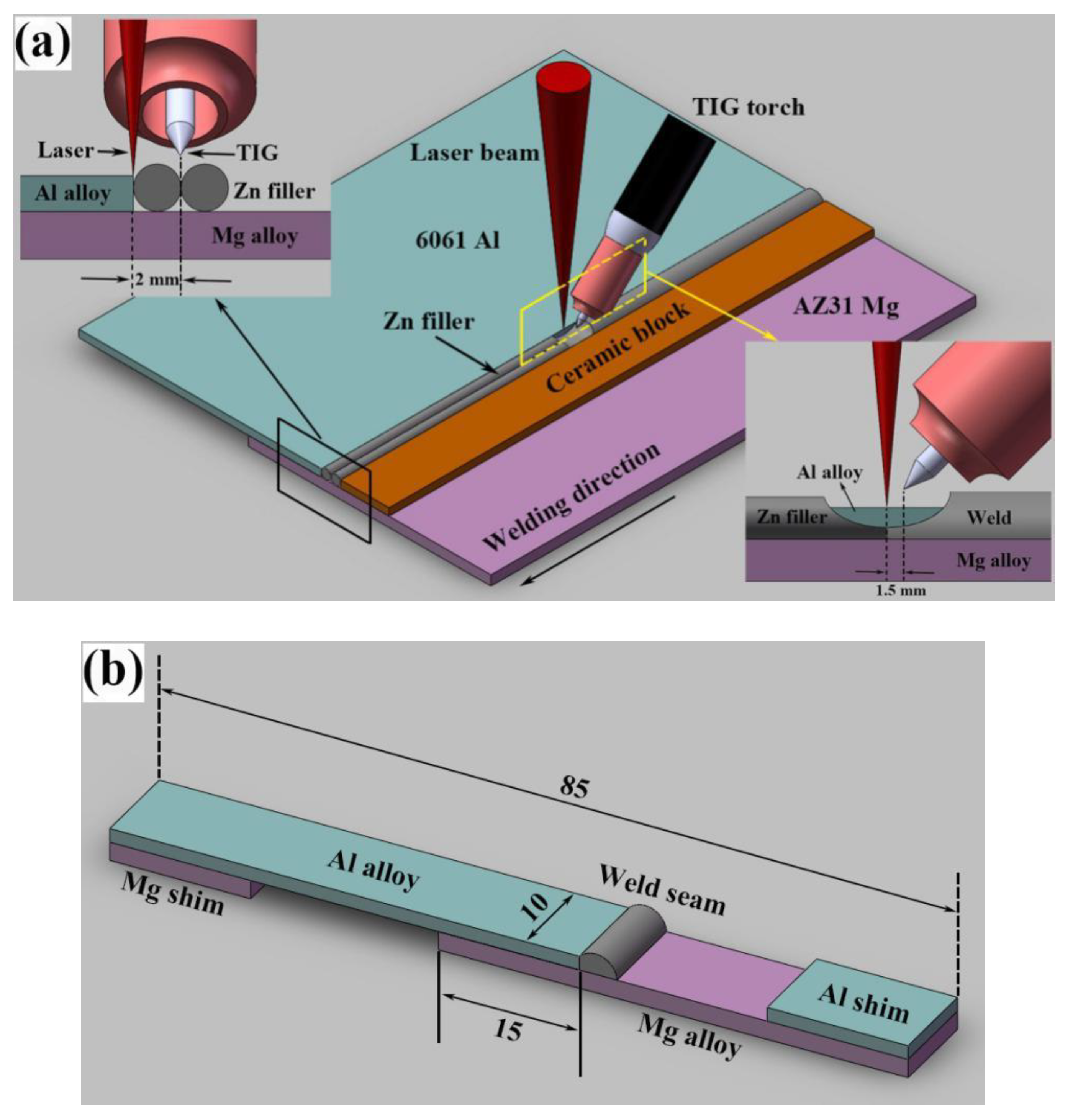
3.1. Design of Heat Source in Dissimilar Welding Process
Based on the characteristics of Mg–Al dissimilar joints, the heat input of laser and TIG in hybrid heat source must be reasonably allocated to control the dissolution of base metals. Single TIG, laser-TIG and non-axisymmetric laser-TIG heat sources were adopted to join Mg–Al dissimilar metals at a relatively high welding speed (800 mm/min), as shown in
Figure 2.
Figure 3 shows the macroscopic morphologies of Mg–Al–Zn dissimilar joints welded by different heat sources. As indicated in
Figure 3a,b, it was impossible to achieve joining between Mg and Al sheets when single TIG was used. No penetration was found on the faying surface due to the relatively low heat input. The Zn filler could not completely melt by arc and remained on the Mg substrate.
When using laser-TIG heat source, the heat input was too concentrated, giving rise to the burn through defect, as shown in
Figure 3c. In this case, most of the energy of arc was used to heat Al base metal, causing its excessive melting (marked in the black box in
Figure 3d). Based on the related study in Mg/Al cold metal transfer welding [
35], it would increase the possibility of forming brittle IMCs because liquid Mg and Al were easier to contact with each other. As a result, obvious cracks were found at the bottom of fusion zone (marked by the white arrows). The tensile testing samples fractured either during mechanical processing or after coupon clamping in tensile test.
Until the non-axisymmetric laser-TIG heat source was adopted, a stable appearance without obvious defect was obtained, as shown in
Figure 3e. In this case, the heat input was dispersed to avoid the excess dissolution of base metals. As a result, an acceptable melting amount of Al was observed from
Figure 3f (marked in the black box). Owing to the offset of the arc, two Zn filler wires were totally melted into the weld pool. This could reduce the dilution ratio of Mg and Al base metals in dissimilar joints and inhibit the formation of brittle Mg–Al IMCs. No crack was found in the cross section of the joint from
Figure 3f. Therefore, a non-axisymmetric laser-TIG heat source is more suitable for joining Mg–Al dissimilar metals. All of the following joints were produced by this heat source.
3.2. Macrostructures and Properties of Joints
From our previous study, the weak area of the Mg–Al–Zn butt joint made by TIG is the fusion zone near Mg alloy base material. Fracture occurred at the interface between the MgZn
2 particles and (Al, Zn) solid solution [
27], as shown in
Figure 4. Through the control of welding speed, it is possible to obtain a joint with more (Al, Zn) solid solution and less MgZn
2 IMCs by increasing the solidification rate of welding pool.
The macroscopic morphologies of Mg–Al–Zn dissimilar joints produced at different welding speeds are shown in
Figure 5. When the welding speed was larger than 900 mm/min, it was hard to obtain a continuous joint because liquid Zn filler spread poorly on Mg substrate. As the welding speed decreased, acceptable joints could be obtained as the weld penetration and width in the Mg substrate increased. Meanwhile, some porosities gradually formed and grow up in the weld seam due to the excessive vaporization of Zn filler. Note that an obvious stratification phenomenon appeared, especially in the case of high welding speed, which resulted from the inhomogeneous microstructures in rapid solidification process. Thus, the joint is divided into three typical areas for following analysis: Part I is fusion zone (FZ). It includes two parts, one is mainly composed of the melting Zn filler (FZ
Zn) and the other is the mixture zone of liquid Zn and Mg (FZ
Mg); Part II is the Al/FZ interface; Part III is the Mg/FZ interface, as shown in
Figure 6.
Figure 7a shows the tensile-shear load as a function of welding speed. As could be seen, the tensile-shear load gradually rose with welding speed increased from 500 mm/min to 800 mm/min and reached a maximum value of 1052.5 N/cm, then slightly dropped to 986.5 N/cm with further increasing welding speed. The typical force-displacement curves are shown in
Figure 7b. A yielding phenomenon (marked in the black box in
Figure 7b) was found in the joint with highest fracture load (made at 800 mm/min). We inferred that plastic deformation occurred in FZ
Zn because the dilution rate in this region was the lowest. The weld metal probably consisted of Zn filler and slight plastic deformation occurred before fracturing. Further analysis could be seen in the fracture mechanism in
Section 3.6.
After tensile-shear strength tests, the fractured joints were re-combined to observe the fracture locations, the results were shown in
Figure 8. For the joint welded at 900 mm/min, the fracture occurred in FZ
Mg near Mg substrate. As the welding speed decreased, all the joints fractured in FZ near Al base metal. The fracture started at FZ
Mg and propagated to FZ
Zn, just as shown in
Figure 8b–d. Seen from
Figure 8c, the fracture had passed through the porosity, indicating that the relatively low welding speed would cause the porosity defect and deteriorate the joint performance. With the help of fracture locations, FZ
Zn and FZ
Mg were identified as the weak areas. Thus, the microstructures in these areas were investigated emphatically.
3.3. Microstructures
The phase constituents of the fusion zones at different welding speeds were examined by XRD detection as shown in
Figure 9. It could be seen that there were no obvious differences of the phase composition among the joints at different welding speeds. The fusion zones mainly consisted of Zn solid solution and MgZn
2 compounds. The Al and Mg solid solution were also detected as base materials. However, whether they exist in the weld seam still needs to be further confirmed by EDS analysis.
Welding speed affects the content of Mg, Al and Zn in liquid metals, which has a main influence on the microstructural evolution.
Figure 10 shows the SEM micrographs of Al/FZ interface at different welding speeds. For the joints made at 900–700 mm/min, some dendrite crystals were found to grow from Al base metal perpendicularly into FZ. Based on the EDS results listed in
Table 4, the dendrites (phase-1A, phase-1D and phase-1F) were identified as Al solid solution; while the phases near these Al dendrites (phase-1B, phase-1C and phase-1E) were confirmed as the mixture of Zn solid solution and Al solid solution (MZAS). Based on the Al–Zn binary diagram, the maximum solid solubility of Zn in Al is 67 at.% at 381 °C, thus Zn solid solution will precipitate from the supersaturated Al grains and form MZAS.
As the welding speed decreased (700–500 mm/min), some phases containing Mg (phase-1G and phase-1I) were found near the Al/FZ interface as more Mg atoms diffused into molten pool. Based on the EDS results in
Table 4, they were identified as MZAS + MgZn
2 eutectic and MgZn
2, respectively. For the joints welded at 500 mm/min, the Al dendrite at the interface completely disappeared, suggesting that its growth mode transformed from dendritic growth to planar growth, which was determined by the low temperature gradient at relatively low welding speed.
Figure 11 shows the microstructures of FZ
Zn at different welding speeds. For the joint welded at 900 mm/min, it was composed of some equiaxed grains (phase-2A) with size of 10–15 μm and the striped phases (phase-2B) distributed at the grain boundaries. Based on the EDS analysis results in
Table 5, they were the Zn solid solution and MZAS, respectively. Furthermore, the MZAS (phase-2B) was identified as the eutectic of Al solid solution and Zn solid solution. It was noticed that the Zn grains could not close contact and the MZAS distributing at the boundary was not continuous. In rapid solidification process, the eutectic liquid formed “liquid films” along the grain boundaries, could not fill in the space caused by the solidification shrinkage, leading to the grain boundary gully, as shown in
Figure 11a.
As the welding speed further decreased to 700 mm/min, some phases with lamellar structure (phase-2E and phase-2H) were found near Zn and Al grains. They exhibited a typical characteristic of eutectic structure as shown in the inset in
Figure 11b. The EDS results indicated that the structure was MZAS + MgZn
2 eutectic. A similar eutectic structure was also found in the Mg–Al butt joints welded by TIG with Zn-30Al filler [
36]. The number of eutectic depended on the Mg content in liquid, which was determined by the heat input. As a result, the amount of eutectic structure increased distinctly with the welding speed decreased, as shown in
Figure 11b,c. Meanwhile the Zn grains were gradually refined, suggesting partial Zn entered the eutectic structure and formed MgZn
2 IMCs.
The microstructure of joint produced at 500 mm/min is shown in
Figure 11d. The dark grey polygynous phases (phase-2I) were surrounded by the light grey irregular phases (phase-2J). Based on the energy-dispersive X-ray spectroscopy (EDS) results in
Table 5, they were confirmed as MgZn
2 and MZAS, respectively. The high heat input promoted the dissolution of Mg base metal, resulting in the formation of Mg-rich phase MgZn
2 in large size.
Figure 12 shows the microstructures of FZ
Mg at different welding speeds, they exhibited the similar microstructures with dark grey particles dispersed into the light grey matrix. In FZ
Mg, it is possible to occur the following reactions based on the Mg–Zn binary diagram:
L→MgZn2 High temperature
L + MgZn2→Mg2Zn11 381 °C
L→Mg2Zn11 + (Zn) 364 °C
L→MgZn2 + (Zn) 364 °C
However, no Mg
2Zn
11 compounds were detected based on the XRD result shown in
Figure 9. The fast cooling rate inhibited the peritectic reaction and the formation of Mg
2Zn
11 compounds. According to the EDS results shown in
Table 6, the dark grey particle (phase-3A) was composed of 20.83 at.% Mg, 6.53 at.% Al and 72.64 at.% Zn, which was identified as MgZn
2 compounds. The light grey matrix (phase-3B), whose content of Mg significantly dropped, was confirmed as (Zn) + little MgZn
2. Similar observation results also appeared in the ultrasonic-assisted semi-solid brazing on dissimilar Al-Mg alloys [
37]. At the extremely fast cooling rate of non-axisymmetric laser-TIG welding, the reaction of Mg and Zn was restrained. As a result, the MgZn
2 IMC particles were visibly refined and dispersed compared with that by single TIG in
Figure 4b. The weak area of the joint also changed from FZ
Mg (by single TIG) to FZ
Zn (by non-axisymmetric laser-TIG).
As the decrease of welding speed, the content of Mg and Al in liquid metal increased. Therefore, the light grey matrix (phase-3D, phase-3F and phase-3H) was prone to excrete Mg and form Al-rich MZAS according to the EDS results in
Table 6.
Despite the similar microstructural characteristics, differences still can be observed from the distribution of IMCs. For the joint welded at 900 mm/min, the dark grey MgZn
2 particles were found to be enriched in some region (marked in the black circles), which was called the “IMCs-rich zone”. As the welding speed decreased, the IMCs-rich zones disappeared and the MgZn
2 IMCs became more dispersed due to the relatively sufficient Mg, Al and Zn liquid mixing. However, extremely low welding speed (500 mm/min) would cause the microstructure coarsening, just like the large MgZn
2 grains shown in
Figure 12d. The distribution of MgZn
2 IMCs had a great influence on the joint performance, which can be seen in fracture mechanism in
Section 3.6.
Figure 13 shows the microstructures of the Mg/FZ interface. A transitional layer was found between the Mg base metal and FZ (marked by the white dotted lines). The thickness of this layer increased distinctly with the decrease of welding speed. For the joint welded at 900 mm/min, the transitional layer (phase-4A) was identified as Mg solid solution + MgZn since it contained 58.42 at.% Mg, 6.08 at.% Al and 35.50 at.% Zn. The MgZn compound was not found by the XRD detection because its content was too low. As the welding speed decreased, a lamellar structure newly appeared in the transitional layer. Base on the EDS results in
Table 7, it was confirmed as Mg solid solution + MgZn eutectic (phase-4B, phase-4D and phase-4F), which corresponded to the observation results when using diffusion bonding of Mg to Al with Zn interlayer [
38]. Note that some polygonal particles (phase-4E), which had the similar composition with phase-4A, were dispersed in the matrix of eutectic layer. They were also confirmed as Mg solid solution + MgZn. The formation of this phase was determined by the composition fluctuation of the eutectic liquid, which changed it into dispersed particles after solidification. These small particles would not deteriorate the property of joint, due to their dispersed distribution characteristic according to the related studies by Tan [
39].
3.5. Formation of Phases and Solidification Process
Based on the microstructural analysis above, the solidification process was expected to be clarified with the assistance of the schematic diagram shown in
Figure 15. At the Al/FZ interface, the upper Al base metal melted under the irradiation of laser and diffused into the Zn liquid (
Figure 15a). As the temperature dropped to 560 °C, Al dendrites precipitated primarily at the solid–liquid interface and grew into FZ (
Figure 15b). When the temperature further decreased to 490 °C, Al-rich MZAS formed at the tip of dendrite (
Figure 15c). Finally as the temperature reached 430 °C, Zn-rich MZAS precipitated from the supersaturated Al dendrites and remaining liquid (
Figure 15d). No Mg–Zn IMCs formed in this region due to the limited diffusion of Mg atoms at relatively high welding speed.
According to
Figure 15e, a small amount of Mg atoms had diffused to FZ
Zn. As the temperature dropped to 550 °C, Al grains first precipitated from the liquid (
Figure 15f). Subsequently based on the Mg–Al–Zn ternary diagram shown in
Figure 16, the composition of liquid reached e
2E
2 line and the eutectic reaction occurred: L→(Al) + MgZn
2 at 490 °C (
Figure 15g). Finally, a large amount of Zn grains formed based on the reaction: L→(Al) + MgZn
2 + (Zn) at 400 °C (
Figure 15h).
Under the heating of the arc, melting and interdiffusion of Mg and Zn took place in FZ
Mg (
Figure 15i). Fine MgZn
2 IMCs particles first precipitated from the liquid at 590 °C. At the same time, a layer of MgZn
2 was deposited at the liquid-solid interface. As the consumption of Mg in liquid, MZAS began to form along the MgZn
2 IMCs boundaries at 460 °C (
Figure 15k). Finally, a eutectic reaction between Mg and Zn occurred at the liquid–solid interface at 340 °C, giving rise to the (Mg) + MgZn eutectic (
Figure 15l). The composition fluctuation of Mg in liquid caused the variation of precipitated phases in the solidification process.
3.6. Microstructure Evolution and Fracture Mechanism
Figure 17 shows the schematic of microstructure evolution at different welding speeds. The distributions of brittle IMCs in weak areas (FZ
Mg and FZ
Zn) were mainly discussed, and the relationship between microstructures and mechanical properties was established.
As the welding speed decreased from 900 mm/min to 800 mm/min, the tensile-shear load of the joint increased with the fracture location changing from FZ
Mg to FZ near Al base metal, indicating that the property of FZ
Mg was improved. Based on the microstructure evolution in
Figure 17, the brittle IMCs particles in FZ
Mg became more dispersed and completely separated by the MZAS phases as the welding speed decreased. The elimination of IMC-rich zones was attributed to the proper solidification rate, which not only ensured the sufficient mixing of liquid Mg and Zn, but also avoided the excessive dissolution of Mg base metal. The plastic MZAS phases could impede the stress concentration caused by brittle MgZn
2 IMCs and reduce the possibility of crack propagation, resulting in the increase of strength.
When further decreasing the welding speed to 700 mm/min, the amount of eutectic in the weak area (FZZn) increased significantly, along with the Zn grains gradually refined. Eventually, at the lowest welding speed of 500 mm/min, FZZn formed primarily MgZn2 IMCs rather than eutectic structure or Zn solid solution. Based on the previous analysis, the eutectic structure is the mixture of MZAS and MgZn2 IMCs, and its plasticity is superior to brittle IMCs but inferior to Al and Zn solid solution. Thus as the welding speed decreased, the joints became brittle gradually, which caused the mechanical performance to deteriorate.
The fracture surface morphologies and corresponding micro-fracture locations are presented in
Figure 18. For the joint welded at 900 mm/min, it was found to exhibit the characteristics of cleavage fracture. Based on the micro-fracture location in
Figure 18b, the fracture occurred along the MgZn
2 IMCs particles and propagated to the MZAS matrix in FZ
Mg. The dense MgZn
2 IMCs were the main crack source due to their intrinsic brittleness.
The joint welded at 800 mm/min displayed a typical quasi-cleavage fracture feature, which usually appeared in HCP (hexagonal close-packed) and BCC (body-centered cubic) structure metals. The micro-fracture location showed that breaking occurred along the Zn grain boundaries. Note that some shallow dimples were found in the fracture surface (pointed by the white arrows), indicating slight deformation occurred before fracture and giving rise to the highest tensile-shear load.
For the joint welded at 700 mm/min, the fracture surface was characterized by plenty of stripped tear edges, which corresponded to the eutectic structure observed in the fracture location in
Figure 18f. In this case, the number of plastic Zn grains was reduced compared with that at 800 mm/min. Moreover, some porosities were also found on the fracture surface (pointed by the white arrows), which deteriorated the joint performance. The joint welded at 500 mm/min displayed a typical brittle fracture feature with many smooth surfaces without adequate plastic deformation. The MgZn
2 IMCs with a large size were mainly responsible for the joint breaking.