Selenium-substituted calcium phosphate powders (CaP_Se) were synthesized by wet precipitation method at mild conditions and subjected to chemical, crystallographic and morphological characterization.
3.1. The Chemical Composition of as-Prepared Powders
The chemical composition of as-prepared powders was determined by ICP-MS analysis. As shown in
Table 2 in addition to Ca, P and Se the presence of strontium (Sr
2+), magnesium (Mg
2+) and sodium (Na
+) ions has been detected. Using a biogenic source (cuttlefish bone) as a precursor of Ca
2+ ions results in a multi-substituted CaP system as already reported in our previous research [
11]. Sr
2+ and Mg
2+ substitution level is ~0.47 and ~0.45 mol%, respectively, while the substitution level of Na
+ ion increase from 1.45 mol% (CaP_Se0) to final 3.49 mol% (CaP_Se10). Increase of Na-substitution along with the increase of SeO
32− ion content could be a result of charge defect compensation, since the substitution of a double-charged selenite ion for a triple-charged phosphate ion creates a negatively charged vacancy. The Sr
2+, Mg
2+ and Na
+ are typical substitutional ions for Ca
2+ in biological apatites. The Sr
2+ ion stimulates the formation of the osteoblast cell line while having an inhibitory effect on osteoclast cells. The Mg
2+ ion is an essential element, highly important in the early stage of bone formation due to grow factor effect. Prolonged Sr
2+ and Mg
2+ deficiency directly result in a decrease in bone density and can cause osteoporosis [
17].
As shown in
Table 2, the Se/(P + Se) molar ratios in as-prepared powders increase along with the selenium content added to the reaction mixture. However, the Se/(P + Se) molar ratios are lower than the nominal ratios indicating that part of SeO
32− ions remained in the mother liquor solution after precipitation. The data from
Table 2 show that 63.6% of nominally added SeO
32− ions were incorporated in CaP_Se1, 59.2% in CaP_Se5, and 60.4% in CaP_Se10. Incomplete incorporation of SeO
32− ions into precipitated calcium phosphates are in accordance with results obtained by Uskoković et al. [
9], Wei et al. [
10], and Liu et al. [
18]. Due to minor substituents, the Ca/(P + Se) molar ratio in starting reaction mixture is lower (~1.61) than the 1.67 required for stoichiometric HAp. In spite of this, for as-prepared powders the values of Ca/(P + Se) molar ratios are between 1.67 and 1.80. It can be hypothesized that the incomplete incorporation of selenite ions is compensated by the substitution of PO
43− with CO
32−, i.e., the formation of type-B carbonated apatites can be expected. As will be shown later, a significant amount of an amorphous phase (ACP) was observed in all as-prepared powders. The ACP can have a Ca/P molar ratio in the range 1.2–2.2, depending on the synthesis conditions and used precursors [
4]. As shown in
Table 3 the powder CaP_Se1 has the highest content of ACP phase. The higher Ca/P ratio of CaP_Se1 sample, compared to other powders, might be a result of the higher ACP content.
3.2. XRD Patterns of as-Prepared Powders and Rietveld Refinements
The XRD patterns of as-prepared non-substituted (CaP_Se0) and Se-substituted (CaP_Se1, CaP_Se5 and CaP_Se10) CaP powders are shown in
Figure 1A,B. Samples CaP_Se0 and CaP_Se1 were identified as biphasic mixtures, with a good match to the line patterns for crystalline HAp (JCPDS No.09-0432) and OCP (JCPDS No. 27-1402), while CaP_Se5 and CaP_Se10 show characteristic diffraction peaks for HAp. No additional peaks characteristic for selenium compounds were observed. With the increase of the Se/P molar ratio, the characteristic diffraction peak intensity and the crystallinity decrease (
Figure 1B), which indicates that incorporation of SeO
32− leads to lattice distortion of obtained CaPs. Compared to the CaP_Se0, the diffraction peaks of the (002) crystal plane of the Se-substituted samples shifted to the left (
Figure 1B). The peak shift of the (002) characteristic peak is related to the change of the HAp and/or OCP lattice constant
c, which means that the incorporation of the SeO
32− ions has a marked impact on the growth of the HAp crystal, particularly in the
c-axis direction as previously described by Wei et al. [
7].
Rietveld refinement analysis (
Figure 1A) was performed on XRD patterns of all prepared samples to determine unit-cell parameters, crystallite size and weight percent proportions of detected phases. Results of quantitative analysis of samples are given in
Table 3.
The Rietveld refinements indicate a significant amount of an ACP phase in all as-prepared powders (
Table 3). The CaP_Se0 and CaP_Se1 powders in addition to HAp and ACP phase contain OCP phase as well. The ACP phase is a hydrated, thermodynamically unstable, transient phase that commonly precipitates during the formation of more stable CaPs in aqueous media. It is considered to be a precursor phase in OCP formation. The OCP is often found as an intermediate phase in the formation of thermodynamically more stable HAp. The final pH values (
Table 3.) of all obtained suspensions were higher than 7. According to the literature [
19], the calcium deficient hydroxyapatite (Ca/P = 1.5–1.67) can be obtained in the pH range 6.5 to 9.5, OCP (Ca/P = 1.33) in the pH range 5.5 to 7.0 and ACP (Ca/P = 1.0–2.2) in the pH range 5.0 to 12.0. The presence of OCP phase within CaP_Se0 and CaP_Se1 systems, even the pH value was higher than 7.0, suggest that foreign ions influence the phase transformation of calcium phosphates from amorphous to crystalline phases.
The lattice parameters, density, and average crystallite size of the HAp and OCP phase in as-prepared powders, determined by Rietveld refinement of the XRD data, are shown in
Table 4.
The variation of unit cell parameters of HAp phase as a function of Se/(P + Se) molar ratio in as prepared powders is shown in
Figure 2.
Lattice parameter
a (=
b) increases with increasing Se-substitution, which can be explained by the larger bond length of Se‒O (0.164 nm) compared to P‒O bond length (0.155 nm). The unit cell parameter
c of substituted HAp phase in all prepared powders is lower than
c of non-substituted HAp but exhibited an increase with the Se substitution level. The lower value of the lattice parameter
c in substituted HAp, compared to non-substituted HAps, may indicate the release of hydroxyl groups and formation of vacancies along the
c axis due to charge compensation since SeO
32− (double charged) substitute PO
43− ion (triple charged), that consequently leads to Ca
2+ and OH
− ion release [
21,
22]:
The difference between the
c parameters of non-substituted and Se-substituted HAps is smaller for higher Se-substitution levels, where the higher content of Na
+ ions was detected, indicating additional charge defect compensation by Na-substitution for Ca according to the formula Ca
10-xNa
x(PO
4)
6-x(SeO
3)
x(OH)
2. The lattice parameters of substituted samples compare fairly well with the literature data reported by Barbanente et al. [
20], showing an almost linear rise of cell parameters with Se substitution up to a Se/(P + Se) molar ratio of 0.1 (
Figure 2). Wei et al. [
10] reported that
a and
b lattice parameters increase along with the Se-substitution level, while lattice parameter
c decreases, while Sun et al. [
21] reported a non-continuous change of
a and
b and decrease of
c lattice parameters. Kolmas et al. [
22] observed an increase of
a and
b and non-continuous change of
c lattice parameters along with the Se-substitution level. The difference in the unit cell paramter
c between the non-substituted (CaP_Se0) powder prepared in this work and those reported in the literature can be attributed to the biogenic precursor (cuttlefish bone) used in this work. The cuttlefish bone in its aragonite structure contains a significant amount of trace elements (e.g., Sr
2+, Mg
2+, Na
+) that can influence the unit cell parameters of prepared CaPs.
As seen from
Table 4, in spite of lower values of parameter
c in substituted HAp, the unit cell volume, V, of the HAp phase in the prepared samples increases and the density consequently decreases with the Se-substitution level. Selenite ion (trigonal pyramidal) has greater volume compared to phosphate ion (tetrahedral) and consequently, the substitution may result in the dilatant unit cell and oxygen voids may be generated [
10]. Very small difference in the average crystallite size L (ranging between 5 and 7 nm) was obtained for the HAp phase in the prepared non-substituted and Se-substituted samples.
The unit cell parameters, unit cell volume, and the average crystallite size of OCP phase in the powder CaP_Se1 differ from the values obtained for non-substituted CaP_Se0 powder, indicating the incorporation of selenite ions in the OCP phase, too. Since the powders CaP_Se5 and CaP_Se10 do not contain an OCP phase, the proper correlation between the mentioned parameters and selenite substitution level cannot be established. It should be noted that the Rietveld refinement is not sensitive enough to determine selenite concentration in each (HAp and OCP) phase.
3.3. XRD Patterns of Heat Treated Powders and Rietveld Refinements
XRD patterns of heat treated powders at 1200 °C are presented in
Figure 3.
Compared to the XRD patterns of as-prepared samples, XRD patterns sharpen after heat treatment, indicating an increase of crystallinity. The XRD data of the sample CaP_Se0 gave a good match to the line pattern of
β-TCP (JCPDS No. 09-0169). Samples CaP_Se1, CaP_Se5, and CaP_Se10 were identified as triphasic mixtures with a good match to the patterns of HAp (JCPDS No.09-0432),
β-TCP, and
α-TCP (JCPDS No. 09-0348). Results of the quantitative phase analysis of the heat-treated samples determined by Rietveld refinement of the XRD data are given in
Table 5.
With the increase of selenite ion content, an increase in the HAp content and a decreasing trend in
β-TCP and
α-TCP content have been observed. Similar findings that the incorporation of selenite ions restricts the phase transformation from HAp to
β-TCP are reported in the literature [
7,
10,
23]. As observed by Wei et al. [
10] the samples with Se/P ≤ 0.05 did not transform to the TCP phase after heat treatment at 1100 °C.
As seen from
Table 5 in the heat-treated sample CaP_Se10 CaO phase was detected, as well. Wei et al. [
10] observed CaO as secondary phase after high-temperature sintering of the sample prepared with a Se/P molar ratio of 0.08. They speculate that the CaO is formed by transformation of CaSeO
3 that eventually precipitates in a very small amount and cannot be detected by XRD.
3.4. FTIR Analysis
The FTIR spectra of as-prepared and heat treated CaP powders are shown in
Figure 4A,B, respectively.
FTIR spectra of as-prepared samples show broad bands in the range 3000‒3700 cm
−1, attributed to adsorbed water. The strong bands in the range 900‒1200 cm
−1 correspond to characteristic stretching vibration modes of the phosphate group. The bands at 1022 and 1074 cm
−1 (ν
3) are attributed to the asymmetric stretching vibration of P–O, at 964 cm
−1 (ν
1) is associated with the symmetric stretching vibration of P–O, and at 565 and 599 (ν
4) cm
−1 are attributed to asymmetric bending vibrations of O–P–O, all of which can be assigned to the HAp phase [
24]. Absorption bands at 525, 912, 1112, and 1193 cm
−1, which are attributed to the characteristic absorption bands of a HPO
42− group, can be assigned to the OCP and ACP phases [
25,
26,
27,
28,
29]. The intensities of HPO
42− bands decrease along with the increase of Se-substitution level, since the content of OCP and ACP phase decrease, as well. The bands at 875, 1419, and 1463 cm
−1 are characteristic vibration modes for carbonate groups substituted for phosphate group (B-type substitution) [
30,
31]. CO
32− substitution was expected since CaCO
3 was used as the source of Ca-ions. Additionally, it could be the result of CO
2 dissolution in the reaction medium during the open-air synthesis. The band at 765 cm
−1 is attributed to O‒Se‒O bending vibration (ν
3) and it becomes stronger as the Se-substitution level increases [
22]. The characteristic vibration modes for Se‒O bands are in the range of 900‒800 cm
−1 [
23]. However, they overlap with vibration modes of carbonate groups. Obtained results indicate that prepared samples are co-substituted with SeO
32− and CO
32− in the position of PO
43− ions. CO
32− ions are considered to be more easily incorporated into the HAp lattice due to having a smaller volume than SeO
32− ions [
10,
32].
In the FTIR spectra of the heat-treated samples (
Figure 4B) the band at 631 cm
−1 is assigned to the hydroxyl group of HAp. Its intensity increases as the Se/P ratio increases, in agreement with the quantitative analysis of the heat-treated CaP powders performed by Rietveld refinement of the XRD data that shows stabilization of the HAp phase during the heat treatment process. Characteristic P‒O bands of HAp appear at 1090, 1023, 965, 600, and 568 cm
−1. A broad band from 950 to 1200 cm
−1 corresponds to the TCP phases. The bands at 1120 and 1004 cm
−1, attributed to stretching vibration of P‒O, at 940 cm
−1, associated with banding modes of P‒O, at 1040, 1062, and 1082 cm
−1 (
ν3), attributed to anti-symmetric P‒O stretching, and at 542, 588, and 600 cm
−1 (
ν3), attributable to anti-symmetric P‒O bending can be assigned to
β-TCP phase. The bands at 940 and 965 cm
−1 arise due to the factor group splitting of the ν
1 fundamental vibration mode corresponding to the symmetric P‒O stretching vibration of the phosphate ion (958 cm
−1). The characteristic bands for
β-TCP are similar to bands of
α-TCP, however, the latter are broader [
33]. This effect can be seen in the FTIR spectrum for CaP_Se1 compared to CaP_Se0 powder. Characteristic band at 765 cm
−1, attributed to O‒Se‒O bending vibration, disappears as a result of decomposition of selenite with the release of SeO
2 in gas form as previously reported by Wei et al. [
10].
3.6. Ion Release and In Vitro Bioactivity
The dissolution of the as-prepared powders and their ability to form apatite like phases in simulated body conditions were followed by ICP-MS and XRD analysis. Release curve of selenite ions for CaP_Se5 powder shown in
Figure 6 indicate an initial burst release (day 1) followed by a stage of slow and mostly steady release. The pH value of the SBF solution varied slightly during incubation time from an initial 7.40 to a maximum of 7.66.
The concentration of released Se ion is the same for CaP and CaP_Se0 powder (selenite-free samples, 0 ppm, green line in
Figure 6).
Results of quantitative XRD analysis of the powders CaP_Se0 and CaP_Se5 after 7 days of immersion in SBF, performed by Rietveld refinement, are given in
Table 6.
As seen, during soaking the partial transformation of OCP and ACP phases into the HAp phase occurred. When apatite samples are immersed in SBF, both dissolution and the precipitation of apatite occurs simultaneously. SBF solution contains calcium, phosphate and carbonate ions, the concentrations of which are close to the solubility limit of carbonated HAp. The dissolution and liberation of ions can result in localized supersaturation that favors precipitation of carbonated HAp. From XRD patterns it was not possible to distinguish between the original powder phases and the newly formed apatite phase.
3.7. Selective Anticancer Activity of Selenium Substituted Calcium Phosphates
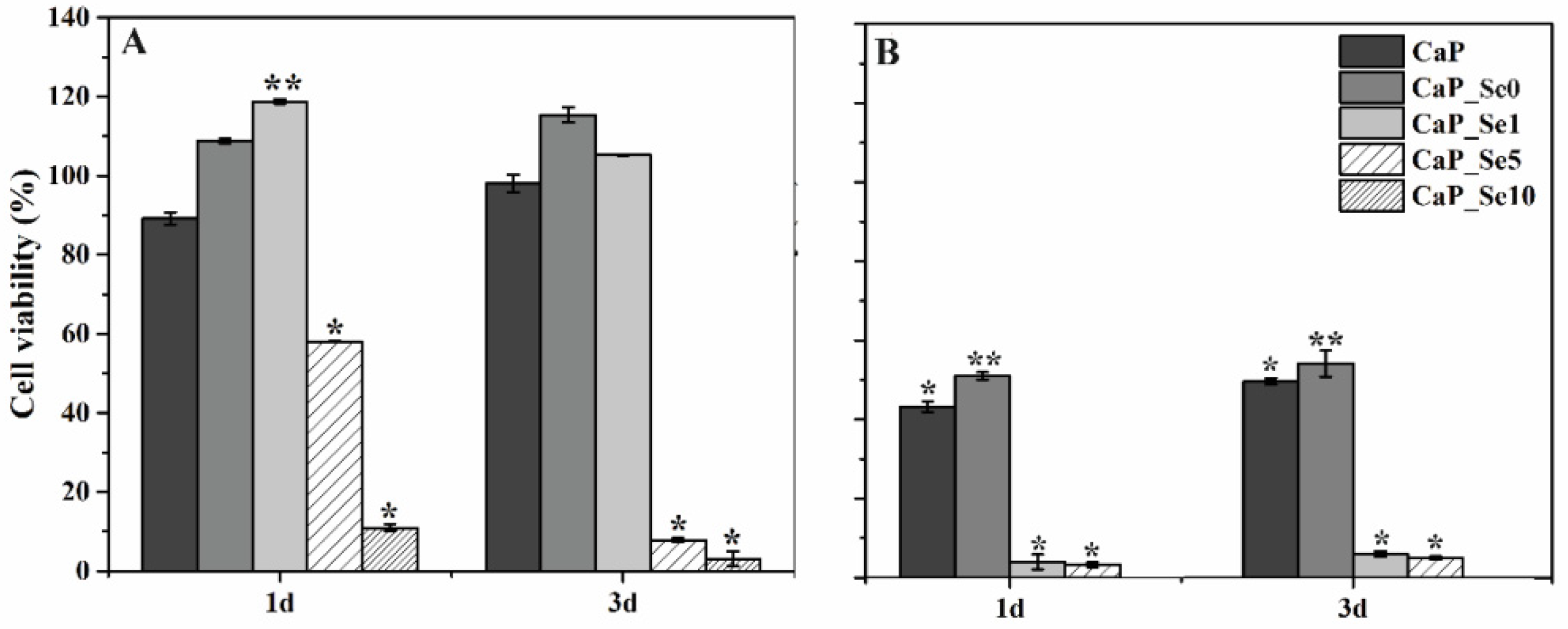
The main goal of preparing selenium substituted CaPs is obtaining a biomaterial that will effectively inhibit the development of bone cancers while showing no cytotoxic activity on healthy cells. The cell viability results are given in
Figure 7.
Figure 7A shows that in comparison with the control group (untreated cells), in which cell viability was estimated to be 100%, extracts of CaP_Se0 and CaP_Se1 powders exert a proliferative effect on the HEK 293 cells. The higher cell viability for CaP_Se0 compared to CaP (non-substituted CaP powder obtained from synthetic CaCO
3) indicates a beneficial effect of foreign ions present in the biogenic Ca-ion precursor on cell proliferation. The treatment with extracts of CaP_Se5 for 1 and 3 days reduced HEK 293 cell viability by ~40 and ~90%, respectively, while the CaP_Se10 powder reduced HEK 293 cell viability by ~90% or more, at both time points. The anticancer activity was examined using U2OS cell line. As
Figure 7B shows, the cell proliferation was significantly suppressed in the presence of the extracts of all investigated powders. The extracts of CaP and CaP_Se0 powders caused a significant reduction in the U2OS cell viability (50% and 54%, respectively) while the extracts of the CaP_Se1 and CaP_Se5 powders reduced U2OS cell viability by more than 95% compared to untreated cells.
Uskoković et al. [
9] also reported that the viability of K7M2 osteosarcoma cells decreased in direct proportion to the amount of selenite in HAp but no reduction was observed in the viability of primary fibroblasts treated with HAp incorporating different amounts of selenite ions, suggesting their potentially selective anticancer activity. Our results suggest that only the CaP_Se1 powder, with low concentration of Se, show selective toxicity to cancer cells (U2OS), without harming non-tumorigenic cells (HEK 293) cells. Results are consistent with the study of Barbanente et al. [
20], who evaluated the cytotoxic activity of Se-doped HAp nano particles using prostate and breast cancer cells as well as healthy human bone marrow stem cells. They found that nanoparticles with a high concentration of Se showed a strong anticancer effect but also caused a significant increase in toxicity towards normal cells.
Additional in vitro osteogenic cell culture experiments and in vivo studies are needed to confirm the suitability of investigated materials for potential application in bone tumor therapy. Furthermore, in our future research, highly porous scaffolds based on a biopolymer and selenite substituted CaPs will be prepared and characterized.