Pectin as Rheology Modifier of a Gelatin-Based Biomaterial Ink
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of GELPect Slurries and GELPectG Microporous Sponges
2.3. Physical Characterization of GELPectG and Microporous Sponges
2.4. Biological Characterization of GELPectG Microporous Sponges
2.5. Preparation and Rheological Investigation of GELPectG Biomaterial Inks
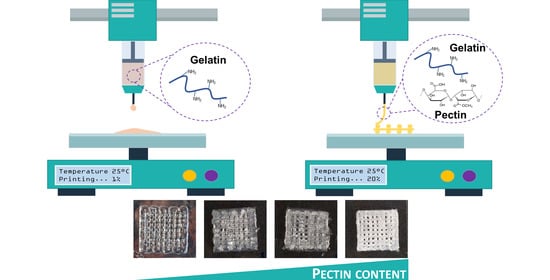
2.6. Extrusion-Based 3D Bioprinting of GELPectG Biomaterial Inks
2.7. Statistical Analysis
3. Results and Discussion
3.1. Preparation of GELPect Slurries
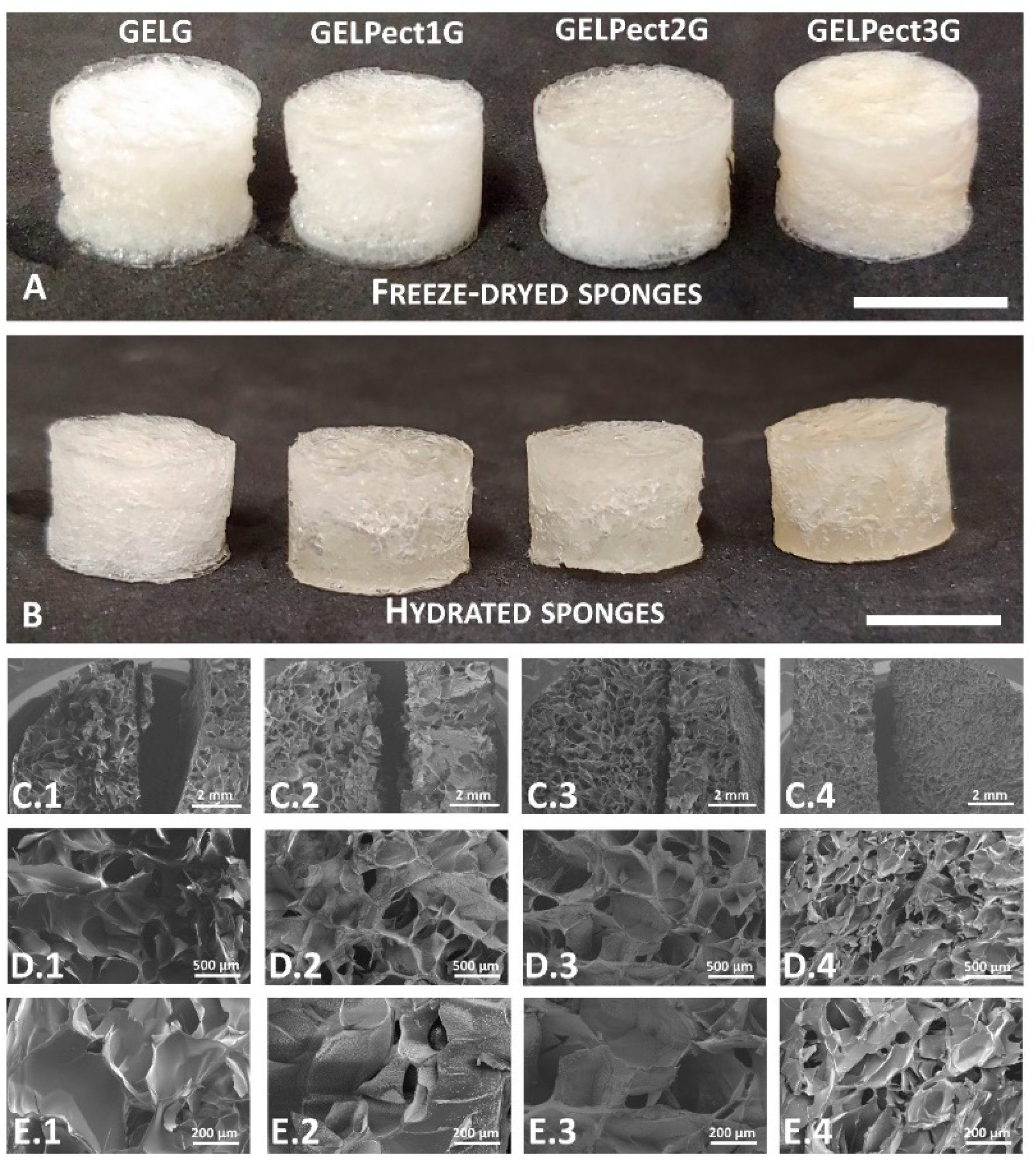
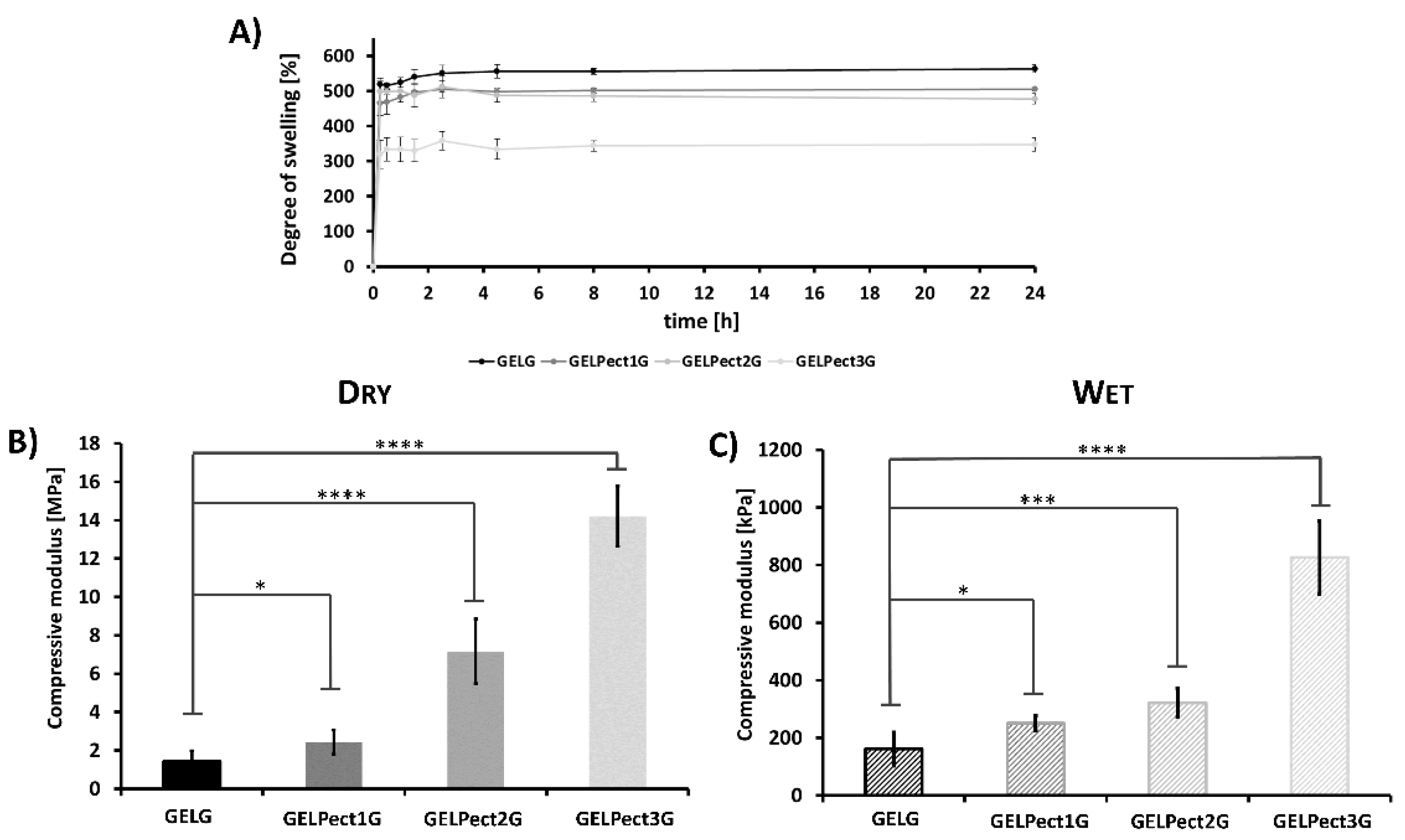
3.2. Physical Characterization of GELPectG Microporous Sponges
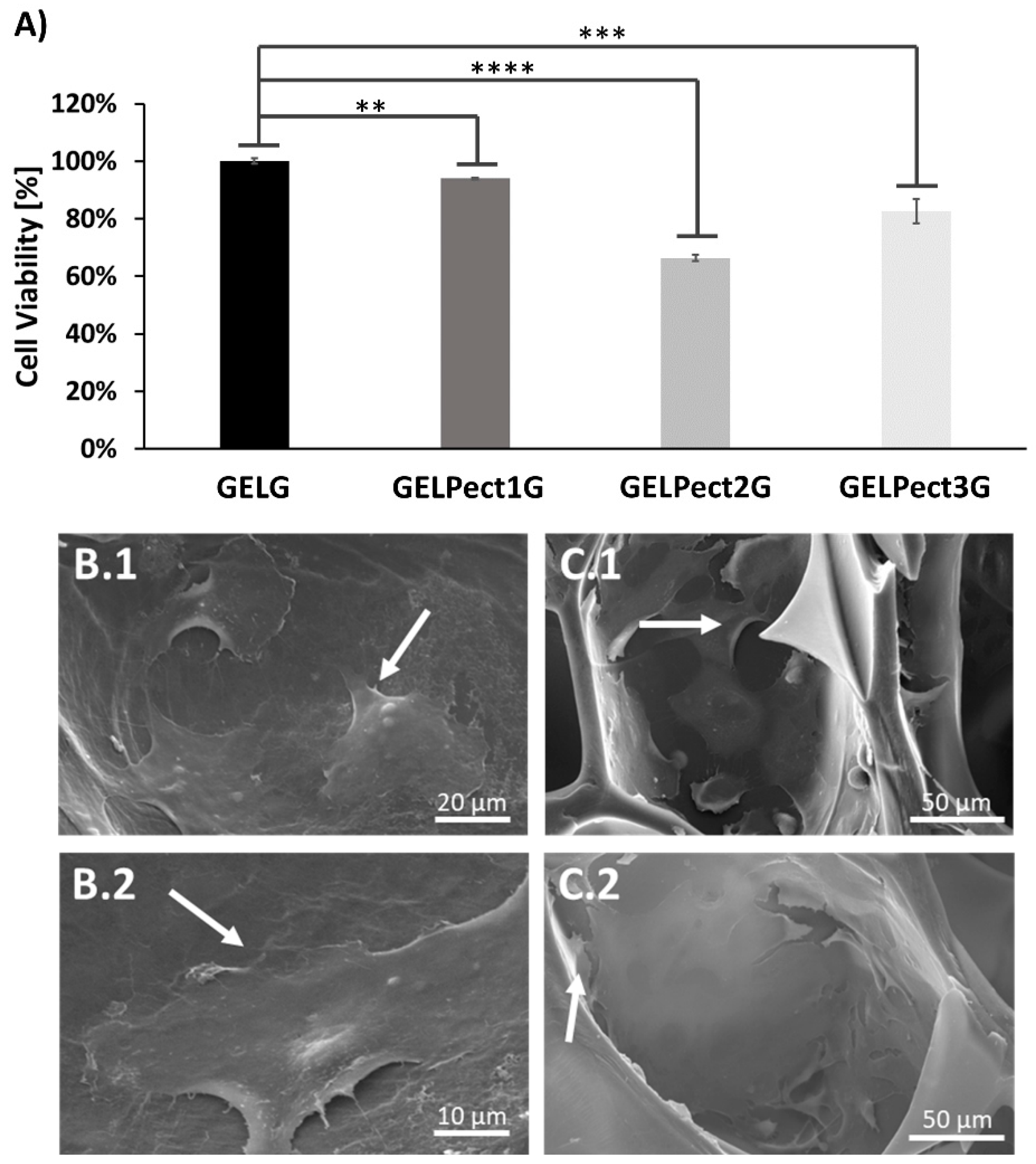
3.3. Biological Characterization of GELPectG Sponges
3.4. Extrusion-Based Bioprinting of GELPectG Biomaterial Inks
3.4.1. Preparation of GELPectG Biomaterial Inks and Rheological Investigation
3.4.2. Bioprinting Assessment of GELPectG Biomaterial Inks
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paxton, N.; Smolan, W.; Böck, T.; Melchels, F.; Groll, J.; Jungst, T. Proposal to assess printability of bioinks for extrusion-based bioprinting and evaluation of rheological properties governing bioprintability. Biofabrication 2017, 9, 044107. [Google Scholar] [CrossRef]
- Levato, R.; Jungst, T.; Scheuring, R.G.; Blunk, T.; Groll, J.; Malda, J. From Shape to Function: The Next Step in Bioprinting. Adv. Mater. 2020, 32, 1906423. [Google Scholar] [CrossRef]
- Groll, J.; Burdick, J.A.; Cho, D.-W.; Derby, B.; Gelinsky, M.; Heilshorn, S.C.; Jüngst, T.; Malda, J.; Mironov, V.A.; Nakayama, K.; et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 2019, 11, 013001. [Google Scholar] [CrossRef] [PubMed]
- Moroni, L.; Boland, T.; Burdick, J.A.; De Maria, C.; Derby, B.; Forgacs, G.; Groll, J.; Li, Q.; Malda, J.; Mironov, V.A.; et al. Biofabrication: A Guide to Technology and Terminology. Trends Biotechnol. 2018, 36.4, 384–402. [Google Scholar] [CrossRef] [Green Version]
- Duty, C.; Ajinjeru, C.; Kishore, V.; Compton, B.; Hmeidat, N.; Chen, X.; Liu, P.; Hassen, A.A.; Lindahl, J.; Kunc, V. What makes a material printable? A viscoelastic model for extrusion-based 3D printing of polymers. J. Manuf. Process. 2018, 35, 526–537. [Google Scholar] [CrossRef]
- Hölzl, K.; Lin, S.; Tytgat, L.; Van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink properties before, during and after 3D bioprinting. Biofabrication 2016, 8, 032002. [Google Scholar] [CrossRef]
- Lee, S.C.; Gillispie, G.; Prim, P.; Lee, S.J. Physical and Chemical Factors Influencing the Printability of Hydrogel-based Extrusion Bioinks. Chem. Rev. 2020, 20, 10834–10886. [Google Scholar] [CrossRef] [PubMed]
- CELLINK Bioink—CELLINK. Available online: https://www.cellink.com/product/cellink-bioink/ (accessed on 30 April 2021).
- Collagen Lifeink® 200|Biomaterials & Bioinks—Allevi. Available online: https://www.allevi3d.com/product/collagen-lifeink-200/ (accessed on 30 April 2021).
- Protocols—Allevi. Available online: https://www.allevi3d.com/protocols/ (accessed on 30 April 2021).
- Kozlov, P.V.; Burdygina, G. The structure and properties of solid gelatin and the principles of their modification. Polym. Rev. 1981, 24, 651–666. [Google Scholar] [CrossRef]
- Echave, M.C.; Burgo, L.S.; Pedraz, J.L.; Orive, G. Gelatin as Biomaterial for Tissue Engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef] [PubMed]
- Hay, E.D. Cell Biology of Extracellular Matrix; Springer Science & Business Media: New York, NY, USA, 2013. [Google Scholar]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.C.; Chang, W.H.; Dong, G.C.; Chen, K.Y.; Chen, Y.S.; Yao, C.H. Cell adhesion and proliferation enhancement by gelatin nanofiber scaffolds. J. Bioact. Compat. Polym. 2011, 26, 565–577. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M.; Yarwood, S.J.; Curtis, A.S.G. Effect of cellular uptake of gelatin nanoparticles on adhesion, morphology and cytoskeleton organisation of human fibroblasts. J. Control. Release 2004, 95, 197–207. [Google Scholar] [CrossRef]
- Madeleine, D.; Leblond, J.; Pierre, P. Gelation of aqueous gelatin solutions. II. Rheology of the sol-gel transition. J. Phys. 1988, 42, 333–334. [Google Scholar] [CrossRef]
- Leuenberger, B.H. Investigation of viscosity and gelation properties of different mammalian and fish gelatins. Top. Catal. 1991, 5, 353–361. [Google Scholar] [CrossRef]
- Van Den Bulcke, A.I.; Bogdanov, B.; De Rooze, N.; Schacht, E.H.; Cornelissen, M.; Berghmans, H. Structural and rheological properties of methacrylamide modified gelatin hydrogels. Biomacromolecules 2000, 1, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ao, Q.; Tian, X.; Fan, J.; Tong, H.; Hou, W.; Bai, S. Gelatin-based hydrogels for organ 3D bioprinting. Polymers (Basel) 2017, 9, 401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alcala-Orozco, C.R.; Mutreja, I.; Cui, X.; Kumar, D.; Hooper, G.J.; Lim, K.S.; Woodfield, T.B.F. Design and characterisation of multi-functional strontium-gelatin nanocomposite bioinks with improved print fidelity and osteogenic capacity. Bioprinting 2020, 18, e00073. [Google Scholar] [CrossRef]
- Chawla, D.; Kaur, T.; Joshi, A.; Singh, N. 3D bioprinted alginate-gelatin based scaffolds for soft tissue engineering. Int. J. Biol. Macromol. 2020, 144, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, I.; De Maria, C.; Lapomarda, A.; Fortunato, G.M.; Montemurro, F.; Di Gesù, R.; Tuan, R.S.; Vozzi, G.; Gottardi, R. Endothelial cells support osteogenesis in an in vitro vascularized bone model developed by 3D bioprinting. Biofabrication 2020, 12, 025013. [Google Scholar] [CrossRef] [PubMed]
- Irvine, S.A.; Agrawal, A.; Lee, B.H.; Chua, H.Y.; Low, K.Y.; Lau, B.C.; Machluf, M.; Venkatraman, S. Printing cell-laden gelatin constructs by free-form fabrication and enzymatic protein crosslinking. Biomed. Microdevices 2015, 17, 1725–1737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nichol, J.W.; Koshy, S.T.; Bae, H.; Hwang, C.M.; Yamanlar, S.; Khademhosseini, A. Cell-laden microengineered gelatin methacrylate hydrogels. Biomaterials 2010, 26, 1211–1218. [Google Scholar] [CrossRef] [Green Version]
- Choi, D.J.; Park, S.J.; Gu, B.K.; Kim, Y.J.; Chung, S.; Kim, C.H. Effect of the pore size in a 3D bioprinted gelatin scaffold on fibroblast proliferation. J. Ind. Eng. Chem. 2018, 67, 388–395. [Google Scholar] [CrossRef]
- Yin, J.; Yan, M.; Wang, Y.; Fu, J.; Suo, H. 3D Bioprinting of Low-Concentration Cell-Laden Gelatin Methacrylate (GelMA) Bioinks with a Two-Step Cross-linking Strategy. ACS Appl. Mater. Interfaces 2018, 10, 6849–6857. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhong, Z.; Hu, N.; Zhou, Y.; Maggio, L.; Miri, A.K.; Fragasso, A.; Jin, X.; Khademhosseini, A.; Zhang, Y.S. Coaxial extrusion bioprinting of 3D microfibrous constructs with cell-favorable gelatin methacryloyl microenvironments. Biofabrication 2018, 10, 024102. [Google Scholar] [CrossRef]
- Munarin, F.; Tanzi, M.C.; Petrini, P. Advances in biomedical applications of pectin gels. Int. J. Biol. Macromol. 2012, 51, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.Y.; Choo, W.S.; Young, D.J.; Loh, X.J. Pectin as a rheology modifier: Origin, structure, commercial production and rheology. Carbohydr. Polym. 2017, 161, 118–139. [Google Scholar] [CrossRef]
- Ma, Y.; Pan, Y.; Xie, Q.; Li, X.; Zhang, B.; Chen, H. Evaluation studies on effects of pectin with different concentrations on the pasting, rheological and digestibility properties of corn starch. Food Chem. 2019, 274, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Kulikouskaya, V.; Kraskouski, A.; Hileuskaya, K.; Zhura, A.; Tratsyak, S.; Agabekov, V. Fabrication and characterization of pectin-based three-dimensional porous scaffolds suitable for treatment of peritoneal adhesions. J. Biomed. Mater. Res. Part A 2019, 107, 1814–1823. [Google Scholar] [CrossRef] [PubMed]
- Alipour, R.; Khorshidi, A.; Shojaei, A.F.; Mashayekhi, F.; Moghaddam, M.J.M. Skin wound healing acceleration by Ag nanoparticles embedded in PVA/PVP/Pectin/Mafenide acetate composite nanofibers. Polym. Test. 2019, 79, 106022. [Google Scholar] [CrossRef]
- Cernencu, A.I.; Lungu, A.; Stancu, I.C.; Serafim, A.; Heggset, E.; Syverud, K.; Iovu, H. Bioinspired 3D printable pectin-nanocellulose ink formulations. Carbohyd. Polym. 2019, 220, 12–21. [Google Scholar] [CrossRef]
- Lapomarda, A.; De Acutis, A.; Chiesa, I.; Fortunato, G.M.; Montemurro, F.; De Maria, C.; Mattioli Belmonte, M.; Gottardi, R.; Vozzi, G. Pectin-GPTMS-Based Biomaterial: Toward a Sustainable Bioprinting of 3D scaffolds for Tissue Engineering Application. Biomacromolecules 2020, 21, 319–327. [Google Scholar] [CrossRef]
- Moreira, H.R.; Munarin, F.; Gentilini, R.; Visai, L.; Granja, P.L.; Tanzi, M.C.; Petrini, P. Injectable pectin hydrogels produced by internal gelation: PH dependence of gelling and rheological properties. Carbohydr. Polym. 2014, 103, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Mezger, T.G. The Rheology Handbook, 4th ed.; Vintcentz Network: Hanover, Germany, 2014. [Google Scholar]
- Warnakulasuriya, S.N.; Nickerson, M.T. Review on plant protein–polysaccharide complex coacervation, and the functionality and applicability of formed complexes. J. Sci. Food Agric. 2018, 98, 5559–5571. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.; Rawat, K.; Bohidar, H.B. pH and ionic strength induced complex coacervation of Pectin and Gelatin A. Food Hydrocoll. 2018, 74, 132–138. [Google Scholar] [CrossRef]
- De Buyl, F.; Kretschmer, A. Understanding hydrolysis and condensation kinetics of γ-glycidoxypropyltrimethoxysilane. J. Adhes. 2008, 84, 125–142. [Google Scholar] [CrossRef]
- Geven, M.A.; Lapomarda, A.; Guillaume, O.; Sprecher, C.M.; Eglin, D.; Vozzi, G.; Grijpma, D.W. Osteogenic differentiation of hBMSCs on porous photo-crosslinked poly (trimethylene carbonate) and nano-hydroxyapatite composites. Eur. Polym. J. 2021, 147, 110335. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- O’Brien, F.J.; Harley, B.A.; Yannas, I.V.; Gibson, L. Influence of freezing rate on pore structure in freeze-dried collagen-GAG scaffolds. Biomaterials 2004, 25, 1077–1086. [Google Scholar] [CrossRef]
- Tonda-Turo, C.; Gentile, P.; Saracino, S.; Chiono, V.; Nandagiri, V.K.; Muzio, G.; Canuto, R.A.; Ciardelli, G. Comparative analysis of gelatin scaffolds crosslinked by genipin and silane coupling agent. Int. J. Biol. Macromol. 2011, 49, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.S.; Liu, C.K.; Fishman, M.L.; Hicks, K.B. Composite films from pectin and fish skin gelatin or soybean flour protein. J. Agric. Food Chem. 2007, 55, 2349–2355. [Google Scholar] [CrossRef]
- Nordby, M.H.; Kjøniksen, A.; Nystrom, B.; Roots, J. Thermoreversible Gelation of Aqueous Mixtures of Pectin and Chitosan. Rheology. Biomacromolecules 2003, 4, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Geven, M.A.; Sprecher, C.; Guillaume, O.; Eglin, D.; Grijpma, D.W. Micro-porous composite scaffolds of photo-crosslinked poly(trimethylene carbonate) and nano-hydroxyapatite prepared by low-temperature extrusion-based additive manufacturing. Polym. Adv. Technol. 2017, 28, 1226–1232. [Google Scholar] [CrossRef]
- Chiesa, I.; Ligorio, C.; Bonatti, A.F.; De Acutis, A.; Smith, A.M.; Saiani, A.; Vozzi, G.; De Maria, C. Modeling the Three-Dimensional Bioprinting Process of β-Sheet Self-Assembling Peptide Hydrogel Scaffolds. Front. Med. Technol. 2020, 2, 571626. [Google Scholar] [CrossRef]



| Gelatin (% w/v) | Pectin (% w/v) | Name without GPTMS/Name with GPTMS |
|---|---|---|
| 5 | 0 | GEL/GELG |
| 5 | 0.5 | GELPect1/GELPect1G |
| 5 | 1 | GELPect2/GELPect2G |
| 5 | 2.5 | GELPect3/GELPect3G |
| Bioprinting Parameters | GELG | GELPect1G | GELPect2G | GELPect3G |
|---|---|---|---|---|
| Time required to induce increase of viscosity before printing (h) | >24 | >10 | 8 | 5 |
| Printing temperature (°C) | 25 | 25 | 25 | 25 |
| Type of tip | Straight | Straight | Straight | Straight |
| Size of the tip (G) | 22 | 22 | 22 | 22 |
| Layer height (µm) | 250 | 250 | 250 | 250 |
| Extrusion multiplier (%) a) | 320 | 380 | 400 | 440 |
| Printing speed (mm/s) | 4 | 4 | 5 | 6 |
| No GPTMS | With GPTMS | |||||||
|---|---|---|---|---|---|---|---|---|
| Parameters | GEL | GELPect1 | GELPect2 | GELPect3 | GELG | GELPect1G | GELPect2G | GELPect3G |
| Yield Stress (Pa) | 3.3 | 3.0 | 11.6 | 41.5 | - | 48.2 | 474.5 | 489.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lapomarda, A.; Pulidori, E.; Cerqueni, G.; Chiesa, I.; De Blasi, M.; Geven, M.A.; Montemurro, F.; Duce, C.; Mattioli-Belmonte, M.; Tiné, M.R.; et al. Pectin as Rheology Modifier of a Gelatin-Based Biomaterial Ink. Materials 2021, 14, 3109. https://doi.org/10.3390/ma14113109
Lapomarda A, Pulidori E, Cerqueni G, Chiesa I, De Blasi M, Geven MA, Montemurro F, Duce C, Mattioli-Belmonte M, Tiné MR, et al. Pectin as Rheology Modifier of a Gelatin-Based Biomaterial Ink. Materials. 2021; 14(11):3109. https://doi.org/10.3390/ma14113109
Chicago/Turabian StyleLapomarda, Anna, Elena Pulidori, Giorgia Cerqueni, Irene Chiesa, Matteo De Blasi, Mike Alexander Geven, Francesca Montemurro, Celia Duce, Monica Mattioli-Belmonte, Maria Rosaria Tiné, and et al. 2021. "Pectin as Rheology Modifier of a Gelatin-Based Biomaterial Ink" Materials 14, no. 11: 3109. https://doi.org/10.3390/ma14113109