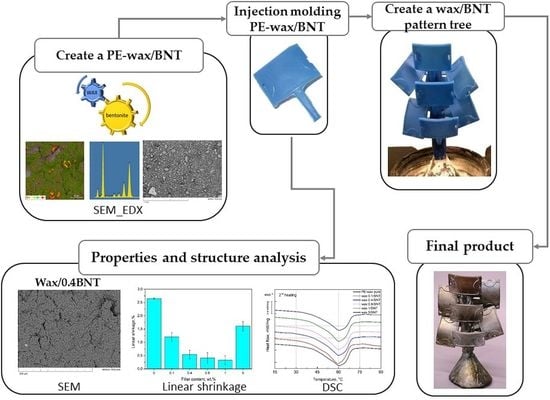
Polyethylene Wax Modified by Organoclay Bentonite Used in the Lost-Wax Casting Process: Processing−Structure−Property Relationships
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of the Wax/Bentonite Blends
2.3. Preparation of the Aluminum Cast
3. Characterization Methods
3.1. Viscosity and Density of Wax/Bentonite Blends
3.2. Linear Shrinkage of Wax/Bentonite Blends
3.3. Thermal Properties (DSC) of Wax/Bentonite Blends
3.4. Mechanical Properties (Shore D Hardness)
3.5. Scanning Electron Microscope (SEM)
3.6. Fourier Transform Infrared (FT-IR) Spectroscopy
4. Results and Discussion
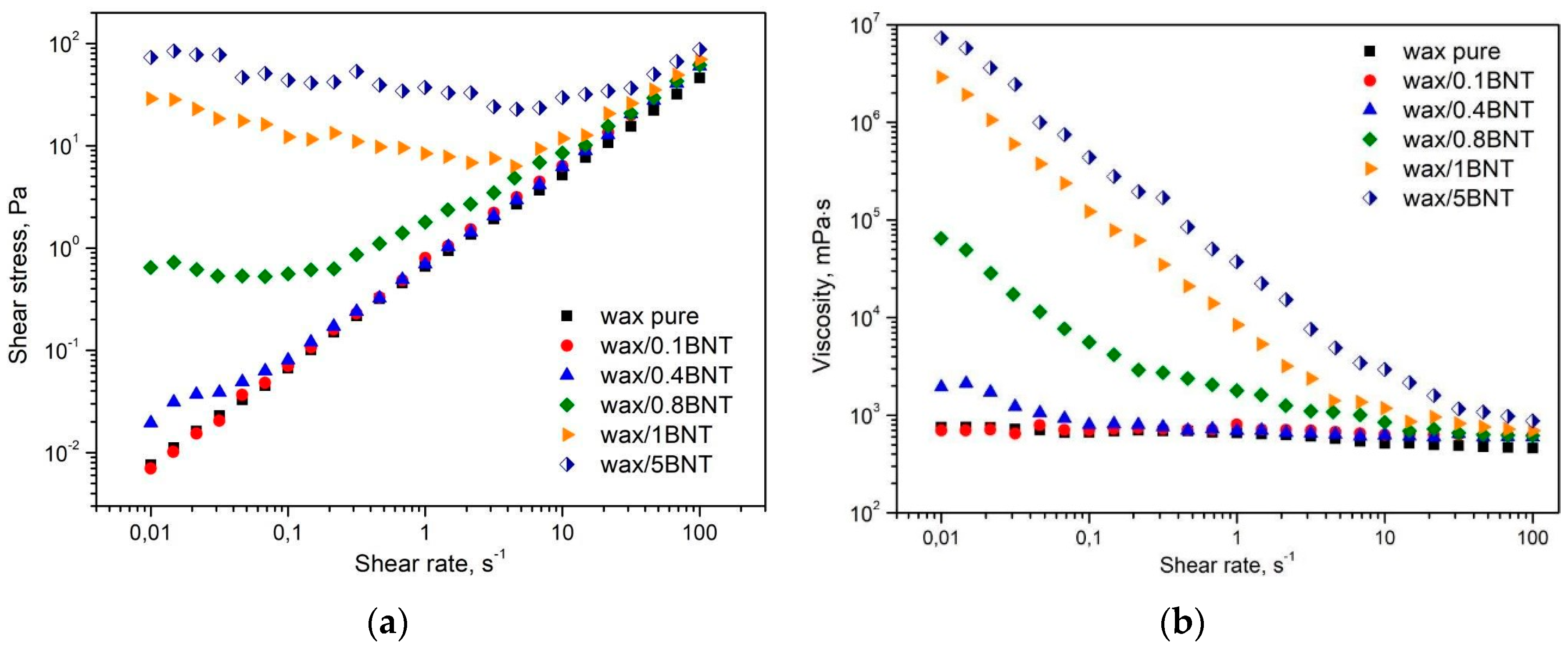
4.1. Effect of the Bentonite on Pure PE-Wax Rheological Behavior
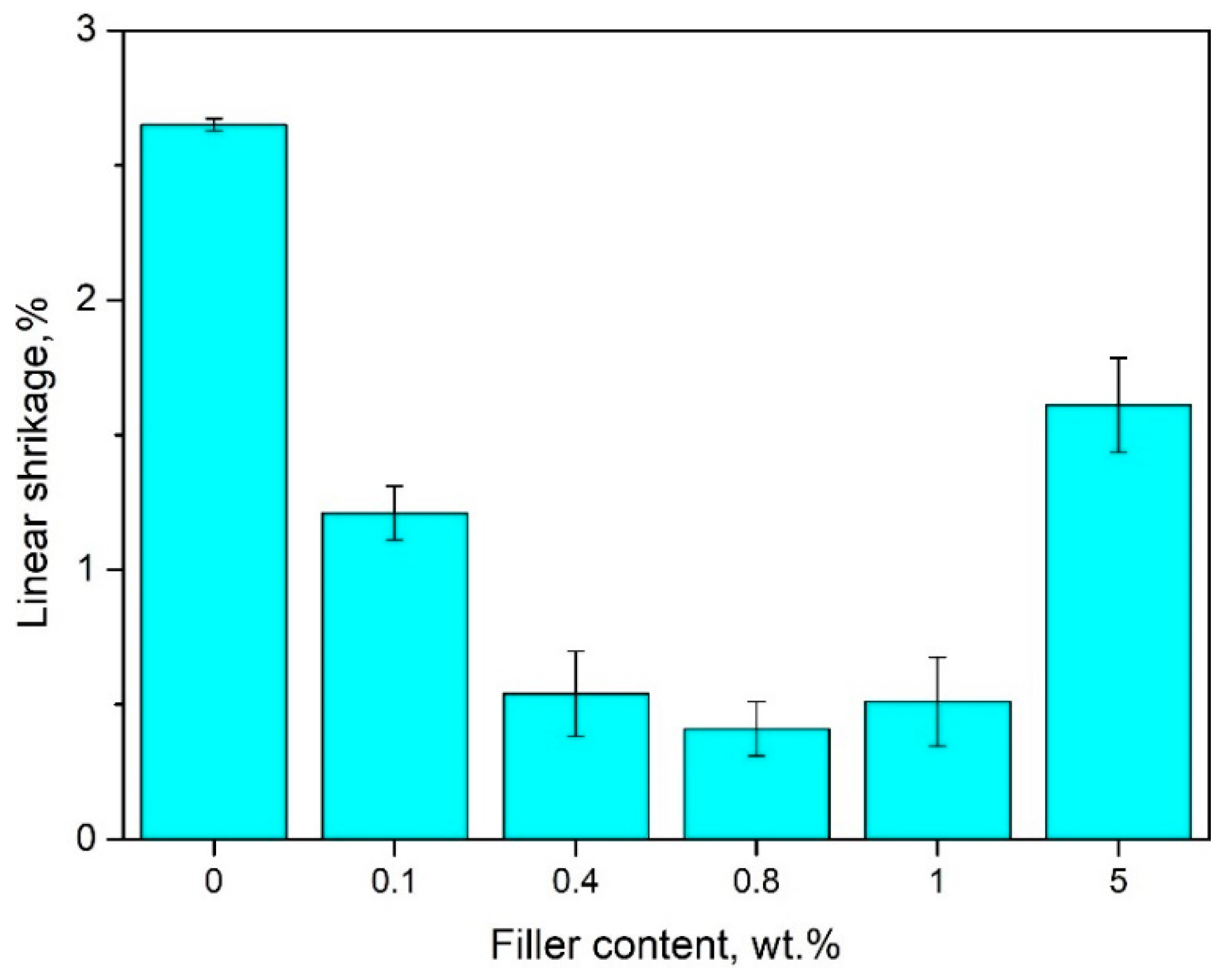
4.2. The Effect of Bentonite on Linear Shrinkage of PE-Wax
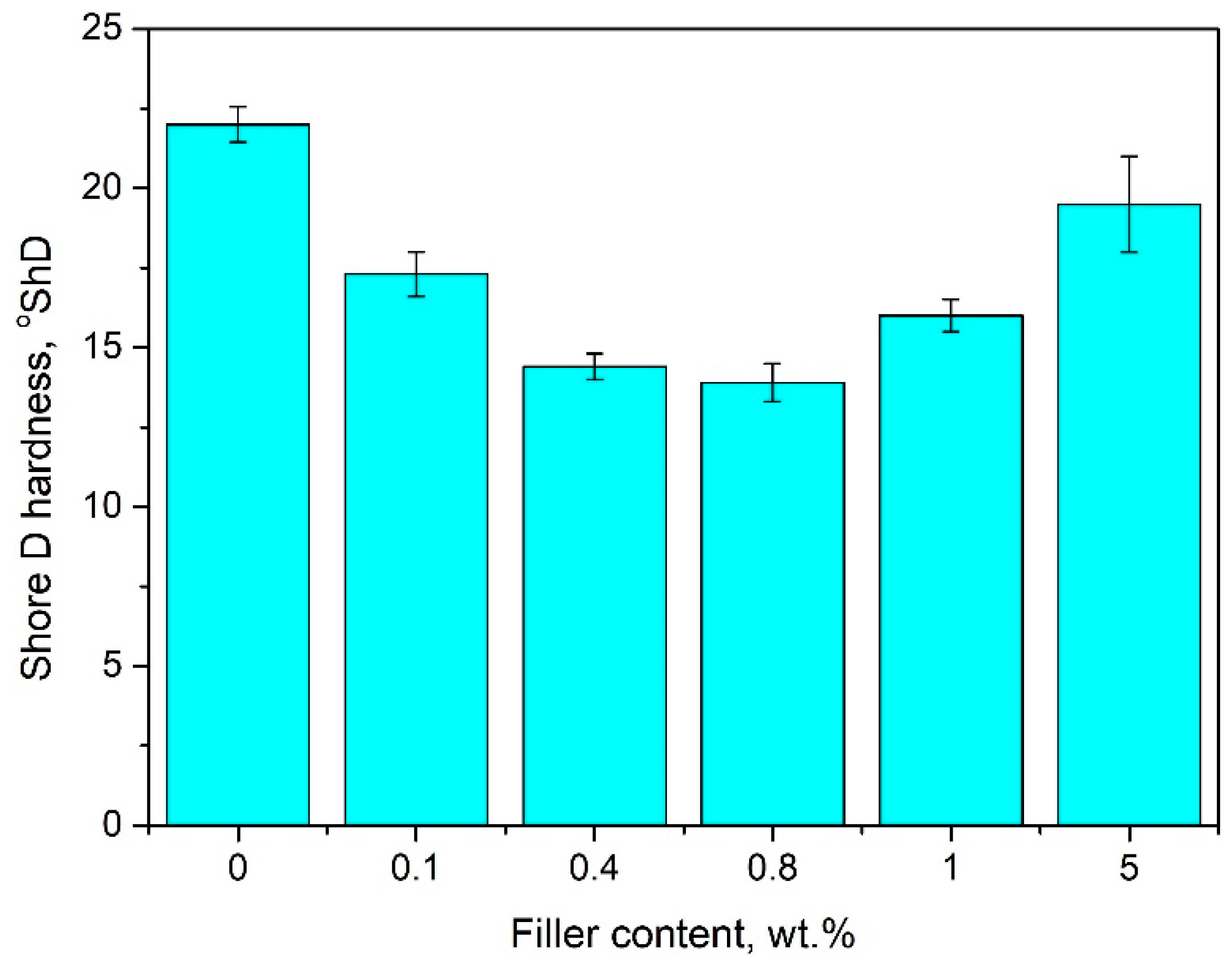
4.3. The Effect of Bentonite Content on the Hardness of Pure PE-Wax
4.4. The Effect of Bentonite Content on the PE-Wax Thermal Properties
4.5. The Effect of Bentonite on the Morphology PE Wax/BNT Blends
4.6. FT-IR Characterization of Pure PE-Wax Filled With Bentonite
4.7. Assessment Casting Produced by the Lost-Wax Method
5. Conclusions
- Bentonite is an effective modifier of linear shrinkage, hardness, and crystallinity in addition to polyethylene wax, when used in lost-wax casting, as distinguished by the lack of sedimentation of the filler from polyethylene wax.
- Modification with bentonite leads to a reduction of linear shrinkage by approximately 90% and hardness by 30% when compared to pure PE-wax, and, thus, improves the dimensional accuracy of wax models and the casting process.
- The new material, based on polyethylene wax modified with bentonite ranging from 0.1 to 0.4 wt.%, results in reduced shrinkage and only a slight increase in viscosity, which, thus, suits it perfectly to lost-wax casting.
- Aluminum alloy castings with excellent surface quality were obtained, as confirmed by macroscopic research based on observing the casting surface with the naked eye or using a magnifying glass. These castings were characterized by a smooth surface. There were no defects in the shape or raw surface, or breaks in continuity. Furthermore, no internal defects such as systolic cavities, scabs, or blisters were recorded.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Harabasz, M. Badania Kinetyki Skurczu Tworzyw Stosowanych Na Modele w Technologii Wytapianych Modeli. Master’s Thesis, AGH University of Science and Technology, Kraków, Poland, 2011. [Google Scholar]
- Stachańczyk, J. Własności Charakteryzujące Masy Modelowe—Proces Kształtowania i Jakości Modeli Wytapianych. Ph.D. Thesis, Warsaw University of Technology, Warsaw, Poland, 2008. [Google Scholar]
- Vasková, I.; Fecko, D.; Malik, J. The Dependence of Castings Quality Produced in Clay Moulding Mixtures from the Properties of Binder Based on a Montmorillonite. Arch. Foundry Eng. 2012, 12, 123–128. [Google Scholar] [CrossRef]
- Zych, J.; Kolczyk, J.; Snopkiewicz, T. Investigations of properties of wax mixtures used in the investment casting technology—New investigation methods. Arch. Foundry Eng. 2012, 12, 199–204. [Google Scholar] [CrossRef]
- Craig, R.G.; Powers, J.M. Restorative Dental Materials, 11th ed.; Mosby Co: St Louis, MO, USA, 2002; pp. 423–448. [Google Scholar]
- Levine, M.N. Ceramic Molds for Mixtec Gold: A New Lost-Wax Casting Technique from Prehispanic Mexico. J. Archaeol. Method Theory 2019, 26, 423–456. [Google Scholar] [CrossRef]
- Smith, A.; Heckelman, P.; Budavari, S.; Obenchain, J.; O’Neil, M. The Merck Index, 12th ed.; Elsevier Health Sciences: London, UK, 1996; pp. 170–171. ISBN 978-09-1191-012-4. [Google Scholar]
- Thoury, M.; Mille, B.; Séverin-Fabiani, T.; Robbiola, L.; Refregiers, M.; Jarrige, J.-F.; Bertrand, L. High spatial dynamics-photoluminescence imaging reveals the metallurgy of the earliest lost-wax cast object. Nat. Commun. 2016, 7, 13356. [Google Scholar] [CrossRef]
- Yuan, C.; Jones, S.; Blackburn, S. Development of a New Ferrous Aluminosilicate Refractory Material for Investment Casting of Aluminum Alloys. Metall. Mater. Trans. A 2012, 43, 5232–5242. [Google Scholar] [CrossRef]
- Wen, J.; Xie, Z.; Cao, W.; Yang, X. Effects of different backbone binders on the characteristics of zirconia parts using wax-based binder system via ceramic injection molding. J. Adv. Ceram. 2016, 5, 321–328. [Google Scholar] [CrossRef]
- Wolff, T.M. Influences which eliminate wax pattern defects. Incast 1999, 12, 22–24. [Google Scholar]
- Marszałek, G.; Majczak, R. Wosk polietylenowy—Otrzymywanie, modyfikacja i zastosowania. Polimery 2012, 57, 640–645. [Google Scholar]
- Chakravorty, S. The Properties of Waxes Used in the Investment Casting Industry: Final Report; NPL Report; CMMT(A)245: Middlesex, UK, 1999; ISSN 1361-4061. [Google Scholar]
- Muschio, H.M. Filler and Wax Composition for Investment Casting. U.S. Patent US5518537A, 21 May 1995. [Google Scholar]
- Widjijono, W.; Agustiono, P.; Irnawati, D. Mechanical properties of carving wax with various Ca-bentonite filter composition. Dent. J. 2009, 42, 114–117. [Google Scholar]
- Bemblage, O.; Karunakar, B.D. A study on the blended wax patterns in investment casting process. In Proceedings of the World Congress on Engineering, London, UK, 6–8 July 2011; Volume 1, ISSN 2078-0966. [Google Scholar]
- Rutto, H.K. Urea-Based Moulding Compounds for Investment Casting. Ph.D. Thesis, University of Pretoria, Pretoria, South Africa, 2006. [Google Scholar]
- Okhuysen, V.F.; Padmanabhan, K.; Voigt, R.C. Tooling allowance practices in investment casting industry. In Proceedings of the 46th Annual Technical Meeting of the Investment Casting Institute, Orlando, FL, USA, 11–14 October 1998. [Google Scholar]
- Gebelin, J.-C.; Jolly, M.R. Modeling of the investment casting process. J. Mater. Process. Tech. 2003, 135, 291–300. [Google Scholar] [CrossRef]
- Bonilla, W.; Masood, S.H.; Iovenitti, P. An investigation of wax patterns for accuracy improvement in investment casting parts. Int. J. Adv. Manuf. Tech. 2001, 18, 348–356. [Google Scholar] [CrossRef]
- Singh, B.; Kumar, P.; Mishra, B.K. Optimization of injection parameters for making wax patterns to be used in ceramic shell investment casting. In Proceedings of the Asian Symposium on Materials and Processing, Bangkok, Thailand, 9–10 November 2006. [Google Scholar] [CrossRef]
- Rezavand, S.A.M.; Behravesh, A.H. An experimental investigation on dimensional stability of injected wax patterns of gas turbine blades. J. Mater. Process. Tech. 2007, 182, 580–587. [Google Scholar] [CrossRef]
- Horacek, M.; Lubos, S. Influence of injection parameters on the dimensional stability of wax patterns. In Proceedings of the Ninth World Conference on Investment Casting, San Francisco, CA, USA, 13–16 October 1996; pp. 1–20. [Google Scholar]
- Kotsiomiti, E.; McCabe, J.F. Experimental wax mixtures for dental use. J. Oral Rehabil. 1997, 24, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Argueso, J.P. Frequently Encountered Problems in a wax room. Mod. Cast. 1993, 83, 32–33. [Google Scholar]
- Aziz, M.N.; Munyensanga, P.; Widyanto, S.A. Application of lost wax casting for manufacturing of orthopedic screw: A review. Procedia CIRP 2018, 78, 149–154. [Google Scholar] [CrossRef]
- Widjijono, W. Correlation between calcium bentonite concentration and paraffin wax to the hardness of carving wax. Dentika Dent. J. 2009, 14, 20–23. [Google Scholar]
- Rydzkowski, T.; Reszka, K.; Szczypiński, M.; Szczypiński, M.M.; Kopczyńska, E.; Thakur, V.K. Manufacturing and Evaluation of Mechanical, Morphological, and Thermal Properties of Reduced Graphene Oxide-Reinforced Expanded Polystyrene (EPS) Nanocomposites. Adv. Polym. Technol. 2020, in press. [Google Scholar] [CrossRef]
- Sutherland, W.M. Wyoming Bentonite—Summary Report: Wyoming State Geological Survey Summary Report. September 2014; 4p. Available online: https://www.wsgs.wyo.gov/products/wsgs-2014-bentonite-summary.pdf (accessed on 2 March 2020).
- Czarnecka-Komorowska, D.; Tomczyk, T. Influence of montmorillonite (MMT) on the mechanical properties in recycled rubber. Arch. Mech. Technol. Autom. 2008, 28, 145–152. [Google Scholar]
- Oleksy, M.; Oliwa, J.; Budzik, G.; Oliwa, R.; Bulanda, K.; Nędzna, M. The use of a mixer with built-in Parshall’s venturi for modification of bentonite designed for the filling of polymer resins. Polimery 2019, 7–8, 499–503. [Google Scholar] [CrossRef]
- Oleksy, M.; Szwarc-Rzepka, K.; Heneczkowski, M.; Oliwa, R.; Jesionowski, T. Epoxy Resin Composite Based on Functional Hybrid Fillers. Materials 2014, 7, 6064–6091. [Google Scholar] [CrossRef]
- Othman, N.; Ismail, H.; Mariatti, M. Effect of compatibilizers on mechanical and thermal properties of bentonite filled polypropylene composites. Polym. Degrad. Stabil. 2006, 91, 1761–1774. [Google Scholar] [CrossRef]
- Piszczyk, Ł.; Hejna, A.; Formela, K.; Danowska, M.; Strankowski, M. Morphology, mechanical and thermal properties of flexible polyurethane foams modified with layered aluminosilicates. Polimery 2014, 11–12, 783–791. [Google Scholar] [CrossRef]
- Wu, J.; Wei, Y.; Lin, J.; Lin, S. Study on starch-graft-acrylamide/mineral powder superabsorbent composite. Polymer 2003, 44, 6513–6520. [Google Scholar] [CrossRef]
- Seyidoglu, T.; Yilmazer, U. Production of modified clays and their use in polypropylene-based nanocomposites. J. Appl. Polym. Sci. 2013, 127, 1257–1267. [Google Scholar] [CrossRef]
- Solomon, M.J.; Almusallam, A.S.; Seefeldt, K.F.; Somwangthanaroj, A.; Varadan, P. Rheology of Polypropylene/Clay Hybrid Materials. Macromolecules 2001, 34, 1864–1872. [Google Scholar] [CrossRef]
- Ruwoldt, J.; Humborstad Sørland, G.; Simon, S.; Oschmann, H.-J.; Sjöblom, J. Inhibitor-wax interactions and PPD effect on wax crystallization: New approaches for GC/MS and NMR, and comparison with DSC, CPM, and rheometry. J. Petrol. Sci. Eng. 2019, 177, 53–68. [Google Scholar] [CrossRef]
- Dentas Neto, A.A.; Gomes, E.A.S.; Barros Neto, E.L.; Dantas, T.N.C.; Moura, C.P.A.M. Determination of wax appearance temperature (WAT) in paraffin/solvent systems by photoelectric signal and viscosimetry. Braz. J. Petrol. Gas 2009, 3, 149–157. [Google Scholar]
- Edwards, R.T. Crystal Habit of Paraffin Wax. Ind. Eng. Chem. 1957, 49, 750–757. [Google Scholar] [CrossRef]
- Simnofske, D.; Mollenhauer, K. Effect of wax crystallization on complex modulus of modified bitumen after varied temperature conditioning rates. IOP Conf. Ser.-Mat. Sci. 2017, 236, 012003. [Google Scholar] [CrossRef]
- Krawiec, P.; Różański, L.; Czarnecka-Komorowska, D.; Warguła, Ł. Evaluation of the Thermal Stability and Surface Characteristics of Thermoplastic Polyurethane V-Belt. Materials 2020, 13, 1502. [Google Scholar] [CrossRef]
- Jakubowska, P.; Sterzyński, T.; Samujło, B. Rheological studies of highly-filled polyolefin composites taking into consideration p-v-T characteristics. Polimery 2010, 55, 379–389. [Google Scholar] [CrossRef]
- Freeman Manufacturing & Supply Company Home Page. Freeman_Catalogue.Pdf. Available online: http://www.freemansupply.com (accessed on 1 March 2020).
- PN-EN ISO 1183-1. Plastics—Methods for Determining the Density of Non-Porous Plastics—Part 1: Immersion Method, Liquid Pycnometer Method and Titration Method; Polish Committee for Standardization: Warsaw, Poland, 2005. [Google Scholar]
- PN-EN ISO 2577. Plastics—Thermosetting Moulding Materials—Determination of Shrinkage; Polish Committee for Standardization: Warsaw, Poland, 2007. [Google Scholar]
- Wunderlich, B. Macromolecular Physics, Volume 2: Crystal Nucleation, Growth, Annealing; Academic Press: New York, NY, USA, 1976; ISBN 978-0-12-765602-1. [Google Scholar]
- PN-EN ISO 868. Plastics and Ebonite—Determination of Indentation Hardness by Means of a Durometer (Shore Hardness); Polish Committee for Standardization: Warsaw, Poland, 2003. [Google Scholar]
- Kurzbeck, S.; Kaschta, J.; Münstedt, H. Rheological behaviour of a filled wax system. Rheol. Acta 1996, 35, 446–457. [Google Scholar] [CrossRef]
- Grzeskowiak, K.; Czarnecka-Komorowska, D.; Sytek, K.; Wojciechowski, M. Influence of waxes remelting used in investment casting on their thermal properties and linear shrinkage. Metalurgija 2015, 54, 350–352. [Google Scholar]
- Fisher, J.M. Handbook of Molded Part Shrinkage and Warpage, 1st ed.; William Andrew Inc.: Norwich, UK, 2003; ISBN 978-1-884207-72-3. [Google Scholar]
- ASM International. Casting Design and Performance; ASM International: Materials Park, OH, USA, 2009; p. 34. ISBN 978-0-87170-724-6. [Google Scholar]
- Baran, P.; Cholewa, M.; Kamińska, K.; Trzeciak, J. An influence of bentonite addition on permeability coefficient of fly ash. Ecol. Eng. 2017, 18, 32–39. [Google Scholar] [CrossRef]
- Gawdzińska, K.; Chybowski, L.; Przetakiewicz, W.; Laskowski, R. Application of FMEA in the Quality Estimation of Metal Matrix Composite Castings Produced by Squeeze Infiltration. Arch. Metall. Mater. 2017, 62, 2171–2182. [Google Scholar] [CrossRef]
- Krupa, I.; Luyt, A.S. Thermal and mechanical properties of extruded LLDPE/wax blends. Poly. Degrad. Stab. 2001, 73, 157–161. [Google Scholar] [CrossRef]
- Czarnecka-Komorowska, D.; Sterzyński, T. Effect of Polyhedral Oligomeric Silsesquioxane on the Melting, Structure, and Mechanical Behavior of Polyoxymethylene. Polymers 2018, 10, 203. [Google Scholar] [CrossRef]
- Chrissopoulou, K.; Anastasiadis, S.H. Polyolefin/layered silicate nanocomposites with functional compatibilizers. Eur. Polym. J. 2011, 47, 600–613. [Google Scholar] [CrossRef]
- Czarnecka-Komorowska, D.; Sterzyński, T.; Dutkiewicz, M. Polyhedral oligomeric silsesquioxanes as modifiers of polyoxymethylene structure. AIP Conf. Proc. 2015, 1695, 020013. [Google Scholar]
- Czarnecka-Komorowska, D.; Sterzyński, T.; Dutkiewicz, M. Polyoxymethylene/polyhedral oligomeric silsesquioxane composites: Processing, crystallization, morphology and thermo-mechanical behavior. Int. Polym. Process. 2016, 31, 598–606. [Google Scholar] [CrossRef]
- ThermoFisher Scientific. Available online: https://blog.phenom-world.com/sem-electrons (accessed on 2 March 2020).
- de Oliveira, T.A.; de Oliveira Mota, I.; Carvalho, L.H.; Barbosa, R.; Alves, T.S. Influence of Carnauba Wax on Films of Poly (Butylene Adipate Co-Terephthalate) and Sugarcane Residue for Application in Soil Cover (Mulching). Mat. Res. 2019, 22 (Suppl. S1), e20190040. [Google Scholar] [CrossRef]
- Lozhechnikova, A.; Bellanger, H.; Michen, B.; Burgert, I.; Österberg, M. Surfactant-free carnauba wax dispersion and its use for layer-by-layer assembled protective surface coatings on wood. Appl. Surf. Sci. 2017, 396, 1273–1281. [Google Scholar] [CrossRef]
- Bonilla-Blancas, A.E.; Romero-Ibarra, I.C.; Vazquez-Arenas, J.; Sanchez-Solis, A.; Manero, O.; Alvarez-Ramirez, J. Molecular interactions arising in polyethylene-bentonite nanocomposites. J. Appl. Polym. Sci. 2019, 136, 46920. [Google Scholar] [CrossRef]
- Hristodor, C.-M.; Vrinceanu, N.; Pui, A.; Novac, O.; Copcia, V.E.; Popovici, E. Textural and morphological characterization of chitosan/bentonite nanocomposite. Environ. Eng. Manag. J. 2012, 11, 573–578. [Google Scholar] [CrossRef]
- Mansor, B.A.; Kamyar, S.; Majid, D.; Yunus, W.M.Z.W.; Nor, A.I. Synthesis and Characterization of Silver/Clay/Chitosan Bionanocomposites by UV Irradiation Method. Am. J. Appl. Sci. 2009, 6, 2030–2035. [Google Scholar]
- Campbell, J. Castings; Butterworth-Heinemann: Oxford, UK, 2003; ISBN 978-0-7506-4790-8. [Google Scholar]
- Hajkowski, J.; Popielarski, P.; Sika, R. Prediction of HPDC Casting Properties Made of Al-Si9Cu3 Alloy. In Advances in Manufacturing, Lecture Notes in Mechanical Engineering; Harmol, A., Ciszak, O., Legutko, S., Jurczyk, M., Eds.; Springer: Cham, Switzerland, 2018; pp. 621–631. [Google Scholar]
- Ignaszak, Z.; Popielarski, P.; Hajkowski, J.; Prunier, J.B. Problem of Acceptability of Internal Porosity in Semi-Finished Cast Product as New Trend “Tolerance of Damage” Present in Modern Design Office. Defect Diffus. Forum 2012, 326, 612–619. [Google Scholar] [CrossRef]
- Ratajczak, M.; Ptak, M.; Chybowski, L.; Gawdzińska, K.; Będziński, R. Material and Structural Modeling Aspects of Brain Tissue Deformation under Dynamic Loads. Materials 2019, 12, 271. [Google Scholar] [CrossRef]



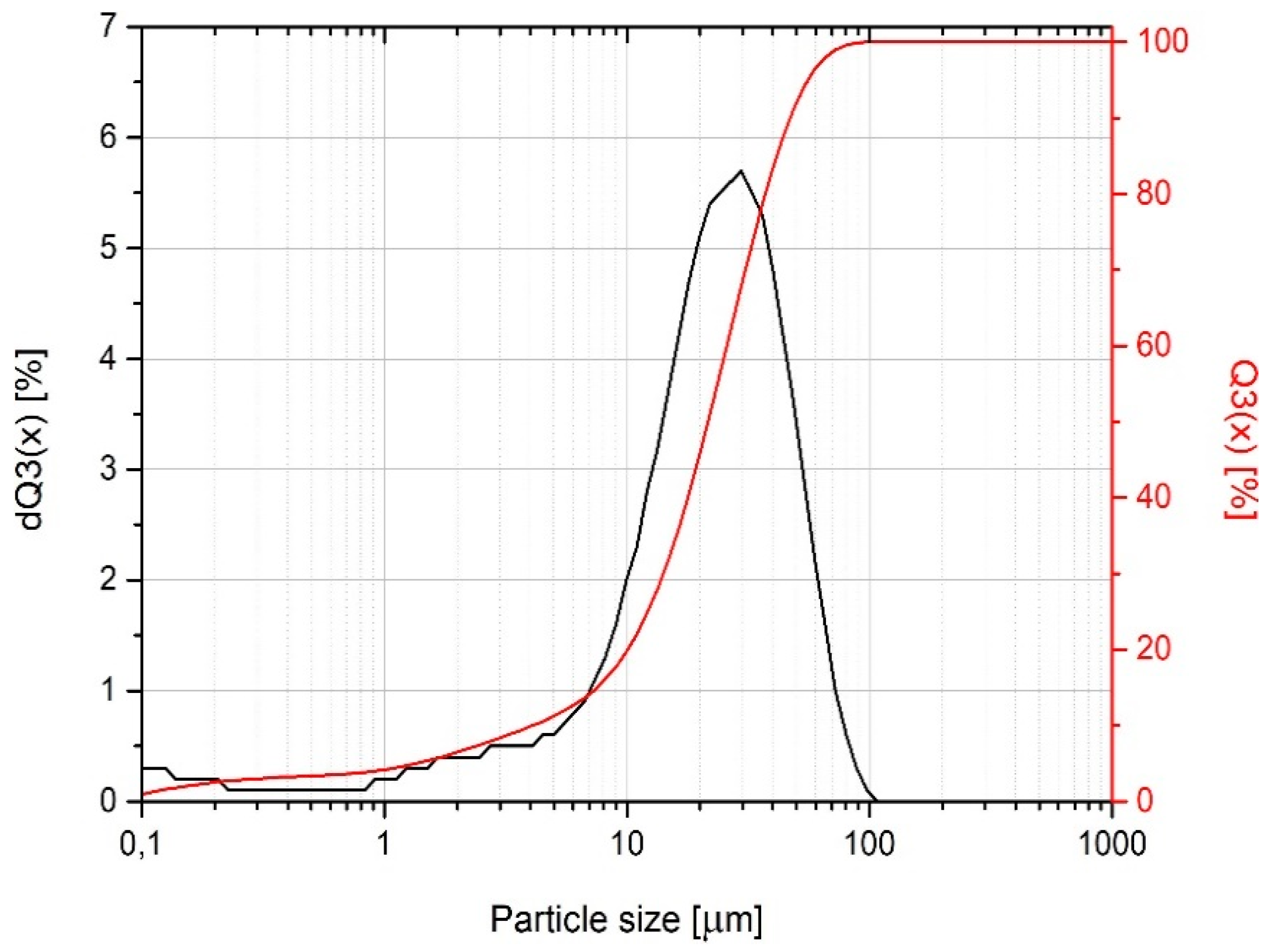

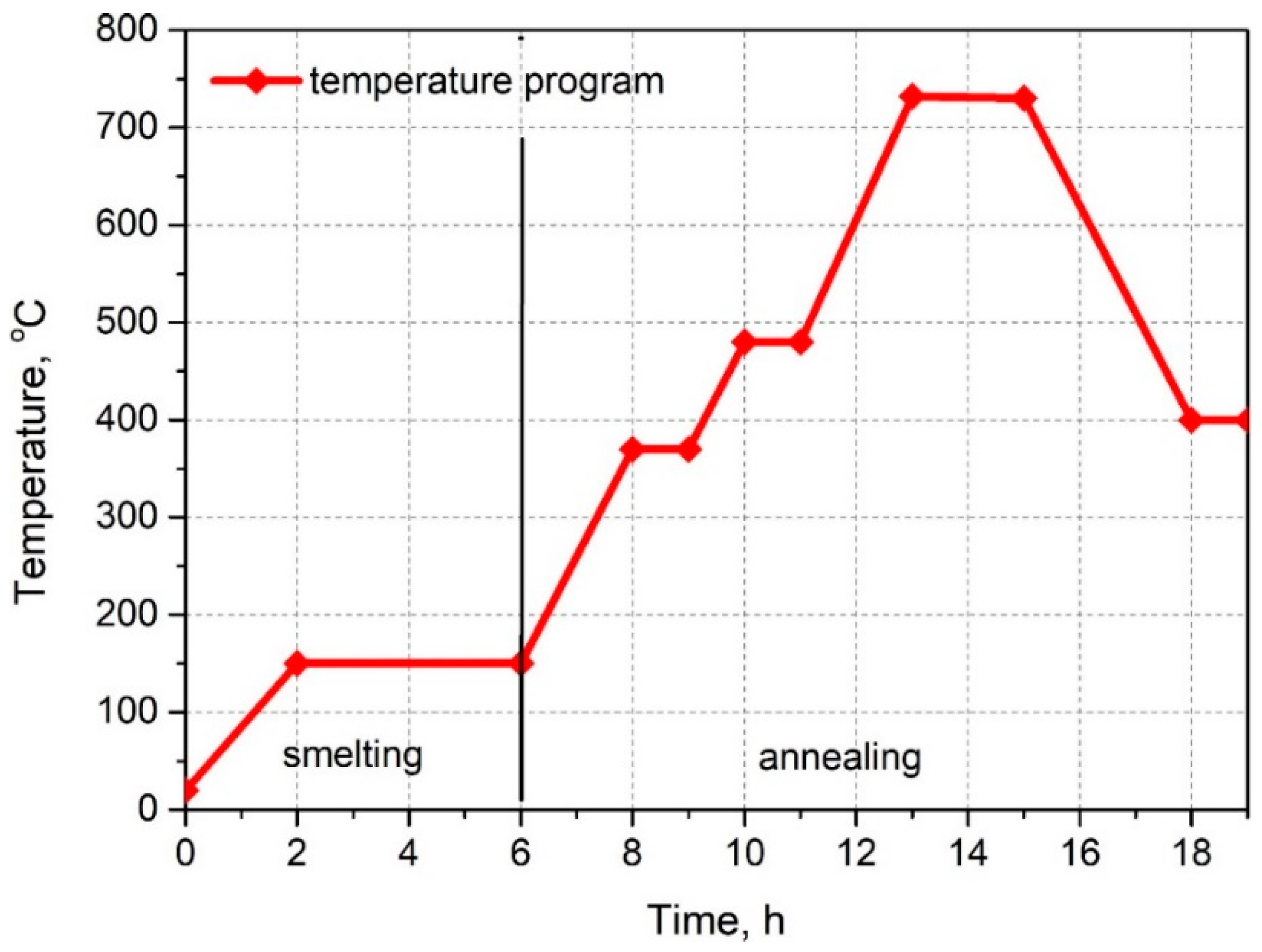
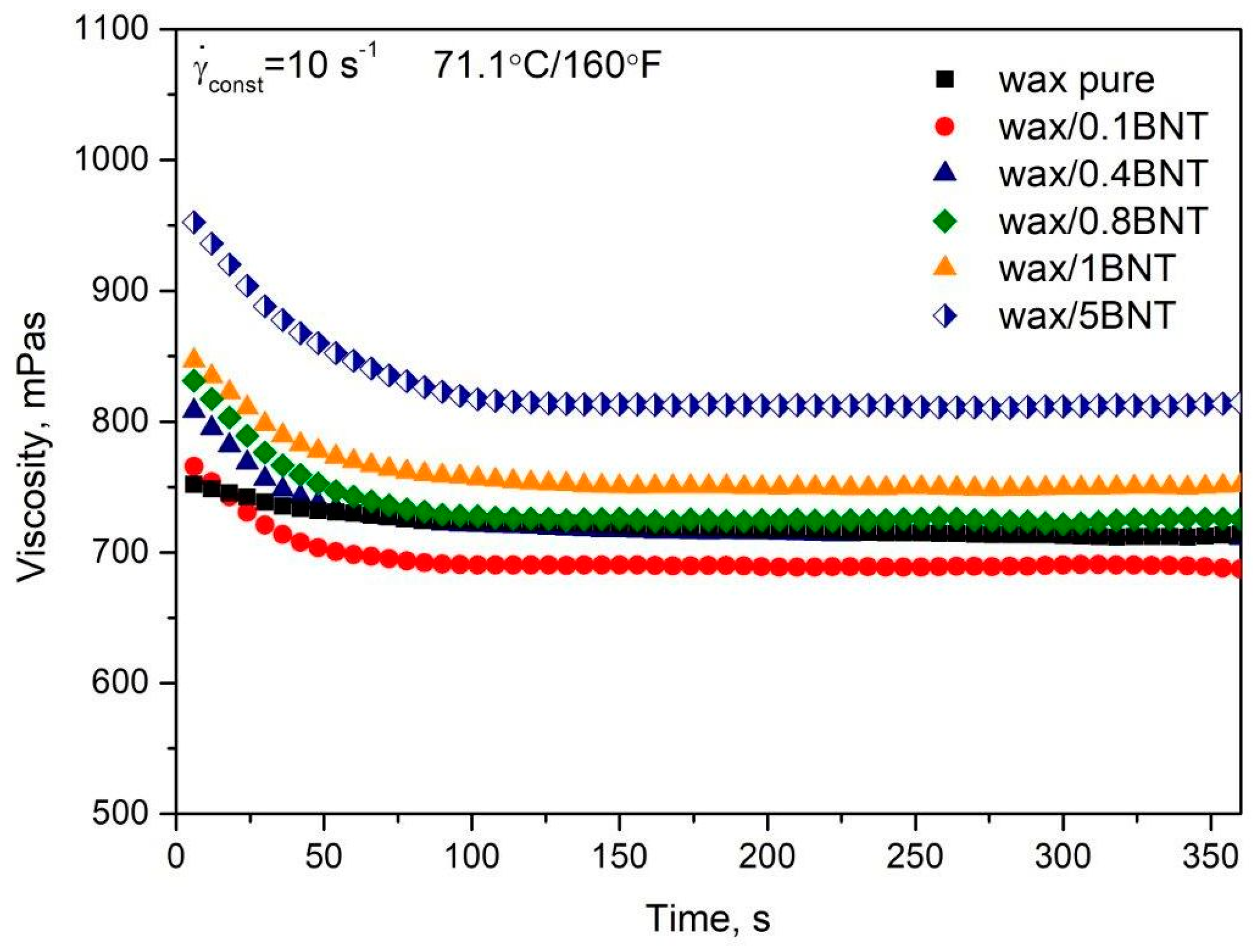






| Properties | Viscosity (160 °F)/71.1 °C (mPa·s) | Injection Temperature (°F/°C) | Geometrical Form | Shore D Hardness (°ShD) |
|---|---|---|---|---|
| Flexible Blue Wax | 615 | 170/76.7 | flakes | 30 |
| Element | O | Si | Al | Fe | Ca | Mg | Na | K |
|---|---|---|---|---|---|---|---|---|
| wt.% | 49.2 | 30.4 | 10.4 | 2.9 | 2.1 | 2.0 | 1.8 | 0.7 |
| Sample | Bentonite (wt.%) | Bentonite (vol.%) |
|---|---|---|
| wax pure | 0 | 0 |
| wax/0.1BNT | 0.1 | 0.13 |
| wax/0.4BNT | 0.4 | 0.5 |
| wax/0.8BNT | 0.8 | 1 |
| wax/1BNT | 1 | 1.25 |
| wax/5BNT | 5 | 6.25 |
| Parameters | Pure Wax | Wax/BNT Blends |
|---|---|---|
| Wax temperature, °C | 72 | 73 |
| Injection temperature, °C | 73 | 74 |
| Injection pressure, kPa | 30 | 31 |
| Holding time, s | 30 | 32 |
| Sample | Density, g/cm3 |
|---|---|
| wax/0.1BNT | 1.026 ± 0.001 |
| wax/0.4BNT | 1.047 ± 0.002 |
| wax/0.8BNT | 1.049 ± 0.003 |
| wax/1BNT | 1.059 ± 0.002 |
| wax/5BNT | 1.070 ± 0.005 |
| Samples | Onset Melting Temperature Tm onset [°C] | Melting Temperature Tm2 [°C] | Heat of Fusion ΔTm2 [J/g] | Crystallization Temperature Tcr2 [°C] | Degree of Crystallinity Xc [%] |
|---|---|---|---|---|---|
| wax pure | 42.0 | 60.0 | −123.2 | 51.0 | 41.5 ± 0.5 |
| wax/0.1BNT | 45.6 | 60.3 | −135.4 | 52.2 | 45.6 ± 0.4 |
| wax/0.4BNT | 47.6 | 60.4 | −138.0 | 52.4 | 46.7 ± 0.7 |
| wax/0.8BNT | 47.3 | 60.0 | −137.0 | 52.3 | 46.5 ± 0.8 |
| wax/1BNT | 48.6 | 60.2 | −139.4 | 52.0 | 47.4 ± 1.0 |
| wax/5BNT | 44.6 | 60.4 | −127.0 | 52.0 | 43.6 ± 1.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czarnecka-Komorowska, D.; Grześkowiak, K.; Popielarski, P.; Barczewski, M.; Gawdzińska, K.; Popławski, M. Polyethylene Wax Modified by Organoclay Bentonite Used in the Lost-Wax Casting Process: Processing−Structure−Property Relationships. Materials 2020, 13, 2255. https://doi.org/10.3390/ma13102255
Czarnecka-Komorowska D, Grześkowiak K, Popielarski P, Barczewski M, Gawdzińska K, Popławski M. Polyethylene Wax Modified by Organoclay Bentonite Used in the Lost-Wax Casting Process: Processing−Structure−Property Relationships. Materials. 2020; 13(10):2255. https://doi.org/10.3390/ma13102255
Chicago/Turabian StyleCzarnecka-Komorowska, Dorota, Krzysztof Grześkowiak, Paweł Popielarski, Mateusz Barczewski, Katarzyna Gawdzińska, and Mikołaj Popławski. 2020. "Polyethylene Wax Modified by Organoclay Bentonite Used in the Lost-Wax Casting Process: Processing−Structure−Property Relationships" Materials 13, no. 10: 2255. https://doi.org/10.3390/ma13102255
APA StyleCzarnecka-Komorowska, D., Grześkowiak, K., Popielarski, P., Barczewski, M., Gawdzińska, K., & Popławski, M. (2020). Polyethylene Wax Modified by Organoclay Bentonite Used in the Lost-Wax Casting Process: Processing−Structure−Property Relationships. Materials, 13(10), 2255. https://doi.org/10.3390/ma13102255