Optimizing Silanization to Functionalize Stainless Steel Wire: Towards Breast Cancer Stem Cell Isolation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pre-Treatment of SS
2.2. Electrodeposition of APTES
2.3. Cyclic Voltammetry
2.4. Scanning Electron Microscopy (SEM) and Energy-Dispersive X-ray Spectroscopy (EDS) Analysis
2.5. FITC Analysis
2.6. Immobilizing Ligands
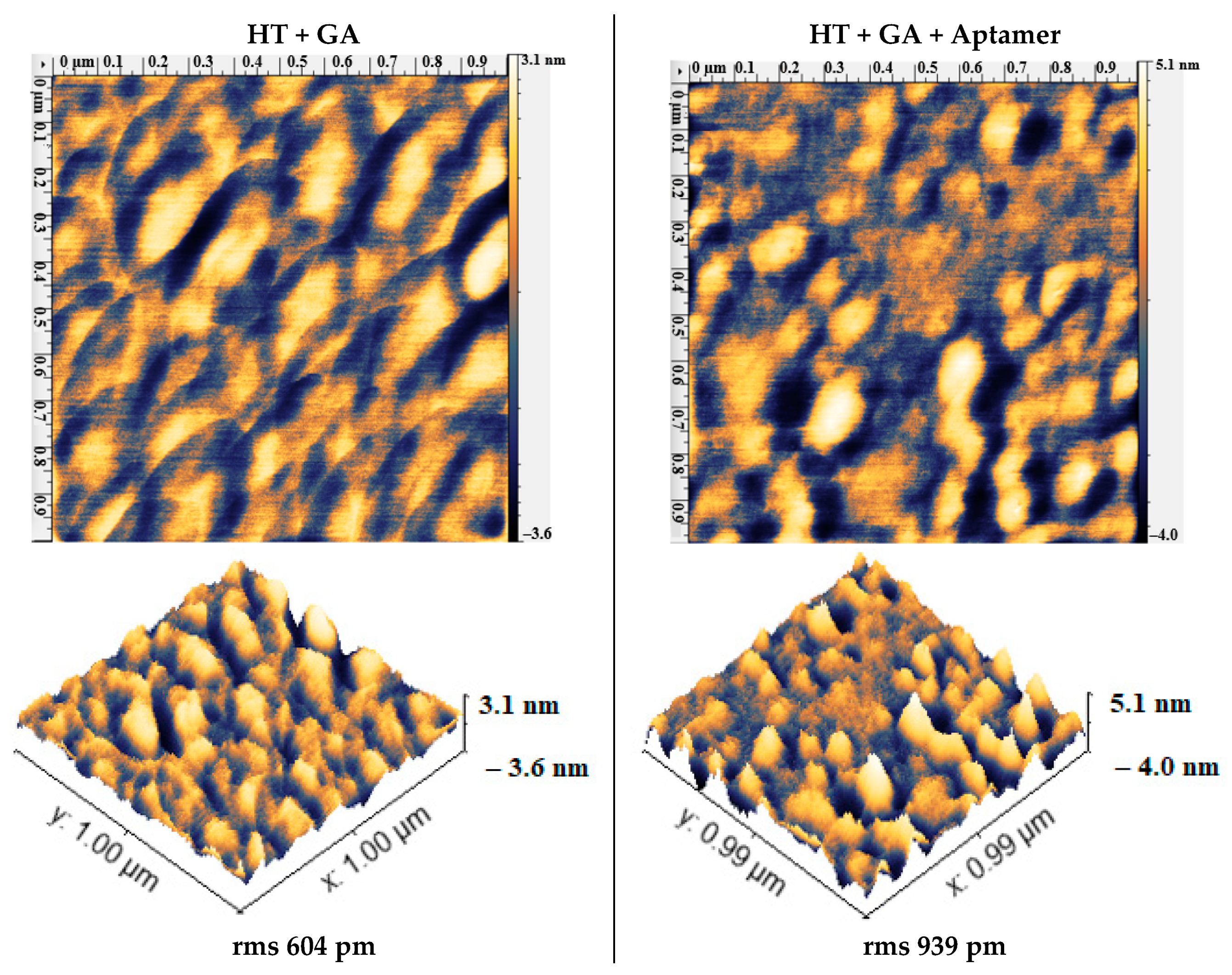
2.7. Atomic Force Microscopy (AFM)
2.8. Testing the Functionalized SS Surface to Capture BCSC
3. Results and Discussion
3.1. Pre-Treatment of SS
3.2. Electrodeposition of APTES: General Consideration
3.3. Electrodeposition of APTES: Applied Potential
3.4. Electrodeposition of APTES: pH of the Solution
3.5. Electrodeposition of APTES: Heat Treatment Temperature
3.6. Attaching Aptamers
3.7. Testing the Surface to Capture Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, H.; Han, J.; Sun, Y.; Huang, Y.; Zhou, M. MC3T3-E1 cell response to stainless steel 316L with different surface treatments. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 56, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H.; Ramdan, D.; Djuansjah, J. Metals for biomedical applications. In Biomedical Engineering—From Theory to Applications; Fazel-Rezai, R., Ed.; BoD–Books on Demand: Norderstedt, Germany, 2011; pp. 411–430. [Google Scholar]
- Saucedo-Zeni, N.; Mewes, S.; Niestroj, R.; Gasiorowski, L.; Murawa, D.; Nowaczyk, P.; Tomasi, T.; Weber, E.; Dworacki, G.; Morgenthaler, N.G.; et al. A novel method for the in vivo isolation of circulating tumor cells from peripheral blood of cancer patients using a functionalized and structured medical wire. Int. J. Oncol. 2012, 41, 1241–1250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weng, W.-H.; Ho, I.-L.; Pang, C.-C.; Pang, S.-N.; Pan, T.-M.; Leung, W.-H. Real-time circulating tumor cells detection via highly sensitive needle-like cytosensor-demonstrated by a blood flow simulation. Biosens. Bioelectron. 2018, 116, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Amiri, A.; Ghaemi, F. Graphene grown on stainless steel mesh as a highly efficient sorbent for sorptive microextraction of polycyclic aromatic hydrocarbons from water samples. Anal. Chim. Acta 2017, 994, 29–37. [Google Scholar] [CrossRef]
- Libertino, S.; Giannazzo, F.; Aiello, V.; Scandurra, A.; Sinatra, F.; Renis, M.; Fichera, M. XPS and AFM characterization of the enzyme glucose oxidase immobilized on SiO2 surfaces. Langmuir 2008, 24, 1965–1972. [Google Scholar] [CrossRef]
- Islam, F.; Gopalan, V.; Smith, R.; Lam, A. Translational potential of cancer stem cells: A review of the detection of cancer stem cells and their roles in cancer recurrence and cancer treatment. Exp. Cell Res. 2015, 335, 135–147. [Google Scholar] [CrossRef]
- Ribatti, D. Cancer stem cells and tumor angiogenesis. Cancer Lett. 2012, 321. [Google Scholar] [CrossRef]
- Sampieri, K.; Fodde, R. Cancer stem cells and metastasis. Semin. Cancer Biol. 2012, 22, 187–193. [Google Scholar] [CrossRef]
- Nowaczyk, P.; Dlugaszewska, S.; Herold, S.; Krahn, T.; Mayer, M.; Morgenthaler, N.; Zabel, M.; Luecke, K.; Murawa, D. A Novel Technology for In Vivo Isolation of circulating Tumor Cells in Breast Cancer Patients. In Proceedings of the 9th European Breast Cancer Conference, Glasgow, UK, 19–21 March 2014. [Google Scholar]
- Heyden, A.; Tomasi, T.; Zeni, N.; Herold, S.; Nowaczyk, P.; Schmitz, A.; Krahn, T.; Zabel, M.; Murawa, D.; Luecke, K. In vivo isolation of circulating tumor cells. Eur. J. Cancer 2011, 47, S19. [Google Scholar] [CrossRef]
- Nowaczyk, P.; Herold, S.; Kim, P.S.; Schmitz, A.; Polom, K.; Murawa, P.; Morgenthaler, N.G.; Zabel, M.; Luecke, K.; Murawa, D. Functionalized and Structured Medical Wire as a Device for In-Vivo Isolation of Circulating Tumor Cells in Breast Cancer Patients. Eur. J. Cancer 2012, 48, S58–S59. [Google Scholar] [CrossRef]
- Li, J.; Geng, C.; Yan, M.; Wang, Y.; Ouyang, Q.; Yin, Y.; Wu, L.; He, J.; Jiang, Z. Circulating tumor cells in patients with breast cancer were detected by a novel device: A multicenter clinical trial in China. Natl. Med. J. China 2017, 97, 1857–1861. [Google Scholar]
- Theil, G.; Wencker, A.; Kersten, F.; Pini, G.; Luecke, K.; Fornara, P. Verification of a functionalized structured medical wire for the isolation of circulating tumor cells (CTC) in patients with renal cell carcinoma. J. Urol. 2013, 189, e192. [Google Scholar] [CrossRef]
- Theil, G.; Fischer, K.; Weber, E.; Medek, R.; Hoda, R.; Lucke, K.; Fornara, P. The Use of a New CellCollector to Isolate Circulating Tumor Cells from the Blood of Patients with Different Stages of Prostate Cancer and Clinical Outcomes—A Proof-of-Concept Study. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Chudak, C.; Herrmann, J.; Lesser, T. Enumeration and molecular characterization of circulating tumor cells in lung cancer patients using the GILUPI CellCollector. J. Thorac. Oncol. 2016, 11, S104–S105. [Google Scholar] [CrossRef] [Green Version]
- Dlugaszewska, B.; Gasiorowski, L.; Herold, S.; Nowack, B.; Dworacki, G.; Luecke, K.; Dyszkiewicz, W. An innovative technology for in vivo isolation of circulating tumor cells in non-small cell lung cancer (NSCLC) patients and immunofluorescent detection of ALK protein. J. Thorac. Oncol. 2014, 9, S14–S15. [Google Scholar]
- Gasiorowski, L.; Herold, S.; Morgenthaler, N.; Dworacki, G.; Luecke, K.; Dyszkiewicz, W. A new medical device for in-vivo isolation of circulating tumor cells in non small cell lung cancer (NSCLC) patients. J. Thorac. Oncol. 2012, 7, S32. [Google Scholar]
- Gallerani, G.; Cocchi, C.; Bocchini, M.; Piccinini, F.; Fabbri, F. Characterization of Tumor Cells Using a Medical Wire for Capturing Circulating Tumor Cells: A 3D Approach Based on Immunofluorescence and DNA FISH. JOVE J. Vis. Exp. 2017. [Google Scholar] [CrossRef]
- Zhang, H.D.; Gong, S.C.; Liu, Y.Q.; Liang, L.J.; He, S.B.; Zhang, Q.X.; Si, M.Y.; Yu, Z.K. Enumeration and molecular characterization of circulating tumor cell using an in vivo capture system in squamous cell carcinoma of head and neck. Chin. J. Cancer Res. 2017, 29, 196–203. [Google Scholar] [CrossRef]
- Herold, S.; Gasiorowski, L.; Nowaczyk, P.; Schumann, A.; Theil, G.; Haubold, K.; Krahn, T.; Dyszkiewicz, W.; Murawa, D.; Lucke, K. An innovative approach for in-vivo isolation of circulating tumor cells (CTCs). Eur. J. Cancer 2013, 49, S199. [Google Scholar]
- Vermesh, O.; Aalipour, A.; Ge, J.; Saenz, Y.; Guo, Y.; Alam, I.; Park, S.-m.; Adelson, C.; Mitsutake, Y.; Vilches-Moure, J.; et al. An intravascular magnetic wire for the high-throughput retrieval of circulating tumour cells in vivo. Nat. Biomed. Eng. 2018, 2, 696–705. [Google Scholar] [CrossRef]
- Pantel, K.; Alix-Panabieres, C. Circulating tumour cells in cancer patients: Challenges and perspectives. Trends Mol. Med. 2010, 16, 398–406. [Google Scholar] [CrossRef]
- Tirino, V.; Desiderio, V.; Paino, F.; Papaccio, G.; De Rosa, M. Methods for cancer stem cell detection and isolation. Methods Mol. Biol. 2012, 879, 513–529. [Google Scholar] [PubMed]
- Malheiro, V.N.; Spear, R.L.; Brooks, R.A.; Markaki, A.E. Osteoblast and monocyte responses to 444 ferritic stainless steel intended for a Magneto-Mechanically Actuated Fibrous Scaffold. Biomaterials 2011, 32, 6883–6892. [Google Scholar] [CrossRef]
- Nazneen, F.; Galvin, P.; Arrigan, D.W.M.; Thompson, M.; Benvenuto, P.; Herzog, G. Electropolishing of medical-grade stainless steel in preparation for surface nano-texturing. J. Solid State Electrochem. 2012, 16, 1389–1397. [Google Scholar] [CrossRef] [Green Version]
- Rezaei, B.; Havakeshian, E.; Ensafi, A.A. Stainless steel modified with an aminosilane layer and gold nanoparticles as a novel disposable substrate for impedimetric immunosensors. Biosens. Bioelectron. 2013, 48, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Somasunderam, A.; Thiviyanathan, V.; Tanaka, T.; Li, X.; Neerathilingam, M.; Lokesh, G.L.R.; Mann, A.; Peng, Y.; Ferrari, M.; Klostergaard, J.; et al. Combinatorial Selection of DNA Thioaptamers Targeted to the HA Binding Domain of Human CD44. Biochemistry 2010, 49, 9106–9112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramanian, N.; Akilandeswari, B.; Bhutra, A.; Alameen, M.; Vetrivel, U.; Khetan, V.; Kanwar, R.K.; Kanwar, J.R.; Krishnakumar, S. Targeting CD44, ABCG2 and CD133 markers using aptamers: In silico analysis of CD133 extracellular domain 2 and its aptamer. RSC Adv. 2016, 6, 32115–32123. [Google Scholar] [CrossRef]
- Ooji, M.; Stacy, M.; Seth, M.; Mugada, T.; Gandhi, J.; Puomi, P. Corrosion Protection Properties of Organofunctional Silanes—An Overview. Tsinghua Sci. Technol. 2005, 6, 639–664. [Google Scholar]
- Collinson, M.M.; Higgins, D.A.; Kommidi, R.; Campbell-Rance, D. Electrodeposited silicate films: Importance of supporting electrolyte. Anal. Chem. 2008, 80, 651–656. [Google Scholar] [CrossRef]
- Woo, H.; Reucroft, P.J.; Jacob, R.J. Electrodeposition of organofunctional silanes and its influence on structural adhesive bonding. J. Adhes. Sci. Technol. 1993, 7, 681–697. [Google Scholar] [CrossRef]
- Okner, R.; Favaro, G.; Radko, A.; Domb, A.J.; Mandler, D. Electrochemical codeposition of sol-gel films on stainless steel: Controlling the chemical and physical coating properties of biomedical implants. Phys. Chem. Chem. Phys. 2010, 12, 15265–15273. [Google Scholar] [CrossRef] [PubMed]
- Moller, R.; Csaki, A.; Kohler, J.M.; Fritzsche, W. DNA probes on chip surfaces studied by scanning force microscopy using specific binding of colloidal gold. Nucleic Acids Res. 2000, 28. [Google Scholar] [CrossRef] [PubMed]
- Gunda, N.S.K.; Singh, M.; Norman, L.; Kaur, K.; Mitra, S.K. Optimization and characterization of biomolecule immobilization on silicon substrates using (3-aminopropyl)triethoxysilane (APTES) and glutaraldehyde linker. Appl. Surf. Sci. 2014, 305, 522–530. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Optimization of silica silanization by 3-aminopropyltriethoxysilane. Langmuir 2006, 22, 11142–11147. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Cho, J.; Seidler, P.M.; Kurland, N.E.; Yadavalli, V.K. Investigations of Chemical Modifications of Amino-Terminated Organic Films on Silicon Substrates and Controlled Protein Immobilization. Langmuir 2010, 26, 2599–2608. [Google Scholar] [CrossRef]
- Ocana, C.; Hayat, A.; Mishra, R.K.; Vasilescu, A.; del Valle, M.; Marty, J.L. Label free aptasensor for Lysozyme detection: A comparison of the analytical performance of two aptamers. Bioelectrochemistry 2015, 105, 72–77. [Google Scholar] [CrossRef] [Green Version]
- Tesoro, G.; Wu, Y.L. Silane coupling agents—The role of the organofunctional group. J. Adhes. Sci. Technol. 1991, 5, 771–784. [Google Scholar] [CrossRef]
- Baumgartel, T.; von Borczyskowski, C.; Graaf, H. Selective surface modification of lithographic silicon oxide nanostructures by organofunctional silanes. Beilstein J. Nanotechnol. 2013, 4, 218–226. [Google Scholar] [CrossRef]
- Seguin, C.; McLachlan, J.M.; Norton, P.R.; Lagugne-Labarthet, F. Surface modification of poly(dimethylsiloxane) for microfluidic assay applications. Appl. Surf. Sci. 2010, 256, 2524–2531. [Google Scholar] [CrossRef]
- Xu, W.J.; Riikonen, J.; Nissinen, T.; Suvanto, M.; Rilla, K.; Li, B.J.; Wang, Q.; Deng, F.; Lehto, V.P. Amine Surface Modifications and Fluorescent Labeling of Thermally Stabilized Mesoporous Silicon Nanoparticles. J. Phys. Chem. C 2012, 116, 22307–22314. [Google Scholar] [CrossRef]
- Heller, M.; Kammerer, P.W.; Al-Nawas, B.; Luszpinski, M.A.; Forch, R.; Brieger, J. The effect of extracellular matrix proteins on the cellular response of HUVECS and HOBS after covalent immobilization onto titanium. J. Biomed. Mater. Res. Part A 2015, 103, 2035–2044. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, F.; Sadjadi, M.; Farhadyar, N. Fe3O4 Nanoparticles Modified with APTES as the Carrier for (+)-(S)-2-(6-methoxynaphthalen-2-yl) Propanoic Acid (Naproxen) and (RS) 2-(3-benzoylphenyl)-propionic Acid (Ketoprofen) Drug. Orient. J. Chem. 2014, 30, 1609–1618. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Gao, F.; Wanjala, B.; Li, Z.Y.; Cernigliaro, G.; Gu, Z.Y. High efficiency reductive degradation of a wide range of azo dyes by SiO2-Co core-shell nanoparticles. Appl. Catal. B Environ. 2016, 199, 504–513. [Google Scholar] [CrossRef]
- Lee, S.H.; Yang, S.W.; Park, E.S.; Hwang, J.Y.; Lee, D.S. High-Performance Adhesives Based on Maleic Anhydride-g-EPDM Rubbers and Polybutene for Laminating Cast Polypropylene Film and Aluminum Foil. Coatings 2019, 9, 61. [Google Scholar] [CrossRef] [Green Version]
- Aziz, M.A.; Patra, S.; Yang, H. A facile method of achieving low surface coverage of Au nanoparticles on an indium tin oxide electrode and its application to protein detection. Chem. Commun. 2008, 4607–4609. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.D.; Ran, Y.; Wang, G.J. Label-Free Detection of Cancer Biomarkers Using an In-Line Taper Fiber-Optic Interferometer and a Fiber Bragg Grating. Sensors 2017, 17, 2559. [Google Scholar] [CrossRef] [Green Version]
- Jannah, F.; Kim, J.H.; Lee, J.W.; Kim, J.M.; Lee, H. Immobilized Polydiacetylene Lipid Vesicles on Polydimethylsiloxane Micropillars as a Surfactin-Based Label-Free Bacterial Sensor Platform. Front. Mater. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Shaimi, R.; Low, S.C. Prolonged protein immobilization of biosensor by chemically cross-linked glutaraldehyde on mixed cellulose membrane. J. Polym. Eng. 2016, 36, 655–661. [Google Scholar] [CrossRef]
- Kemmegne-Mbouguen, J.C.; Ngameni, E.; Baker, P.G.; Waryo, T.T.; Kgarebe, B.; Iwuoha, E.I. Carcinoembryonic Antigen Immunosensor Developed with Organoclay Nanogold Composite Film. Int. J. Electrochem. Sci. 2014, 9, 478–492. [Google Scholar]
- Arya, S.C.; Agarwal, N.; Agarwal, S. Use of polymerase chain reaction to diagnose tubercular arthritis from joint tissues and synovial fluid. Arch. Pathol. Lab. Med. 2004, 128, 1326–1327. [Google Scholar]
- Shin, D.S.; Kang, C.K.; Kim, J.K.; Chung, W.J.; Jang, K.H.; Lee, Y.S.; Ieee, I. Surface modification technology for bio-MEMS. In Proceedings of the Boston Transducers’03: Digest of Technical Papers, Boston, MA, USA, 8–12 June 2003; IEEE: New York, NY, USA, 2003; Volumes 1–2, pp. 1746–1749. [Google Scholar]
- Yazdanparast, S.; Benvidi, A.; Banaei, M.; Nikukar, H.; Tezerjani, M.D.; Azimzadeh, M. Dual-aptamer based electrochemical sandwich biosensor for MCF-7 human breast cancer cells using silver nanoparticle labels and a poly(glutamic acid)/MWNT nanocomposite. Microchim. Acta 2018, 185. [Google Scholar] [CrossRef] [PubMed]
- Aghajari, R.; Azadbakht, A. Amplified detection of streptomycin using aptamer-conjugated palladium nanoparticles decorated on chitosan-carbon nanotube. Anal. Biochem. 2018, 547, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Sefah, K.; Shangguan, D.; Xiong, X.; O’Donoghue, M.B.; Tan, W. Development of DNA aptamers using Cell-SELEX. Nat. Protoc. 2010, 5, 1169–1185. [Google Scholar] [CrossRef] [PubMed]









© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bekmurzayeva, A.; Dukenbayev, K.; Azevedo, H.S.; Marsili, E.; Tosi, D.; Kanayeva, D. Optimizing Silanization to Functionalize Stainless Steel Wire: Towards Breast Cancer Stem Cell Isolation. Materials 2020, 13, 3693. https://doi.org/10.3390/ma13173693
Bekmurzayeva A, Dukenbayev K, Azevedo HS, Marsili E, Tosi D, Kanayeva D. Optimizing Silanization to Functionalize Stainless Steel Wire: Towards Breast Cancer Stem Cell Isolation. Materials. 2020; 13(17):3693. https://doi.org/10.3390/ma13173693
Chicago/Turabian StyleBekmurzayeva, Aliya, Kanat Dukenbayev, Helena S. Azevedo, Enrico Marsili, Daniele Tosi, and Damira Kanayeva. 2020. "Optimizing Silanization to Functionalize Stainless Steel Wire: Towards Breast Cancer Stem Cell Isolation" Materials 13, no. 17: 3693. https://doi.org/10.3390/ma13173693







