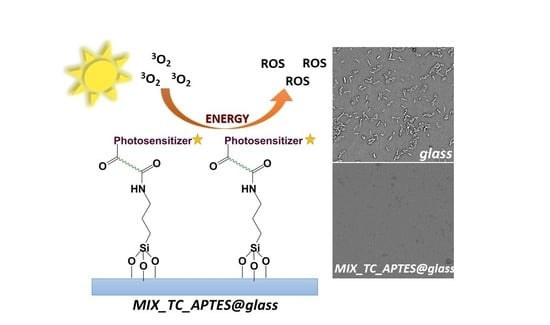
Covalent Immobilization of Organic Photosensitizers on the Glass Surface: Toward the Formation of the Light-Activated Antimicrobial Nanocoating
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Formation and Characterization of the Photoactive Layer
2.2.1. Glass Substrate Pretreatment
2.2.2. Photoactive Monolayer Formation
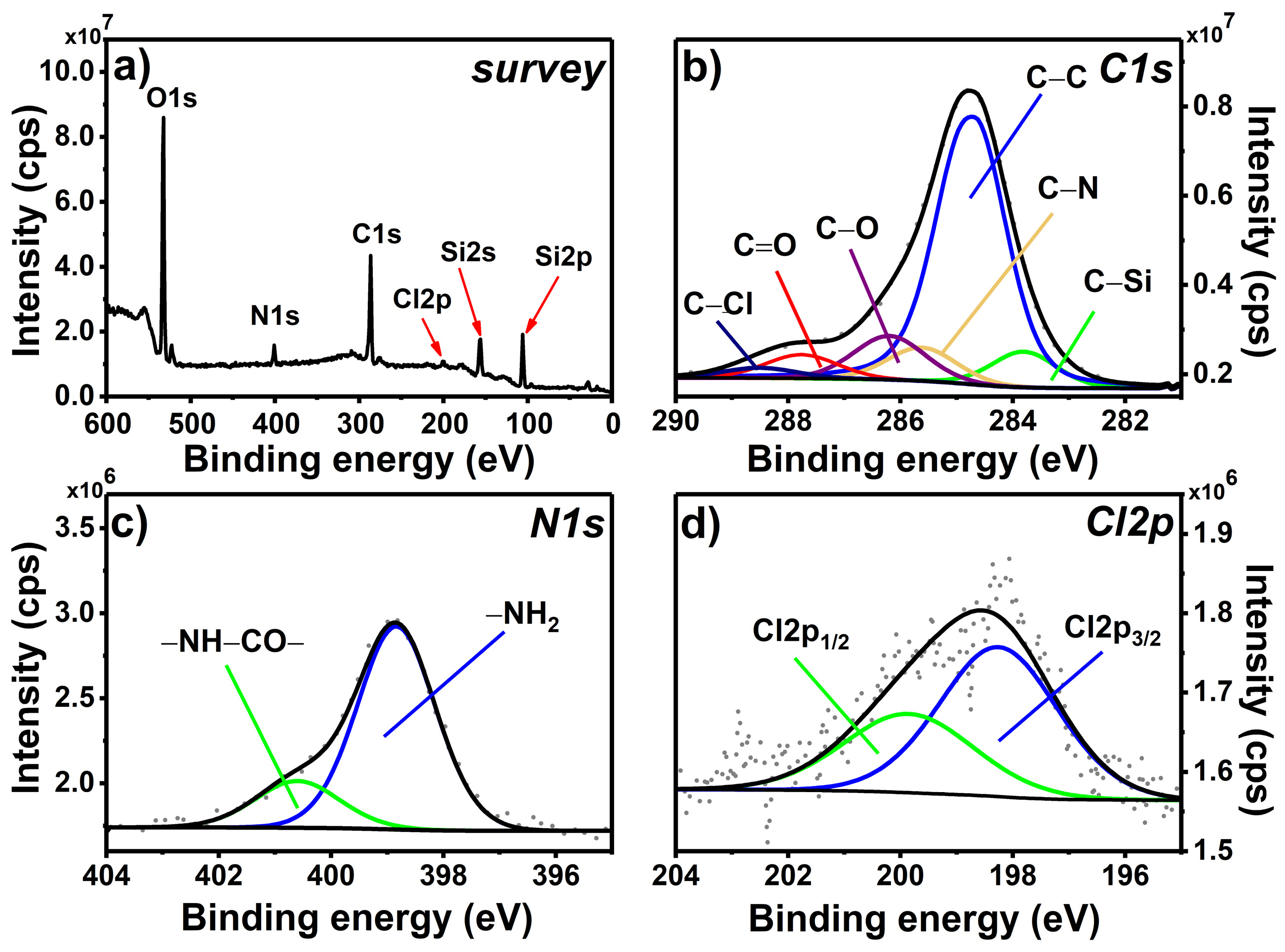
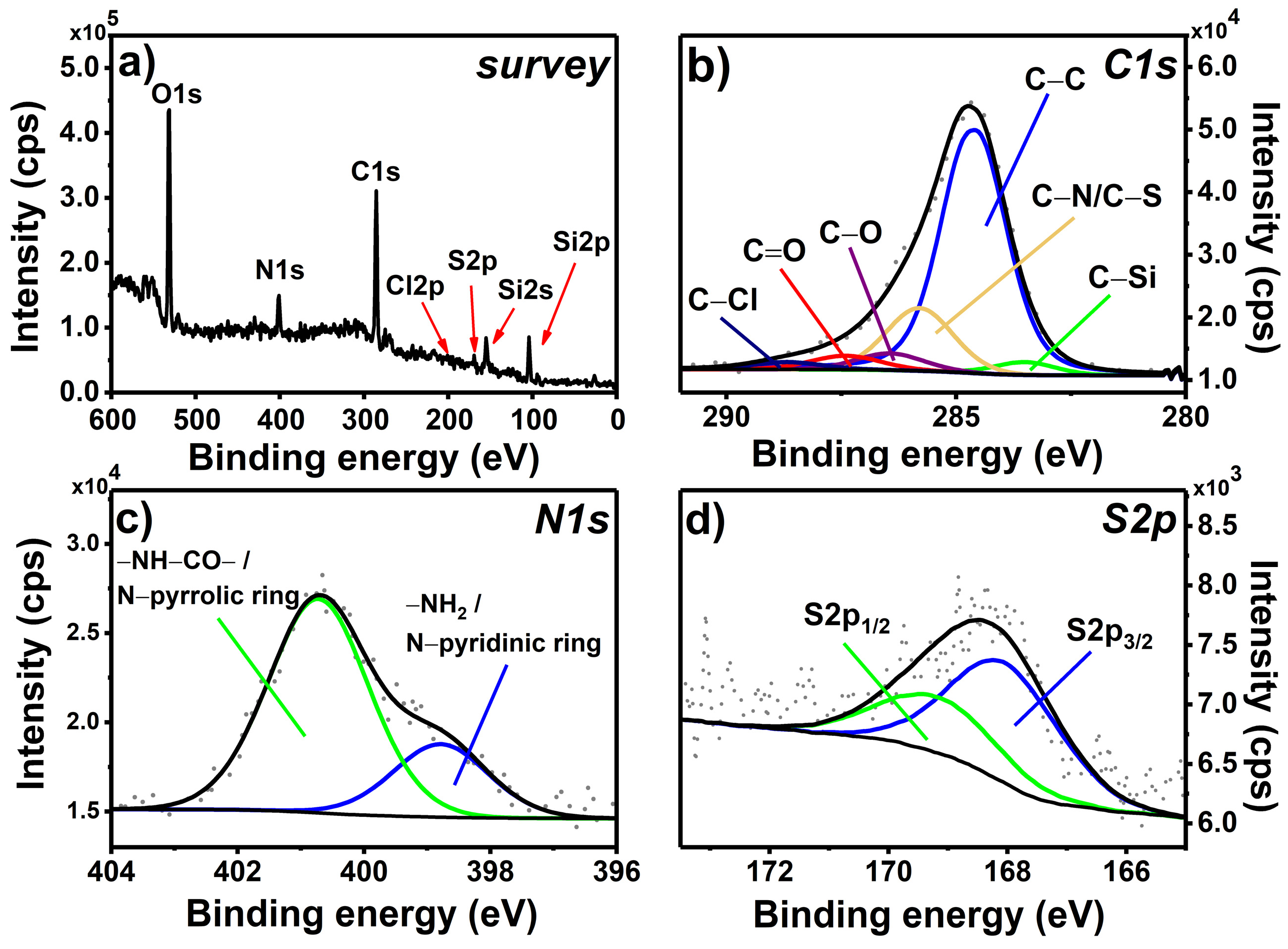
2.2.3. X-ray Photoelectron Spectroscopy
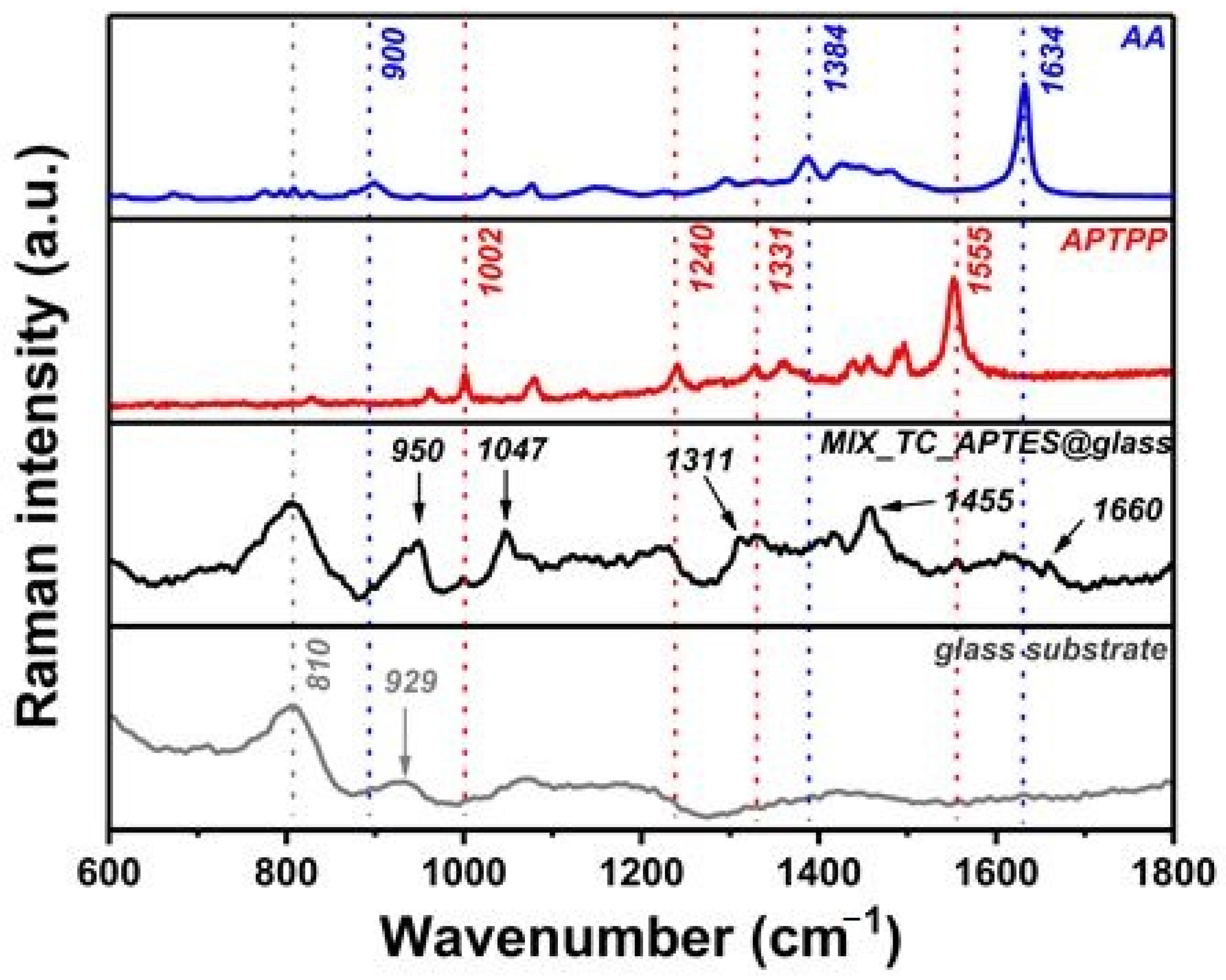
2.2.4. Raman Spectroscopy
2.3. Photogeneration of Reactive Oxygen Species (ROS)
2.4. Microbiological Analysis
2.4.1. Bacterial Strain and Culture Conditions
2.4.2. Bacterial Cell Staining and Imaging
3. Results and Discussion
3.1. Chemical Grafting and Spectroscopic Characterization of the Photoactive Mixed Monolayer (MIX_TC_APTES@glass)
3.1.1. Chemical Grafting and XPS Spectroscopy
3.1.2. Raman Spectroscopy
3.2. Reactive Oxygen Species (ROS) Photogeneration by the Chemically Grafted Mixed Monolayer
3.3. Antimicrobial Properties of the Chemically Grafted Mixed Monolayer
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allegranzi, B.; Bagheri, S.N.; Castillejos, G.G.; Kilpatrick, C.; Kelley, E.; Mathiai, E. Report on the Burden of Endemic Health Care-Associated Infection Worldwide-Clean Care Is Safer Care; WHO: Geneva, Switzerland, 2011; Volume 3, pp. 1–34. [Google Scholar]
- Grammatikova, N.E.; George, L.; Ahmed, Z.; Candeias, N.R.; Durandin, N.A.; Efimov, A. Zinc phthalocyanine activated by conventional indoor light makes a highly efficient antimicrobial material from regular cellulose. J. Mater. Chem. B 2019, 7, 4379–4384. [Google Scholar] [CrossRef]
- Noimark, S.; Dunnill, C.W.; Parkin, I.P. Shining light on materials — A self-sterilising revolution. Adv. Drug Deliv. Rev. 2013, 65, 570–580. [Google Scholar] [CrossRef]
- Sautrot-Ba, P.; Malval, J.-P.; Weiss-Maurin, M.; Paul, J.; Blacha-Grzechnik, A.; Tomane, S.; Mazeran, P.-E.; Lalevée, J.; Langlois, V.; Versace, D.-L. Paprika, Gallic Acid, and Visible Light: The Green Combination for the Synthesis of Biocide Coatings. ACS Sustain. Chem. Eng. 2017, 6, 104–109. [Google Scholar] [CrossRef]
- Hwang, G.B.; Allan, E.; Parkin, I.P. White Light-Activated Antimicrobial Paint using Crystal Violet. ACS Appl. Mater. Interfaces 2015, 8, 15033–15039. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Canales, M.; Noimark, S.; Page, K.; Parkin, I.; Faull, J.; Bhatti, M.; Ciric, L. A Light-Activated Antimicrobial Surface Is Active Against Bacterial, Viral and Fungal Organisms. Sci. Rep. 2017, 7, 15298. [Google Scholar] [CrossRef] [PubMed]
- DeRosa, M.C. Photosensitized singlet oxygen and its applications. Coord. Chem. Rev. 2002, 233-234, 351–371. [Google Scholar] [CrossRef]
- Wainwright, M.; Crossley, K.B. Photosensitising agents—Circumventing resistance and breaking down biofilms: A review. Int. Biodeterior. Biodegrad. 2004, 53, 119–126. [Google Scholar] [CrossRef]
- Spagnul, C.; Turner, L.C.; Boyle, R.W. Immobilized photosensitizers for antimicrobial applications. J. Photochem. Photobiol. B Biol. 2015, 150, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Mangoni, M.L.; Epand, R.F.; Rosenfeld, Y.; Peleg, A.; Barra, D.; Epand, R.M.; Shai, Y. Lipopolysaccharide, a Key Molecule Involved in the Synergism between Temporins in Inhibiting Bacterial Growth and in Endotoxin Neutralization. J. Biol. Chem. 2008, 283, 22907–22917. [Google Scholar] [CrossRef] [PubMed]
- Dahl, T.A.; Midden, W.R.; Hartman, P.E. Pure Singlet Oxygen Cytotoxicity for Bacteria. Photochem. Photobiol. 1987, 46, 345–352. [Google Scholar] [CrossRef]
- Laurentius, L.; Stoyanov, S.R.; Gusarov, S.; Kovalenko, A.; Du, R.; Lopinski, G.P.; McDermott, M. Diazonium-Derived Aryl Films on Gold Nanoparticles: Evidence for a Carbon–Gold Covalent Bond. ACS Nano 2011, 5, 4219–4227. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, A. Surface Treatments for Biological, Chemical and Physical Applications. Johns. Matthey Technol. Rev. 2018, 62, 259–262. [Google Scholar] [CrossRef]
- Namba, N.; Yoshida, Y.; Nagaoka, N.; Takashima, S.; Matsuura-Yoshimoto, K.; Maeda, H.; Van Meerbeek, B.; Suzuki, K.; Takashiba, S. Antibacterial effect of bactericide immobilized in resin matrix. Dent. Mater. 2009, 25, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Page, K.; Correia, A.; Wilson, M.; Allan, E.; Parkin, I.P. Light-activated antibacterial screen protectors for mobile telephones and tablet computers. J. Photochem. Photobiol. A Chem. 2015, 296, 19–24. [Google Scholar] [CrossRef]
- Peveler, W.J.; Noimark, S.; Al-Azawi, H.; Hwang, G.B.; Crick, C.R.; Allan, E.; Edel, J.B.; Ivanov, A.; MacRobert, A.J.; Parkin, I.P. Covalently Attached Antimicrobial Surfaces Using BODIPY: Improving Efficiency and Effectiveness. ACS Appl. Mater. Interfaces 2018, 10, 98–104. [Google Scholar] [CrossRef]
- Piccirillo, C.; Perni, S.; Gil-Thomas, J.; Prokopovich, P.; Wilson, M.; Pratten, J.; Parkin, I.P. Antimicrobial activity of methylene blue and toluidine blue O covalently bound to a modified silicone polymer surface. J. Mater. Chem. 2009, 19, 6167–6171. [Google Scholar] [CrossRef][Green Version]
- Sehmi, S.K.; Noimark, S.; Pike, S.; Bear, J.C.; Peveler, W.J.; Williams, C.K.; Shaffer, M.S.P.; Allan, E.; Parkin, I.P.; MacRobert, A.J. Enhancing the Antibacterial Activity of Light-Activated Surfaces Containing Crystal Violet and ZnO Nanoparticles: Investigation of Nanoparticle Size, Capping Ligand, and Dopants. ACS Omega 2016, 1, 334–343. [Google Scholar] [CrossRef]
- Decraene, V.; Pratten, J.; Wilson, M. Cellulose Acetate Containing Toluidine Blue and Rose Bengal Is an Effective Antimicrobial Coating when Exposed to White Light. Appl. Environ. Microbiol. 2006, 72, 4436–4439. [Google Scholar] [CrossRef] [PubMed]
- Decraene, V.; Pratten, J.; Wilson, M. Novel Light-Activated Antimicrobial Coatings Are Effective Against Surface-Deposited Staphylococcus aureus. Curr. Microbiol. 2008, 57, 269–273. [Google Scholar] [CrossRef]
- Ballatore, M.B.; Durantini, J.; Gsponer, N.S.; Suarez, M.B.; Gervaldo, M.; Otero, L.; Spesia, M.B.; Milanesio, M.E.; Durantini, E.N. Photodynamic Inactivation of Bacteria Using Novel Electrogenerated Porphyrin-Fullerene C60 Polymeric Films. Environ. Sci. Technol. 2015, 49, 7456–7463. [Google Scholar] [CrossRef] [PubMed]
- Page, K.; Wilson, M.; Parkin, I.P. Antimicrobial surfaces and their potential in reducing the role of the inanimate environment in the incidence of hospital-acquired infections. J. Mater. Chem. 2009, 19, 3819–3831. [Google Scholar] [CrossRef]
- Sangermano, M.; Periolatto, M.; Castellino, M.; Wang, J.; Dietliker, K.; Grützmacher, J.L.; Grützmacher, H. A Simple Preparation of Photoactive Glass Surfaces Allowing Coatings via the “Grafting-from” Method. ACS Appl. Mater. Interfaces 2016, 8, 19764–19771. [Google Scholar] [CrossRef]
- Ringot, C.; Sol, V.; Barrière, M.; Saad, N.; Bressollier, P.; Granet, R.; Couleaud, P.; Frochot, C.; Krausz, P. Triazinyl Porphyrin-Based Photoactive Cotton Fabrics: Preparation, Characterization, and Antibacterial Activity. Biomacromolecules 2011, 12, 1716–1723. [Google Scholar] [CrossRef]
- Ribeiro, S.M.; Serra, A.C.; Gonsalves, A.R. Covalently immobilized porphyrins as photooxidation catalysts. Tetrahedron 2007, 63, 7885–7891. [Google Scholar] [CrossRef][Green Version]
- Krouit, M.; Granet, R.; Krausz, P. Photobactericidal films from porphyrins grafted to alkylated cellulose—Synthesis and bactericidal properties. Eur. Polym. J. 2009, 45, 1250–1259. [Google Scholar] [CrossRef]
- Bélanger, D.; Pinson, J. Electrografting: A powerful method for surface modification. Chem. Soc. Rev. 2011, 40, 3995–4048. [Google Scholar] [CrossRef]
- Blacha-Grzechnik, A.; Piwowar, K.; Kościelniak, P.; Kwoka, M.; Szuber, J.; Zak, J. Phenothiazines grafted on the electrode surface from diazonium salts as molecular layers for photochemical generation of singlet oxygen. Electrochim. Acta 2015, 182, 1085–1092. [Google Scholar] [CrossRef]
- Lv, Y.; Liu, H.; Wang, Z.; Hao, L.; Liu, J.; Wang, Y.; Du, G.; Liu, D.; Zhan, J.; Wang, J. Antibiotic glass slide coated with silver nanoparticles and its antimicrobial capabilities. Polym. Adv. Technol. 2008, 19, 1455–1460. [Google Scholar] [CrossRef]
- Saravanan, P.; Jayamoorthy, K.; Anandakumar, S. Fluorescence quenching of APTES by Fe2O3 nanoparticles—Sensor and antibacterial applications. J. Lumin. 2016, 178, 241–248. [Google Scholar] [CrossRef]
- Metwalli, E.; Haines, D.; Becker, O.; Conzone, S.; Pantano, C. Surface characterizations of mono-, di-, and tri-aminosilane treated glass substrates. J. Colloid Interface Sci. 2006, 298, 825–831. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Optimization of Silica Silanization by 3-Aminopropyltriethoxysilane. Langmuir 2006, 22, 11142–11147. [Google Scholar] [CrossRef]
- Acres, R.; Ellis, A.V.; Alvino, J.; Lenahan, C.E.; Khodakov, D.A.; Metha, G.F.; Andersson, G. Molecular Structure of 3-Aminopropyltriethoxysilane Layers Formed on Silanol-Terminated Silicon Surfaces. J. Phys. Chem. C 2012, 116, 6289–6297. [Google Scholar] [CrossRef]
- Cui, N.-Y.; Liu, C.; Yang, W. XPS and AFM characterization of the self-assembled molecular monolayers of a 3-aminopropyltrimethoxysilane on silicon surface, and effects of substrate pretreatment by UV-irradiation. Surf. Interface Anal. 2010, 43, 1082–1088. [Google Scholar] [CrossRef]
- Chang, C.-C.; Imae, T.; Chen, L.-Y.; Ujihara, M. Efficient surface enhanced Raman scattering on confeito-like gold nanoparticle-adsorbed self-assembled monolayers. Phys. Chem. Chem. Phys. 2015, 17, 32328–32334. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K.; Lam, E.; Hrapovic, S.; Male, K.B.; Luong, J. Immobilization of Antibodies and Enzymes on 3-Aminopropyltriethoxysilane-Functionalized Bioanalytical Platforms for Biosensors and Diagnostics. Chem. Rev. 2014, 114, 11083–11130. [Google Scholar] [CrossRef]
- Beamson, G.; Briggs, D. XPS of Organic Polymers; John Wiley & Sons: Chichester, UK, 1992. [Google Scholar]
- Shircliff, R.A.; Stradins, P.; Moutinho, H.; Fennell, J.; Ghirardi, M.L.; Cowley, S.W.; Branz, H.M.; Martin, I. Angle-Resolved XPS Analysis and Characterization of Monolayer and Multilayer Silane Films for DNA Coupling to Silica. Langmuir 2013, 29, 4057–4067. [Google Scholar] [CrossRef]
- Marbach, H. Surface-Mediated in Situ Metalation of Porphyrins at the Solid–Vacuum Interface. Acc. Chem. Res. 2015, 48, 2649–2658. [Google Scholar] [CrossRef]
- Sun, Z.; Li, J.; Zheng, H.; Liu, X.; Ye, S.; Du, P. Pyrolyzed cobalt porphyrin-modified carbon nanomaterial as an active catalyst for electrocatalytic water oxidation. Int. J. Hydrogen Energy 2015, 40, 6538–6545. [Google Scholar] [CrossRef]
- Thi, N.; Hoan, V.; Minh, N.N.; Thi, T.; Nhi, K.; Thang, N.V.; Tuan, V.A.; Nguyen, V.T.; Thanh, N.M.; Hung, N. Van TiO2/Diazonium/Graphene Oxide Composites: Synthesis and Visible-Light-Driven Photocatalytic Degradation of Methylene Blue. J. Nanomater. 2020, 2020. [Google Scholar] [CrossRef]
- Manghnani, M.H.; Hushur, A.; Sekine, T.; Wu, J.; Stebbins, J.F.; Williams, Q. Raman, Brillouin, and nuclear magnetic resonance spectroscopic studies on shocked borosilicate glass. J. Appl. Phys. 2011, 109, 113509. [Google Scholar] [CrossRef]
- Sun, Y.; Yanagisawa, M.; Kunimoto, M.; Nakamura, M.; Homma, T. Depth profiling of APTES self-assembled monolayers using surface-enhanced confocal Raman microspectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 184, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gunda, N.S.K.; Singh, M.; Norman, L.; Kaur, K.; Mitra, S. Optimization and characterization of biomolecule immobilization on silicon substrates using (3-aminopropyl)triethoxysilane (APTES) and glutaraldehyde linker. Appl. Surf. Sci. 2014, 305, 522–530. [Google Scholar] [CrossRef]
- Foggia, M.D.; Taddei, P.; Torreggiani, A.; National, I.; Dettin, M. Self-assembling peptides for biomedical applications: IR and Raman spectroscopies for the study of secondary structure. J. Proteome Res. 2012, 2, 231–272. [Google Scholar]
- Snehalatha, M.; Joe, I.H.; Ravikumar, C.; Jayakumar, V.S. Azure A chloride: Computational and spectroscopic study. J. Raman Spectrosc. 2009, 40, 176–182. [Google Scholar] [CrossRef]
- Lokesh, K.S.; De Keersmaecker, M.; Adriaens, A. Self Assembled Films of Porphyrins with Amine Groups at Different Positions: Influence of Their Orientation on the Corrosion Inhibition and the Electrocatalytic Activity. Molecules 2012, 17, 7824–7842. [Google Scholar] [CrossRef]
- Aydin, M. DFT and Raman spectroscopy of porphyrin derivatives: Tetraphenylporphine (TPP). Vib. Spectrosc. 2013, 68, 141–152. [Google Scholar] [CrossRef]
- Nyga, A.; Motyka, R.; Bussetti, G.; Calloni, A.; Jagadeesh, M.S.; Fijak, S.; Pluczyk-Malek, S.; Data, P.; Blacha-Grzechnik, A. Electrochemically deposited poly(selenophene)-fullerene photoactive layer: Tuning of the spectroscopic properties towards visible light-driven generation of singlet oxygen. Appl. Surf. Sci. 2020, 525, 146594. [Google Scholar] [CrossRef]
- Blacha-Grzechnik, A.; Piwowar, K.; Zdyb, T.; Krzywiecki, M. Formation of poly(Azure A)-C60 photoactive layer as a novel approach in the heterogeneous photogeneration of singlet oxygen. Appl. Surf. Sci. 2018, 457, 221–228. [Google Scholar] [CrossRef]
- Toval, F.; Köhler, C.-D.; Vogel, U.; Wagenlehner, F.; Mellmann, A.; Fruth, A.; Schmidt, M.A.; Karch, H.; Bielaszewska, M.; Dobrindt, U.; et al. Characterization of Escherichia coli Isolates from Hospital Inpatients or Outpatients with Urinary Tract Infection. J. Clin. Microbiol. 2014, 52, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Peleg, A.Y.; Hooper, D.C. Hospital-Acquired Infections Due to Gram-Negative Bacteria Anton. J. Med. N. Engl. 2010, 362, 1804–1813. [Google Scholar] [CrossRef]
- Nagarjuna, D.; Mittal, G.S.; Dhanda, R.; Verma, P.; Gaind, R.; Yadav, M. Faecal Escherichia coli isolates show potential to cause endogenous infection in patients admitted to the ICU in a tertiary care hospital. New Microbes New Infect. 2015, 7, 57–66. [Google Scholar] [CrossRef]
- Wang, K.-K.; Kim, B.-J.; Heo, I.-; Jung, S.-J.; Hwang, J.-W.; Kim, Y.-R. Fabrication and characterization of antimicrobial surface-modified stainless steel for bio-application. Surf. Coat. Technol. 2017, 310, 256–262. [Google Scholar] [CrossRef]
- Zhou, X.; Chen, Z.; Wang, Y.; Guo, Y.; Tung, C.-H.; Zhang, F.; Liu, X. Honeycomb-patterned phthalocyanine films with photo-active antibacterial activities. Chem. Commun. 2013, 49, 10614–10616. [Google Scholar] [CrossRef]
- Ringot, C.; Sol, V.; Granet, R.; Krausz, P. Porphyrin-grafted cellulose fabric: New photobactericidal material obtained by “Click-Chemistry” reaction. Mater. Lett. 2009, 63, 1889–1891. [Google Scholar] [CrossRef]
- Modjinou, T.; Versace, D.L.; Andaloussi, S.A.; Langlois, V.; Renard, E. Co-Networks Poly(hydroxyalkanoates)-Terpenes to Enhance Antibacterial Properties. Bioengineering 2020, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Mobeen, A.; Magdalane, C.M.; Shahina, S.J.; Lakshmi, D.; Sundaram, R.; Ramalingam, G.; Raja, A.; Madhavan, J.; Letsholathebe, D.; Bashir, A.; et al. Investigation on antibacterial and photocatalytic degradation of Rhodamine-B dye under visible light irradiation by titanium molybdate nanoparticles prepared via microwave method. Surf. Interfaces 2019, 17, 100381. [Google Scholar] [CrossRef]
- Nuñez, J.A.P.; Salapare, H.S.; Villamayor, M.M.S.; Siringan, M.A.T.; Ramos, H.J. Antibacterial efficiency of magnetron sputtered TiO2 on poly(methyl methacrylate). Surf. Interfaces 2017, 8, 28–35. [Google Scholar] [CrossRef]
- Cardoso, V.; Rittmeyer, T.; Correa, R.J.; Brêda, G.C.; Almeida, R.V.; Simões, G.; de França, B.M.; de Azevedo, P.N.; Forero, J.S.B. Photoactive cotton fabric: Synthesis, characterization and antibacterial evaluation of anthraquinone-based dyes linked to cellulose. Dyes Pigm. 2019, 161, 16–23. [Google Scholar] [CrossRef]
- Tu, Q.; Zhang, Q.; Wang, Y.; Jiao, Y.; Xiao, J.; Peng, T.; Wang, J. Antibacterial properties of poly(dimethylsiloxane) surfaces modified with graphene oxide-catechol composite. Prog. Org. Coat. 2019, 129, 247–253. [Google Scholar] [CrossRef]
- Ferreira, A.M.; Carmagnola, I.; Chiono, V.; Gentile, P.; Fracchia, L.; Ceresa, C.; Georgiev, G.; Ciardelli, G. Surface modification of poly(dimethylsiloxane) by two-step plasma treatment for further grafting with chitosan–Rose Bengal photosensitizer. Surf. Coat. Technol. 2013, 223, 92–97. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nyga, A.; Czerwińska-Główka, D.; Krzywiecki, M.; Przystaś, W.; Zabłocka-Godlewska, E.; Student, S.; Kwoka, M.; Data, P.; Blacha-Grzechnik, A. Covalent Immobilization of Organic Photosensitizers on the Glass Surface: Toward the Formation of the Light-Activated Antimicrobial Nanocoating. Materials 2021, 14, 3093. https://doi.org/10.3390/ma14113093
Nyga A, Czerwińska-Główka D, Krzywiecki M, Przystaś W, Zabłocka-Godlewska E, Student S, Kwoka M, Data P, Blacha-Grzechnik A. Covalent Immobilization of Organic Photosensitizers on the Glass Surface: Toward the Formation of the Light-Activated Antimicrobial Nanocoating. Materials. 2021; 14(11):3093. https://doi.org/10.3390/ma14113093
Chicago/Turabian StyleNyga, Aleksandra, Dominika Czerwińska-Główka, Maciej Krzywiecki, Wioletta Przystaś, Ewa Zabłocka-Godlewska, Sebastian Student, Monika Kwoka, Przemysław Data, and Agata Blacha-Grzechnik. 2021. "Covalent Immobilization of Organic Photosensitizers on the Glass Surface: Toward the Formation of the Light-Activated Antimicrobial Nanocoating" Materials 14, no. 11: 3093. https://doi.org/10.3390/ma14113093
APA StyleNyga, A., Czerwińska-Główka, D., Krzywiecki, M., Przystaś, W., Zabłocka-Godlewska, E., Student, S., Kwoka, M., Data, P., & Blacha-Grzechnik, A. (2021). Covalent Immobilization of Organic Photosensitizers on the Glass Surface: Toward the Formation of the Light-Activated Antimicrobial Nanocoating. Materials, 14(11), 3093. https://doi.org/10.3390/ma14113093