A 4 Year Human, Randomized, Radiographic Study of Scalloped versus Non-Scalloped Cemented Implants
Abstract
1. Introduction
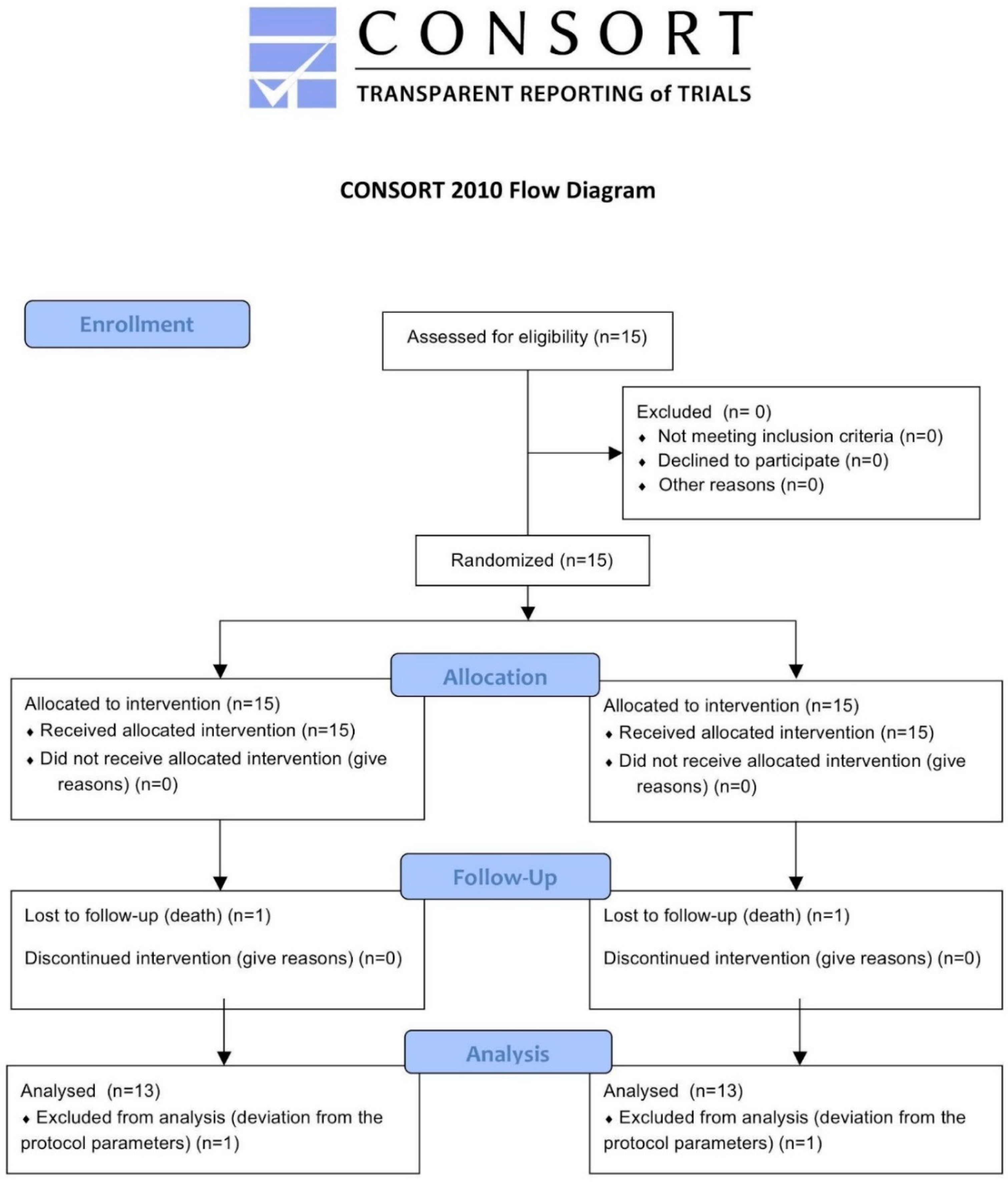
2. Materials and Methods
2.1. Study Design
2.2. Patient Selection
2.3. Randomization
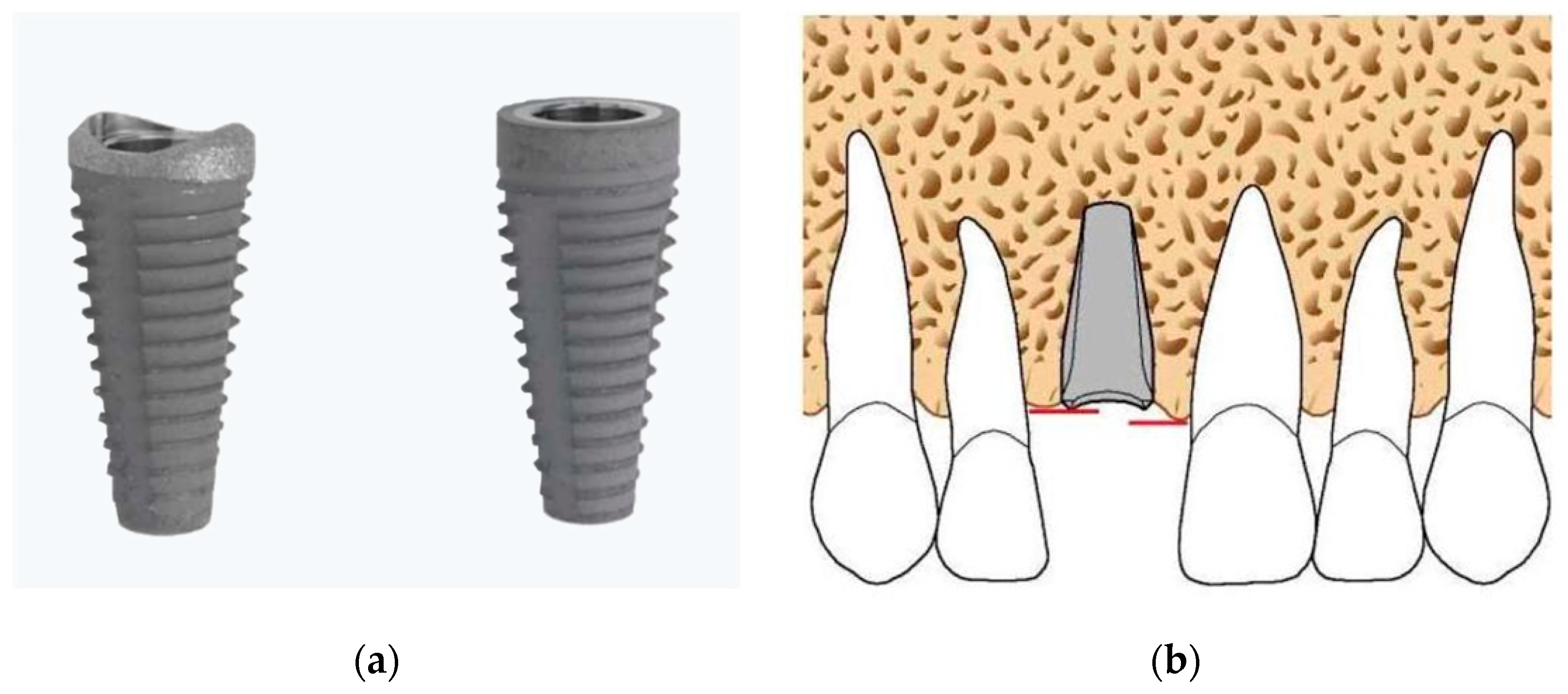
2.4. Surgical and Prosthetic Treatment
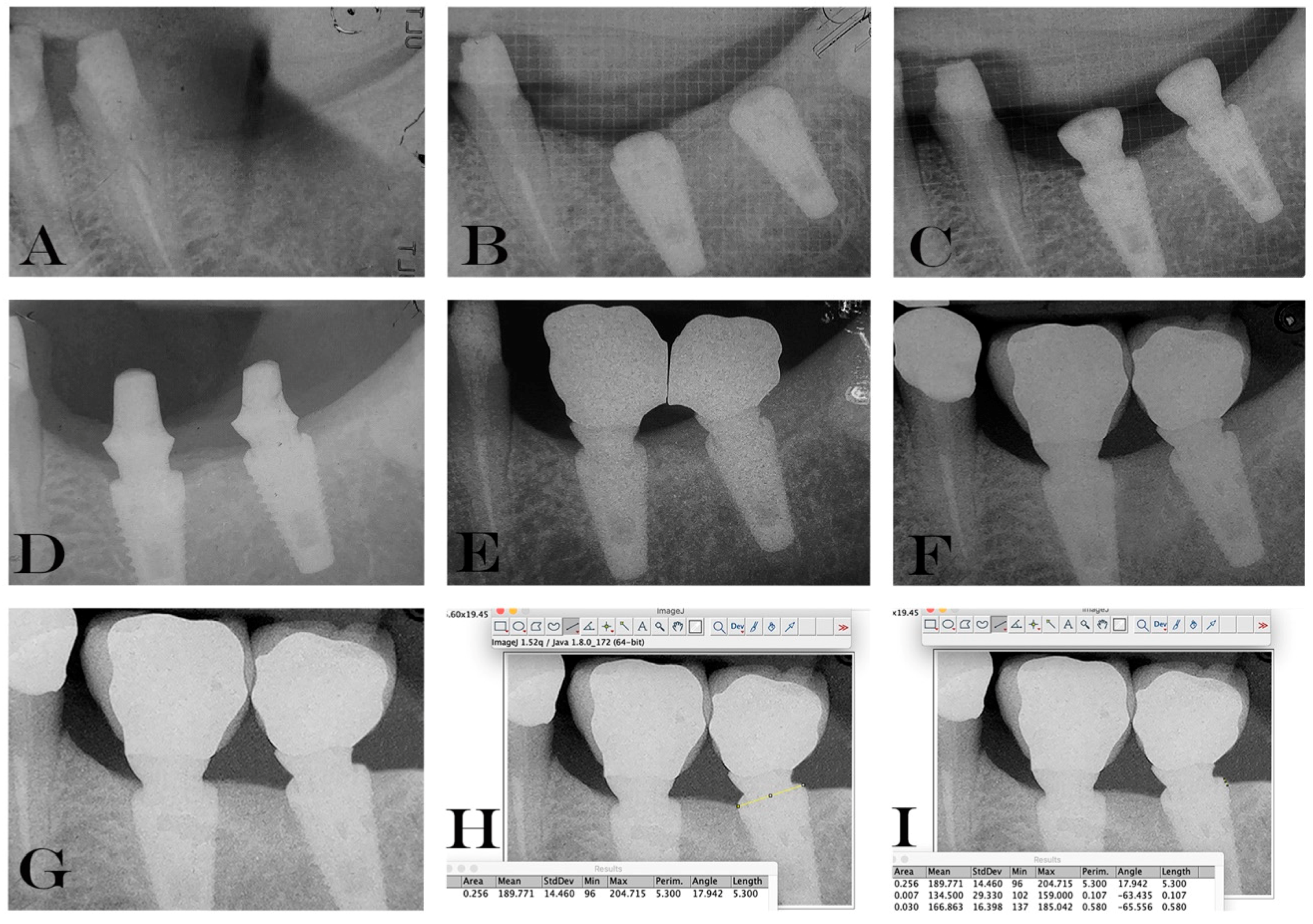
2.5. Data Handling and Radiographic Analysis of Marginal Bone Level Changes.
2.6. Statistical Analysis
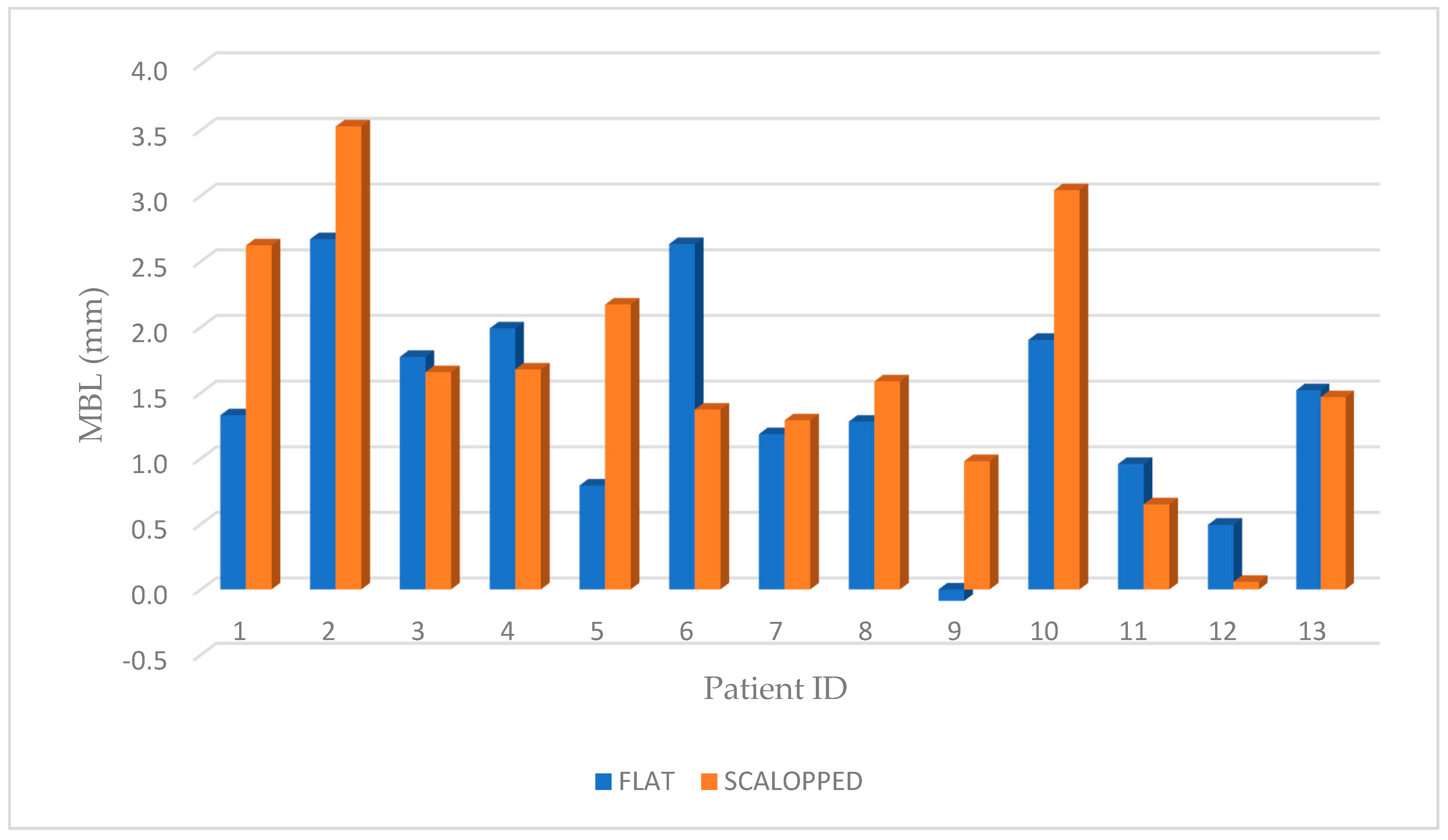
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Caricasulo, R.; Malchiodi, L.; Ghensi, P.; Fantozzi, G.; Cucchi, A. The influence of implant-abutment connection to peri-implant bone loss: A systematic review and meta-analysis. Clin. Implant Dent. Relat. Res. 2018, 20, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Sinjari, B.; D’Addazio, G.; De Tullio, I.; Traini, T.; Caputi, S. Peri-Implant Bone Resorption during Healing Abutment Placement: The Effect of a 0.20% Chlorhexidine Gel vs. Placebo-A Randomized Double Blind Controlled Human Study. BioMed Res. Int. 2018, 16, 5326340. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Buser, D.; Sennerby, L. On crestal/marginal bone loss around dental implants. Int. J. Oral Maxillofac. Implants 2012, 27, 736–738. [Google Scholar] [PubMed]
- Sinjari, B.; D’Addazio, G.; Traini, T.; Varvara, G.; Scarano, A.; Murmura, G.; Caputi, S. A 10-year retrospective comparative human study on screw-retained versus cemented dental implant abutments. J. Boil. Regul. Homeost. Agents 2019, 33, 787–797. [Google Scholar]
- Leonard, G.; Coelho, P.; Polyzois, I.; Stassen, L.; Claffey, N. A study of the bone healing kinetics of plateau versus screw root design titanium dental implants. Clin. Oral Implant. Res. 2009, 20, 232–239. [Google Scholar] [CrossRef]
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Figuero, E.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S313–S318. [Google Scholar] [CrossRef]
- Sinjari, B.; Traini, T.; Caputi, S.; Mortellaro, C.; Scarano, A. Evaluation of Fibrin Clot Attachment on Titanium Laser-Conditioned Surface Using Scanning Electron Microscopy. J. Craniofacial Surg. 2018, 29, 2277–2281. [Google Scholar] [CrossRef]
- Oh, T.J.; Yoon, J.; Misch, C.E.; Wang, H.L. The causes of early implant bone loss: Myth or science? J. Periodontol. 2002, 73, 322–333. [Google Scholar] [CrossRef]
- Marconcini, S.; Giammarinaro, E.; Covani, U.; Mijiritsky, E.; Vela, X.; Rodríguez, X. The Effect of Tapered Abutments on Marginal Bone Level: A Retrospective Cohort Study. J. Clin. Med. 2019, 8, 1305. [Google Scholar] [CrossRef]
- Kitamura, E.; Stegaroiu, R.; Nomura, S.; Miyakawa, O. Influence of marginal bone resorption on stress around an implant—A three-dimensional finite element analysis. J. Oral Rehabilitation 2005, 32, 279–286. [Google Scholar] [CrossRef]
- Renvert, S.; Persson, G.R.; Pirih, F.Q.; Camargo, P.M. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J. Periodontol. 2018, 89, S304–S312. [Google Scholar] [CrossRef] [PubMed]
- D’Ercole, S.; D’Addazio, G.; Di Lodovico, S.; Traini, T.; Di Giulio, M.; Sinjari, B. Porphyromonas Gingivalis Load is Balanced by 0.20% Chlorhexidine Gel. A Randomized, Double-Blind, Controlled, Microbiological and Immunohistochemical Human Study. J. Clin. Med. 2020, 20, 284. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Murmura, G.; Sinjari, B.; Sollazzo, V.; Spinelli, G.; Carinci, F. Analysis and structural examination of screw loosening in oral implants. Int. J. Immunopathol. Pharmacol. 2011, 24, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Penarrocha-Diago, M.A.; Flichy-Fernandez, A.J.; Alonso-Gonzalez, R.; Penarrocha-Oltra, D.; Balaguer-Martínez, J.; Penarrocha-Diago, M. Influence of implant neck design and implant-abutment connection type on peri-implant health. Radiological study. Clin. Oral Implant. Res. 2013, 24, 1192–1200. [Google Scholar] [CrossRef]
- Sinjari, B.; D’Addazio, G.; Bozzi, M.; Celletti, R.; Traini, T.; Mavriqi, L.; Caputi, S. Comparison of a Novel Ultrasonic Scaler Tip vs. Conventional Design on a Titanium Surface. Materials 2018, 22, 2345. [Google Scholar] [CrossRef]
- Ghinassi, B.; D’Addazio, G.; Di Baldassarre, A.; Femminella, B.; Di Vincenzo, G.; Piattelli, M.; Gaggi, G.; Sinjari, B. Immunohistochemical Results of Soft Tissues Around a New Implant Healing-Abutment Surface: A Human Study. J. Clin. Med. 2020, 9, 1009. [Google Scholar] [CrossRef]
- Annibali, S.; Bignozzi, I.; Cristalli, M.P.; Graziani, F.; La Monaca, G.; Polimeni, A. Peri-implant marginal bone level: A systematic review and meta- analysis of studies comparing platform switching versus convention- ally restored implants. J. Clin. Periodontol. 2012, 39, 1097–1113. [Google Scholar] [CrossRef]
- Tallarico, M.; Caneva, M.; Meloni, S.M.; Xhanari, E.; Omori, Y.; Canullo, L. Survival and Success Rates of Different Shoulder Designs: A Systematic Review of the Literature. Int. J. Dent. 2018, 26, 6812875. [Google Scholar] [CrossRef]
- Starch-Jensen, T.; Christensen, A.E.; Lorenzen, H. Scalloped Implant-Abutment Connection Compared to Conventional Flat Implant-Abutment Connection: A Systematic Review and Meta-Analysis. J. Oral Maxillofac. Res. 2017, 8, e2. [Google Scholar] [CrossRef]
- Pozzi, A.; Tallarico, M.; Moy, P.K. Three-year post-loading results of a randomised, controlled, split-mouth trial comparing implants with different prosthetic interfaces and design in partially posterior edentulous mandibles. Eur. J. Oral Implant. 2014, 7, 47–61. [Google Scholar]
- Löe, H. The Gingival Index, the Plaque Index and the Retention Index Systems. J. Periodontol. 1967, 38, 610–616. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; CONSORT Group. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. Int. J. Surg. 2011, 9, 672–677. [Google Scholar] [PubMed]
- Sclar, A.G. Surgical techniques for management of peri-implant soft tissues. In Soft Tissue Esthetic Considerations in Implant Therapy; Motamedi, M., Ed.; Quintessence: Chicago, IL, USA, 2003; pp. 47–51. [Google Scholar]
- Scarano, A.; Murmura, G.; Sinjari, B.; Assenza, B.; Sollazzo, V.; Spinelli, G.; Carinci, F. Expansion of the alveolar bone crest with ultrasonic surgery device: Clinical study in mandible. Int. J. Immunopathol. Pharmacol. 2011, 24, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Papaspyridakos, P.; Chen, C.J.; Singh, M.; Weber, H.P.; Gallucci, G.O. Success criteria in implant dentistry: A systematic review. J. Dent. Res. 2012, 91, 242–248. [Google Scholar] [CrossRef] [PubMed]
- De Smet, E.; Jacobs, R.; Gijbels, F.; Naert, I. The accuracy and reliability of radiographic methods for the assessment of marginal bone level around oral implants. Dentomaxillofac. Radiol. 2002, 31, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Lee, S.P.; Han, C.H.; Kwon, J.H.; Jung, Y.C. The microtomographic evaluation of marginal bone resorption of immediately loaded scalloped design implant with various microthread configurations in canine mandible: Pilot study. J. Oral Implant. 2010, 36, 357–362. [Google Scholar] [CrossRef]
- McAllister, B.S. Scalloped implant designs enhance interproximal bone levels. Int. J. Periodontics Restor. Dent. 2007, 27, 9–15. [Google Scholar]
- Kan, J.Y.; Rungcharassaeng, K.; Liddelow, G.; Henry, P.; Goodacre, C.J. Periimplant tissue response following immediate provisional restoration of scalloped implants in the esthetic zone: A one-year pilot prospective multicenter study. J. Prosthet. Dent. 2007, 97, S109–S118. [Google Scholar] [CrossRef]
- Nowzari, H.; Chee, W.; Yi, K.; Pak, M.; Chung, W.H.; Rich, S. Scalloped dental implants: A retrospective analysis of radiographic and clinical outcomes of 17 NobelPerfect implants in 6 patients. Clin. Implant Dent. Relat. Res. 2006, 8, 1–10. [Google Scholar] [CrossRef]
- Khatami, A.H.; Al-Ajmi, M.; Kleinman, A. Preservation of the gingival architecture with the scalloped implant design: A clinical report. J. Oral Implant. 2006, 32, 167–170. [Google Scholar] [CrossRef]
- Den Hartog, L.; Raghoebar, G.M.; Slater, J.J.; Stellingsma, K.; Vissink, A.; Meijer, H.J. Single-tooth implants with different neck designs: A randomized clinical trial evaluating the aesthetic outcome. Clin. Implant Dent. Relat. Res. 2013, 15, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Khraisat, A.; Zembic, A.; Jung, R.E.; Hammerle, C.H. Marginal bone levels and soft tissue conditions around single- tooth implants with a scalloped neck design: Results of a prospective 3-year study. Int. J. Oral Maxillofac. Implant. 2013, 28, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Van Nimwegen, W.G.; Raghoebar, G.M.; Stellingsma, K.; Tymstra, N.; Vissink, A.; Meijer, H.J. Treatment Outcome of Two Adjacent Implant-Supported Restorations with Different Implant Platform Designs in the Esthetic Region: A Five-Year Randomized Clinical Trial. Int. J. Prosthodont. 2015, 28, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Assenza, B.; Tripodi, D.; Scarano, A.; Perrotti, V.; Piattelli, A.; Iezzi, G.; D’Ercole, S. Bacterial leakage in implants with different implant-abutment connections: An in vitro study. J. Periodontol. 2012, 83, 491–497. [Google Scholar] [CrossRef]
- Assenza, B.; Artese, L.; Scarano, A.; Rubini, C.; Perrotti, V.; Piattelli, M.; Thams, U.; San Roman, F.; Piccirilli, M.; Piattelli, A. Screw vs cement-implant-retained restorations: An experimental study in the beagle. Part 2. Immunohistochemical evaluation of the peri-implant tissues. J. Oral Implant. 2006, 32, 1–7. [Google Scholar] [CrossRef]
- Staubli, N.; Walter, C.; Schmidt, J.C.; Weiger, R.; Zitzmann, N.U. Excess cement and the risk of peri-implant disease—A systematic review. Clin. Oral Implant. Res. 2017, 28, 1278–1290. [Google Scholar] [CrossRef]
- Berglundh, T.; Lindhe, J. Dimensions of perimplants mucosa. Biological width revisited. J. Clin. Periodontol. 1996, 23, 971–973. [Google Scholar] [CrossRef]
- Berglundh, T.; Lindhe, J. The mucosal attachment at different abutments. An experimental study in dogs. J. Clin. Periodontol. 1998, 25, 721–727. [Google Scholar]
- Ericsson, I.; Nilner, K.; Klinge, B.; Glantz, P.O. Radiographical and histological characteristics of submerged and nonsubmerged titanium implants. An experimental study in the Labrador dog. Clin. Oral Implant. Res. 1996, 7, 20–26. [Google Scholar] [CrossRef]
- Hermann, J.S.; Cochran, D.L.; Nummikoski, P.V.; Buser, D. Crestal bone changes around titanium implants. A radiographic evaluation of unloaded nonsubmerged and submerged implants in the canine mandible. J. Periodontol. 1997, 68, 1117–1130. [Google Scholar] [CrossRef]
- Veis, A.; Parissis, N.; Tsirlis, A.; Papadeli, C.; Marinis, G.; Zogakis, A. Evaluation of peri-implant marginal bone loss using modified abutment connections at various crestal level placements. Int. J. Periodontics Restor. Dent. 2010, 30, 609–617. [Google Scholar]
- Papaspyridakos, P.; Bordin, T.B.; Natto, Z.S.; El-Rafie, K.; Pagni, S.E.; Chochlidakis, K.; Ercoli, C.; Weber, H.P. Complications and survival rates of 55 metal-ceramic implant-supported fixed complete-arch prostheses: A cohort study with mean 5-year follow-up. J. Prosthet. Dent. 2019, 122, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.L.; Rosenberg, M.M.; Zinser, P.J.; Ganeles, J. Concept for a biologically derived, parabolic implant design. Int. J. Periodontics Restor. Dent. 2002, 22, 473–481. [Google Scholar]
- Gadhia, M.H.; Holt, R.L. A new implant design for optimal esthetics and retention of interproximal papillae. Implant. Dent. 2003, 12, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Wohrle, P.S. NobelPerfectTM esthetic scalloped implant: Rationale for a new design. Clin. Implant. Dent. Relat. Res. 2003, 5, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Nordland, W.P.; Tarnow, D.P. A classification system for loss of papillary height. J. Periodontol. 1998, 69, 1124–1126. [Google Scholar] [CrossRef]
- Tarnow, D.P.; Magner, A.W.; Fletcher, P. The effect of the distance from the contact point to the crest of bone on the presence or absence of the interproximal dental papilla. J. Periodontol. 1992, 63, 995–996. [Google Scholar] [CrossRef]
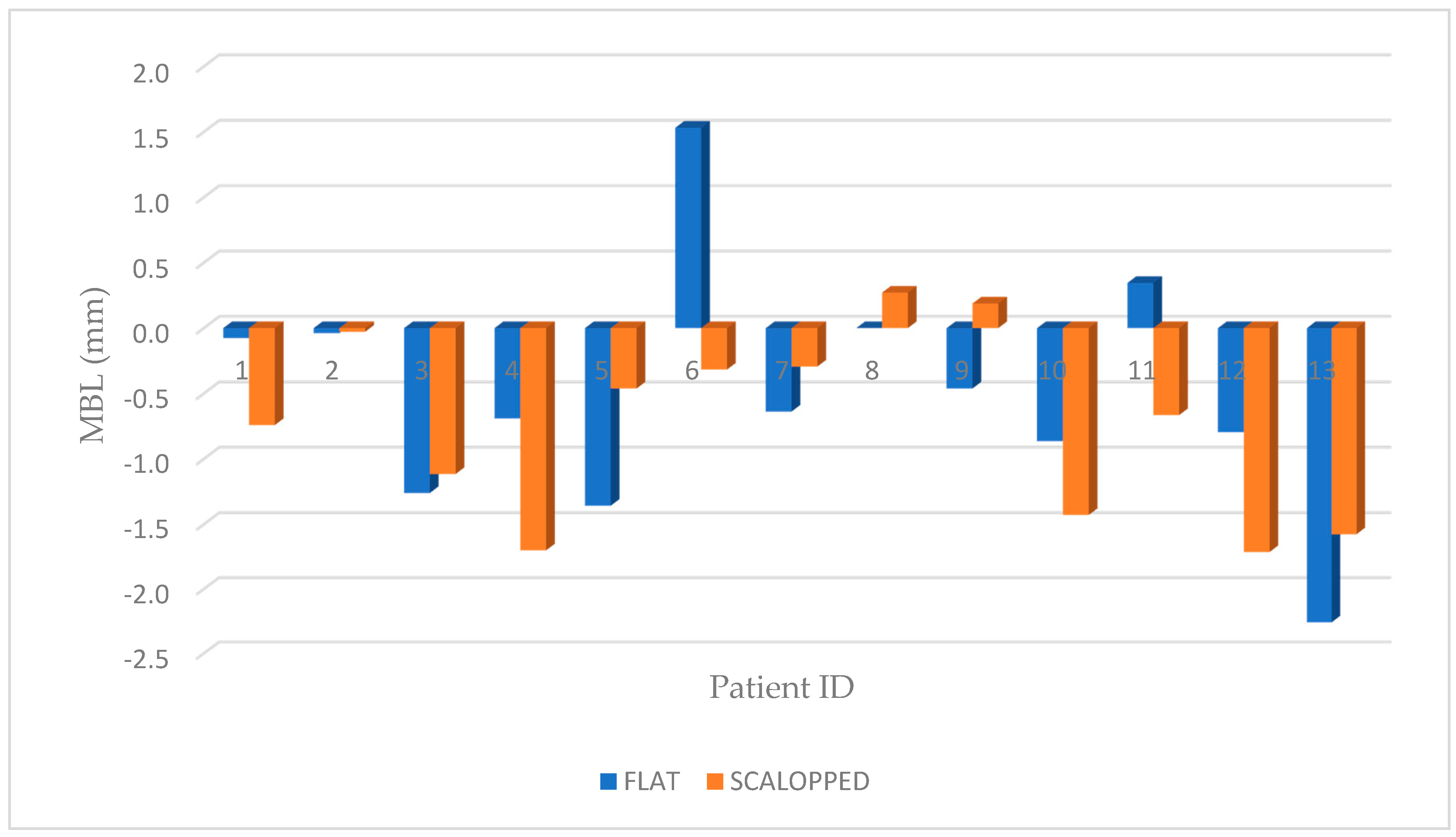


| Id | Age | Sex | Group | Site | Bone Density | Final Torque NCM | T0 Rx (mm) | T1 Rx (mm) | Rx T1-T0 |
|---|---|---|---|---|---|---|---|---|---|
| 1A | 78 | M | CONTROL | 46 | Normal | 45 | 1.329 | −0.076 | 1.404 |
| 1B | 78 | M | TEST | 47 | Dense | 35 | 2.622 | −0.741 | 3.363 |
| 2A | 67 | F | CONTROL | 16 | Poor | 25 | 2.669 | −0.039 | 2.708 |
| 2B | 66 | F | TEST | 14 | Normal | 40 | 3.528 | −0.028 | 3.555 |
| 3A | 48 | F | CONTROL | 25 | Poor | 30 | 1.773 | −1.261 | 3.034 |
| 3B | 47 | F | TEST | 24 | Dense | 45 | 1.656 | −1.115 | 2.771 |
| 4A | 67 | M | CONTROL | 24 | Normal | 40 | 1.990 | −0.690 | 2.680 |
| 4B | 68 | M | TEST | 26 | Normal | 45 | 1.677 | −1.700 | 3.377 |
| 5A | 76 | M | CONTROL | 35 | Dense | 50 | 0.794 | −1.358 | 2.152 |
| 5B | 77 | M | TEST | 36 | Normal | 35 | 2.171 | −0.462 | 2.633 |
| 6A | 59 | F | CONTROL | 37 | Normal | 30 | 2.633 | 1.532 | 1.101 |
| 6B | 60 | F | TEST | 46 | Dense | 40 | 1.371 | −0.317 | 1.688 |
| 7A | 45 | M | CONTROL | 25 | Poor | 25 | 1.185 | −0.638 | 1.823 |
| 7B | 44 | M | TEST | 24 | Normal | 30 | 1.290 | −0.294 | 1.584 |
| 8A | 68 | M | CONTROL | 26 | Normal | 25 | 1.280 | 0.000 | 1.280 |
| 8B | 69 | M | TEST | 27 | Poor | 30 | 1.587 | 0.272 | 1.315 |
| 9A | 56 | M | CONTROL | 46 | Dense | 50 | −0.089 | −0.462 | 0.374 |
| 9B | 57 | M | TEST | 47 | Normal | 55 | 0.979 | 0.188 | 0.791 |
| 10A | 73 | M | CONTROL | 27 | Normal | 40 | 1.902 | −0.864 | 2.766 |
| 10B | 72 | M | TEST | 26 | Normal | 50 | 3.042 | −1.429 | 4.470 |
| 11A | 66 | F | CONTROL | 16 | Poor | 25 | 0.958 | 0.346 | 0.612 |
| 11B | 65 | F | TEST | 15 | Normal | 25 | 0.650 | −0.665 | 1.315 |
| 12A | 51 | F | CONTROL | 37 | Dense | 35 | 0.495 | −0.795 | 1.289 |
| 12B | 50 | F | TEST | 35 | Normal | 30 | 0.058 | −1.713 | 1.771 |
| 13A | 32 | M | CONTROL | 26 | Poor | 40 | 1.519 | −2.259 | 3.777 |
| 13B | 31 | M | TEST | 16 | Normal | 35 | 1.466 | −1.576 | 3.041 |
| Categories (Test Group) | T0 | T1 | T1-T0 | |
| Sex | Male | 1.854 ± 0.700 | −0.717 ± 0.78 | 2.571 ± 1.243 |
| Female | 1.452 ± 1.32 | −0.76 ± 0.66 | 2.22 ± 0.92 | |
| Location | Posterior maxilla | 1.861 ± 0.94 | −0.816 ± 0.748 | 2.678 ± 1.166 |
| Posterior mandible | 1.44 ± 1.007 | −0.61 ± 0.703 | 2.05 ± 0.98 | |
| Categories (Control Group) | T0 | T1 | T1-T0 | |
| Sex | Male | 1.239 ± 0.661 | −0.793 ± 0.733 | 2.032 ± 1.054 |
| Female | 1.705 ± 0.977 | −0.043 ± 1.082 | 1.749 ± 1.060 | |
| Location | Posterior maxilla | 1.659 ± 0,545 | −0.676 ± 0.827 | 2.335 ± 1.025 |
| Posterior mandible | 1.032 ± 1.031 | −0.232 ± 1.092 | 1.264 ± 0.638 | |
| Categories (All Inserted Implants) | T0 | T1 | T1-T0 | |
| Sex | Male | 1.546 ± 0.73 | −0.755 ± 0.73 | 2.30 ± 1.147 |
| Female | 1.578 ± 1.10 | −0.40 ± 0.93 | 1.984 ± 0.968 | |
| Location | Posterior maxilla | 1.760 ± 0.753 | −0.746 ± 0.7655 | 2.506 ± 1.075 |
| Posterior mandible | 1.236 ± 0.984 | −0.42 ± 0.888 | 1.656 ± 0.883 | |
| Groups- | Abutment Decementation | Temporary Crown Decementation | Crown Decementation | Ceramic Chipping |
|---|---|---|---|---|
| TEST | 0 | 0 | 0 | 3 (23.07%) |
| CONTROL | 0 | 1 (7.69%) | 0 | 2 (15.38%) |
| TOTAL | 0 | 1 (3.84%) | 0 | 5 (19.23%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinjari, B.; D’Addazio, G.; Santilli, M.; D’Avanzo, B.; Rexhepi, I.; Scarano, A.; Traini, T.; Piattelli, M.; Caputi, S. A 4 Year Human, Randomized, Radiographic Study of Scalloped versus Non-Scalloped Cemented Implants. Materials 2020, 13, 2190. https://doi.org/10.3390/ma13092190
Sinjari B, D’Addazio G, Santilli M, D’Avanzo B, Rexhepi I, Scarano A, Traini T, Piattelli M, Caputi S. A 4 Year Human, Randomized, Radiographic Study of Scalloped versus Non-Scalloped Cemented Implants. Materials. 2020; 13(9):2190. https://doi.org/10.3390/ma13092190
Chicago/Turabian StyleSinjari, Bruna, Gianmaria D’Addazio, Manlio Santilli, Barbara D’Avanzo, Imena Rexhepi, Antonio Scarano, Tonino Traini, Maurizio Piattelli, and Sergio Caputi. 2020. "A 4 Year Human, Randomized, Radiographic Study of Scalloped versus Non-Scalloped Cemented Implants" Materials 13, no. 9: 2190. https://doi.org/10.3390/ma13092190
APA StyleSinjari, B., D’Addazio, G., Santilli, M., D’Avanzo, B., Rexhepi, I., Scarano, A., Traini, T., Piattelli, M., & Caputi, S. (2020). A 4 Year Human, Randomized, Radiographic Study of Scalloped versus Non-Scalloped Cemented Implants. Materials, 13(9), 2190. https://doi.org/10.3390/ma13092190












