Bactericidal and Biocompatible Properties of Plasma Chemical Oxidized Titanium (TiOB®) with Antimicrobial Surface Functionalization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Antibacterial Functionalization of TiOB®
2.2.1. TiOB® Gta
2.2.2. TiOB® SiOx Ag
2.2.3. TiOB® Zn
2.3. X-ray Photoelectron Spectroscopy (XPS)
2.4. Experimental Section
2.4.1. Bactericidal Properties of Functionalized TiOB®
2.4.1.1. Agar Diffusion Test
2.4.1.2. Proliferation Test
2.4.2. Biocompatible Properties of Functionalized TiOB®
2.5. Statistics
3. Results
3.1. Surface Characterization by Scanning Electron Microscopy
3.2. XPS Analysis
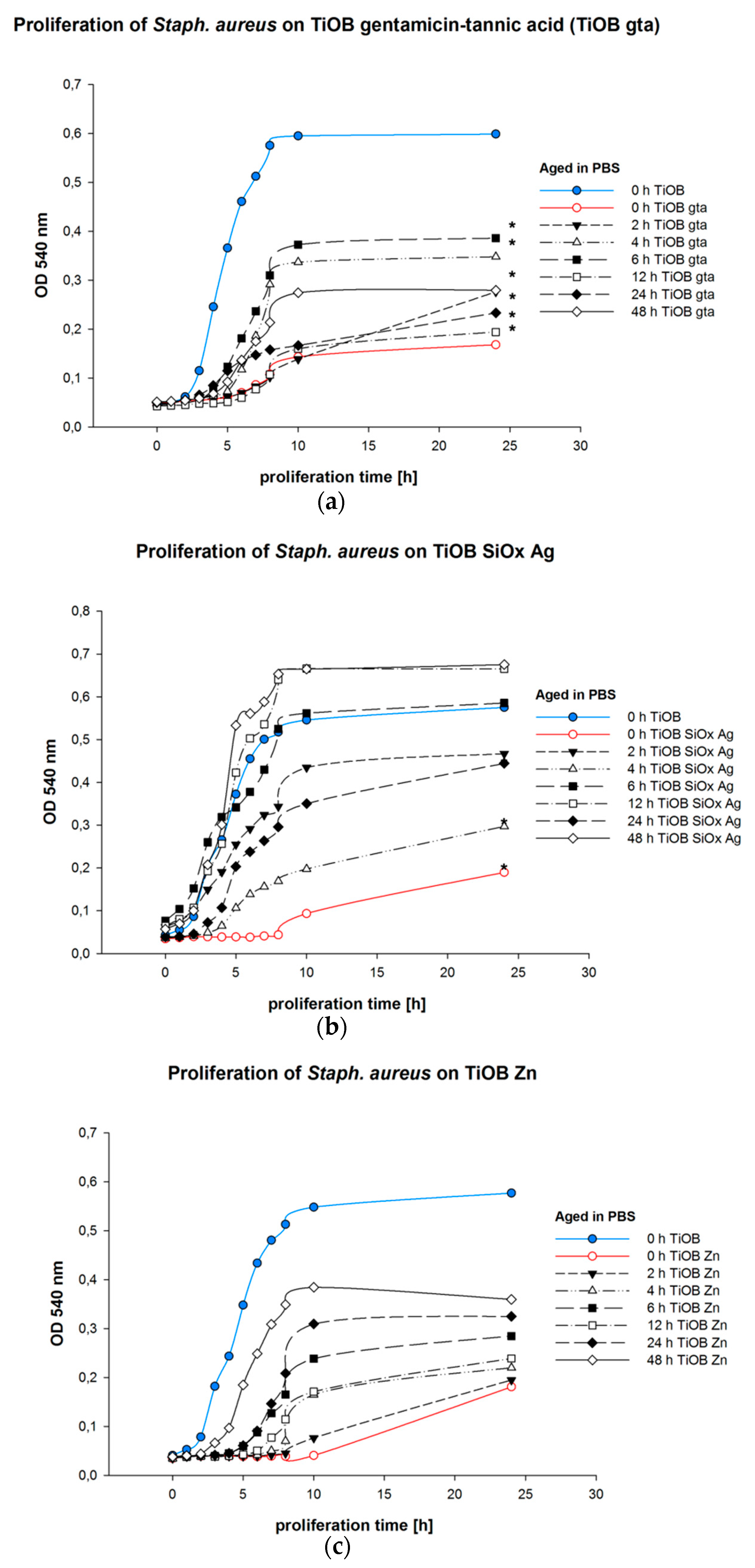
3.3. Bactericidal Properties of Functionalized TiOB®
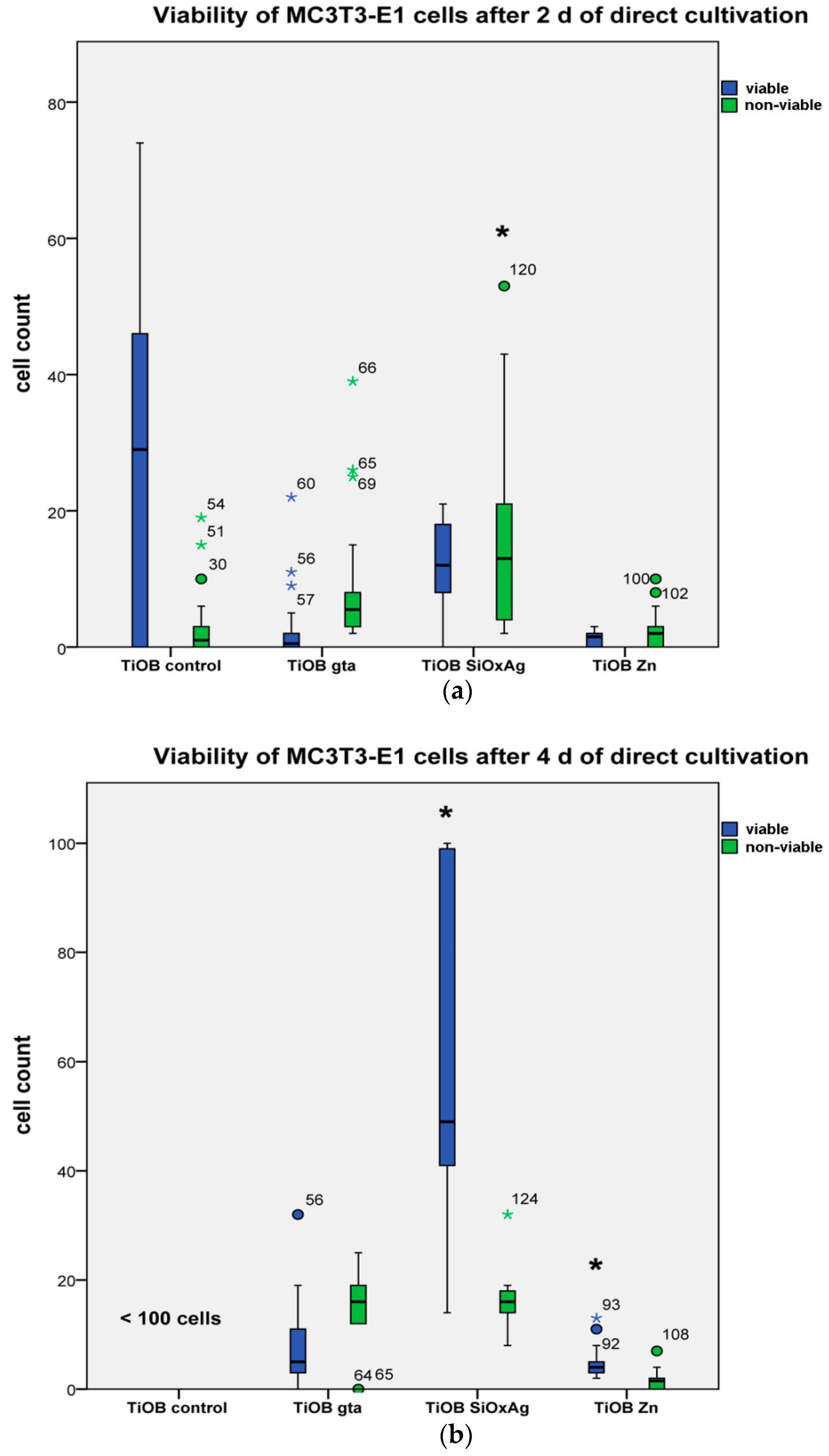
3.4. Biocompatible Properties of Functionalized TiOB®
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jager, M.; Jennissen, H.P.; Dittrich, F.; Fischer, A.; Kohling, H.L. Antimicrobial and osseointegration properties of nanostructured titanium orthopaedic implants. Materials 2017, 10, 1302. [Google Scholar] [CrossRef] [PubMed]
- Spriano, S.; Yamaguchi, S.; Baino, F.; Ferraris, S. A critical review of multifunctional titanium surfaces: New frontiers for improving osseointegration and host response, avoiding bacteria contamination. Acta Biomater. 2018, 79, 1–22. [Google Scholar] [CrossRef]
- Su, Y.; Luo, C.; Zhang, Z.; Hermawan, H.; Zhu, D.; Huang, J.; Liang, Y.; Li, G.; Ren, L. Bioinspired surface functionalization of metallic biomaterials. J. Mech. Behav. Biomed. Mater. 2018, 77, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Lugovskoy, A.; Lugovskoy, S. Production of hydroxyapatite layers on the plasma electrolytically oxidized surface of titanium alloys. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 43, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Fiorellini, J.P.; Glindmann, S.; Salcedo, J.; Weber, H.P.; Park, C.J.; Sarmiento, H.L. The effect of osteopontin and an osteopontin-derived synthetic peptide coating on osseointegration of implants in a canine model. Int. J. Periodontics Restor. Dent. 2016, 36, e88–e94. [Google Scholar] [CrossRef]
- Gao, Y.; Zou, S.; Liu, X.; Bao, C.; Hu, J. The effect of surface immobilized bisphosphonates on the fixation of hydroxyapatite-coated titanium implants in ovariectomized rats. Biomaterials 2009, 30, 1790–1796. [Google Scholar] [CrossRef] [PubMed]
- Diefenbeck, M.; Muckley, T.; Schrader, C.; Schmidt, J.; Zankovych, S.; Bossert, J.; Jandt, K.D.; Faucon, M.; Finger, U. The effect of plasma chemical oxidation of titanium alloy on bone-implant contact in rats. Biomaterials 2011, 32, 8041–8047. [Google Scholar] [CrossRef] [PubMed]
- Schrader, C.; Schmidt, J.; Diefenbeck, M.; Muckley, T.; Zankovych, S.; Bossert, J.; Jandt, K.D.; Faucon, M.; Finger, U. Bioactive tiob-coating on titanium alloy implants enhances osseointegration in a rat model. Adv. Eng. Mater. 2012, 14, B21–B27. [Google Scholar] [CrossRef]
- De Avila, E.D.; de Molon, R.S.; Vergani, C.E.; de Assis Mollo, F., Jr.; Salih, V. The relationship between biofilm and physical-chemical properties of implant abutment materials for successful dental implants. Materials 2014, 7, 3651–3662. [Google Scholar] [CrossRef]
- Dapunt, U.; Radzuweit-Mihaljevic, S.; Lehner, B.; Haensch, G.M.; Ewerbeck, V. Bacterial infection and implant loosening in hip and knee arthroplasty: Evaluation of 209 cases. Materials 2016, 9, 871. [Google Scholar] [CrossRef] [PubMed]
- Laffer, R.R.; Graber, P.; Ochsner, P.E.; Zimmerli, W. Outcome of prosthetic knee-associated infection: Evaluation of 40 consecutive episodes at a single centre. Clin. Microbio. Infect. Off. Publ. Eur. Soc. Clin. Microbio. Infect. Dis. 2006, 12, 433–439. [Google Scholar] [CrossRef]
- Diefenbeck, M.; Schrader, C.; Gras, F.; Muckley, T.; Schmidt, J.; Zankovych, S.; Bossert, J.; Jandt, K.D.; Volpel, A.; Sigusch, B.W.; et al. Gentamicin coating of plasma chemical oxidized titanium alloy prevents implant-related osteomyelitis in rats. Biomaterials 2016, 101, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Webster, T.J. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2018, 36, 22–32. [Google Scholar] [CrossRef]
- Radulescu, M.; Andronescu, E.; Dolete, G.; Popescu, R.C.; Fufa, O.; Chifiriuc, M.C.; Mogoanta, L.; Balseanu, T.A.; Mogosanu, G.D.; Grumezescu, A.M.; et al. Silver nanocoatings for reducing the exogenous microbial colonization of wound dressings. Materials 2016, 9, 345. [Google Scholar] [CrossRef] [PubMed]
- Bechert, T.; Boswald, M.; Lugauer, S.; Regenfus, A.; Greil, J.; Guggenbichler, J.P. The erlanger silver catheter: In vitro results for antimicrobial activity. Infection 1999, 27 (Suppl. 1), S24–S29. [Google Scholar] [CrossRef]
- Alt, V.; Bechert, T.; Steinrucke, P.; Wagener, M.; Seidel, P.; Dingeldein, E.; Domann, E.; Schnettler, R. An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement. Biomaterials 2004, 25, 4383–4391. [Google Scholar] [CrossRef]
- Mei, S.; Wang, H.; Wang, W.; Tong, L.; Pan, H.; Ruan, C.; Ma, Q.; Liu, M.; Yang, H.; Zhang, L.; et al. Antibacterial effects and biocompatibility of titanium surfaces with graded silver incorporation in titania nanotubes. Biomaterials 2014, 35, 4255–4265. [Google Scholar] [CrossRef]
- Toledano-Osorio, M.; Babu, J.P.; Osorio, R.; Medina-Castillo, A.L.; Garcia-Godoy, F.; Toledano, M. Modified polymeric nanoparticles exert in vitro antimicrobial activity against oral bacteria. Materials 2018, 11, 1013. [Google Scholar] [CrossRef]
- Liu, L.; Pushalkar, S.; Saxena, D.; LeGeros, R.Z.; Zhang, Y. Antibacterial property expressed by a novel calcium phosphate glass. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 423–429. [Google Scholar] [CrossRef]
- Yu, J.; Li, K.; Zheng, X.; He, D.; Ye, X.; Wang, M. In vitro and in vivo evaluation of zinc-modified ca-si-based ceramic coating for bone implants. PLoS ONE 2013, 8, e57564. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Low, H.T.; Kasiri-Asgarani, M.; Farahany, S.; Akbari, E.; Cho, M.H. Fabrication of biodegradable zn-al-mg alloy: Mechanical properties, corrosion behavior, cytotoxicity and antibacterial activities. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 73, 215–219. [Google Scholar] [CrossRef]
- Beier, O.; Pfuch, A.; Horn, K.; Weisser, J.; Schnabelrauch, M.; Schimanski, A. Low temperature deposition of antibacterially active silicon oxide layers containing silver nanoparticles, prepared by atmospheric pressure plasma chemical vapor deposition. Plasma Process. Polym. 2013, 10, 77–87. [Google Scholar] [CrossRef]
- Bechert, T.; Steinrucke, P.; Guggenbichler, J.P. A new method for screening anti-infective biomaterials. Nat. Med. 2000, 6, 1053–1056. [Google Scholar] [CrossRef]
- Lin, W.T.; Zhang, Y.Y.; Tan, H.L.; Ao, H.Y.; Duan, Z.L.; He, G.; Tang, T.T. Inhibited bacterial adhesion and biofilm formation on quaternized chitosan-loaded titania nanotubes with various diameters. Materials 2016, 9, 155. [Google Scholar] [CrossRef]
- Dapunt, U.; Hansch, G.M.; Arciola, C.R. Innate immune response in implant-associated infections: Neutrophils against biofilms. Materials 2016, 9, 387. [Google Scholar] [CrossRef]
- Alt, V. Antimicrobial coated implants in trauma and orthopaedics-a clinical review and risk-benefit analysis. Injury 2017, 48, 599–607. [Google Scholar] [CrossRef]
- Popat, K.C.; Eltgroth, M.; Latempa, T.J.; Grimes, C.A.; Desai, T.A. Decreased staphylococcus epidermis adhesion and increased osteoblast functionality on antibiotic-loaded titania nanotubes. Biomaterials 2007, 28, 4880–4888. [Google Scholar] [CrossRef]
- Babuska, V.; Palan, J.; Kolaja Dobra, J.; Kulda, V.; Duchek, M.; Cerny, J.; Hrusak, D. Proliferation of osteoblasts on laser-modified nanostructured titanium surfaces. Materials 2018, 11, 1827. [Google Scholar] [CrossRef]
- Sahiner, N.; Sagbas, S.; Aktas, N. Single step natural poly(tannic acid) particle preparation as multitalented biomaterial. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 49, 824–834. [Google Scholar] [CrossRef]
- Vester, H.; Wildemann, B.; Schmidmaier, G.; Stockle, U.; Lucke, M. Gentamycin delivered from a pdlla coating of metallic implants: In vivo and in vitro characterisation for local prophylaxis of implant-related osteomyelitis. Injury 2010, 41, 1053–1059. [Google Scholar] [CrossRef]
- Caldas, I.P.; Alves, G.G.; Barbosa, I.B.; Scelza, P.; de Noronha, F.; Scelza, M.Z. In vitro cytotoxicity of dental adhesives: A systematic review. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2019, 35, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Marinucci, L.; Balloni, S.; Bodo, M.; Carinci, F.; Pezzetti, F.; Stabellini, G.; Conte, C.; Lumare, E. Patterns of some extracellular matrix gene expression are similar in cells from cleft lip-palate patients and in human palatal fibroblasts exposed to diazepam in culture. Toxicology 2009, 257, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Wiesli, M.G.; Ozcan, M. High-performance polymers and their potential application as medical and oral implant materials: A review. Implant Dent. 2015, 24, 448–457. [Google Scholar] [CrossRef]
- Kheur, S.; Singh, N.; Bodas, D.; Rauch, J.Y.; Jambhekar, S.; Kheur, M.; Rajwade, J. Nanoscale silver depositions inhibit microbial colonization and improve biocompatibility of titanium abutments. Colloids Surf. B Biointerfaces 2017, 159, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Smeets, R.; Precht, C.; Hahn, M.; Jung, O.; Hartjen, P.; Heiland, M.; Grobe, A.; Holthaus, M.G.; Hanken, H. Biocompatibility and osseointegration of titanium implants with a silver-doped polysiloxane coating: An in vivo pig model. Int. J. Oral Maxillofac. Implant. 2017, 32, 1338–1345. [Google Scholar] [CrossRef] [PubMed]
- Lischer, S.; Korner, E.; Balazs, D.J.; Shen, D.; Wick, P.; Grieder, K.; Haas, D.; Heuberger, M.; Hegemann, D. Antibacterial burst-release from minimal ag-containing plasma polymer coatings. J. R. Soc. Interface 2011, 8, 1019–1030. [Google Scholar] [CrossRef]
- Van Hengel, I.A.J.; Riool, M.; Fratila-Apachitei, L.E.; Witte-Bouma, J.; Farrell, E.; Zadpoor, A.A.; Zaat, S.A.J.; Apachitei, I. Selective laser melting porous metallic implants with immobilized silver nanoparticles kill and prevent biofilm formation by methicillin-resistant staphylococcus aureus. Biomaterials 2017, 140, 1–15. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on escherichia coli and staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Jaiswal, S.; Duffy, B.; Jaiswal, A.K.; Stobie, N.; McHale, P. Enhancement of the antibacterial properties of silver nanoparticles using beta-cyclodextrin as a capping agent. Int. J. Antimicrob. Agent. 2010, 36, 280–283. [Google Scholar] [CrossRef]
- Dror-Ehre, A.; Mamane, H.; Belenkova, T.; Markovich, G.; Adin, A. Silver nanoparticle-e. Coli colloidal interaction in water and effect on e. Coli survival. J. Colloid Interface Sci. 2009, 339, 521–526. [Google Scholar] [CrossRef]
- Sondi, I.; Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: A case study on e. Coli as a model for gram-negative bacteria. J. Colloid Interface Sci. 2004, 275, 177–182. [Google Scholar] [CrossRef]
- Li, J.; Xie, B.; Xia, K.; Li, Y.; Han, J.; Zhao, C. Enhanced antibacterial activity of silver doped titanium dioxide-chitosan composites under visible light. Materials 2018, 11, 1403. [Google Scholar] [CrossRef]
- Deshmukh, S.P.; Mullani, S.B.; Koli, V.B.; Patil, S.M.; Kasabe, P.J.; Dandge, P.B.; Pawar, S.A.; Delekar, S.D. Ag nanoparticles connected to the surface of TiO2 electrostatically for antibacterial photoinactivation studies. Photochem. Photobiol. 2018, 94, 1249–1262. [Google Scholar] [CrossRef]
- Zhu, D.; Su, Y.; Young, M.L.; Ma, J.; Zheng, Y.; Tang, L. Biological responses and mechanisms of human bone marrow mesenchymal stem cells to zn and mg biomaterials. ACS Appl. Mater. Interfaces 2017, 9, 27453–27461. [Google Scholar] [CrossRef]
- Yu, H.; Huang, X.; Yang, X.; Liu, H.; Zhang, M.; Zhang, X.; Hang, R.; Tang, B. Synthesis and biological properties of zn-incorporated micro/nano-textured surface on ti by high current anodization. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 78, 175–184. [Google Scholar] [CrossRef]
- Shen, X.; Hu, Y.; Xu, G.; Chen, W.; Xu, K.; Ran, Q.; Ma, P.; Zhang, Y.; Li, J.; Cai, K. Regulation of the biological functions of osteoblasts and bone formation by zn-incorporated coating on microrough titanium. ACS Appl. Mater. Interfaces 2014, 6, 16426–16440. [Google Scholar] [CrossRef]
- Pagano, S.; Chieruzzi, M.; Balloni, S.; Lombardo, G.; Torre, L.; Bodo, M.; Cianetti, S.; Marinucci, L. Biological, thermal and mechanical characterization of modified glass ionomer cements: The role of nanohydroxyapatite, ciprofloxacin and zinc l-carnosine. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 94, 76–85. [Google Scholar] [CrossRef]
- Fiedot-Tobola, M.; Ciesielska, M.; Maliszewska, I.; Rac-Rumijowska, O.; Suchorska-Wozniak, P.; Teterycz, H.; Bryjak, M. Deposition of zinc oxide on different polymer textiles and their antibacterial properties. Materials 2018, 11, 707. [Google Scholar] [CrossRef]
- Xie, Y.; He, Y.; Irwin, P.L.; Jin, T.; Shi, X. Antibacterial activity and mechanism of action of zinc oxide nanoparticles against campylobacter jejuni. Appl. Environ. Microbiol. 2011, 77, 2325–2331. [Google Scholar] [CrossRef]




| Group Assignment | Technical Information |
|---|---|
| TiOB control | Ti6Al4V, PCO (280 V), TiOB surface |
| TiOB gentamicin-tannic acid (TiOB gta) | Ti6Al4V, PCO (280 V), TiOB surface, dip coating |
| TiOB SiOx Ag | Ti6Al4V, PCO (280 V), TiOB surface, APCVD |
| TiOB Zn | Ti6Al4V, 1st PCO (200 V), TiOB surface, 2nd PCO (350 V), Zn electrolyte |
| Agar Diffusion Test- - | ||||||||
|---|---|---|---|---|---|---|---|---|
| Eluates Collecting Time | ||||||||
| 0 h | 2 h | 4 h | 6 h | 12 h | 24 h | 48 h | ||
| TiOB® control | 0 | 0 | 0 | 0 | 0 | 0 | 0 | mean inhibition zones [mm] |
| TiOB® gta | 0 | 1.94 | 1.88 | 0.50 | 0.94 | 1.50 | 1.25 | |
| TiOB8® SiOx Ag | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| TiOB® Zn | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kranz, S.; Guellmar, A.; Voelpel, A.; Lesser, T.; Tonndorf-Martini, S.; Schmidt, J.; Schrader, C.; Faucon, M.; Finger, U.; Pfister, W.; et al. Bactericidal and Biocompatible Properties of Plasma Chemical Oxidized Titanium (TiOB®) with Antimicrobial Surface Functionalization. Materials 2019, 12, 866. https://doi.org/10.3390/ma12060866
Kranz S, Guellmar A, Voelpel A, Lesser T, Tonndorf-Martini S, Schmidt J, Schrader C, Faucon M, Finger U, Pfister W, et al. Bactericidal and Biocompatible Properties of Plasma Chemical Oxidized Titanium (TiOB®) with Antimicrobial Surface Functionalization. Materials. 2019; 12(6):866. https://doi.org/10.3390/ma12060866
Chicago/Turabian StyleKranz, Stefan, André Guellmar, Andrea Voelpel, Tobias Lesser, Silke Tonndorf-Martini, Juergen Schmidt, Christian Schrader, Mathilde Faucon, Ulrich Finger, Wolfgang Pfister, and et al. 2019. "Bactericidal and Biocompatible Properties of Plasma Chemical Oxidized Titanium (TiOB®) with Antimicrobial Surface Functionalization" Materials 12, no. 6: 866. https://doi.org/10.3390/ma12060866
APA StyleKranz, S., Guellmar, A., Voelpel, A., Lesser, T., Tonndorf-Martini, S., Schmidt, J., Schrader, C., Faucon, M., Finger, U., Pfister, W., Diefenbeck, M., & Sigusch, B. (2019). Bactericidal and Biocompatible Properties of Plasma Chemical Oxidized Titanium (TiOB®) with Antimicrobial Surface Functionalization. Materials, 12(6), 866. https://doi.org/10.3390/ma12060866






