Phototherapy Combined with Carbon Nanomaterials (1D and 2D) and Their Applications in Cancer Therapy
Abstract
1. Introduction
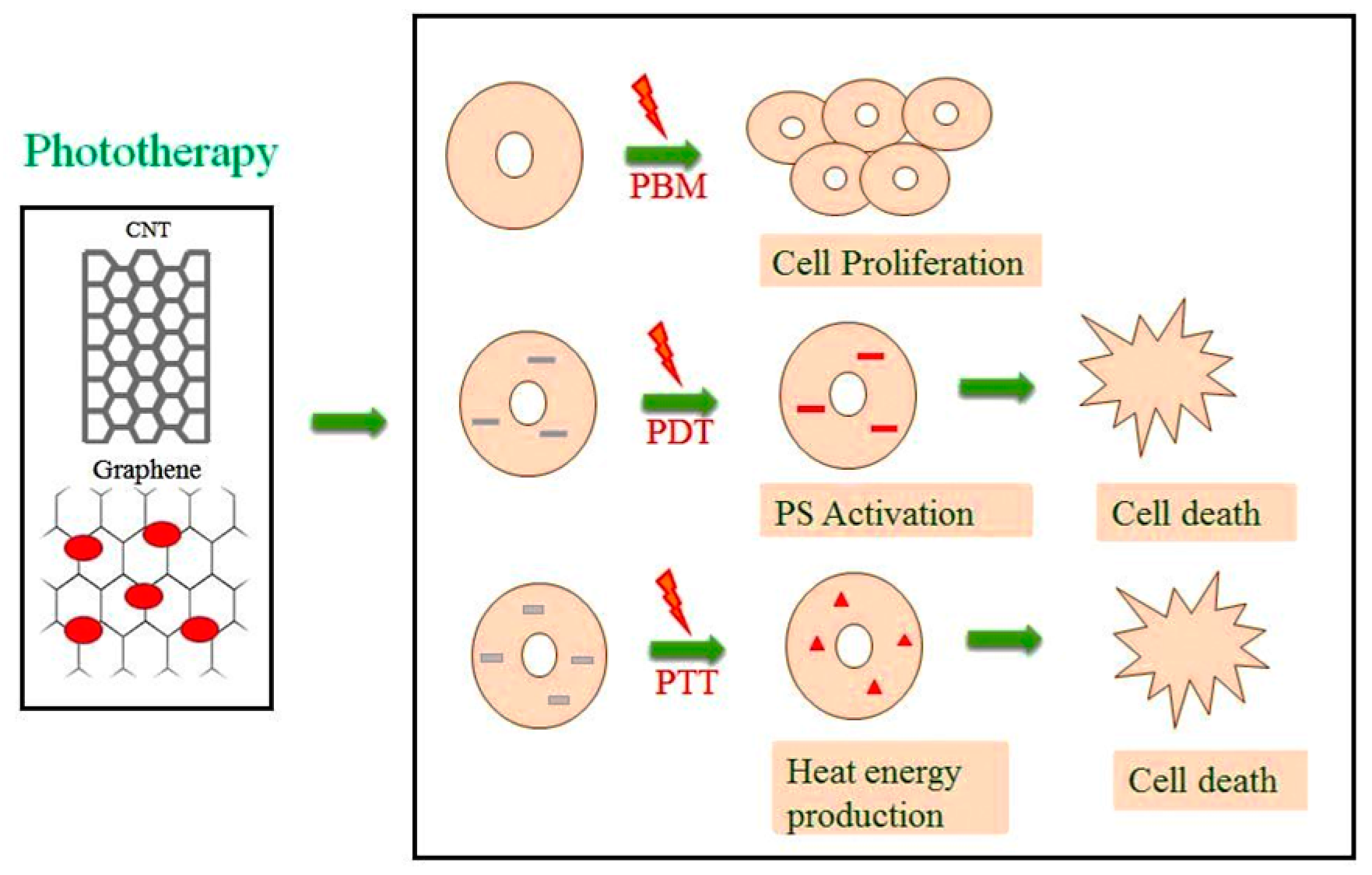
2. Types of Phototherapy
2.1. Photobiomodulation (PBM)
2.2. Photodynamic Therapy (PDT)
2.3. Photothermal Therapy (PTT)
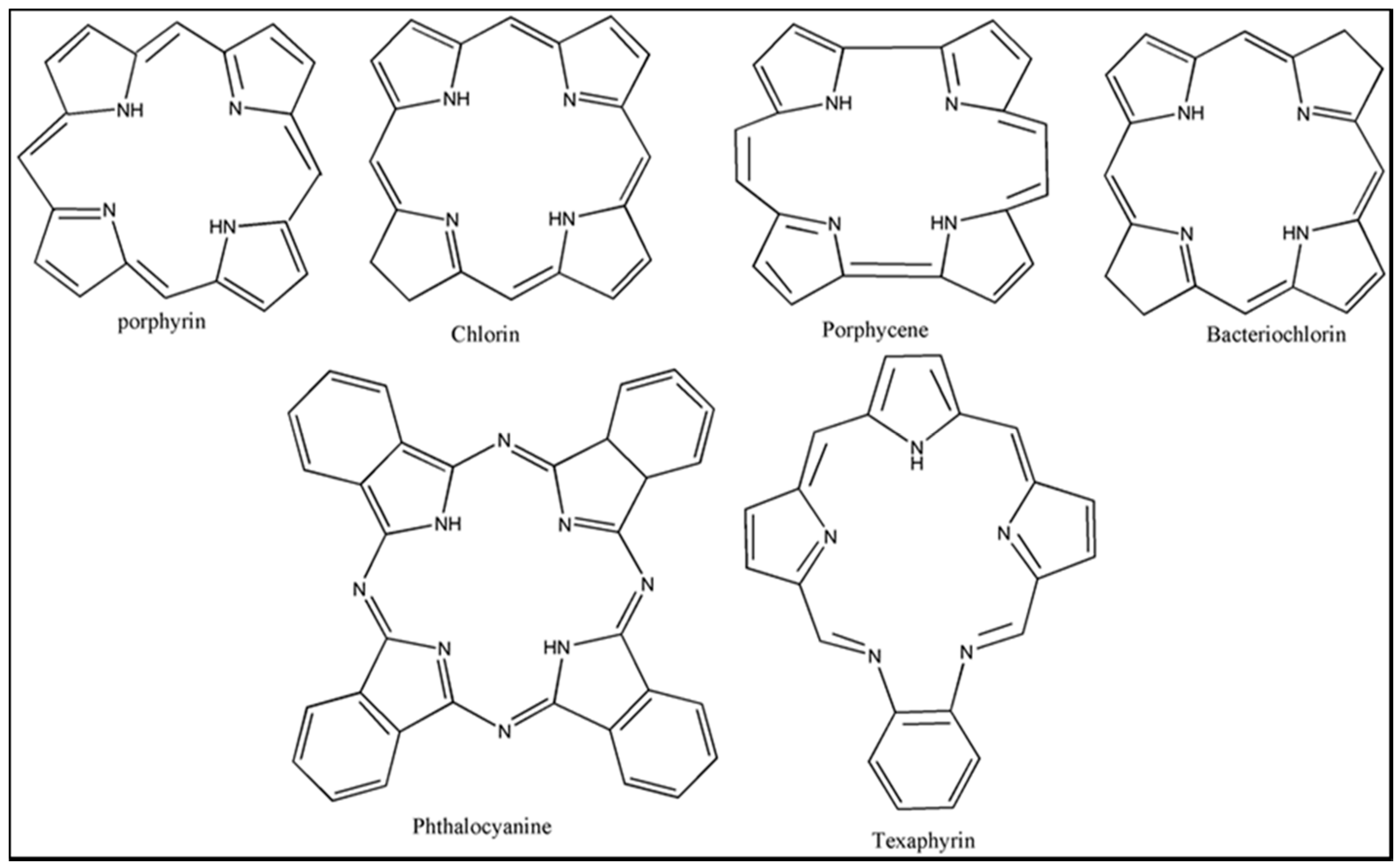
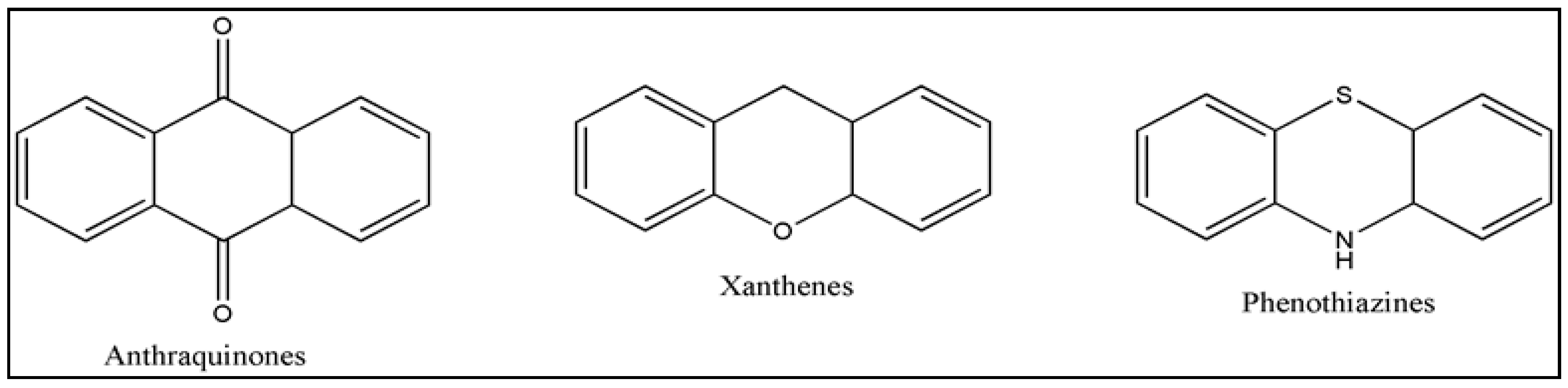
3. Photosensitizers (PS)
4. Carbon Nanomaterials
4.1. One Dimensional Carbon Nanotubes (CNT)
4.2. Two-Dimensional Graphene
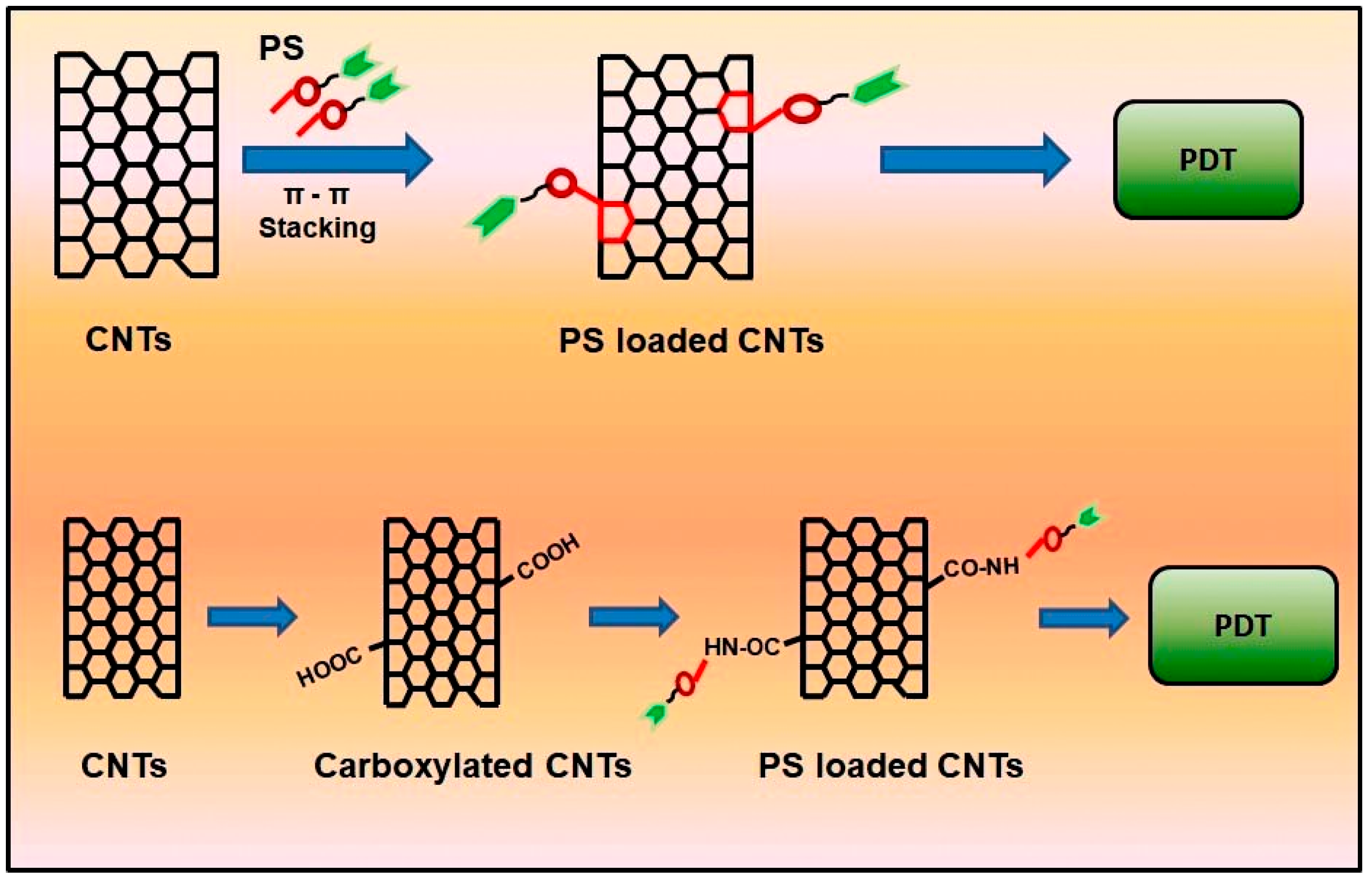
5. Mechanism and Loading of PS on Nanomaterials
5.1. Physical Loading of PS
5.2. Chemical Loading of PS
6. Application of Phototherapy Using CNT and Graphene on Cancer Therapy
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 0D | Zero dimensions |
| 1D | One dimension |
| 2D | Two dimensions |
| CD | Compact disk |
| CNT | Carbon nanotubes |
| COOH | Carboxyl group |
| CTCL | Cutaneous T-cell lymphoma |
| CVD | Chemical vapor deposition |
| DVD | Digital video disk |
| ECP | Extracorporeal photopheresis |
| EDC | Ethyl(dimethylaminopropyl) carbodiimide |
| FDA | Food and drug administration |
| GCOOH | Carboxylated graphene |
| GO | Graphene oxide |
| He-Ne | Helium and neon |
| HpD | Haematoporphyrin Derivative |
| IEC | International Engineering Consortium |
| LLLT | Low-level laser therapy |
| M−1 cm−1 | Per moles per centimetre |
| mTHPC | m-tetra hydroxyl phenylchlorin |
| MWCNT | Multi-walled carbon nanotubes |
| Nd:YAG | Neodymium-doped yttrium aluminium garnet |
| NHS | N -Hydroxysuccinimide |
| NIR | Near-infrared |
| Nm | Nanometre |
| OH | Hydroxyl group |
| PBM | Photobiomodulation |
| PDT | Photodynamic therapy |
| PEG | Polyethylene glycol |
| PEI | Polyetherimide |
| PS | Photosensitizers |
| PTT | Photothermal therapy |
| PUVA | Psoralens and ultraviolet A |
| PVPk30 | Polyvinylpyrrolidone |
| rGO | Reduced graphene oxide |
| ROS | Reactive oxygen species |
| siRNA | Small interfering ribonucleic acid |
| DNA | Double-stranded nucleic acid |
| SWCNT | Single-walled carbon nanotubes |
| TRITC | Tetramethylrhodamine |
| UCNP | Upconverting nanoparticles |
| UVA | Ultraviolet A |
| UVB | Ultraviolet B |
| εmax | Molar extinction coefficient |
References
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Naira, M.; Sandhub, S.S.; Sharma, A.K. Cancer molecular markers: A guide to cancer detection and management. Semin. Cancer Biol. 2018, 52, 39–55. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Cancer Fact Sheet; No. 297; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Grzybowski, A.; Sak, J.; Pawlikowski, J. A brief report on the history of phototherapy. Clin. Dermatol. 2016, 34, 532–537. [Google Scholar] [CrossRef]
- Pathak, M.A.; Fitzpatrick, T.B. The evolution of photochemotherapy with psoralens and UVA (PUVA): 2000 BC to 1992 AD. J. Photochem. Photobiol. B 1992, 14, 3–22. [Google Scholar] [CrossRef]
- Hönigsmann, H. History of phototherapy in dermatology. Photochem. Photobiol. Sci. 2013, 12, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Wiskemann, A. Die neuere Entwicklung der Lichttherapie [Recent developments in light therapy]. Dermatol. Wochenschr. 1963, 20, 377–383. [Google Scholar]
- Wolff, K.W.; Fitzpatrick, T.B.; Parrish, J.A.; Gschnait, F.; Gilchrest, B.; Hönigsmann, H.; Pathak, M.A.; Tanenbaum, L. Photochemotherapy for psoriasis with orally administered methoxsalen. Arch. Dermatol. 1976, 112, 943–950. [Google Scholar] [CrossRef]
- Trautinger, F.; Knobler, R.; Willemze, R.; Peris, K.; Stadler, R.; Laroche, L.; D’Incan, M.; Ranki, A.; Pimpinelli, N.; Ortiz-Romero, P.; et al. EORTC consensus recommendations for the treatment of mycosis fungoides/Sézary syndrome. Eur. J. Cancer. 2006, 42, 1014–1030. [Google Scholar] [CrossRef]
- Bjordal, J.M.; Lopes-Martins, R.A.; Joensen, J.; Couppe, C.; Ljunggren, A.E.; Stergioulas, A.; Johnson, M.I. A systematic review with procedural assessments and meta-analysis of Low Level Laser Therapy in lateral elbow tendinopathy (tennis elbow). BMC Musculoskelet. Disord. 2008, 9. [Google Scholar] [CrossRef]
- Mester, E.; Ludany, G.; Sellyei, M.; Szende, B.; Gyenes, G.; Tota, G.J. Studies on the inhibiting and activating effects of laser beams. Langenbecks Arch. Chir. 1968, 322, 1022–1027. [Google Scholar] [CrossRef]
- Da Silva, J.P.; Da Silva, M.A.; Almeida, A.P.F.; Junior, I.L.; Matos, A.P. Laser Therapy in the Tissue Repair Process: A Literature Review. Photomed. Laser Surg. 2010, 28, 17–21. [Google Scholar] [CrossRef]
- Cobb, C.M. Lasers in Periodontics: A Review of the Literature. J. Periodontol. 2006, 77, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Antunes, H.S.; Herchenhorn, D.; Small, I.A.; Araújo, C.M.M.; Viégas, C.M.P.; de Assis Ramos, G.; Dias, F.L.; Ferreira, C.G. Long-term survival of a randomized phase III trial of head and neck cancer patients receiving concurrent chemoradiation therapy with or without low-level laser therapy (LLLT) to prevent oral mucositis. Oral Oncol. 2017, 71, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Willis, G.L.; Moore, C.; Armstrong, S.M. Parkinson’s disease, lights and melanocytes: Looking beyond the retina. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhi-Jun, L.; Yao, W.; Hua-Feng, Z.; Xin-Long, M.; Peng, T.; Yuting, H. Effectiveness of low-level laser on carpal tunnel syndrome. Medicine 2016, 95. [Google Scholar] [CrossRef]
- Cotler, H.B.; Chow, R.T.; Hamblin, M.R.; Carroll, J. The Use of Low Level Laser Therapy (LLLT) For Musculoskeletal Pain. MOJ Orthop. Rheumatol. 2015, 2. [Google Scholar] [CrossRef]
- Mester, E.; Mester, A.F.; Mester, A. The biomedical effects of laser application. Lasers Surg. Med. 1985, 5, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Rezende, A.B.; Ribeiro, M.S.; Nunez, S.C.; Garcia, V.G.; Maldonando, M.P. Effects of a single near-infrared laser treatment on cutaneous wound healing: Biometrical and histological study in rats. J. Photochem. Photobiol. B. 2007, 87, 145–153. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, Y.Y.; Wang, Y.; Lyu, P.; Hamblin, M.R. Photobiomodulation (blue and green light) encourages osteoblastic differentiation of human adipose-derived stem cells: Role of intracellular calcium and light-gated ion channels. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Preece, D.; Chow, K.W.; Gomez-Godinez, V.; Gustafson, K.; Esener, S.; Ravida, N.; Durrant, B.; Berns, M.W. Red light improves spermatozoa motility and does not induce oxidative DNA damage. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Bohil, C.J.; Alicea, B.; Biocca, F.A. Virtual reality in neuroscience research and therapy. Nat. Rev. Neurol. 2011, 12, 752. [Google Scholar] [CrossRef]
- Frigo, L.; Fávero, G.M.; Lima, H.J.; Maria, D.A.; Bjordal, J.M. Low-level laser irradiation (InGaAlP-660 nm) increases fibroblast cell proliferation and reduces cell death in a dose-dependent manner. Photomed. Laser Surg. 2010. [Google Scholar] [CrossRef]
- Basso, F.G.; Oliveira, C.F.; Kurachi, C.; Hebling, J.; Costa, C.A. Biostimulatory effect of low-level laser therapy on keratinocytes in vitro. Lasers Med. Sci. 2013. [Google Scholar] [CrossRef] [PubMed]
- Saygun, I.; Nizam, N.; Ural, A.U.; Serdar, M.A.; Avcu, F. Low-level laser irradiation affects the release of basic fibroblast growth factor (bFGF), insulin-like growth factor-I (IGF-I), and receptor of IGF-I (IGFBP3) from osteoblasts. Photomed. Laser Surg. 2012, 30, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Guedes, C.D.C.F.V.; de Freitas Filho, S.A.J.; de Faria, P.R.; Loyola, A.M.; Sabino-Silva, R.; Cardoso, S.V. Variation of Energy in Photobiomodulation for the Control of Radiotherapy-Induced Oral Mucositis: A Clinical Study in Head and Neck Cancer Patients. Int. J. Dent. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dolmans, D.E.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer. 2003. [Google Scholar] [CrossRef] [PubMed]
- Hopper, C. Photodynamic therapy: A clinical reality in the treatment of cancer. Lancet Oncol. 2000, 1, 212–219. [Google Scholar] [CrossRef]
- Detty, M.R.; Gibson, S.L.; Wagner, S.J. Current clinical and preclinical photosensitizers for use in photodynamic therapy. J. Med. Chem. 2004, 47, 3897–3915. [Google Scholar] [CrossRef]
- Patrice, T.; Moan, J.; Peng, Q. An outline of the history of PDT. In Comprehensive Series in Photochemical and Photobiological Sciences; Patrice, T., Ed.; Royal Society of Chemistry Publishing: Cambridge, UK, 2003; pp. 1–18. [Google Scholar] [CrossRef]
- Bechet, D.; Couleaud, P.; Frochot, C.; Viriot, M.L.; Guillemin, F.; Barberi-Heyob, M. Nanoparticles as vehicles for delivery of photodynamic therapy agents. Trends Biotechnol. 2008, 26, 612–621. [Google Scholar] [CrossRef]
- Konan, Y.N.; Gurny, R.; Allémann, E. State of the art in the delivery of photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B 2002, 66, 89–106. [Google Scholar] [CrossRef]
- Lim, C.K.; Heo, J.; Shin, S.; Jeong, K.; Seo, Y.H.; Jang, W.D.; Park, C.R.; Park, S.Y.; Kim, S.; Kwon, I.C. Nanophotosensitizers toward advanced photo-dynamic therapy of Cancer. Cancer Lett. 2013, 334, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.; Wang, L.; Cao, Y.; Ma, Y.; Shen, H.; Zhang, M.; Zhang, Z. Efficient cancer ablation by combined photothermal and enhanced chemo-therapy based on carbon nanoparticles/doxorubicin@SiO2 nanocomposites. Carbon 2016, 97, 35–44. [Google Scholar] [CrossRef]
- Yu-Wei, C.; Yu-Lin, S.; Shang-Hsiu, H.; San-Yuan, C. Functionalized graphene nanocomposites for enhancing photothermal therapy in tumor treatment. Adv. Drug Deliv. Rev. 2016, 105, 190–204. [Google Scholar]
- Chen, L.; Feng, W.; Zhou, X.; Qiu, K.; Miao, Y.; Zhang, Q.; Qin, M.; Li, L.; Zhang, Y.; He, C. Facile synthesis of novel albumin-functionalized flower-like MoS2 nanoparticles for in vitro chemo photothermal synergistic therapy. RSC Adv. 2016, 6, 13040–13049. [Google Scholar] [CrossRef]
- Houreld, N.; Abrahamse, H. Low-Intensity Laser Irradiation Stimulates Wound Healing in Diabetic Wounded Fibroblast Cells (WS1). Diabetes Technol. Ther. 2010, 12, 971–978. [Google Scholar] [CrossRef]
- Asgari, M.; Gazor, R.; Abdollahifar, M.; Fathabady, F.F.; Zare, F.; Norouzian, M.; Amini, A.; Khosravipour, A.; Kiani, P.; Atashgah, R.B.; et al. Combined therapy of adipose-derived stem cells and photobiomodulation on accelerated bone healing of a critical size defect in an osteoporotic rat model. Biochem. Biophys. Res. Commun. 2020, 530, 173–180. [Google Scholar] [CrossRef]
- Cherkasova, E.; Babak, K.; Belotelov, A.; Labutina, J.; Yusupov, V.; Vorobieva, N.; Nerush, A.; Maslennikova, A. Effects of photobiomodulation in relation to HeLa kyoto tumor cells exposed to ionizing radiation. J. Photochem. Photobiol. B Biol. 2020, 209, 111936. [Google Scholar] [CrossRef]
- Dhilip Kumar, S.S.; Houreld, N.N.; Abrahamse, H. Selective laser efficiency of green-synthesized silver nanoparticles by aloe arborescens and its wound healing activities in normal wounded and diabetic wounded fibroblast cells: In vitro studies. Int. J. Nanomed. 2020, 15, 6855–6870. [Google Scholar] [CrossRef]
- Hong, E.J.; Choi, D.G.; Shim, M.S. Targeted and effective photodynamic therapy for cancer using functionalized nanomaterials. Acta Pharm. Sin. B. 2016, 6, 297–307. [Google Scholar] [CrossRef]
- Chen, G.; Zhao, Y.; Xu, Y.; Zhu, C.; Liu, T.; Wang, K. Chitosan nanoparticles for oral photothermally enhanced photodynamic therapy of colon cancer. Int. J. Pharm. 2020, 589, 119763. [Google Scholar] [CrossRef]
- Zhang, C.; Qin, W.; Bai, X.; Zhang, X. Nanomaterials to relieve tumor hypoxia for enhanced photodynamic therapy. Nano Today 2020, 35, 100960. [Google Scholar] [CrossRef]
- Xu, D.D.; Leong, M.M.L.; Wong, F.; Lam, H.M.; Hoeven, R. Photodynamic therapy on prostate cancer cells involve mitochondria membrane proteins. Photodiagn. Photodyn. Ther. 2020, 31, 101933. [Google Scholar] [CrossRef] [PubMed]
- Nassar, A.; Mostafa, M.; Khashaba, S.A. Photodynamic therapy versus candida antigen immunotherapy in plane wart treatment: A comparative controlled study. Photodiagn. Photodyn. Ther. 2020, 32, 101973. [Google Scholar] [CrossRef] [PubMed]
- Qamar, Z.; Bamousa, B.; AlSheikh, R.; Ali, S.; Ali Baeshen, H.; Noushad, M.; Niazi, F.H. A bibliometric review of top 50 cited articles published in the photodiagnosis and photodynamic therapy from 2004 to 2019. Photodiagn. Photodyn. Ther. 2020, 31, 101927. [Google Scholar] [CrossRef]
- Junqi, C.; Chengyun, N.; Zhengnan, Z.; Peng, Y.; Ye, Z.; Guoxin, T.; Chuanbin, M. Nanomaterials as photothermal therapeutic agents. Prog. Mater. Sci. 2019, 99, 1–26. [Google Scholar]
- Zhi, D.; Yang, T.; O’Hagan, J.; Zhang, S.; Donnelly, R.F. Photothermal therapy. J. Control. Release 2020, 325, 52–71. [Google Scholar] [CrossRef]
- Tian, L.; Tao, L.; Li, H.; Zhao, S.; Zhang, Y.; Yang, S.; Xue, J.; Zhang, X. Hollow mesoporous carbon modified with cRGD peptide nanoplatform for targeted drug delivery and chemo-photothermal therapy of prostatic carcinoma. Colloids Surf. A Physicochem. Eng. Asp. 2019, 570, 386–395. [Google Scholar] [CrossRef]
- Allison, R.R.; Downie, G.H.; Cuenca, R.; Hu, X.-H.; Childs, C.J.; Sibata, C.H. Photosensitizers in clinical PDT. Photodiagn. Photodyn. Ther. 2004, 1, 27–42. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef]
- Kou, J.; Dou, D.; Yang, L. Porphyrin photosensitizers in photodynamic therapy and its applications. Oncotarget 2017, 8, 81591–81603. [Google Scholar] [CrossRef]
- Nyman, E.S.; Hynninen, P.H. Research advances in the use of tetrapyrrolic photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B 2004, 73, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Allison, R.R.; Sibata, C.H. Oncologic photodynamic therapy photosensitizers: A clinical review. Photodiagn. Photodyn. Ther. 2010, 7, 61–75. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, T.J.; Kaufman, J.E.; Goldfarb, A.; Weishaupt, K.R.; Boyle, D.; Mittleman, A. Photoradiation therapy for the treatment of malignant tumours. Cancer Res. 1978, 38, 2628–2635. [Google Scholar]
- Yoon, I.; Li, J.Z.; Shim, Y.K. Advance in photosensitizers and light delivery for photodynamic therapy. Clin. Endosc. 2013, 46, 7–23. [Google Scholar] [CrossRef]
- Nowak-Stepniowska, A.; Pergoł, P.; Padzik-Graczyk, A. Photodynamic method of cancer diagnosis and therapy–mechanisms and applications. Postepy Biochem. 2013, 59, 53–63. [Google Scholar]
- Kataoka, H.; Nishie, H.; Hayashi, N.; Tanaka, M.; Nomoto, A.; Yano, S.; Joh, T. New photodynamic therapy with next-generation photosensitizers. Ann. Transl. Med. 2017, 5. [Google Scholar] [CrossRef]
- Ormond, A.B.; Freeman, H.S. Dye Sensitizers for Photodynamic Therapy. Materials 2013, 6, 817–840. [Google Scholar] [CrossRef]
- Kato, H.; Romka, C.; Ono, J.; Saito, T.; Matsushima, Y.; Iimura, I.; Tahara, M.; Kawate, N.; Yoneyama, K.; Nishimiya, K.; et al. Cancer localization by detection of fluorescence by means of HpD administration and krypton ion laser photoradiation in canine lung cancer. Lung Cancer 1981, 21, 439–445. [Google Scholar]
- Madan, V.; Loncaster, J.; Allan, D.; Lear, J.; Sheridan, L.; Leach, C.; Allan, E. Systemic photodynamic therapy with photofrin for naevoid basal cell carcinoma syndrome—A pilot study. Photodiagn. Photodyn. Ther. 2005, 2, 273–281. [Google Scholar] [CrossRef]
- Pushpan, S.K.; Venkatraman, S.; Anand, V.G.; Sankar, J.; Parmeswaran, D.; Ganesan, S.; Chandrashekar, T.K. Porphyrins in photodynamic therapy—A search for ideal photosensitizers. Curr. Med. Chem. Anti-Cancer Agents 2002, 2, 187–207. [Google Scholar] [CrossRef] [PubMed]
- Batlle, A. Porphyrins, porphyrias, cancer and photodynamic therapy—A model for carcinogenesis. J. Photochem. Photobiol. B 1993, 20, 5–22. [Google Scholar] [CrossRef]
- Morton, C.A.; Brown, S.B.; Collins, S.; Ibbotson, S.; Jenkinson, H.; Kurwa, H.; Langmack, K.; McKenna, K.; Moseley, H.; Pearse, A.D.; et al. Guidelines for topical photodynamic therapy: Report of a workshop of the British Photodermatology Group. J. Dermatol. 2002, 146, 552–567. [Google Scholar] [CrossRef]
- Peng, Q.; Berg, K.; Moan, J.; Kongshaug, M.; Nesland, J.M. 5-Aminolevulinic acid-based photodynamic therapy: Principles and experimental research. Photochem. Photobiol. 1997, 65, 235–251. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration, Center for Drug Evaluation and Research. Metvixia NDA 21-415 Approval Letter, 25 May 2004. Retrieved 5 January 2013. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/nda/2004/021415s000_Metvixia_Aprov.pdf (accessed on 28 February 2013).
- Gilchrest, B.A. Photodynamic Therapy and Selected Off-label Uses. In Proceedings of the Winter Clinical Dermatology Conference, Kohala Coast, HI, USA, 23–28 January 2010; Tuleya, S., Ed.; HMP Communications, LLC: Malvern, PA, USA, 2010; pp. 10–12. [Google Scholar]
- PhotoCure. A Double Blinded, Prospective, Randomized, Vehicle Controlled Multi-Center Study of Photodynamic Therapy with Visonac® Cream in Patients with Acne Vulgaris. In ClinicalTrials.Gov [Internet]; National Library of Medicine (US): Bethesda, MD, USA, NLM Identifier: NCT01347879. Available online: http://clinicaltrials.gov/show/NCT01347879 (accessed on 6 January 2013).
- Furre, I.E.; Shahzidi, S.; Luksiene, Z.; Moller, M.T.N.; Borgen, E.; Morgan, J.; Tkacz-Stachowska, K.; Nesland, J.M.; Peng, Q. Targeting PBR by hexaminolevulinate-mediated photodynamic therapy induces apoptosis through translocation of apoptosis-inducing factor in human leukemia cells. Cancer Res. 2005, 65, 11051–11060. [Google Scholar] [CrossRef] [PubMed]
- PhotoCure. Dose-Finding Study of Hexaminolevulinate (HAL) Photodynamic Therapy (PDT) to Treat Cervical Neoplasia. In ClinicalTrials.Gov [Internet]; National Library of Medicine (US): Bethesda, MD, USA, NLM Identifier: NCT01256424. Available online: http://clinicaltrials.gov/show/NCT01256424 (accessed on 5 January 2012).
- Oslo University Hospital. Treatment of Female Genital Erosive Lichen Planus(GELP) with Hexaminolevulinate PDT. In ClinicalTrials.Gov [Internet]; National Library of Materials 2013, 6 837 Medicine (US): Bethesda, MD, USA, NLM Identifier: NCT01282515. Available online: http://clinicaltrials.gov/show/NCT01282515 (accessed on 6 January 2012).
- Patel, H.; Mick, R.; Finlay, J.C.; Zhu, T.C.; Rickter, E.; Cengel, K.A.; Malkowicz, S.B.; Hahn, S.M.; Busch, T.M. Motexafin lutetium-photodynamic therapy of prostate cancer: Short- and long-term effects of prostate-specific antigen. Clin. Cancer Res. 2008, 14, 4869–4876. [Google Scholar] [CrossRef] [PubMed]
- University of Pittsburgh. Photodynamic Therapy using Lutetium Texaphyrin in Treating Patients with Cervical Intraepithelial Neoplasia. In ClinicalTrials.gov [Internet]; National Library of Medicine (US): Bethesda, MD, USA, NLM Identifier: NCT00005808. Available online: http://clinicaltrials.gov/show/NCT00005808 (accessed on 5 January 2013).
- Josefsen, L.B.; Boyle, R.W. Photodynamic therapy and the development of metal-based photosensitizers. Met. Based Drugs 2008, 1–24. [Google Scholar] [CrossRef]
- Mody, T.D.; Sessler, J.L. Texaphyrins: A new approach to drug development. J. Porphyr. Phthalocyanines 2001, 5, 134–142. [Google Scholar] [CrossRef]
- Rousset, N.; Bourre, L.; Thibaud, S. Sensitizers in Photodynamic Therapy. In Photodynamic Therapy; Patrice, T., Ed.; Royal Society of Chemistry: Cambridge, UK, 2003. [Google Scholar]
- Triesscheijn, M.; Ruevekamp, M.; Aalders, M.; Baas, P.; Stewart, F.A. Outcome of mTHPC mediated photodynamic therapy is primarily determined by the vascular response. Photochem. Photobiol. 2005, 81, 1161–1167. [Google Scholar] [CrossRef]
- Stapleton, M.; Rhodes, L.E. Photosensitizers for photodynamic therapy of cutaneous disease. J. Dermatol. Treat. 2003, 14, 107–112. [Google Scholar] [CrossRef]
- Bonnett, R. Photosensitizers of the porphyrin and phthalocyanine series for photodynamic therapy. Chem. Soc. Rev. 1995, 24, 19–33. [Google Scholar] [CrossRef]
- Santoro, O.; Bandieramonte, G.; Melloni, E.; Marchesini, R.; Zunino, F.; Lepera, P.; de Palo, G. Photodynamic therapy by topical meso-tetraphenylporphinesulfonate tetrasodium salt administration in superficial basal cell carcinoma. Cancer Res. 1990, 50, 4501–4503. [Google Scholar]
- Usuda, J.; Kato, H.; Okunaka, T.; Furukawa, K.; Tsutsi, H.; Yamada, K.; Suga, Y.; Honda, H.; Nagatsuka, Y.; Ohira, T.; et al. Photodynamic therapy (PDT) for lung cancers. J. Thorac. Oncol. 2006, 1, 489–493. [Google Scholar] [CrossRef]
- Kobayashi, W.; Liu, Q.; Nakagawa, H.; Sakaki, H.; Teh, B.; Matsumiya, T.; Yoshida, H.; Imaizumi, T.; Satoh, K.; Kimura, H. Photodynamic therapy with mono-L-aspartyl chlorin e6 can cause necrosis of squamous cell carcinoma of tongue: Experimental study on an animal model of nude mouse. Oral Oncol. 2006, 42, 46–50. [Google Scholar] [CrossRef]
- Lobel, J.; MacDonald, I.J.; Ciesielski, M.J.; Barone, T.; Potter, W.R.; Pollina, J.; Plunkett, R.J.; Fenstermaker, R.A.; Dougherty, T.J. 2-[1-Hexyloxyethyl]-2-Devinyl Pyropheophorbide-a (HPPH) in a nude rat glioma model: Implications for photodynamic therapy. Lasers Surg. Med. 2001, 29, 397–405. [Google Scholar] [CrossRef]
- Roswell Park Cancer Institute. Photodynamic Therapy using HPPH in Treating Patients with Recurrent Dysplasia, Carcinoma in Situ, or Stage I Oral Cavity Cancer. In ClinicalTrials.gov [Internet]; National Library of Medicine (US): Bethesda, MD, USA, NLM Identifier: NCT00670397. Available online: http://clinicaltrials.gov/show/NCT00670397 (accessed on 5 January 2013).
- Chen, Y.; Zheng, W.; Li, Y.; Zhong, J.; Ji, J.; Shen, P. Apoptosis induced by methylene-blue-mediated photodynamic therapy in melanolmas and the involvement of mitochondrial dysfunction revealed by proteomics. Cancer Sci. 2008, 99, 2019–2027. [Google Scholar]
- Lu, Y.; Jiao, R.; Chen, X.; Zhong, J.; Ji, J.; Shen, P. Methylene blue-mediated photodynamic therapy induces mitochondria-dependent apoptosis in HeLa cell. J. Cell. Biochem. 2008, 105, 1451–1460. [Google Scholar] [CrossRef]
- Brun, P.H.; DeGroot, J.L.; Dickson, E.F.G.; Farahani, M.; Pottier, R.H. Determination of the in vivo pharmacokinetics of palladium-bacteriopheophorbide (WST09) in EMT6 tumor-bearing Balb/c mice using graphite furnace atomic absorption spectroscopy. Photochem. Photobiol. Sci. 2004, 3, 1006–1010. [Google Scholar] [CrossRef]
- Zimcik, P.; Miletin, M. Photodynamic Therapy. In Dyes and Pigments: New Research; Lang, A.R., Ed.; Nova Science Publishers: New York, NY, USA, 2008. [Google Scholar]
- Waranyoupalin, R.; Wongnawa, S.; Wongnawa, M.; Pakawatchai, C.; Panichayupakaranant, P.; Sherdshoopongse, P. Studies on complex formation between curcumin and Hg(II) ion by spectrophotometric method: A new approach to overcome peak overlap. Cent. Eur. J. Chem. 2009, 7, 388–394. [Google Scholar] [CrossRef]
- Araujo, N.C.; Fontana, C.R.; Gerbi, M.E.M.; Bagnato, V.S. Overall-mouth disinfection by photodynamic therapy using curcumin. Photomed. Laser Surg. 2012, 30, 96–101. [Google Scholar] [CrossRef]
- Mousavi, S.H.; Tavakkol-Afshari, J.; Brook, A.; Jafari-Anarkooli, I. Direct toxicity of rose Bengal in MCF-7 cell line: Roloe of apoptosis. Food Chem. Toxicol. 2009, 47, 855–859. [Google Scholar] [CrossRef]
- Guimond, M.; Balassy, A.; Barrette, M.; Brochu, S.; Perreault, C.; Roy, D.C. P-glycoprotein targeting: A unique strategy to selectively eliminate immunoreactive T cells. Blood 2002, 100, 375–382. [Google Scholar] [CrossRef]
- Sanovic, R.; Krammer, B.; Grumboeck, S.; Verwanger, T. Time-resolved gene expression profiling of human squamous cell carcinoma cells during the apoptosis process induced by photodynamic treatment with hypericin. Int. J. Oncol. 2009, 35, 921–939. [Google Scholar]
- Kacerovska, D.; Pizinger, K.; Majer, F.; Smid, F. Photodynamic therapy of nonmelanoma skin cancer with topical hypericum perforatum extract-a pilot study. Photochem. Photobiol. 2008, 84, 779–785. [Google Scholar] [CrossRef]
- Erola, O.; Uyana, I.; Hatipa, M.; Yilmaza, C.; Tekinaya, A.; Gulera, M.O. Recent advances in bioactive 1D and 2D carbon nanomaterials for biomedical applications. Nanomedicine 2018, 14, 2433–2454. [Google Scholar] [CrossRef]
- Mehra, N.K.; Palakurthi, S. Interactions between carbon nanotubes and bioactives: A drug delivery perspective. Drug Discov. Today 2016, 21, 585–597. [Google Scholar] [CrossRef]
- Singh, B.; Baburao, C.; Pispati, V.; Pathipati, H.; Muthy, N.; Prassana, S.; Rathode, B.G. Carbon nanotubes: A novel drug delivery system. Int. J. Res. Pharm. Chem. 2012, 2, 523–532. [Google Scholar]
- He, H.; Pham-Huy, L.A.; Dramou, P.; Xiao, D.; Zuo, P.; Pham-Huy, C. Carbon nanotubes: Applications in pharmacy and medicine. Biomed Res. Int. 2013. [Google Scholar] [CrossRef]
- Donaldson, K.; Aitken, R.; Tran, L.; Stone, V.; Duffin, R.; Forrest, G.; Alexander, A. Carbon nanotubes: A review of their properties in relation to pulmonary toxicology and workplace safety. Toxicol. Sci. 2006, 92, 5–22. [Google Scholar] [CrossRef]
- Hou, P.; Bai, S.; Yang, Q.; Liu, C.; Cheng, H. Multi-step purification of carbon nanotubes. Carbon 2002, 40, 81–85. [Google Scholar] [CrossRef]
- Lei, W.; Jinjin, S.; Ruiyuan, L.; Yan, L.; Jing, Z.; Xiaoyuan, Y.; Jun, G.; Chaofeng, Z.; Zhenzhong, Z. Photodynamic effect of functionalized singlewalled carbon nanotubes: A potential sensitizer for photodynamic therapy. Nanoscale 2014, 6, 4642–4652. [Google Scholar]
- Marangon, I.; Ménard-Moyon, C.; Silva, A.K.A.; Bianco, A.; Luciani, N.; Gazeau, F. Synergic mechanisms of photothermal and photodynamic therapies mediated by photosensitizer/carbon nanotube complexes. Carbon 2016, 97, 110–123. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.; Wang, J.; Li, J.; Lin, Y. Graphene and graphene oxide: Biofunctionalization and applications in biotechnology. Trends Biotechnol. 2011, 29, 205–212. [Google Scholar] [CrossRef]
- Kalluru, P.; Vankayala, R.; Chiang, C.S.; Hwang, K.C. Nano-graphene oxide-mediated In vivo fluorescence imaging and bimodal photodynamic and photothermal destruction of tumors. Biomaterials 2016, 95, 1–10. [Google Scholar] [CrossRef]
- Shi, S.; Chen, F.; Ehlerding, E.B.; Cai, W. Surface engineering of graphene-based nanomaterials for biomedical applications. Bioconjug. Chem. 2014, 25, 1609–1619. [Google Scholar] [CrossRef]
- Chen, R.J.; Zhang, Y.; Wang, D.; Dai, H. Noncovalent sidewall functionalization of single-walled carbon nanotubes for protein immobilization. J. Am. Chem. Soc. 2001, 123, 3838–3839. [Google Scholar] [CrossRef]
- Lee, D.J.; Park, S.Y.; Oh, Y.T.; Oh, N.M.; Oh, K.T.; Youn, Y.S. Preparation of chlorine e6-conjugated single walled carbon nanotube for photodynamic therapy. Macromol Res. 2011, 19, 848–852. [Google Scholar] [CrossRef]
- Bilalis, P.; Katsigiannopoulos, D.; Avgeropoulos, A.; Sakellario, G. Noncovalent functionalization of carbon nanotubes with polymers. RSC Adv. 2014, 4, 2911–2934. [Google Scholar] [CrossRef]
- Besteman, K.; Lee, J.O.; Wiertz, F.G.; Heering, H.A.; Dekker, C. Enzyme-coated carbon nanotubes as single-molecule biosensors. Nano Lett. 2003, 3, 727–730. [Google Scholar] [CrossRef]
- Siu, K.S.; Chen, D.; Zheng, X.; Zhang, X.; Johnston, N.; Liu, Y.; Yuan, K.; Koropatnick, J.; Gillies, E.R.; Min, W.P. Non-covalently functionalized single-walled carbon nanotube for topical siRNA delivery into melanoma. Biomaterials 2014, 35, 3435–3442. [Google Scholar] [CrossRef] [PubMed]
- Paloniemi, H.; Ääritalo, T.; Laih, T.; Like, H.; Kocharova, N.; Haapakka, K.; Terzi, F.; Seeber, R.; Lukkari, J. Water-soluble full-length single-wall carbon nanotube polyelectrolytes: Preparation and characterization. J. Phys. Chem. B 2005, 109, 8634–8642. [Google Scholar] [CrossRef]
- Ogbodu, R.O.; Antunes, E.; Nyokong, T. Physicochemical properties of zinc monoaminophthalocyanine conjugated to folic acid and single walled carbon nanotubes. Polyhedron 2013, 60, 59–67. [Google Scholar] [CrossRef]
- Wu, C.; Li, D.; Wang, L.; Guan, X.; Tian, Y.; Yang, H.; Li, S.; Liu, Y. Single wavelength light-mediated synergistic bimodal cancer photoablation and amplified photothermal performance by graphene/gold nanostar/photosensitizer theranostics. Acta Biomater. 2017, 53, 631–642. [Google Scholar] [CrossRef]
- Xie, L.; Wang, G.; Zhou, H.; Zhang, F.; Guo, Z.; Liu, C.; Zhang, X.; Zhu, L. Functional long circulating single walled carbon nanotubes for Fluorescent/photoacoustic imaging-guided enhanced phototherapy. Biomaterials 2016, 103, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, N.G.; Cheng, H.K.F.; Cai, J.; Li, L.; Chan, S.H.; Zhao, J.; Yo, S. Improvement of mechanical and thermal properties of carbon nanotube composites through nanotube functionalization and processing methods. Mater. Chem. Phys. 2009, 117, 313–320. [Google Scholar] [CrossRef]
- Park, H.; Zhao, J.; Lu, J.P. Effects of Sidewall Functionalization on Conducting Properties of Single Wall Carbon Nanotubes. Nano Lett. 2006, 5, 916–919. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Liu, B.; Chen, Y.L.; Jiang, H.; Hwang, K.C.; Huang, Y. Mechanical properties of functionalized carbon nanotubes. Nanotechnology 2008, 19, 395702. [Google Scholar] [CrossRef]
- Banerjee, S.; Hemraj-Benny, T.; Wong, S.S. Covalent surface chemistry of single walled carbon nanotubes. Adv. Mater. 2005, 17, 17–29. [Google Scholar] [CrossRef]
- Carrillo, A.; Swartz, J.A.; Gamba, J.M.; Kane, R.S. Noncovalent Functionalization of Graphite and Carbon Nanotubes with Polymer Multilayers and Gold Nanoparticles. Nano Lett. 2003, 3, 1437–1440. [Google Scholar] [CrossRef]
- Xie, J.; Lee, S.; Chen, X. Nanoparticle-based theranostic agents. Adv. Drug Deliv. Rev. 2010, 62, 1064–1079. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Gu, Z.; Huang, D.; Liu, Z.; Guo, X.; Zhong, H. Synergistic effect of chemo-photothermal therapy using PEGylated graphene Oxide. Biomaterials 2011, 32, 8555–8561. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Chiou, S.; Chou, C.; Chen, Y.; Huang, Y.; Peng, C. Photothermolysis of glioblastoma stem-like cells targeted by carbon nanotubes conjugated with CD133 monoclonal antibody. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Nakatsuji, H.; Inada, M.; Matoba, Y.; Umeyama, T.; Tsujimoto, M.; Isoda, S.; Hashida, M.; Imahori, H. Photodynamic and Photothermal Effects of Semiconducting and Metallic-Enriched Single-Walled Carbon Nanotubes. J. Am. Chem. Soc. 2012, 134, 17862–17865. [Google Scholar] [CrossRef]
- Ogbodu, R.O.; Limson, J.L.; Prinsloo, E.; Nyokong, T. Photophysical properties and photodynamic therapy effect of zinc phthalocyanine-spermine-single walled carbon nanotube conjugate on MCF-7 breast cancer cell line. Synth. Metals 2015, 204, 122–132. [Google Scholar] [CrossRef]
- Xiao, H.; Zhu, B.; Wang, D.; Pang, Y.; He, L.; Ma, X.; Wang, R.; Jin, C.; Chen, Y.; Zhu, X. Photodynamic effects of chlorin e6 attached to single wall carbon nanotubes through noncovalent interactions. Carbon 2012, 50, 1681–1689. [Google Scholar] [CrossRef]
- Hou, L.; Yuan, Y.; Ren, J.; Zhang, Y.; Wang, Y.; Shan, X.; Liu, Q.; Zhang, Z. In vitro and in vivo comparative study of the phototherapy anticancer activity of hyaluronic acid-modified single-walled carbon nanotubes, graphene oxide, and fullerene. J. Nanopart. Res. 2017, 19, 286. [Google Scholar] [CrossRef]
- Ogbodu, R.O.; Ndhundhuma, I.; Karsten, A.; Nyokong, T. Photodynamic therapy effect of zinc monoaminophthalocyanine–folic acid conjugate adsorbed on single walled carbon nanotubes on melanoma cells. Spectrochim. Acta A 2015, 137, 1120–1125. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, M.; Zhang, N.; Shi, J.; Zhang, H.; Li, M.; Lu, C.; Zhang, Z. Synergistic enhancement of cancer therapy using a combination of docetaxel and photothermal ablation induced by single-walled carbon nanotubes. Int. J. Nanomed. 2011, 2642–2652. [Google Scholar] [CrossRef]
- Neelgund, G.M.; Oki Aderemi, R. Influence of carbon nanotubes and graphene nanosheets on photothermal effect of hydroxyapatite. J. Colloid Interface Sci. 2016, 484, 135–145. [Google Scholar] [CrossRef]
- Khan, M.S.; Pandey, S.; Bhaisare, M.L.; Gedda, G.; Talib, A.; Wu, H.F. Graphene oxide @gold nanorods for chemo-photothermal treatment and controlled release of doxorubicin in mice Tumor. Colloids Surf. B Biointerfaces 2017, 160, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Lima-Sousa, R.; de Melo-Diogo, D.; Alves, C.G.; Costa, E.C.; Ferreira, P.; Louro, R.O.; Correia, I.J. Hyaluronic acid functionalized green reduced graphene oxide for targeted cancer photothermal therapy. Carbohydr. Polym. 2018, 200, 93–99. [Google Scholar] [CrossRef]
- Thapa, R.K.; Soe, Z.C.; Ou, W.; Poudel, K.; Jeong, J.-H.; Jin, S.G.; Kwang Ku, S.; Choi, H.-G.; Lee, Y.M.; Yong, C.S.; et al. Palladium nanoparticle-decorated 2-D graphene oxide for effective photodynamic and photothermal therapy of prostate solid tumors. Collides Surf. B Biointerfaces 2018, 169, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Hong, L.; Li, H.; Liu, C. Graphene oxide-fullerene C60 (GO-C60) hybrid for photodynamic and photothermal therapy triggered by near-infrared light. Biosens. Bioelectron. 2017, 89, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wen, L.; Huang, R.; Wang, H.; Hu, X.L.; Xing, D. Mitochondrial specific photodynamic therapy by rare-earth nanoparticles mediated near-infrared graphene quantum dots. Biomaterials 2018, 153, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.S.; Kong, W.H.; Sung, D.K.; Lee, M.Y.; Beack, S.E.; Keumdo, H.; Kim, K.S.; Yun, S.H.; Hahn, S.K. Nanographene oxide–hyaluronic acid conjugate for photothermal ablation therapy of skin cancer. ACS Nano 2014, 8, 260–268. [Google Scholar] [CrossRef]
- Mauro, N.; Scialabba, C.; Agnello, S.; Cavallaro, G.; Giammona, G. Folic acid-functionalized graphene oxide nanosheets via plasma etching as a platform to combine NIR anticancer phototherapy and targeted drug delivery. Mater. Sci. Eng. C 2020, 107, 110201. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, P.; Abrahamse, H. Effective photodynamic therapy for colon cancer cells using chlorin e6 coated hyaluronic acid-based carbon nanotubes. Int. J. Mol. Sci. 2020, 21, 4745. [Google Scholar] [CrossRef]
| Wavelengths | Lamps | Mechanism of Action | Applications | Reference | |
|---|---|---|---|---|---|
| PBM | 500–1100 nm | Neodymium:yttrium-aluminium-garnet (Nd:YAG) laser, helium-neon laser (He-Ne), Diode laser. | The low-level light source at a particular wavelength applied into the cells will stimulate or enhance the cells. | Rheumatoid arthritis, osteoarthritis, wound healing, low back pain. | [37,38,39,40] |
| PDT | 400–800 nm | NIR lasers, diode lasers, UV lights. | The PS molecule in the ground state; when the molecule activated by the light source reaches the excited the state, it converts to the triplet state by electron spinning. The triplet state interacts with the surrounding oxygen molecule and produces ROS through type I and type II reaction | Antimicrobial, fungal, viral activity, acne vulgaris, malignant tumour (lung, skin, head and neck, prostate cancer), wound healing. | [41,42,43,44,45,46] |
| PTT | 800–980 nm | NIR lasers, UV lights. | The particle (PS or nanoparticles) will be activated by the light source and produce heat energy. | Prostate cancer, melanoma skin cancer, Alzheimer’s disease. | [47,48,49] |
| PSs | Structure | Λ(nm) and (Εmax) | Application |
|---|---|---|---|
| HpD |  | 630 nm and 3000 (M−1 cm−1) | Brain, Lung cancer [60,61] |
| Photofrin |  | 630 nm and 3000 (M−1 cm−1) | Bladder, Lung, Oesophagus cancer [62,63] |
| 5-Aminole-vulinic acid |  | 632 nm and 5000 (M−1 cm−1) | Cancer diagnosis [64,65,66] |
| Methyl aminolevulinate | 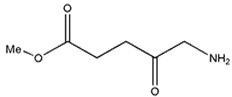 | 632 nm and 5000 (M−1 cm−1) | Nonmelanoma cancer, Basal cell carcinoma [67,68,69] |
| Hexaminolevulinate | 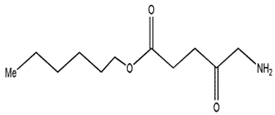 | 632 nm and 5000 (M−1 cm−1) | Bladder cancer diagnosis [70,71,72] |
| Lu-Tex | 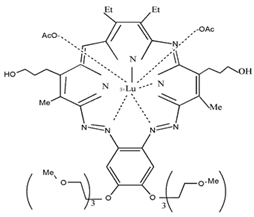 | 732 nm and 42,000 (M−1 cm−1) | Prostate cancer, Breast cancer, Cervical cancer [73,74,75] |
| Meta-tetra(hy-droxyphe-nyl)por-phyrin |  | 648 nm | Basal cell Carcinoma, Head and neck cancer and Skin cancer [76,77,78] |
| 5,10,15,20-tetrakis(4-sulfanato-phenyl)-21H,23H-porphyrin | 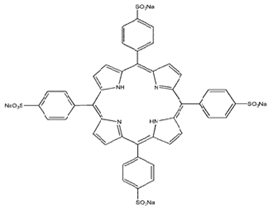 | 645 nm | Basal cell Carcinoma [27,63,79,80] |
| N-aspartyl chlorin e6 | 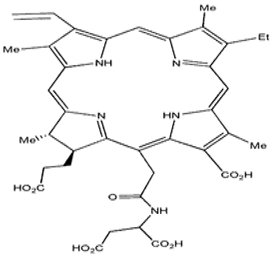 | 664 nm and 40,000 (M−1 cm−1) | Lung cancer [81,82] |
| 2-(1-hexylo-xyethyl)-2-devinyl Pyropheo-phorbide | 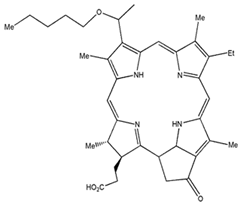 | 665 nm and 47,000 (M−1 cm−1) | Oesophageal cancer, Basal cell carcinoma, Lung cancer [83,84] |
| Phenothia-zines | 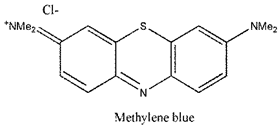 | 666 nm and 82,000 (M−1 cm−1) | Basal cell Carcinoma, Kaposi’s sarcoma, Cervical cancer [85,86] |
| Padoporfin | 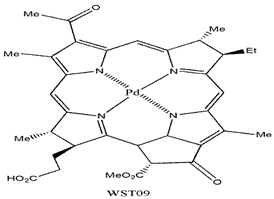 | 763 nm and 88,000 (M−1 cm−1) | Prostate cancer [87] |
| Aluminium phthalo-cyanine-tetrasulfo-nate | 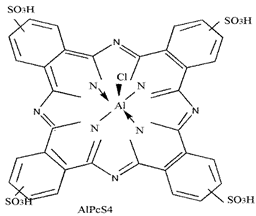 | 676 nm and 200,000 (M−1 cm−1) | Stomach, Skin, Oral, Breast cancer [88] |
| Curcumi-noids | 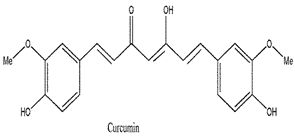 | 420 nm and 55,000 (M−1 cm−1) | Breast, Skin cancer [89,90] |
| Xanthenes | 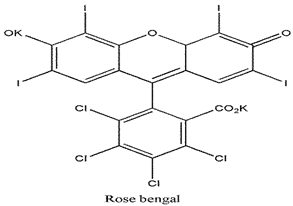 | 549 nm and 100,000 (M−1 cm−1) | Breast carcinoma and Metastatic melanoma [91] |
| 4,5-Dibromorhodamine methyl ester |  | 514 nm and 100,000 (M−1 cm−1) | Breast cancer [92] |
| Anthraquinones |  | 590 nm and 44,000 (M−1 cm−1) | Squamous cell carcinoma and Basal cell carcinoma [93,94] |
| Meta-tetra(hydroxyphenyl)chlorin | 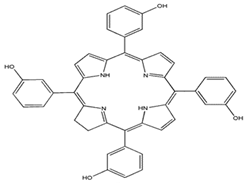 | 652 nm and 35,000 (M−1 cm−1) | Breast, Prostate, Pancreatic cancer, Head and neck cancer [77,80,95] |
| Nanoparticle | Photosensitizer | Bonding | Photo Therapy | Applications | Targeted | References |
|---|---|---|---|---|---|---|
| SWCNTs | - | Covalent (Carboxyl-amine cross-linking) | PTT | Glioblastoma cells | Anti CD133 | [124] |
| SWCNTs | SWCNT-PEI SWCNT-PVPk30 | Covalent (cationic polymerization) and noncovalent (physical attachment) | PDT | Mus musculus skin melanoma cells (B16-F10 cells) | - | [102] |
| Metallic-SWCNT Semiconducting-SWCNT | - | - | PDT PTT | Lung cancer cells (NCI-H460) | - | [125] |
| MWCNT | m-tetrahydroxylphenylcholrin | Non-covalent (physical attachment) | PDT PTT | Ovarian cancer cells (SKOV3) | - | [103] |
| SWCNT (Evans blue and albumin) | Chlorin e6 | Non-covalent (physical attachment) | PDT/PTT | Mouse squamous cancer cells (SCC-7) and In vivo | - | [116] |
| SWCNT | Zinc phthalocyanine | Covalent (carboxyl-amine cross-linking) and non-covalent (physical attachment) | PDT | Breast cancer cells (MCF 7) | Spermine | [126] |
| SWCNT (chitosan) | Chlorin e6 | Non-covalent (π-π interaction) | PDT | HeLa cancer cells | - | [127] |
| SWCNT, GO, Fullerene | - | Covalent (Carboxyl-amine cross linking) | PTT | Breast cancer cells (MCF 7) and In vivo | Hyaluronic acid | [128] |
| SWCNT | Zinc monoamino phthalocyanine | Covalent (Carboxyl-amine cross linking) and non- covalent (π-π interaction) | PDT/PTT | Melanoma cells (A375) | Folic acid | [129] |
| SWCNT (Docetaxel NGR peptide) | - | Non-covalent (physical attachment) | PTT | Human prostate cancer (PC3) and in vivo | - | [130] |
| CNT Graphene sheet | Hydroxyapatite | Non-Covalent (physical attachment) | PTT | - | - | [131] |
| Graphene oxide-PEG-DOX | - | Covalent (Carboxyl-amine cross-linking) | PTT | Murine mammary cancer cells (EMT6) and in vivo | - | [123] |
| Graphene oxide-gold nanorods-Doxorubicin | - | Noncovalent (physical attachment) | PTT | Lung cancer cells (A549) | - | [132] |
| Reduced graphene oxide | - | Noncovalent (physical attachment) | PTT | Human breast cancer (MCF 7) | Hyaluronic acid | [133] |
| Graphene oxide Palladium | - | Covalent (Carboxyl-amine cross-linking) | PDT PTT | Human prostate cancer (PC3) | - | [134] |
| Graphene | - | - | PDT PTT | Cervical cancer cells (HeLa) | - | [135] |
| Graphene oxide-PEG | - | Covalent (Carboxyl-amine cross-linking) | PDT PTT | Melanoma cells (B16F0) | Folate | [106] |
| Graphene oxide (Quantum dots) | TRITC | Covalent (Carboxyl-amine cross-linking) Noncovalent (physical attachments) | PDT | Mouse mammary tumour cells (4T1) | Upconversion nanoparticle (UCNP) | [136] |
| Graphene | - | Covalent (Carboxyl-amine cross-linking) | PTT | Skin cancer (in vivo) | Hyaluronic acid | [137] |
| Graphene oxide | Chlorin e6 | Noncovalent (π-π interaction) | PDT/ PTT | Mouse breast mammary carcinoma cells (EMT6) | - | [115] |
| Graphene oxide-PEG-Folic acid | - | Covalent (Carboxyl-amine cross-linking) | PTT | Human breast cancer (MCF 7 and MDA-MB-231) | Folate | [138] |
| SWCNTs | Chlorin e6 | Non-covalent (π-π interaction) | PDT | Human colon cancer cells (Caco 2) | Hyaluronic acid | [139] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sundaram, P.; Abrahamse, H. Phototherapy Combined with Carbon Nanomaterials (1D and 2D) and Their Applications in Cancer Therapy. Materials 2020, 13, 4830. https://doi.org/10.3390/ma13214830
Sundaram P, Abrahamse H. Phototherapy Combined with Carbon Nanomaterials (1D and 2D) and Their Applications in Cancer Therapy. Materials. 2020; 13(21):4830. https://doi.org/10.3390/ma13214830
Chicago/Turabian StyleSundaram, Prabhavathi, and Heidi Abrahamse. 2020. "Phototherapy Combined with Carbon Nanomaterials (1D and 2D) and Their Applications in Cancer Therapy" Materials 13, no. 21: 4830. https://doi.org/10.3390/ma13214830
APA StyleSundaram, P., & Abrahamse, H. (2020). Phototherapy Combined with Carbon Nanomaterials (1D and 2D) and Their Applications in Cancer Therapy. Materials, 13(21), 4830. https://doi.org/10.3390/ma13214830






