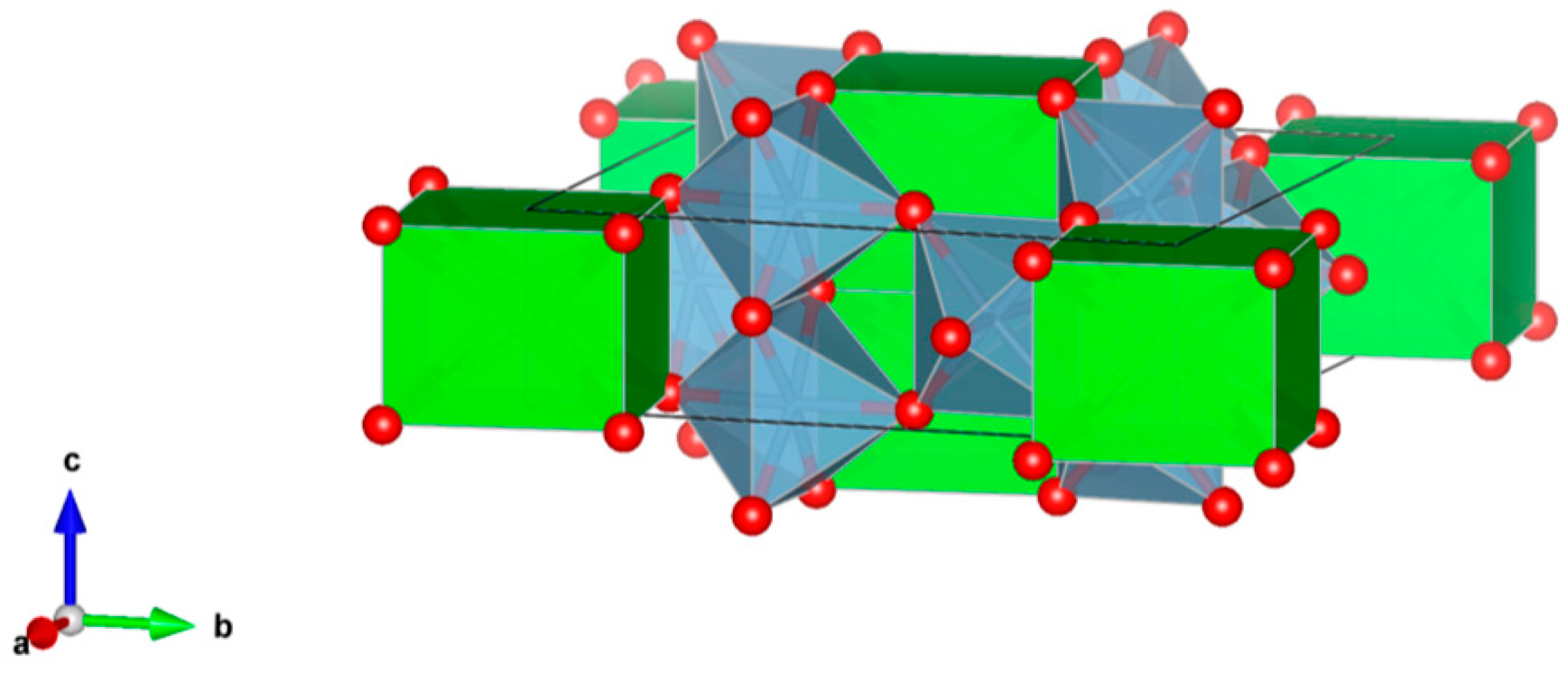
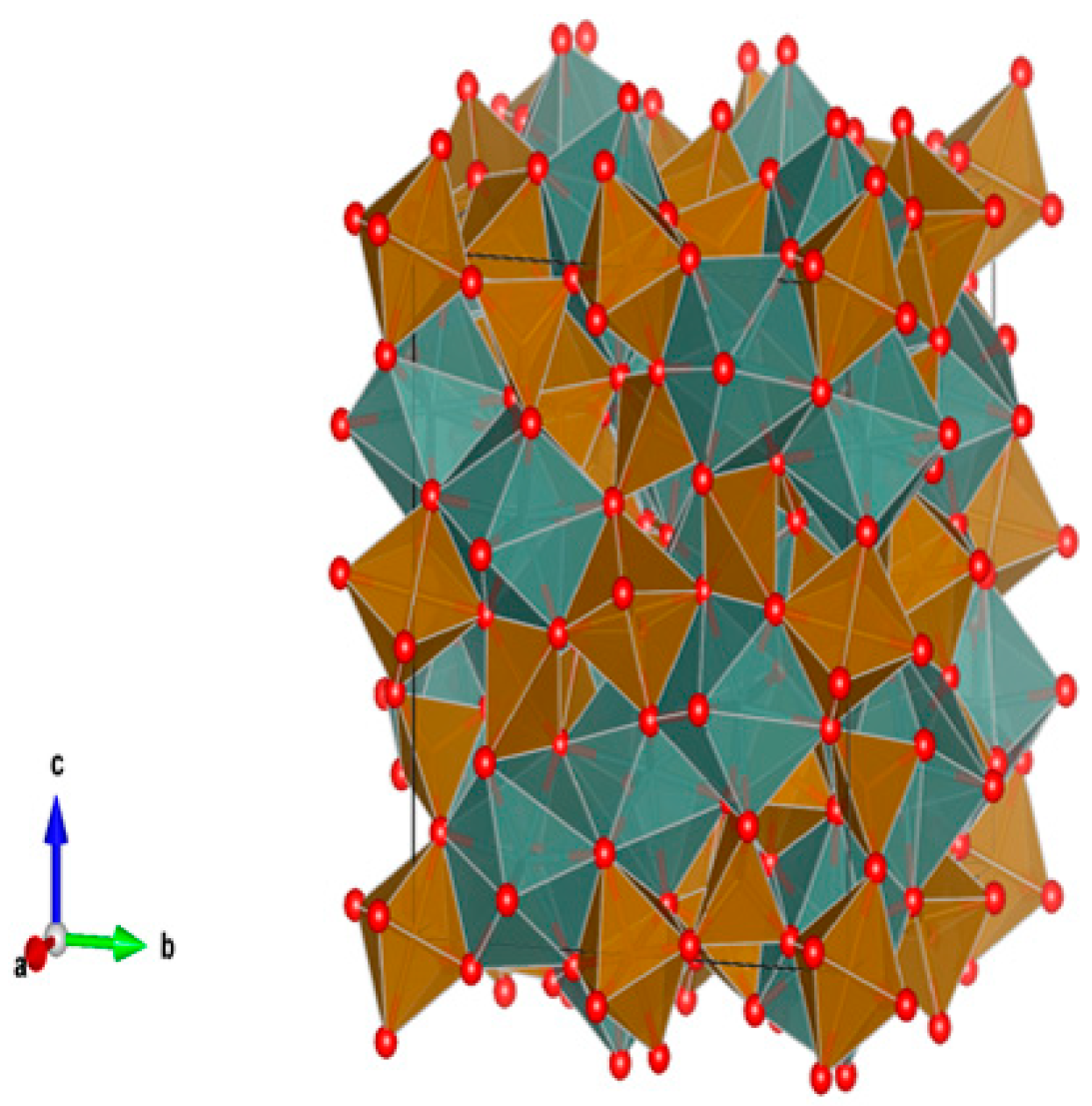
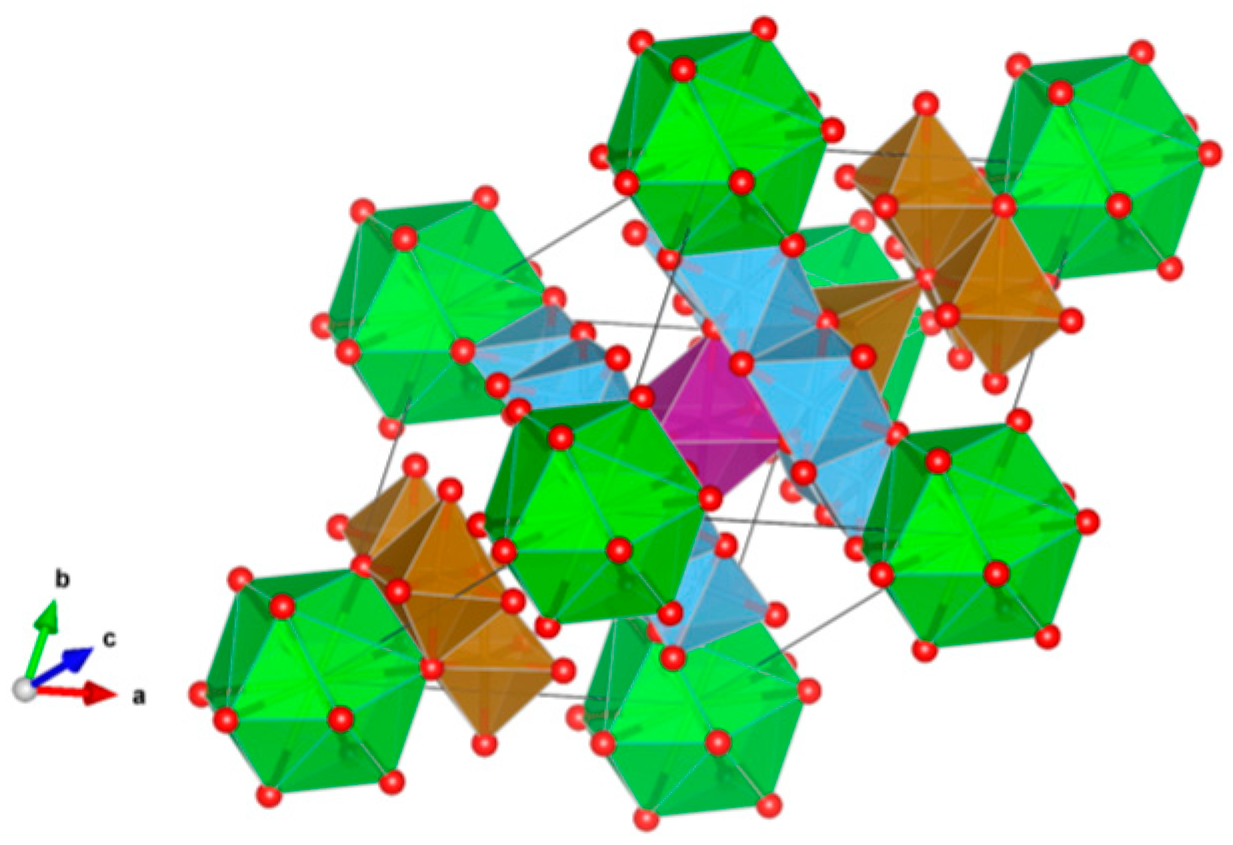
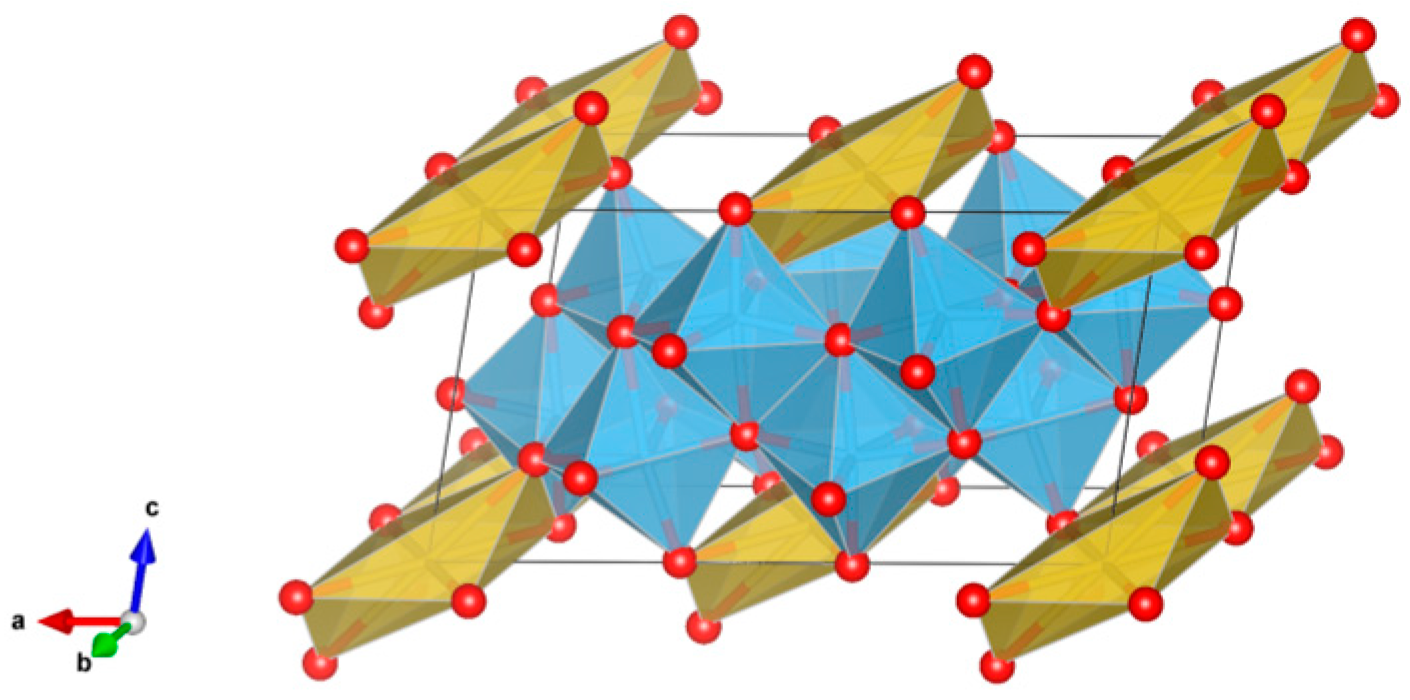
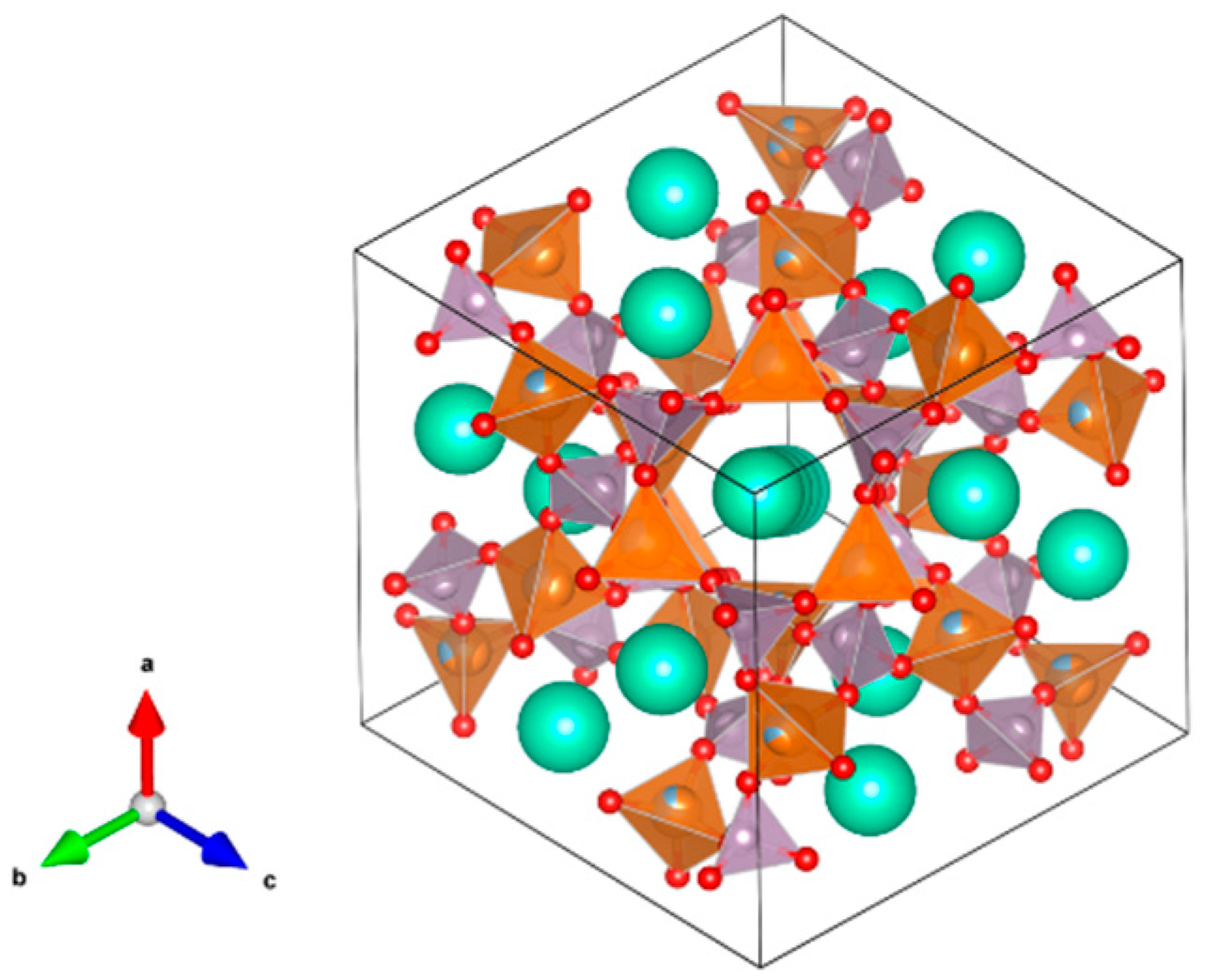

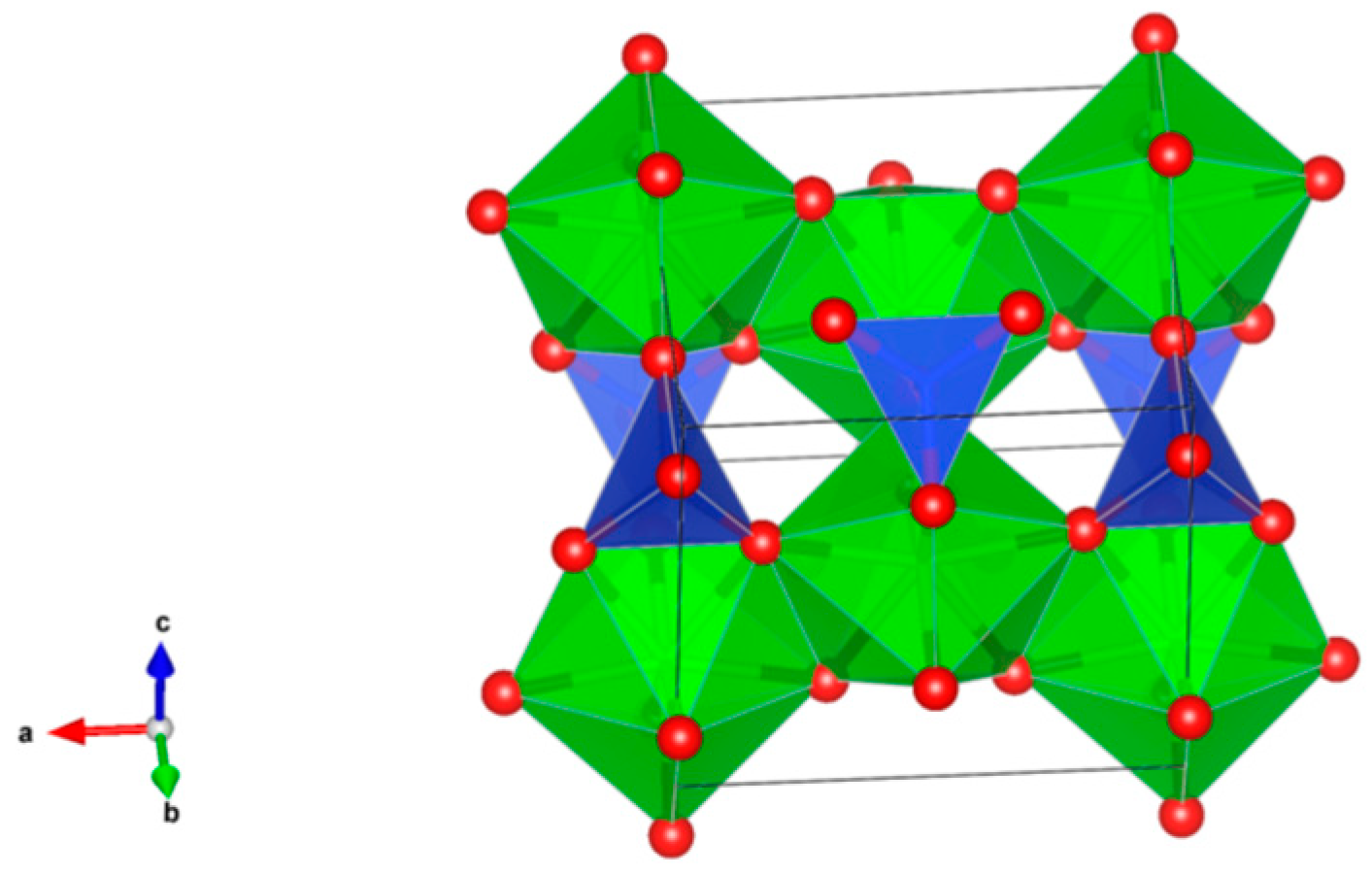
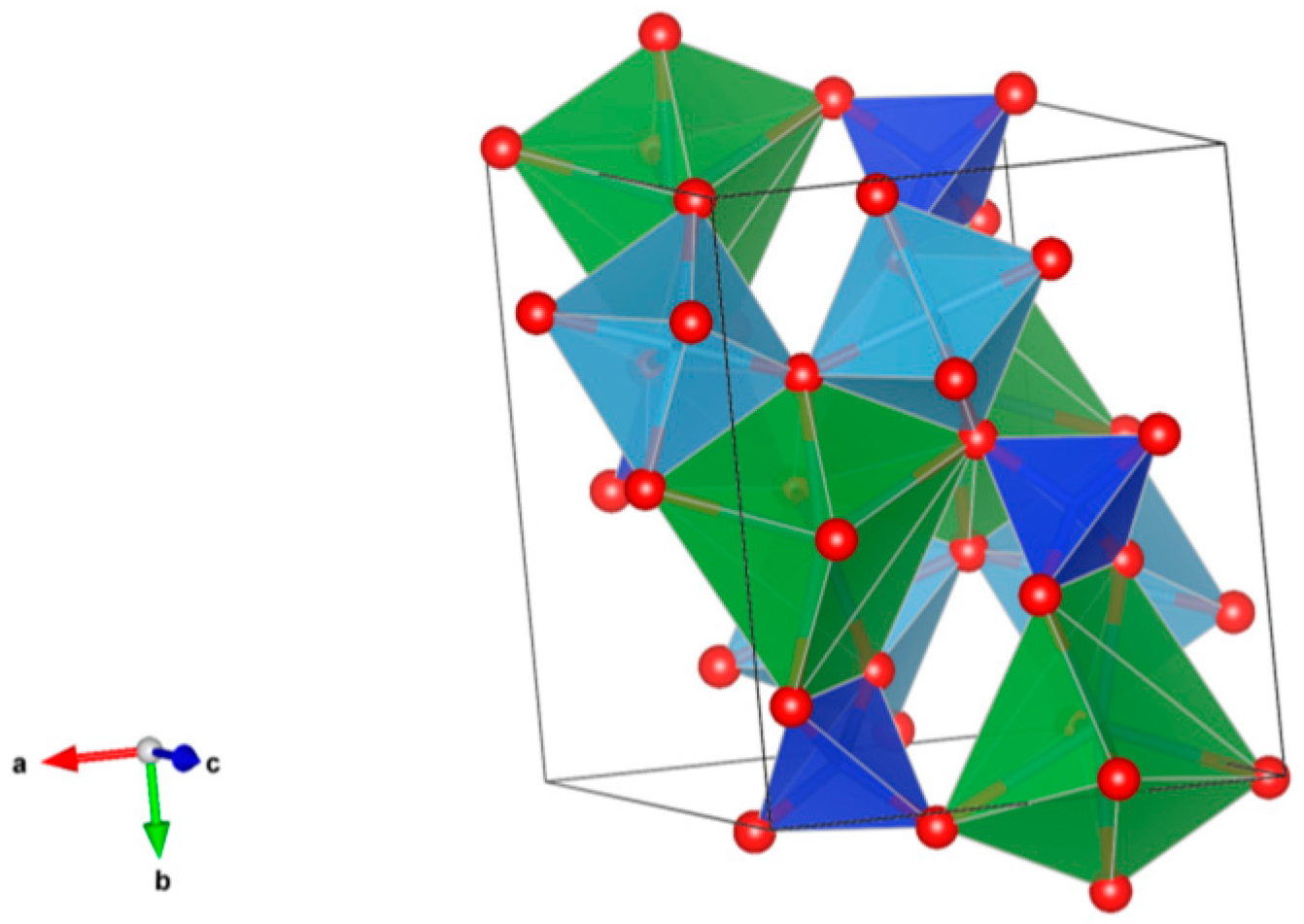


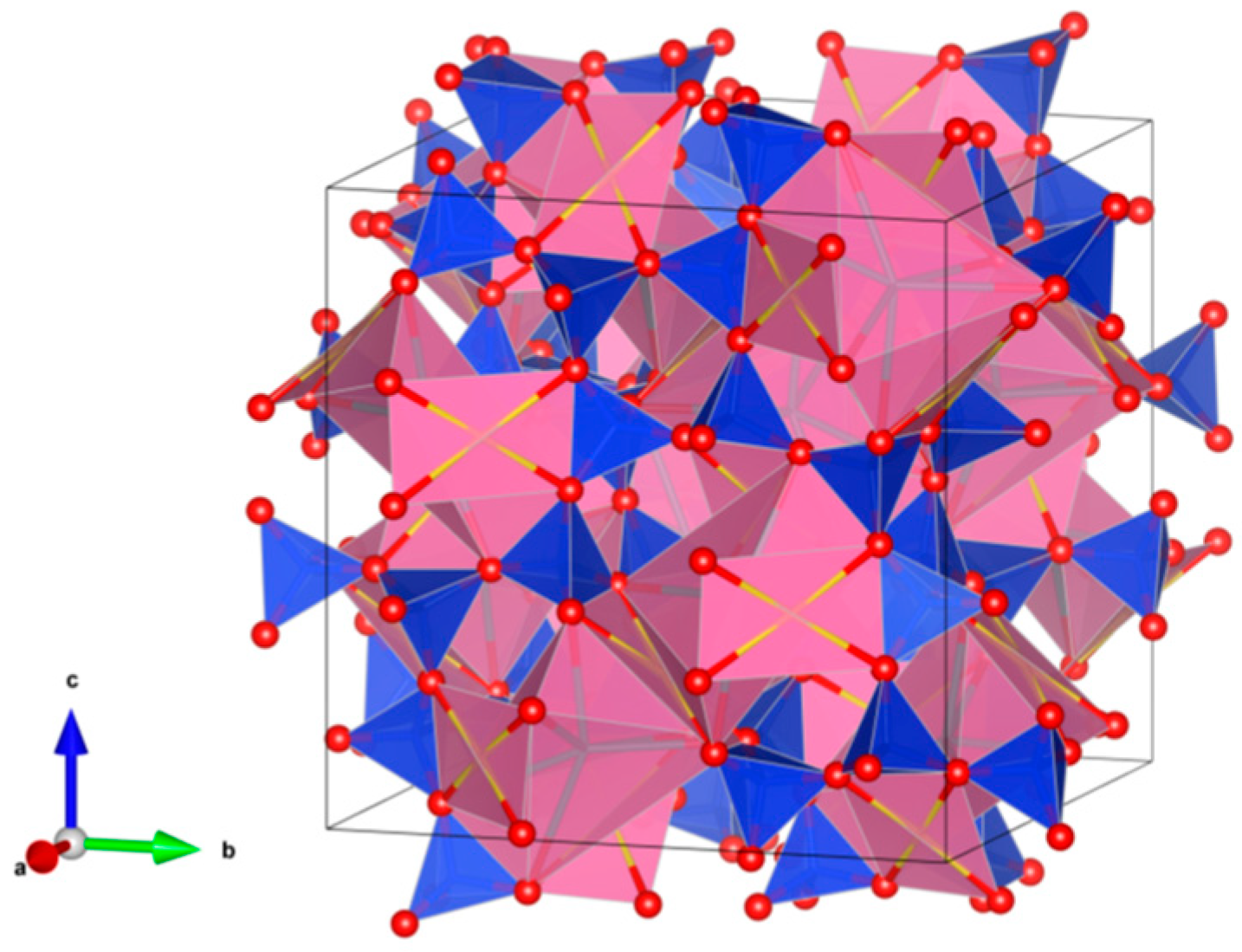



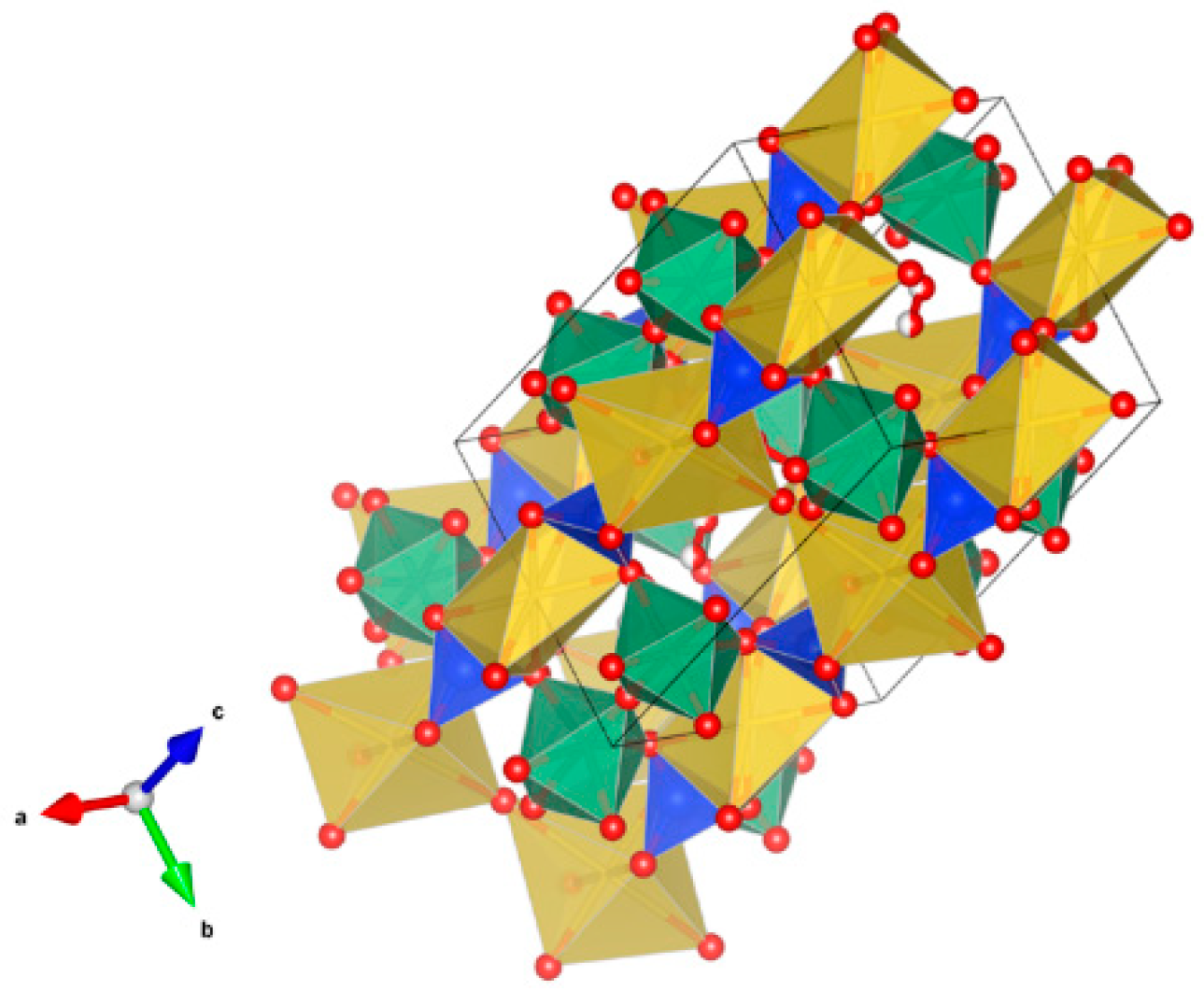
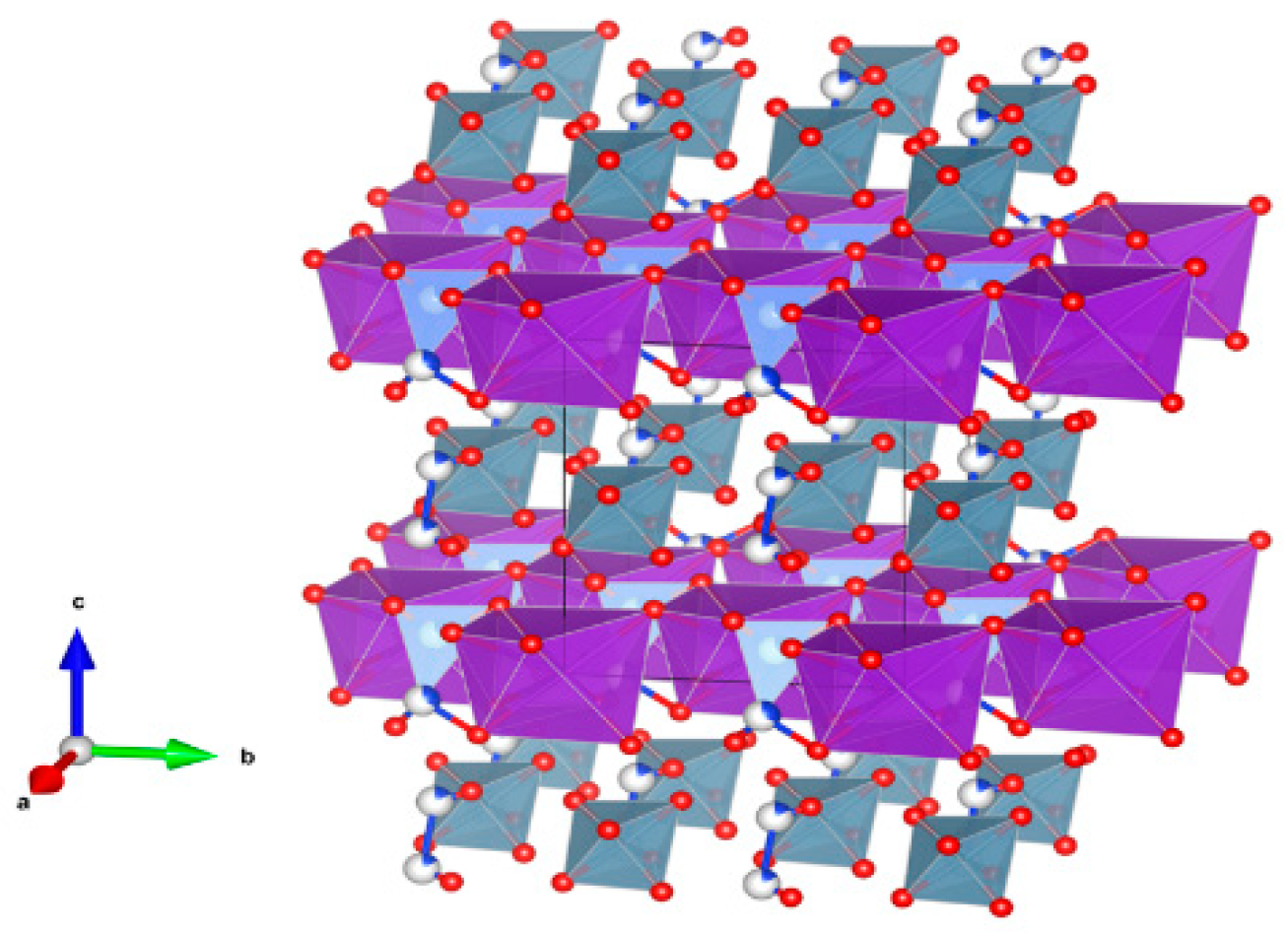

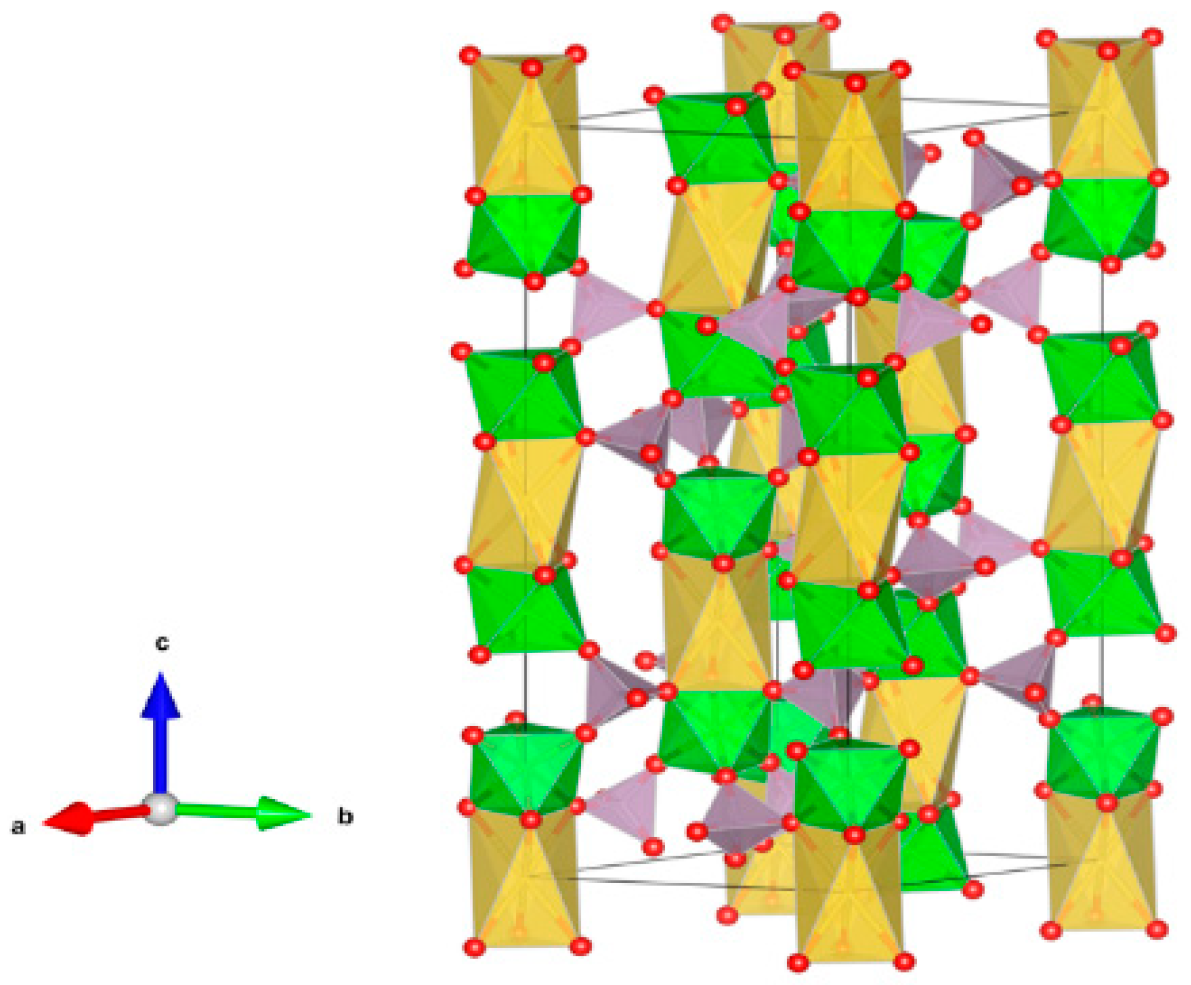
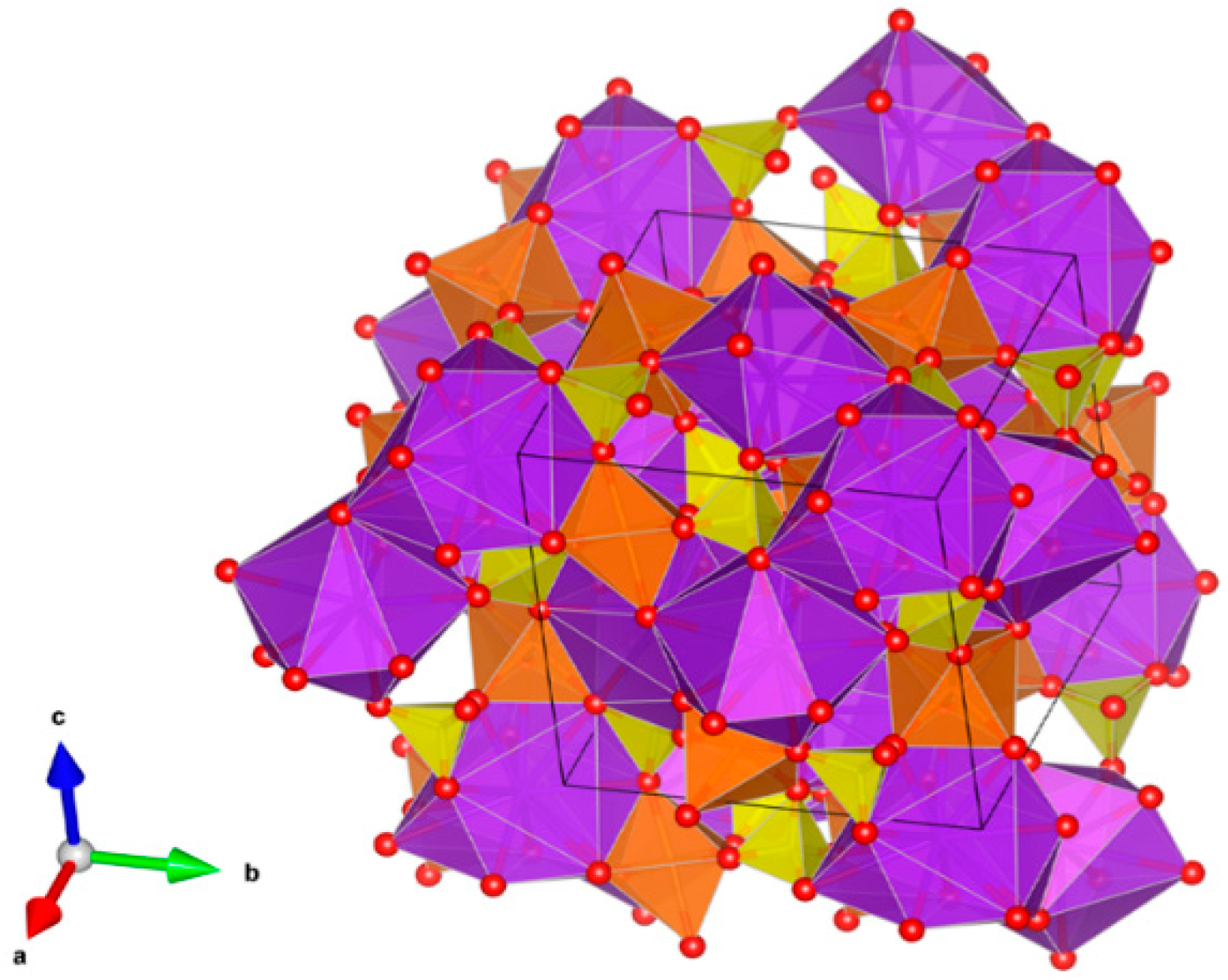
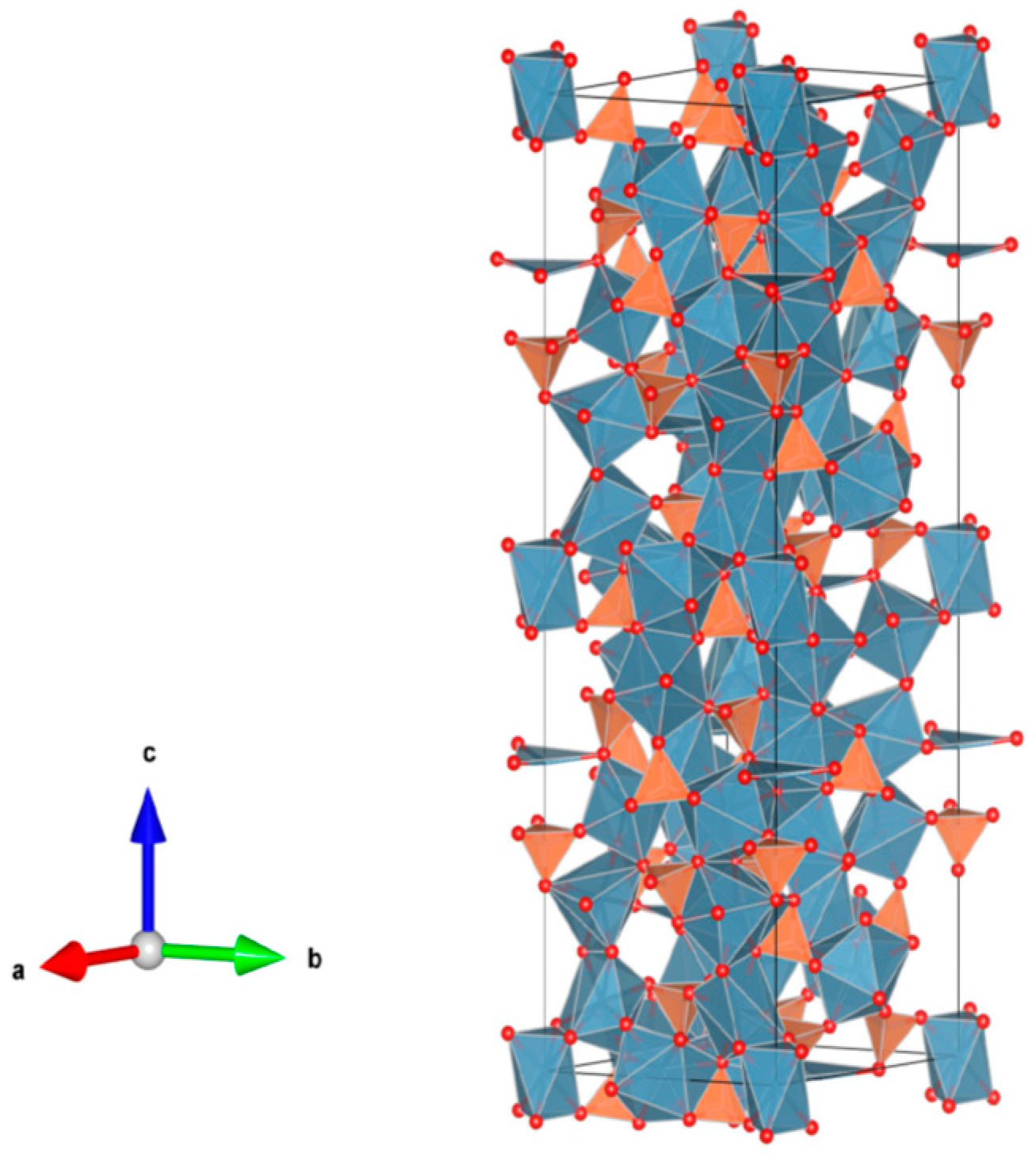
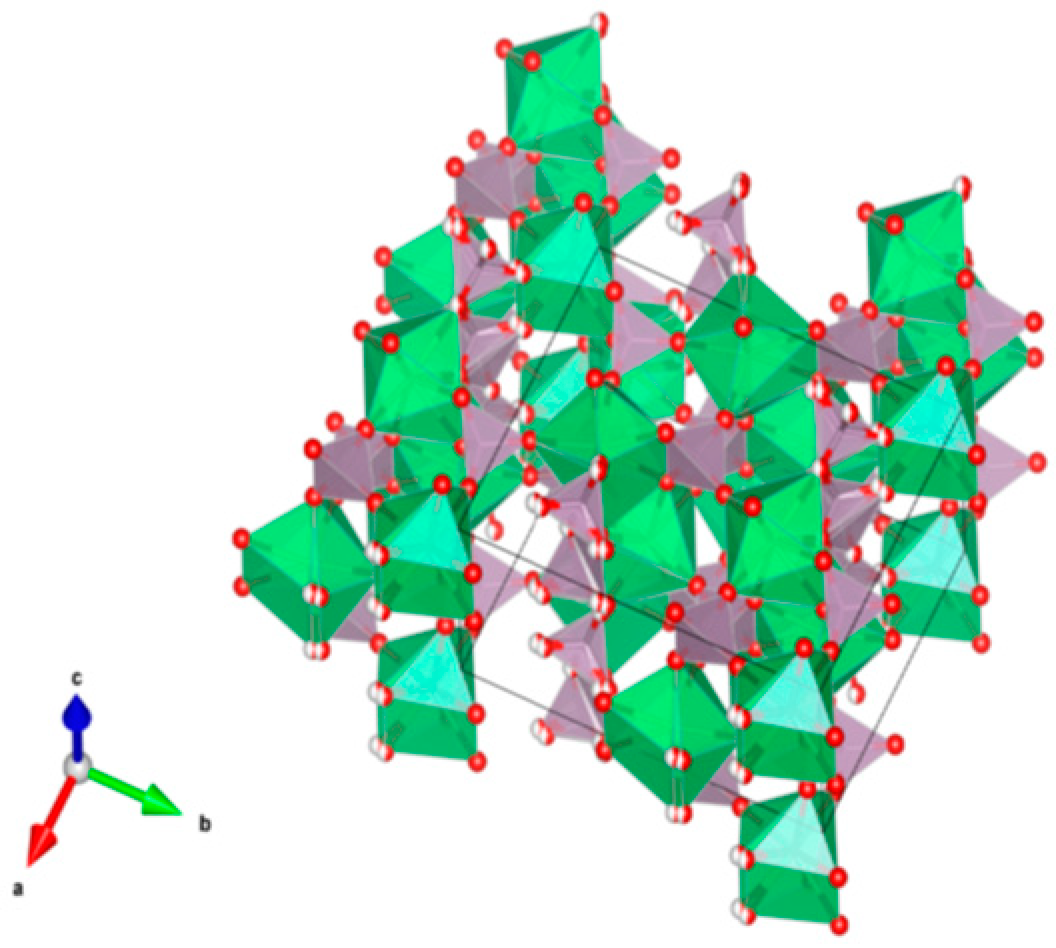

| Type of Chemical Compound | Structure | Compound Cations | ||
|---|---|---|---|---|
| Structural Type | Syngony, Sp. gr. | |||
| Oxide Compounds | ||||
| Simple oxides | SiO2 | Silica | rhombohedral, R3 | Li, Na, K. Mg, Ca, Mn, Cu, Ni, Pb B, Al, Fe, Cr, Ti, Zr, Te |
| CeO2 | Fluorite | cubic, Fm3m | Cs, Sr, Ce, Y, Zr, U, Th, Hf, Pu, U, Np | |
| Complex oxides | A2B2O7 | Pyrochlore | cubic, | A: Na, Ca, U, Th, Y, Ln; B: Nb, Ta, Ti, Zr, Fe3+ |
| A6B12C5TX40-x | Murataite | cubic, | U, Np, Pu, Am, Cm, REE | |
| CaZrTi2O7 | Zirconolite | trigonal C2/c | Gd, Hf, Ce, Th, U, Pu, Nb | |
| CaTiO3 | Perovskite | cubic, Pm3m; rhombohedral, Pnma | Ca, Y, REE, Ti, Zr, U, Pu | |
| Ba1.2(Al,Ti)8O16 | Hollandite | tetragonal, I4/m | Na, K, Cs, Mg, Ca, Ba, Al, Fe, Mn3+, Si, Ti, Mn4+ | |
| [8]A3[6]B2[TiO4]3 .[8](Ca,Gd, actinides)[6]Fe2[4]Fe3O12 | Garnet | cubic, Ia3d | A, B: REE, An, Y, Mg, Ca, Fe2+, Mn2+; X: Cr3+, Fe3+, Al3+, Ga3+, Si4+, Ge4+, V5+ | |
| (Sr,Pb,La,Ce,Y)(Ti,Fe3+,Mn,Mg,Zn,Cr,Al,Zr,Hf,U,V,Nb,Sn,Cu,Ni)21O38 | Crichtonite | rhombohedral, R3 | ||
| Na2Al2(Ti,Fe)6O16 | Freudenbergitespinel based phase | monoclinic, C12/m1 | Mg, Co, Ni, Zn, Al, Ti3+, Cr, Fe, Ga, Si, Nb | |
| P-Pollucite | cubic, I4132 | Li, Na, K, Rb, Cs, Tl, Be, Mg, Sr, Ba, Cd, Mn, Co, Ni, Cu, Zn, B, Al, Fe, Si, Ti, P, V, Nb, Ta | ||
| ZrSiO4/ThSiO4/USiO4 | Zircon/Thorite/Coffinite | tetragonal, I41/amd | REE, Th, U, Pu; Na, Mg, Ca, Mn, Co, Fe, Ti, P, V, Se, Mo | |
| CaTiSiO5 [CaTiO(SiO4)] | Titanite (sphene) | monoclinic, P2I/a | Mg, Ca, Sr, Ba, Mn, Al, Fe, Cr, Ce, Y, Zr, Th, F | |
| (REE,Ca)5(SiO4,PO4)3(OH,F) | Britholite (oxy-apatite) | monoclinic, sp. gr. P21, hexagonal, P63/m | Cs, Sr, B, Th, U, Np, Nd3+, La3+, Pu3+ | |
| Salt compounds | ||||
| Framework Silicates | (Xx/n[(AlO2)x(SiO2)y] | Zeolites | Na, K, NH4+, Cs, Mg, Ca, Sr, Co, Fe, Ga, REE, Ti | |
| (Ca,Na)2Al2Si4O12·2H2O | Pollucite | cubic, Ia3d | Li, Na, K, Rb, Cs, Tl, Be, Mg, Sr, Ba, Cd, Mn, Co, Ni, Cu, Zn, B, Al, Fe, Si, Ti, P, V, Nb | |
| NaAlSiO4 | Nepheline/Leucite | Nepheline: hexagonal, P63; Leucite: tetragonal, I41/a, I41/acd; cubic, Ia3d | Li, Na, K, Rb, Cs, Be, Mg, Ca, Ba, Pb, Mn, Co, Ni, Al, Fe, Cr, Si, Ti, V | |
| Na8Cl2Al6Si6O24 | Sodalite | cubic, | Na, K, Mg, Ca, Mn, Fe, Al, Si, Ti, Cl, SO4, CO3 | |
| (Na,Ca,K)6[Al6Si6O24]((Na,Ca,K)2CO3)1.6·2.1H2O | Cancrinite | hexagonal, P63 | Cl−, SO42−, | |
| [(Ca,Na,K,Ba)AlSiO4 | Crystalline SilicoTitanate (CST) | cubic, sp. gr. Pm3m up to 105 °C, after tetragon. symm., sp. gr. I4/mcm or P42/mcm | Na, K, Cs, Ca, Sr, Ba, Pb, Al, REE, Bi, Ti, Zr, Nb, Ta | |
| LiAl3Si3O11, NaAl3Si3O11, KAl3Si3O11, RbAl3Si3O11, CsAl3Si3O11, TlAl3Si3O11, Ca0.5□0.5Al3Si3O11, Sr0.5□0.5Al3Si3O11, Ba0.5□0.5Al3Si3O11, La0.33□0.66Al3Si3O11 | Micas (Dehydroxylated) | monoclinic, C2/c | Cs, Rb, Ba, Mg, Fe2+, Fe3+, Mn, Li, Cr, Ti, V | |
| Phosphates | CePO4 | Monazite | monoclinic, P21/n | Ce: Li, Na, K, Rb, Mg, Ca, Sr, Ba, Cd, Pb, Bi, Y, La, Pr, Nd, Sm, Eu, Gd, Tb, Yb, Am, Cm, Cf, Es, Ge, Zr, Th, Np, U, Pu; P: Cr, Si, Se, V, As, S |
| YPO4 | Xenotime | tetragonal, I41/amd | Be, Ca, Al, Sc, La, Ce, Er, Dy–Lu, Zr, Th, U | |
| Ca4-xRE6+x(SiO4)6-y(PO4)y(O,F)2 | Apatite | hexagonal, P63/m; monoclinic, P21/b | Na, K, Cs, Mg, Ca, Sr, Ba, Mn, Ni, Cd, Hg, Pb, Cr, Y, REE, Th, U, Si, P, V, As, S, F, Cl, OH, CO3 | |
| NaZr2(PO4)3 | Sodium zirconium phosphate (NZP) | rhombohedral, R3c, R3 | Li, Na, K, Rb, Cs; H, Cu(I), Ag, Mg, Ca, Sr, Ba, Mn, Co, Ni, Cu, Zn, Cd, Hg, Al, Ga, In, Sc, Y, La, Ce-Lu, Am, Cm, V, Cr, Fe, Sb, Bi, Ge, Sn, Ti, Zr, Hf, Mo, Ce, Th, U, Np, Pu, Sb, Nb, Ta | |
| K2Mg2(SO4)3 | Langbeinite | cubic, P213 | Na, K, Rb, Cs, Tl, NH4, Mg, Sr, Ba, Pb, Mn, Co, Ni, Zn, Al, Fe Cr, Ti3+, Ga, V3+, Rh, In, REE, Bi, Sn, Ti, Zr, Hf, P, Nb, Ta, S | |
| β-Ca3(PO4)2 | Whitlockite | trigonal, R3c | H, Li, Na, K, Cu, Mg, Ca, Sr, Ba, Al, Sc, Cr, Fe, Ga, In, La, Ce, Sm, Eu, Gd, Lu, Th, Pu | |
| Th4(PO4)4P2O7 | Thorium phosphate/Diphosphate (TPD) | orthorhombic, Pbcm, Pcam | U, Np, Pu, Am, Cm | |
| Tungstates | CaWO4 | Scheelite | tetragonal, I4/c | Ca: Li, Na, K, Rb, Cs, Tl; Ca, Sr, Ba, Mn, Cu; Fe, Ce, La-Lu, Y; Th, U, Np, Pu; Nb, Ta; W: Mo, Re, I, V, Ge |
| Aluminates | X(Al,Fe)12O19 | Magnetoplumbite | hexagonal, P63/mmc | Na, Cs, Mg, Sr, Ba, Pb, Mn, Co, Cu, Al, Fe, Sc, Y, La, Ce, Sm, Gd, Yb, Lu, An, Si, Ti, Sn |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orlova, A.I.; Ojovan, M.I. Ceramic Mineral Waste-Forms for Nuclear Waste Immobilization. Materials 2019, 12, 2638. https://doi.org/10.3390/ma12162638
Orlova AI, Ojovan MI. Ceramic Mineral Waste-Forms for Nuclear Waste Immobilization. Materials. 2019; 12(16):2638. https://doi.org/10.3390/ma12162638
Chicago/Turabian StyleOrlova, Albina I., and Michael I. Ojovan. 2019. "Ceramic Mineral Waste-Forms for Nuclear Waste Immobilization" Materials 12, no. 16: 2638. https://doi.org/10.3390/ma12162638
APA StyleOrlova, A. I., & Ojovan, M. I. (2019). Ceramic Mineral Waste-Forms for Nuclear Waste Immobilization. Materials, 12(16), 2638. https://doi.org/10.3390/ma12162638





