X-ray Absorption (XRA): A New Technique for the Characterization of Granular Activated Carbons
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Adsorbent
2.3. Preparation of Metal and Dye Solutions
2.4. Langmuir Isotherm
2.5. Freundlich Isotherm
2.6. Effective Atomic Number
2.7. X-ray Digital Radiography Experiments (XRA)
2.8. Relationship between Grey-Scale Intensity (GSI) and Mass Attenuation Coefficient Using XRA. Spatial Concentration (SC)
2.9. Relationship between Energy (to Achieve Maximum Photon Attenuation), Adsorbate Mass, Molar Mass and Atomic Number
2.10. Probabilistic Approach for Comparison between Elements/Molecules
3. Results and Discussion
3.1. Isotherms for the Sorption Process
3.1.1. Langmuir Isotherms
3.1.2. Freundlich Isotherms
3.2. X-ray Absorption (XRA)
3.3. Probability Approach
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van Grieken, R.; Markowicz, A. Handbook of X-ray Spectrometry; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Depristo, A.E.; Augustin, S.D.; Ramaswamy, R.; Rabitz, H. Quantum number and energy scaling for nonreactive collisions. J. Chem. Phys. 1979, 71, 850–865. [Google Scholar] [CrossRef]
- Massimi, M. Pauli’s Exclusion Principle: The Origin and Validation of a Scientific Principle; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Gonzalez, R.C.; Woods, R.E.; Eddins, S.L. Digital Image Processing Using MATLAB; Person: London, UK, 2004. [Google Scholar]
- Vanderheyden, S.R.H.; Yperman, J.; Carleer, R.; Schreurs, S. Enhanced cesium removal from real matrices by nickel-hexacyanoferrate modified activated carbons. Chemosphere 2018, 202, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Vanreppelen, K.; Vanderheyden, S.; Schreurs, S.; Yperman, J.; Băbeanu, N.; Carleer, R. Activated carbon from pyrolysis of brewer’s spent grain: Production and adsorption properties. Waste Manag. Res. 2014, 32, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Veith, J.; Sposito, G. On the use of the Langmuir equation in the interpretation of “adsorption” phenomena. Soil. Sci. Soc. Am. J. 1977, 41, 697–702. [Google Scholar] [CrossRef]
- Harter, R.D.; Smith, G. Langmuir equation and alternate methods of studying “adsorption” reactions in soils. Chem. Soil Environ. 1981, 40, 167–182. [Google Scholar]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.E.; Matsumoto, M. Modeling Cadmium Adsorption by Activated Carbon Using the Langmuir and Freundlich Isotherm Expressions. Sep. Sci. Technol. 1993, 28, 2179–2195. [Google Scholar] [CrossRef]
- Johns, H.; Cunningham, J. The Physics of Radiology, 4th ed.; Charles C Thomas Publisher: Springfield, IL, USA, 1983. [Google Scholar]
- Kumar, A. Studies on effective atomic numbers and electron densities of nucleobases in DNA. Radiat. Phys. Chem. 2016, 127, 48–55. [Google Scholar] [CrossRef]
- Taylor, M.L.; Smith, R.L.; Dossing, F.; Franich, R.D. Robust calculation of effective atomic numbers: The Auto-Zeff software. Med. Phys. 2012, 39, 1769–1778. [Google Scholar] [CrossRef] [PubMed]
- Tonguc, B.T.; Arslan, H.; Al-Buriahi, M. Studies on mass attenuation coefficients, effective atomic numbers and electron densities for some biomolecules. Radiat. Phys. Chem. 2018, 153, 86–91. [Google Scholar] [CrossRef]
- Torres, J.P.; Sariol, H.C.; Yperman, J.; Sauvanell, Á.B.; Carleer, R.; Campa, J.N. A novel X-ray radiography approach for the characterization of granular activated carbons used in the rum production. J. Anal. Sci. Technol. 2018, 9, 1. [Google Scholar] [CrossRef]
- Torres, J.P.; Sariol, H.C.; Yperman, J.; Adriaensens, P.; Carleer, R.; Peacok, T.M.; Sauvanell, Á.B.; Thijssen, E.; Reggers, G.; Haeldermans, T.; et al. X-ray absorption as an alternative method to determine the exhausting degree of activated carbon layers in water treatment system for medical services. Talanta 2019, 205, 120058. [Google Scholar] [CrossRef] [PubMed]




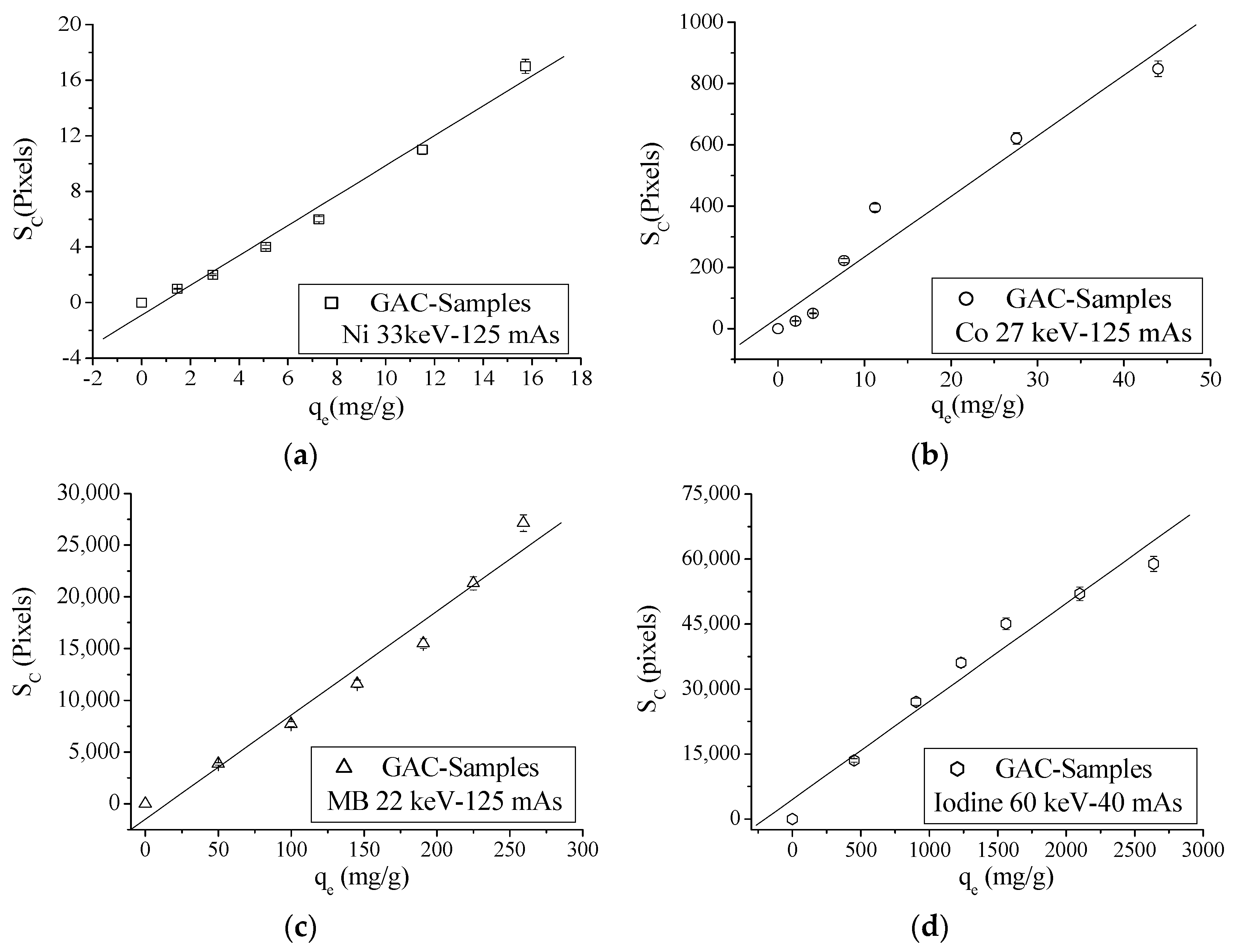



| Element | W | Z | A |
|---|---|---|---|
| H | 0.056722 | 1 | 1 |
| C | 0.600817 | 6 | 12 |
| N | 0.131372 | 7 | 14 |
| S | 0.100251 | 16 | 32 |
| Nickel | Cobalt | Methylene Blue | Iodine | ||||
|---|---|---|---|---|---|---|---|
| Ce | qe | Ce | qe | Ce | qe | Ce | qe |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10.4 | 1.46 | 14.9 | 2.01 | 0.0395 | 49.97 | 665 | 451 |
| 20.8 | 2.92 | 29.8 | 4.03 | 0.079 | 99.94 | 1330 | 903 |
| 74.05 | 5.09 | 86.85 | 7.62 | 23.22 | 145.31 | 9077 | 1232 |
| 127.3 | 7.27 | 143.9 | 11.21 | 46.36 | 190.68 | 16,825 | 1561 |
| 335 | 11.51 | 312.15 | 27.58 | 374.93 | 224.99 | 39,036 | 2099 |
| 542.7 | 15.73 | 480.4 | 43.95 | 703.5 | 259.3 | 61,248 | 2638 |
| Element/Molecule | qm | e (qm) | KL | e (KL) | R2 |
|---|---|---|---|---|---|
| Nickel | 22 | 3 | 0.0040 | ±0.0010 | 0.98 |
| Cobalt | 23 | 4 | 0.0015 | ±0.0001 | 0.92 |
| Methylene Blue | 250 | 40 | 0.063 | ±0.055 | 0.86 |
| Iodine | 2680 | 400 | 0.00001 | ±0.00001 | 0.89 |
| Element/Molecule | qm | e(qm) | KL | e(KL) | R2 |
|---|---|---|---|---|---|
| Nickel | 13 | ±3 | 5 | ±1 | 0.71 |
| Cobalt | 30 | ±4 | 3 | ±1 | 0.28 |
| Methylene Blue | 251 | ±9 | 5 | ±1 | 0.99 |
| Iodine | 2366 | ±3 | 12 | ±4 | 0.92 |
| Element/Molecule | Co (mg/L) | RL |
|---|---|---|
| Nickel | 25–700 | 0.91–0.26 |
| Cobalt | 25–700 | 0.96–0.49 |
| Methylene Blue | 250–2000 | 0.059–0.008 |
| Iodine | 3585–74,438 | 0.96–0.57 |
| Element/Molecule | KF | e(KF) | N | e(n) | R2 |
|---|---|---|---|---|---|
| (a) | |||||
| Nickel | 0.72 | ±0.14 | 0.51 | ±0.01 | 0.99 |
| Cobalt | 0.13 | ±0.04 | 1.00 | ±0.07 | 0.99 |
| Methylene Blue | 106 | ±10 | 0.13 | ±0.01 | 0.99 |
| Iodine | 53 | ±16 | 0.35 | ±0.02 | 0.99 |
| (b) | |||||
| Nickel | 0.44 | ±0.06 | 0.57 | ±0.01 | 0.99 |
| Cobalt | 0.19 | ±0.05 | 0.86 | ±0.05 | 0.99 |
| Methylene Blue | 89 | ±3 | 0.165 | ±0.008 | 0.99 |
| Iodine | 40 | ±3 | 0.377 | ±0.008 | 0.99 |
| Nickel | Pixels in | “1” | |||||
| qe (mg/g) | SC (22 keV) | SC (27 keV) | SC (33 keV) | SC (40 keV) | SC (50 keV) | SC (60 keV) | |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 1.46 | 3205 | 2 | 1 | 0 | 0 | 0 | |
| 2.92 | 6410 | 4 | 2 | 0 | 0 | 0 | |
| 5.09 | 12,365 | 371 | 4 | 0 | 0 | 0 | |
| 7.27 | 18,320 | 738 | 6 | 0 | 0 | 0 | |
| 11.51 | 19,186 | 1040 | 11 | 0 | 0 | 0 | |
| 15.73 | 20,052 | 1342 | 17 | 0 | 0 | 0 | |
| Cobalt | Pixels in | “1” | |||||
| qe (mg/g) | SC (22 keV) | SC (27 keV) | SC (33 keV) | SC (40 keV) | SC (50 keV) | SC (60 keV) | |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 2.01 | 418 | 25 | 2 | 0 | 0 | 0 | |
| 4.03 | 835 | 50 | 5 | 0 | 0 | 0 | |
| 7.62 | 7192 | 222 | 97 | 0 | 0 | 0 | |
| 11.21 | 13,550 | 395 | 190 | 0 | 0 | 0 | |
| 27.58 | 13,800 | 621 | 227 | 0 | 0 | 0 | |
| 43.95 | 14,050 | 848 | 265 | 0 | 0 | 0 | |
| MB | Pixels in | “1” | |||||
| qe (mg/g) | SC (22 keV) | SC (27 keV) | SC (33 keV) | SC (40 keV) | SC (50 keV) | SC (60 keV) | |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 49.97 | 3857 | 15 | 0 | 0 | 0 | 0 | |
| 99.94 | 7714 | 30 | 0 | 0 | 0 | 0 | |
| 145.31 | 11,607 | 51 | 0 | 0 | 0 | 0 | |
| 190.68 | 15,500 | 73 | 0 | 0 | 0 | 0 | |
| 224.99 | 21,315 | 637 | 0 | 0 | 0 | 0 | |
| 259.3 | 27,130 | 1201 | 15 | 0 | 0 | 0 | |
| Iodine | Pixels in | “1” | |||||
| qe (mg/g) | SC (40 keV) | SC (50 keV) | SC (60 keV) | SC (70 keV) | SC (80 keV) | SC (90 keV) | |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 451 | 31,545 | 34,810 | 13,525 | 0 | 0 | 0 | |
| 903 | 63,090 | 69,620 | 27,050 | 0 | 0 | 0 | |
| 1232 | 51,470 | 74,755 | 36,060 | 0 | 0 | 0 | |
| 1561 | 69,850 | 79,890 | 45,070 | 0 | 0 | 0 | |
| 2099 | 57,575 | 81,285 | 51,985 | 0 | 0 | 0 | |
| 2638 | 75,300 | 82,680 | 58,900 | 0 | 0 | 0 |
| Nickel | |||||
| Evaluated Parameter | A (Pixels) | e(A) | B (Pixels·g/mg) | e(B) | R2 |
| SC (22 keV–125 mAs) | 3027 | ±4 | 1326 | ±162 | 0.91 |
| SC (27 keV–125 mAs) | −98 | ±7 | 95 | ±8 | 0.98 |
| SC (33 keV–125 mAs) | −0.900 | ±0.001 | 1.03 | ±0.05 | 0.99 |
| Cobalt | |||||
| Evaluated Parameter | A (Pixels) | e(A) | B (Pixels·g/mg) | e(B) | R2 |
| SC (22 keV–125 mAs) | 2533 | ±200 | 333 | ±10 | 0.81 |
| SC (27 keV–125 mAs) | 36 | ±10 | 20 | ±2 | 0.97 |
| SC (33 keV–125 mAs) | 25 | ±12 | 6 | ±1 | 0.89 |
| Methylene Blue | |||||
| Evaluated Parameter | A (Pixels) | e(A) | B (Pixels·g/mg) | e(B) | R2 |
| SC (22 keV–125 mAs) | −1481 | ±129 | 100 | ±8 | 0.98 |
| SC (27 keV–125 mAs) | −237 | ±23 | 4 | ±1 | 0.76 |
| SC (33 keV–125 mAs) | −2.56 | ±0.54 | 0.03 | ±0.02 | 0.56 |
| Iodine | |||||
| Evaluated Parameter | A (Pixels) | e(A) | B (Pixels·g/mg) | e(B) | R2 |
| SC (40 keV–40 mAs) | 19,647 | ±160 | 24 | ±7 | 0.83 |
| SC (50 keV–40 mAs) | 23,536 | ±122 | 29 | ±8 | 0.85 |
| SC (60 keV–40 mAs) | 4498 | ±50 | 22 | ±2 | 0.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puente Torres, J.; Crespo Sariol, H.; Mariño Peacok, T.; Yperman, J.; Adriaensens, P.; Carleer, R.; Brito Sauvanell, Á. X-ray Absorption (XRA): A New Technique for the Characterization of Granular Activated Carbons. Materials 2021, 14, 91. https://doi.org/10.3390/ma14010091
Puente Torres J, Crespo Sariol H, Mariño Peacok T, Yperman J, Adriaensens P, Carleer R, Brito Sauvanell Á. X-ray Absorption (XRA): A New Technique for the Characterization of Granular Activated Carbons. Materials. 2021; 14(1):91. https://doi.org/10.3390/ma14010091
Chicago/Turabian StylePuente Torres, Jeamichel, Harold Crespo Sariol, Thayset Mariño Peacok, Jan Yperman, Peter Adriaensens, Robert Carleer, and Ángel Brito Sauvanell. 2021. "X-ray Absorption (XRA): A New Technique for the Characterization of Granular Activated Carbons" Materials 14, no. 1: 91. https://doi.org/10.3390/ma14010091
APA StylePuente Torres, J., Crespo Sariol, H., Mariño Peacok, T., Yperman, J., Adriaensens, P., Carleer, R., & Brito Sauvanell, Á. (2021). X-ray Absorption (XRA): A New Technique for the Characterization of Granular Activated Carbons. Materials, 14(1), 91. https://doi.org/10.3390/ma14010091







