Regeneration Mechanism of Sulfur Absorption Via Samarium-doped Cerium Adsorbents in the Gas Atmosphere of O2/N2
Abstract
1. Introduction
2. Experimental
2.1. Preparation of the Sorbents
2.2. Preparation of the Ce-O-S Powder and Corresponding Regenerated Powders
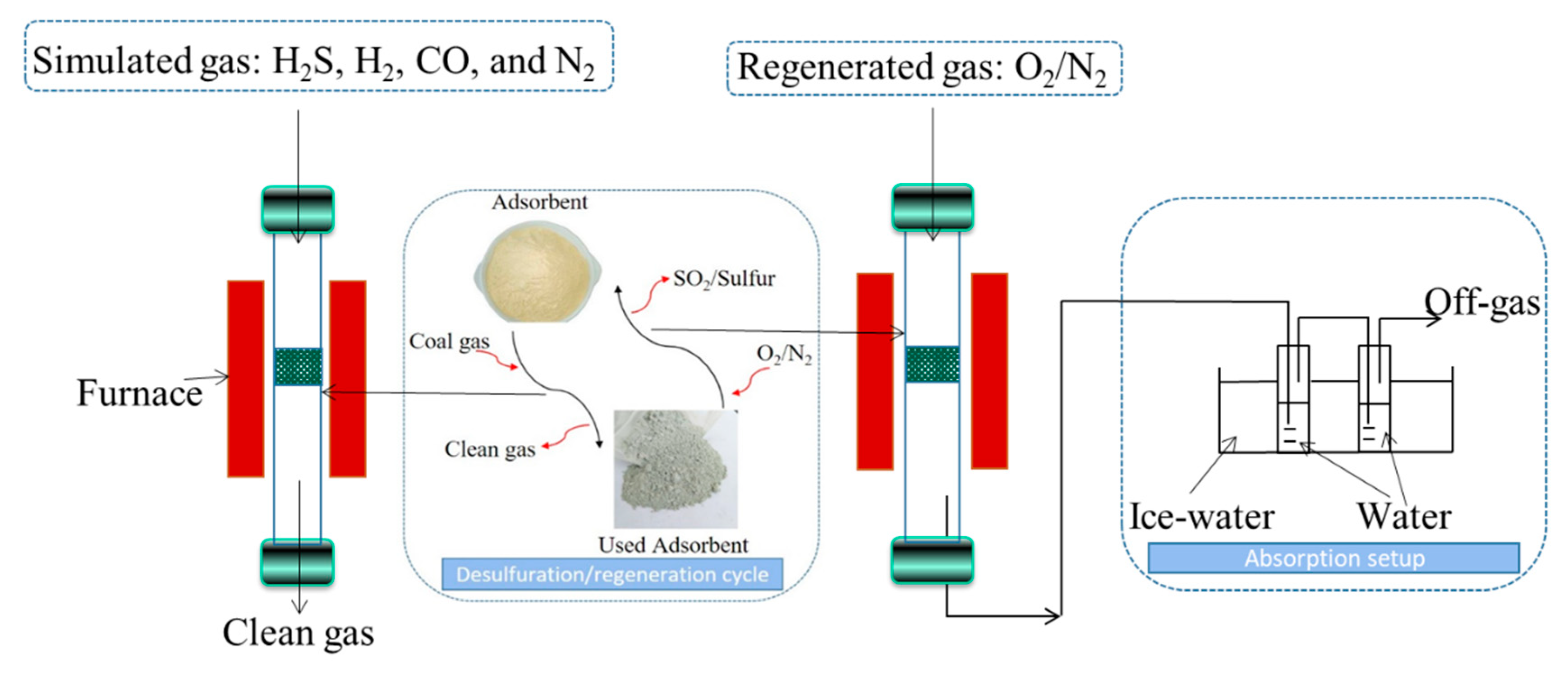
2.3. Desulfurization and Regeneration Assessments
2.4. Sulfur Collection Test
2.5. Characterizations
3. Results and Discussion
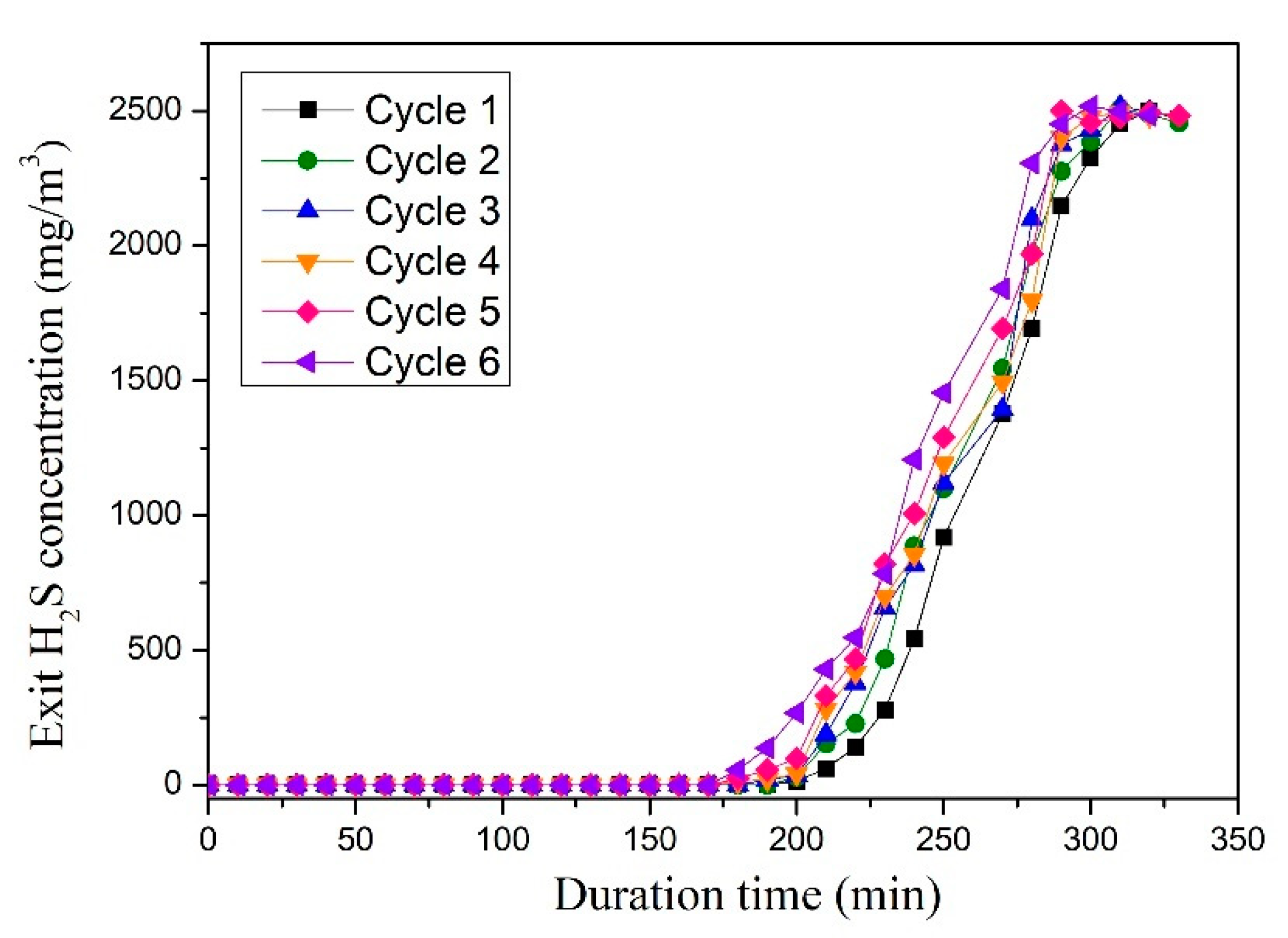
3.1. Performance of the Sorbents
3.2. Regeneration Mechanism of the SDC Sorbent
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.; Yang, X.; Tighe, S. Evaluation of mechanical properties and microscopic structure of coal gangue after aqueous solution treatment. Materials 2019, 12, 3207. [Google Scholar] [CrossRef]
- Cecilia, J.A.; Soriano, M.D.; Natoli, A.; Rodríguez-Castellón, E.; Nieto, J.M.L. Selective oxidation of hydrogen sulfide to sulfur using vanadium oxide supported on porous clay heterostructures (PCHs) formed by pillars silica, silica-zirconia or silica-titania. Materials 2018, 11, 1562. [Google Scholar] [CrossRef]
- Flytzani-Stephanopoulous, M.; Sakbodin, M.; Zheng, W. Regenerative adsorption and removal of H2S from hot fuel gas streams by rare earth oxides. Science 2006, 312, 1508–1518. [Google Scholar] [CrossRef]
- Huang, Y.; Rezvani, S.; Mcllveen-Wright, D.; Minchener, A.; Hewitt, N. Techno-economic study of CO2 capture and storage in coal fired oxygen fed entrained flow IGCC power plants. Fuel Process. Technol. 2008, 9, 916–925. [Google Scholar] [CrossRef]
- Huang, Z.; Liu, B.; Wang, F.; Amin, R. Performance of Zn–Fe–Mn/MCM-48 sorbents for high temperature H2S removal and analysis of regeneration process. Appl. Surf. Sci. 2015, 353, 1–10. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, B.; Wang, F.; Wang, W.; Xia, C.; Zheng, S.; Amin, R. Hydrogen sulfide removal from hot coal gas by various mesoporous silica supported Mn2O3 sorbents. Appl. Surf. Sci. 2014, 313, 961–969. [Google Scholar] [CrossRef]
- Pishahang, M.; Larring, Y.; Dijk, E.V.; Berkel, F.V.; Dahl, P.I.; Cobden, P.; Mccann, M.; Bakken, E. Regenerative copper-alumina H2S sorbent for hot gas cleaning through chemical swing adsorption. Ind. Eng. Chem. Res. 2016, 55, 1024–1032. [Google Scholar] [CrossRef]
- Giuffrida, A.; Romano, M.C.; Lozza, G.G. Thermodynamic assessment of IGCC power plants with hot fuel gas desulfurization. Appl. Energy 2010, 87, 3374–3383. [Google Scholar] [CrossRef]
- Westmoreland, P.R.; Gibsion, J.B.; Harrison, D.P. Evaluation of candidate solids for high temperature desulfurization of low-Btu gases. Environ. Sci. Technol. 1976, 10, 659–661. [Google Scholar] [CrossRef]
- Wang, J.; Liang, B.; Parnas, R. Manganese-based regenerable sorbents for high temperature H2S removal. Fuel 2013, 107, 539–546. [Google Scholar] [CrossRef]
- Bulavchenko, O.A.; Afonasenko, T.N.; Tsyrulńikov, P.G.; Tsybulya, S.V. Effect of heat treatment conditions on the structure and catalytic properties of MnOx/Al2O3 in the reaction of CO oxidation. Appl. Catal. A 2013, 459, 73–80. [Google Scholar] [CrossRef]
- Wang, F.; Liu, B.S.; Zhang, Z.F.; Zheng, S. High-temperature desulfurization of coal gas over Sm doped Mn-based/MSU-S sorbents. Ind. Eng. Chem. Res. 2015, 54, 8405–8416. [Google Scholar] [CrossRef]
- Xia, H.; Liu, B.S.; Li, Q.; Huang, Z.; Cheung, A.S.-C. High capacity Mn-Fe-Mo/FSM-16 sorbents in hot coal gas desulfurization and mechanism of elemental sulfur formation. Appl. Catal. B 2017, 200, 552–565. [Google Scholar] [CrossRef]
- Dilek, K.; Timur, D.; Sena, Y.; Gulsen, D. Mn-Cu and Mn-Cu-V mixed-oxide regenerable sorbents for hot gas desulfurization. Ind. Eng. Chem. Res. 2005, 44, 5221–5226. [Google Scholar]
- Jung, S.Y.; Lee, S.J.; Park, J.J.; Lee, S.C.; Jun, H.K.; Lee, T.J.; Ryu, C.K.; Kim, J.C. Properties of nanosize zinc titanium desulfurization sorbents promoted with iron and cerium oxides. Ind. Eng. Chem. Res. 2008, 47, 4909–4916. [Google Scholar] [CrossRef]
- Alonso, L.; Palacios, J.M. Performance and recovering of a Zn-doped manganese oxide as a regenerable sorbent for hot coal gas. Energy Fuels 2002, 16, 1550–1556. [Google Scholar] [CrossRef]
- Gibson, J.B.; Harrison, D.P. The reaction between hydrogen sulfide and spherical pellets of zinc oxide. Ind. Eng. Chem. Process. Des. Dev. 1980, 19, 231–237. [Google Scholar] [CrossRef]
- Liu, D.J.; Wang, Q.; Wu, J.; Liu, Y.X. A review of sorbents for high-temperature hydrogen sulfide removal from hot coal gas. Environ. Chem. Lett. 2019, 17, 259–276. [Google Scholar] [CrossRef]
- Cheah, S.F.; Parent, Y.O.; Jablonski, W.S.; Vinzant, T.; Olstad, J.L. Manganese and ceria sorbents for high temperature sulfur removal from biomass-derived syngas—The impact of steam on capacity and sorption mode. Fuel 2012, 97, 612–620. [Google Scholar] [CrossRef]
- Pan, Y.G.; Perales, J.F.; Velo, E.; Puigjaner, L. Kinetic behaviour of iron oxide sorbent in hot gas desulfurization. Fuel 2005, 84, 1105–1109. [Google Scholar] [CrossRef]
- Wang, J.; Guo, J.; Parnas, R.; Liang, B. Calcium-based regenerable sorbents for high temperature H2S removal. Fuel 2015, 154, 17–23. [Google Scholar] [CrossRef]
- Luo, T.; Vohs, J.M.; Gorte, R.J. An examination of sulfur poisoning on Pd/ceria catalysts. J. Catal. 2002, 210, 397–404. [Google Scholar] [CrossRef]
- He, H.P.; Gorte, R.J.; Vohs, J.M. Highly sulfur tolerant Cu-ceria anodes for SOFCs. Electrochem. Solid-State Lett. 2005, 8, A279–A280. [Google Scholar] [CrossRef]
- Park, S.; Vohs, J.M.; Gorte, R.J. Direct oxidation of hydrocarbons in a solid-oxide fuel cell. Nature 2000, 404, 265–267. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhang, S.; Groves, F.R.; Harrison, D.P. High temperature gas desulfurization with elemental sulfur production. Chem. Eng. Sci. 1999, 54, 3007–3017. [Google Scholar] [CrossRef]
- Zeng, Y.; Kaytakoglu, S.; Harrison, D.P. Reduced cerium oxide as an efficient and durable high temperature desulfurization sorbent. Chem. Eng. Sci. 2000, 55, 4893–4900. [Google Scholar] [CrossRef]
- Yi, K.B.; Podlaha, E.J.; Harrison, D.P. Ceria-zirconia high-temperature desulfurization sorbents. Ind. Eng. Chem. Res. 2005, 44, 7086–7091. [Google Scholar] [CrossRef]
- Yu, Q.; Zhang, S.; Wang, X.; Zhang, J.; Lu, Z. Study on sulfation of CeO2/γ-Al2O3 sorbent in simulated flue gas. J. Rare Earth 2007, 25, 184–188. [Google Scholar] [CrossRef]
- Jiang, X.Y.; Zhou, R.X.; Yuan, J.; Lu, G.L.; Zheng, X.M. Catalytic oxidative properties and characterization of CuO/CeO2 catalysts. J. Rare Earths 2003, 1, 55–59. [Google Scholar]
- Yu, B.; Zhao, Y.; Li, Y. A SnO2-samarium doped ceria additional anode layer in a direct carbon fuel cell. J. Power Sour. 2016, 306, 387–393. [Google Scholar] [CrossRef]
- Tao, R.; Xu, J.S.; Zhong, H.; Wen, W.; Pan, Q.F.; Liu, Y.; Chen, J. Finely tuned structure and catalytic performance of cerium oxides by a continuous samarium doping from 0 to 100%. Inorg Chem. 2019, 58, 13066–13076. [Google Scholar] [CrossRef] [PubMed]
- Coduri, M.; Masala, P.; Allieta, M.; Peral, I.; Brunelli, M.; Biffi, C.A.; Scavini, M. Phase transformations in the CeO2-Sm2O3 system: A multiscale powder diffraction investigation. Inorg. Chem. 2018, 57, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Laosiripojana, N.; Charojrochkul, S.; Kim-Lohsoontorn, P.; Assabumrungrat, S. Role and advantages of H2S in catalytic steam reforming over nanoscale CeO2-based catalysts. J. Catal. 2010, 276, 6–15. [Google Scholar] [CrossRef]
- Girija, T.S.; Ganesh, K.; Mangalaraj, V.; Viswanathan, D.; Ponpandian, C.N. Novel Synthesis of LaFeO3 Nanostructure Dendrites: A Systematic Investigation of Growth Mechanism, Properties, and Biosensing for Highly Selective Determination of Neurotransmitter Compounds. Cryst. Growth Des. 2013, 13, 291–302. [Google Scholar]
- Ferrizz, R.M.; Gorte, R.J.; Vohs, J.M. TPD and XPS investigation of the interaction of SO2 with model ceria catalysts. Catal. Lett. 2002, 82, 123–129. [Google Scholar] [CrossRef]
- Fang, S.M.; Bi, L.; Wu, X.S.; Gao, H.Y.; Chen, C.S.; Liu, W. Chemical stability and hydrogen permeation performance of Ni–BaZr0.1Ce0.7Y0.2O3−δ in an H2S-containing atmosphere. J. Power Sour. 2008, 183, 126–132. [Google Scholar] [CrossRef]
- Kay, D.A.R.; Subramanian, S.V.; Kuma, V.; Meng, V. The use of RICO-S phase stability diagrams in gaseous desulfurisation and iron and steel production. Inorg. Chim. Acta 1984, 94, 132–134. [Google Scholar] [CrossRef]
- Kay, D.A.R.; Wilson, W.G. Method for the Regeneration of Sulfded Cerumoxde Back to a Form that Sagan Capable of Removing Sulfur from Fluid Materals. U.S. Patent No.: 4857280, 15 August 1989. [Google Scholar]
- Larquet, C.; Nguyen, A.M.; Ávila-Gutiérrez, M.; Tinat, L.; Lassalle-Kaiser, B.; Gallet, J.J.; Bournel, F.; Gauzzi, A.; Sanchez, C.; Carenco, S. Synthesis of Ce2O2S and Gd2(1–y)Ce2yO2S nanoparticles and reactivity from in situ X-ray absorption spectroscopy and X-ray photoelectron spectroscopy. Inorg. Chem. 2017, 56, 14227–14236. [Google Scholar] [CrossRef]
- Leskelä, M. Thermal stability of Ce2O2S. DOES Ce2O2SO4 exist? Thermochim. Acta 1985, 92, 739–742. [Google Scholar] [CrossRef]
- Schriver-mazzuoli, L.; Chaabouni, H.; Schriver, A. Infrared spectra of SO2 and SO2: H2O ices at low temperature. J. Mol. Struct. 2003, 644, 151–164. [Google Scholar] [CrossRef]
- Zasova, L.V.; Moroz, V.I.; Esposito, L.W.; Na, C.Y. SO2 in the middle atmosphere of venus: IR measurements from venera-15 and comparison to UV data. Icarus 1993, 105, 92–109. [Google Scholar] [CrossRef]
- Bonfim, V.S.; Castilho, R.B.; Baptista, L.; Pilling, S. SO3 formation from the X-ray photolysis of SO2 astrophysical ice analogues: FTIR spectroscopy and thermodynamic investigations. Phys. Chem. Chem. Phys. 2017, 19, 26906–26917. [Google Scholar] [CrossRef] [PubMed]
- Khanna, R.K. Infrared spectra and structure of solid phases of sulfur trioxide: Possible identification of solid SO3 on io’s surface. Icarus 1995, 115, 250–257. [Google Scholar] [CrossRef]
- Szargan, R.; Karthe, S.; Suoninen, E. XPS studies of xanthate adsorption on pyrite. Appl. Surf. Sci. 1992, 55, 227–232. [Google Scholar] [CrossRef]
- Mycroft, J.R.; Bancroft, G.M.; McIntyre, N.S. Detection of sulphur and polysulphides on electrochemically oxidized pyrite surfaces by X-ray photoelectron spectroscopy and Raman spectroscopy. J. Electroanal. Chem. 1990, 292, 139–152. [Google Scholar] [CrossRef]









| Samples | S (m2 g−1) | D (nm) | V (cm3 g−1) | P (nm) |
|---|---|---|---|---|
| CeO2 | 64 | 9.1 | 0.117 | 9.86 |
| SDC | 271 | 5.2 | 0.362 | 5.46 |
| SDC a (cycle 1) | 269 | 5.7 | 0.347 | 6.53 |
| SDC a (cycle 2) | 260 | 6.6 | 0.339 | 6.91 |
| SDC a (cycle 3) | 256 | 7.2 | 0.341 | 7.45 |
| SDC a (cycle 4) | 249 | 7.5 | 0.325 | 7.58 |
| SDC a (cycle 5) | 251 | 8.0 | 0.317 | 8.16 |
| SDC a (cycle 6) | 245 | 8.2 | 0.311 | 8.24 |
| Samples | O 1s | S 2p | ||
|---|---|---|---|---|
| OL Position (eV) | OA Position (eV) | OL/(OL + OA) % | S-species Mass % | |
| fresh CeO2 | 529.1 | 531.1 | 91.5 | - |
| Ce-O-S powder | 528.9 | 531.0 | 4.7 | 13.8 |
| Re1 powder | 529.0 | 531.1 | 75.9 | 1.7 |
| Ce-O-S Powder (mg) | NaOH Dosage 0.05 mol/L (mL) | Actual Yield (AY) of Acid (mol) | Theoretical Yield (TY) of Acid (mol) | AY/TY (%) |
|---|---|---|---|---|
| 250 | 28.3 | 0.71 × 10−3 | 0.87 × 10−3 | 81.6 |
| 500 | 55.6 | 1.39 × 10−3 | 1.73 × 10−3 | 80.1 |
| 1000 | 106.4 | 2.66 × 10−3 | 3.47 × 10−3 | 76.7 |
| 1500 | 155.2 | 3.88 × 10−3 | 5.20 × 10−3 | 74.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.; Dong, J. Regeneration Mechanism of Sulfur Absorption Via Samarium-doped Cerium Adsorbents in the Gas Atmosphere of O2/N2. Materials 2020, 13, 1225. https://doi.org/10.3390/ma13051225
Hu X, Dong J. Regeneration Mechanism of Sulfur Absorption Via Samarium-doped Cerium Adsorbents in the Gas Atmosphere of O2/N2. Materials. 2020; 13(5):1225. https://doi.org/10.3390/ma13051225
Chicago/Turabian StyleHu, Xuechao, and Junhui Dong. 2020. "Regeneration Mechanism of Sulfur Absorption Via Samarium-doped Cerium Adsorbents in the Gas Atmosphere of O2/N2" Materials 13, no. 5: 1225. https://doi.org/10.3390/ma13051225
APA StyleHu, X., & Dong, J. (2020). Regeneration Mechanism of Sulfur Absorption Via Samarium-doped Cerium Adsorbents in the Gas Atmosphere of O2/N2. Materials, 13(5), 1225. https://doi.org/10.3390/ma13051225




