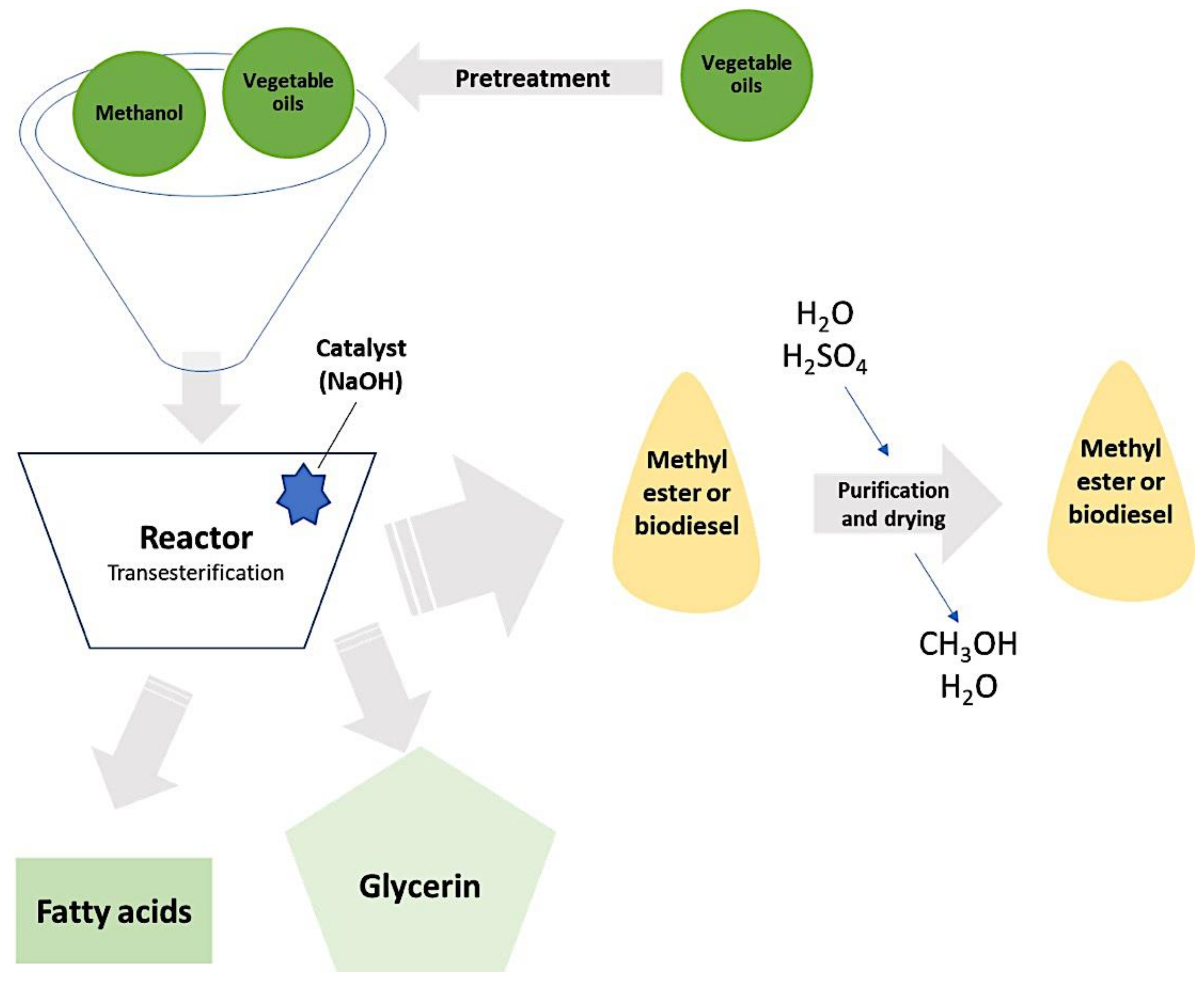
4.2.4. Hydrothermal Liquefaction
The thermochemical liquefaction of biomass is the process that has generated the most interest in recent years since it generates higher energy density, requires a shorter reaction period, and can be applied to a wider range of materials [
55,
71]. In addition, the liquid product resulting from techniques such as pyrolysis generally has a high oxygen content, which increases instability and makes it difficult to use as a fuel [
72]. Instead, liquefaction is potentially capable of producing a liquid with considerably lower oxygen content [
55,
73].
One of the most promising thermochemical liquefaction processes is hydrothermal liquefaction (HTL). HTL is a technology that is capable of efficiently processing wet and dry biomass without restriction of lipid content, from lignocellulosics to organic waste. The product generated in this process is called bio-crude, which is the renewable equivalent to oil, since it is an energy-dense intermediate that can be upgraded to a variety of liquid fuels [
7,
74]. HTL generates bio-crude from organic matter thanks to specific characteristics, such as the presence of water in hydrothermal conditions, with temperatures ranging from 250 to 450 °C and pressures between 100 and 300 bar. Under these conditions, the water remains in a liquid state or in a very dense supercritical state [
12,
55,
71,
75,
76,
77,
78,
79]. This technique uses the specific characteristics of compressed hot water [
76]. Throughout the liquefaction process, the biomass undergoes a series of depolymerization reactions such as hydrolysis, dehydration or decarboxylation, giving rise to insoluble products such as bio-crude oil or bio-carbon. In addition, other products are also generated, such as gases (CO
2, CO, H
2 or CH
4) or soluble organic substances (mainly acids or phenols) [
12,
71].
Most of the studies that have been carried out to date focus on the reaction mechanism [
80,
81], including the effect of biomass types, reaction temperature and heating rate [
82], the pressure, type and nature of the catalysts or pH modifiers [
83], biofuel performance and final product characteristics [
73]. In both the reviews carried out by Gollakota et al. [
55] and Castello et al. [
12], it is possible to find information about the operation of hydrothermal liquefaction based on the conditions mentioned above, as well as the historical evolution of the technology. According to these studies, different types of biomass give rise to different reaction patterns and respond differently to each of the reaction conditions.
Water is the most widely used solvent to carry out organic reactions, since it is considered a safe, economical and environmentally friendly medium. The vapor pressure curve that separates the liquid and gas phases ends at the so-called critical point (374 °C, 221 bar) [
56]. From this point on, the properties of the water can be modified without any phase changes. This results in the supercritical state, which refers to the temperature and pressure zone at the critical point at which water acts as a reactant and a catalyst. In this state, properties of water, such as ionic product, density, viscosity, and dielectric constant, vary rapidly, becoming a liquid without phase limits of high miscibility. Thanks to these properties, supercritical water becomes an apolar solvent for most organic reactions [
55,
84]. Despite this, in most studies in which hydrothermal liquefaction is analyzed, subcritical conditions (350 °C and >200 bar) have been preferred, i.e., conditions that limit the supercritical state in which the properties of water have changed enough to facilitate liquefaction: the dielectric constant has been reduced by approximately 80% allowing better solubility of apolar compounds.
The elemental composition of the biofuel generated by hydrothermal liquefaction varies as a function of the reaction temperature. The hydrogen content remains constant, but the percentage of oxygen is lower as the process temperature increases, since the increase in temperature favors deoxygenation. The percentage of carbon is higher at higher temperatures. Therefore, the H/C and O/C ratios of the biocrude decrease with the increase in temperature, which is consistent with the increase in the calorific value of the fuel observed (see
Table 4). However, production decreases at high temperatures, resulting in less biofuel. Therefore, depending on the needs (quantity or quality), one temperature or another may be of interest, since the ideal temperature to obtain maximum quality fuel is not the ideal temperature to obtain maximum production. It has also been suggested that the heating rate could be an important parameter in hydrothermal liquefaction. In addition, evidence has been found that indicates that the shorter the time in which the reaction is at maximum temperature, the greater the bio-crude production [
71,
76,
77,
83].
In subcritical conditions, temperature is the dominant parameter, it becomes less decisive in conditions close to or above the supercritical point [
12]. One of the main advances that have been made in the field of hydrothermal liquefaction in recent years is to characterize the effect of pressure in these types of conditions [
87,
88]. High pressures modify the properties of water, favoring liquefaction over gasification. For example, at a temperature of 400 °C and a pressure of 350 bar, the ionic product is practically the same as at 350 °C and 250 bar. This is interesting, since the high temperatures favor the reactions and their kinetics, achieving greater deoxygenation [
12].
The type of biomass used is an important parameter to consider when carrying out hydrothermal liquefaction, since biomass can be made up of different components, which react differently to hydrotreatment. Among the types of biomass that are most commonly used in hydrothermal liquefaction, dry lignocellulosic biomass (composed of cellulose, hemicellulose and lignin) and wet biomass from algae (proteins) and oils (lipids) can be highlighted [
55]. According to literature, the components that can most efficiently be converted into biofuel by hydrothermal liquefaction are lipids, proteins and carbohydrates. Among these three components, lipids are the ones that give the best results and carbohydrates the ones that give the poorest results [
71].
OFUSW, WWTP, and biomethanization digestate have been considered as low-value materials in the past. Recent studies consider that they have been wasted since their value as an energy source has been increasing, and today, they are considered very valuable resources for the generation of biofuels [
41,
89,
90]. In terms of chemical composition, biomass from bio-waste is highly varied [
41]. The chemical compositions of the most commonly used bio-wastes are summarized in
Table 5. The waste from WWTP contains a great variety of organic waste, such as oils and fats, that can be used to generate biofuels [
49]. In addition, microorganisms, such as algae or bacteria, that grow in this type of wastewater can also be used [
89]. Waste from WWTP has also been shown to be a good candidate for bio-crude production due to their cheap price, availability and reasonable calorific value [
91]. OFUSW is a biomass with a high lignocellulosic content that is also present in other sources of energy such as food waste [
44].
A descriptive scheme of the experimental methodology for the treatment of urban solid waste by hydrothermal liquefaction is included in
Figure 2. The initial fraction of urban solid waste comes from three main sources of waste, namely food waste, agroforestry waste, and compound waste. It is a wet raw material for which a drying pre-treatment is carried out first and the product obtained constitutes the hydrothermal liquefaction feed. Specifically [
92], HTL experiments were carried out near the subcritical regions of water (200 °C and 100 bar) in order to obtain a reaction time of 60 min from hydrogen-induced reducing conditions. Several variants of the process were studied and basically four main products were obtained: HTL aqueous fraction (45–50%, HTLAF), organic fraction (45–50%, HTLOF), gas fraction (15–20%, HTLG), and solid fraction of biochar (30–35%, HTLBC) [
92,
93].
Another parameter that influences the liquefaction process is the catalyst. The use of catalysts in hydrothermal liquefaction can lower the pressure and temperature of the reaction while improving the yield and reducing the solid waste produced. Homogeneous acid or base catalysis is widely used in HTL. Regarding acid catalysis, the most used catalysts for biomass HTL are formic acid, acetic and other weak acids as catalyst solvents, which leads to a bio-oil with a high oxygen content [
93]. Although strong acids appear to be effective catalysts, their industrial application is hampered by their strong corrosivity. The basic catalysts are considered better catalysts, specifically, those that are in the form of alkaline salts, such as Na
2CO
3, K
2CO
3, NaOH, or KOH. A disadvantage of alkaline catalysts is that they can inhibit the dehydration of biological molecules and promote the decarboxylation process due to an increase in the pH of the liquid phase [
71]. Many heterogeneous catalysts, mainly in the form of various metals, such as Pd, Ru, Pt, Mo, Ni, Co, and Ni, on SiO
2, Al
2O
3, and zeolite, have been investigated. However, some drawbacks have been found that limit their application, such as poisoning, sintering, or intraparticular diffusion [
71]. Biller et al. [
94] carried out an investigation of the HTL of humid biomass with heterogeneous catalysts Co/Mo, Ni/Al and Pt/Al. In their conclusions, it appears that these catalysts promote the calorific value, the oxygen content, and the performance of the bio-oil. In addition, it has been reported that they also play a very important role in reducing the nitrogen content of the bio-oil. Wang et al. [
95] studied the effect of HTL of microalgae
Nannochloropsis (NAS) over various transition metal/TiO
2 catalysts. As can be seen in
Table 6, with the Ni/TiO
2 catalyst, a liquefaction conversion of 85.19% and a bio-crude yield of 42.40% were obtained, which was 10% higher than the blank experiment.
Operation Mode: Batch Reactors and Continuous Hydrothermal Liquefaction
Hydrothermal liquefaction has been widely studied in batch type discontinuous reactors [
73]. By means of this procedure, any type of material can be screened, and a wide range of process conditions can be evaluated [
83,
96]. Another great advantage of the process is that relatively high concentrations of dry matter can be obtained. In addition, there are no complications, e.g., plugging in the pipes or difficulties in pressurizing and pumping the raw material. However, a number of important drawbacks have been described concerning continuous hydrothermal liquefaction [
12]:
Thermal fugacity: In batch type reactors, the process conditions are not constant, because the system has to go from ambient conditions to the desired temperature. This transience makes it difficult to separate the effects of temperature and time. The faster the heating, the less this problem is present.
Difficulty decoupling temperature and pressure: In most batch experiments, pressure is obtained by heating the reagents. Pre-pressurizing the system with an inert gas can partially solve this problem. However, the elevated pressures increase the solubility of the inert gas, reducing the usefulness of the pre-pressurization. In a continuous system, pressure and temperature can be controlled completely independently.
Initial contact pattern: In a batch reactor, the reactants are usually mixed by stirring the reactor itself or thanks to an impeller. This contact pattern is different from that observed in continuous reactors. That is, in continuous flow reactors, new reagents are continuously supplied while products are removed. In a continuous tubular reactor, the flow pattern (laminar or turbulent) can significantly change the results of the process.
Difficulty for its implementation at the industrial level: The industrial use of batch reactors is normally justified only for the production of high added value products, often produced in limited quantities. This is definitely not the case for biofuel production, as the goal is to produce large quantities. Furthermore, hydrothermal liquefaction requires extensive optimization to reduce the energy consumption of the process, which can only be carried out effectively in a continuous configuration.
Biofuel Optimization
In general, it can be said that the most severe conditions cause a high degree of deoxygenation and denitrogenation. At the same time, a higher quality of the bio-oil is associated with a significant decrease in its yields. The best results, considering both quantity and quality, are obtained when using a system as a catalyst based on Co/Mo [
97].
In a study to obtain bio-crude from
Nannochloropsis algae in a continuous process, López Barreiro et al. [
7] report different yields depending on the catalyst used. The best results in terms of deoxygenation were obtained with a Pt/Al
2O
3 catalyst reaching 1.6% oxygen and 63.5% yield. The highest yield values (96.6%) were obtained with a NiMo/Al
2O
3 catalyst. There are some less important parameters that have a direct relationship with the yield and quality of the bio-crude. They are as follows:
Type of biomass: Biomass with a higher percentage of cellulose produces a higher conversion than that with a higher proportion of lignin (see
Figure 3a) [
98].
Effect of temperature: Temperature is one of the decisive parameters of the HTL process. When working at subcritical temperatures, the break of chemical bonds and the depolymerization of the biomass are increased. As a consequence, the concentration of free radicals and the probability of repolymerization of molecular fragments are also increased to increase the total yield of the bio-crude and the HHV (see
Figure 3b). When the temperature approaches or exceeds the critical point of water, the repolymerization of intermediate products increases, therefore the performance decreases [
71].
Heating rate: Slow heating rates generally lead to the formation of carbon residues due to the repolymerization of intermediates, while fast heating rates lead to the formation of gaseous products due to redecomposition reactions. Therefore, proper heating rates are an important parameter in the HTL process. More research on heating rates is necessary to fully understand the mechanism of this conversion process [
71].
Time of retention/residence: A large number of investigations have been carried out on the effect of residence times on the yield of bio-crudes. It is expected that the short retention times produce a large amount of bio-oil, and conversely, a long retention time causes the repolymerization of the intermediate products, which therefore reduces the yield of the biocrude (see
Figure 3c) [
71].
Particle size: A reduction in the size of the particles seems to indicate an improvement in the performance of the bio-oil, as well as a decrease in the amount of solid waste. An optimal particle size for the HTL process is considered to be between 4 and 10 mm [
71].
Although hydrothermal liquefaction is a hopeful process with great potential, the bio-crude generated usually has a high content of heteroatoms, mainly oxygen, nitrogen and sulfur. Therefore, in its original form, bio-crude is not compatible with most of the uses for which it is intended in the transport sector and requires improvement or optimization [
102]. Oxygen reduces the calorific value and energy density of bio-crude, while nitrogen and sulfur corrode equipment and pollute the atmosphere [
103].
One of the most promising techniques for improving the quality of biocrude and its conversion to hydrocarbons is hydrotreating. This process, which represents a serious technological challenge, removes heteroatoms from the bio-crude in the presence of heterogeneous catalysts like CoMo/γ-Al
2O
3 or NiMo/γ-Al
2O
3 at high temperatures and pressures (300–450 °C and 30–170 bar) [
74]. The presence of considerable amounts of oxygenated compounds makes the bio-crude obtained by liquefaction unstable under severe conditions, which means that it could be susceptible to polymerization or condensation reactions, as observed in bio-oils obtained by pyrolysis [
104]. Hydrotreating removes oxygen through dehydration, decarboxylation, and decarbonylation reactions and removes nitrogen mainly in the form of ammonia. Nitrogen-containing compounds found in biocrude increase chemical complexity and lead to higher H
2 consumption, as they are recalcitrant to denitrogenation. Nitrogen compounds can be difficult to remove, so more severe conditions are required in terms of temperature and partial pressure of H
2 to be able to remove them [
105,
106,
107].
As bio-crudes obtained by liquefaction are generally more stable than bio-oils obtained by pyrolysis due to their lower oxygen content, less attention has been paid to the problem of the formation of coke at such high temperatures (400 °C) [
74]. That is to say, severe thermal conditions are associated with effective hydrodenitrogenation. However, these conditions promote unwanted polymerization or coking reactions, due to the presence of a considerable amount of reactive oxygen-containing functional groups. Haider et al. [
74] have proposed the use of a multi-stage hydrotreatment to overcome this problem. According to their study, by performing a first hydrotreating stage at 350 °C, practically complete deoxygenation is achieved, resulting in a much lower amount of coke (see
Figure 4). As a next step, they carried out a second stage at a higher temperature at which a denitrogenation of 92% was obtained.