3.1. Physiochemical Properties
According to the objective of the present work, the materials based on amidoxime functional groups, in particular, the Se-derivative of N’-hydroxy-1,2,5-oxadiazole-3-carboximidamide (Se-init), were studied as highly effective sorbents for uranium recovery. The choice of Se was determined by the mild selective oxidizing properties of SeO
2 towards N’-hydroxy-1,2,5-oxadiazole-3-carboximidamide. The method of preparation is based on the reaction of oxidative polycondensation of N’-hydroxy-1,2,5-oxadiazole-3-carboximidamide with SeO
2 in the presence of polar organic solvents followed by the formation of a polymer structure, whose elementary units are linked to each other due to the formation of strong diselenide bridges. This original method offers a very simple implementation by a single-stage reaction, as well as the use of an organic radical with a large number of reactive groups (hydroxyl and oxime) that remain in the final product. A number of physical–chemical methods were used to determine the molecular structure of the resulting compound. In particular,
Figure 1 shows the IR spectrum of Se-init.
The bands observed in the spectrum indicate that the initial compounds are built into the general structure and preserve functional radicals. An intense absorption band is observed at 3454 cm
−1, which corresponds to the O–H vibrations at the oxime group. The absorption band at 3311 cm
−1 corresponds to N–H vibrations. The absorption band at 1623 cm
−1 corresponds to C=N vibrations and the one at 1357 cm
−1 to vibrations of C–N bonds. In the Se-init spectrum, one observes narrowing of absorption bands at 3311 and 3454 cm
−1, which indicates the absence of expressed hydrogen bonds. The specific feature of the Se-init spectrum (
Figure 1b) consists in the presence of peaks at 412 and 489 cm
−1 corresponding to vibrations of Se–Se and N–Se bonds, respectively. In addition, an important feature consists in the presence of the absorption bands at 1548 cm
−1, corresponding to the Se=C-N bond vibrations, and 1105 cm
−1, corresponding to the C–NO bond vibrations.
The X-ray diffraction analysis showed the presence of peaks corresponding to the initial N’-hydroxy-1,2,5-oxadiazole-3-carboximidamide, which indicated the complete binding of the functional organic part with partial preservation of the original structure (27 Å—
Figure 2, X-ray Pattern b). The syngony and the spatial group P 3(1)21 were established—a structure similar to gray selenium (β-Se) but different from it (
Figure 2, X-ray pattern b). In addition, the Se-init X-ray pattern shows signals corresponding to the Se phases that were not earlier found in the published data.
The presence of the crystal structure can be indirectly corroborated by the images of scanning electron microscopy (
Figure 3). One can see that the morphology of the Se-init surface is represented by clearly expressed crystallites of irregular structures and sizes.
The material was analyzed by the method of solid-state NMR on
13C and
1H nuclei. In the
13C spectrum (
Figure 4a), there is a clearly expressed signal at 138 ppm corresponding to carbon at the oxime group—C=NOH, as well as signals of 146 ppm corresponding to the -HN–C= and 154 ppm typical for =C–C= (
Table 1). In the PMR spectrum (
Figure 4b), there are signals of 1.5 ppm corresponding to the proton at aliphatic C–NH, and 7.1 ppm corresponding to H–N(C)–Se.
The additional PMR analysis in the liquid phase was performed: it showed the presence of an additional signal with an intensity of 3 at 2.73 ppm, which probably corresponded to three acetylene protons.
Figure 5 shows the XPS spectra of the prepared material. In order to obtain a more accurate understanding of the structure of the compound, the material was pre-washed with distilled water and ethanol alcohol to remove the synthesis by-products.
The XPS spectrum shows the presence of a C 1s peak (
Figure 5b) around 285.0 eV, which corresponds to the aliphatically bound carbon at the position CH
2–CH-; the peak of 286.9 eV corresponds to the tri-substituted carbon NH
2–CH=N–OH. A signal of energy of 288.5 eV corresponds to the groups C–N/C=N. In the region corresponding to N 1s (
Figure 5c), there are peaks with binding energies of 399.2 eV typical for triazine-type nitrogen C–N–OH and of 400.4 eV corresponding to thiadiazole-type nitrogen, probably bound to the selenium atom (Se–NH–C). The signals of the O 1s peak (
Figure 5d) with binding energies of 533.6 eV and 531.2 eV correspond to an oxygen atom with surroundings of NH
2–C=N–O–N and N–O–H, respectively. The position of the Se 3p peak (
Figure 5e) of the binding energy of 164.0 eV is typical for tetravalent selenium, probably bound to the second selenium atom by a double bond (>Se=Se<). The spectrum also contains a signal at 169.3 eV corresponding to a selenium atom with the C–NH–Se surrounding.
Based on the performed physical and chemical analysis, we suggested the mechanism of binding of compound
1 (
Figure 6) to selenium dioxide and suggested the molecular structure of the resulting material (
Figure 6).
It was found that, as a result of the polycondensation reaction, the degree of oxidation of Se (IV) is preserved with the formation of a strong double bond >Se=Se< bound by functional amidoxime radicals.
During the synthesis, compounds 2 and 3 remained in the dissolved phase and were then completely removed by repeated washing with distilled water and organic solvents, as evidenced by the results of the analysis of the material (
Table 1).
Table 1 shows the data calculated for an elementary unit of Se-init (
Figure 6). The experimental percent values of the elements match the theoretically calculated ones with insignificant deviations, which is probably related to the presence of non-removable monomeric or oligomeric structures.
3.2. Sorption Selective Properties
Figure 7 shows the experimental results of the determination of the zero charge point (pH
pzc) using a NaNO
3 solution of a concentration of 0.01 mol L
−1. The results are shown in the form of a dependence of the equilibrium pH of solutions after contact with a sorbent on the initial pH of these solutions. The bend point on curve 3 corresponds to the zero charge point that is the value of the solution pH, at which the sorbent surface is uncharged. In our case, the pH
pzc value is equal to 4.3. Below this value of solution pH, the sorbent surface is charged positively, which prevents uranium adsorption due to electrostatic repulsion. Above pH
pzc, the sorbent surface is charged negatively, which positively affects the efficiency of recovery of uranium and its hydrated cationic forms.
The results of the determination of pH
pzc are in good agreement with the dependence of the efficiency of uranium recovery on the solution pH (
Figure 8). The solution pH correction was carried out using solutions of HNO
3 and NaOH. In acidic solutions, low recovery efficiency is related to positively charged sorbent surface as, probably, to the predominant reaction of protonation.
The form of uranium presence in a solution depends significantly on the solution pH. In solutions with pH above 4, one observes the increase in the uranium recovery due to strong binding by the adsorbent negatively charged surface and predominant cationic hydrate forms of uranium. The dependence of the composition of the hydrated uranium cation with the pH increase (from 4 to 8) is expressed in the row UO
2OH
+ → (UO
2)
2(OH)
22+ → (UO
2)
3(OH)
5+ [
37]. The recovery efficiency maximum is attained at pH 8, with a subsequent decrease at the transition to the alkaline range. The decrease in the efficiency of uranium recovery in solutions with pH above 8 is probably related to the increased formation of the anionic form of uranium UO
2(OH)
3− [
37] and the uranium adsorption on the flask walls, which was found in the control experiment without the sorbent (curve 3,
Figure 8). The high efficiency of the uranium recovery is explained by the presence of a large number of sorption sites forming at polymer formation as a result of polycondensation of cyclic molecules through the ˃Se=Se< bond.
Table 2 shows the values of the sorption-selective properties of the sorbent, depending on the initial pH of the solution. According to the obtained data, these materials can be recommended for the recovery of uranium from neutral and slightly alkaline solutions. A change in the value of pH of the model solution as compared to the initial value was observed. This was probably the result of deprotonation of the =N–O–H group during the adsorption.
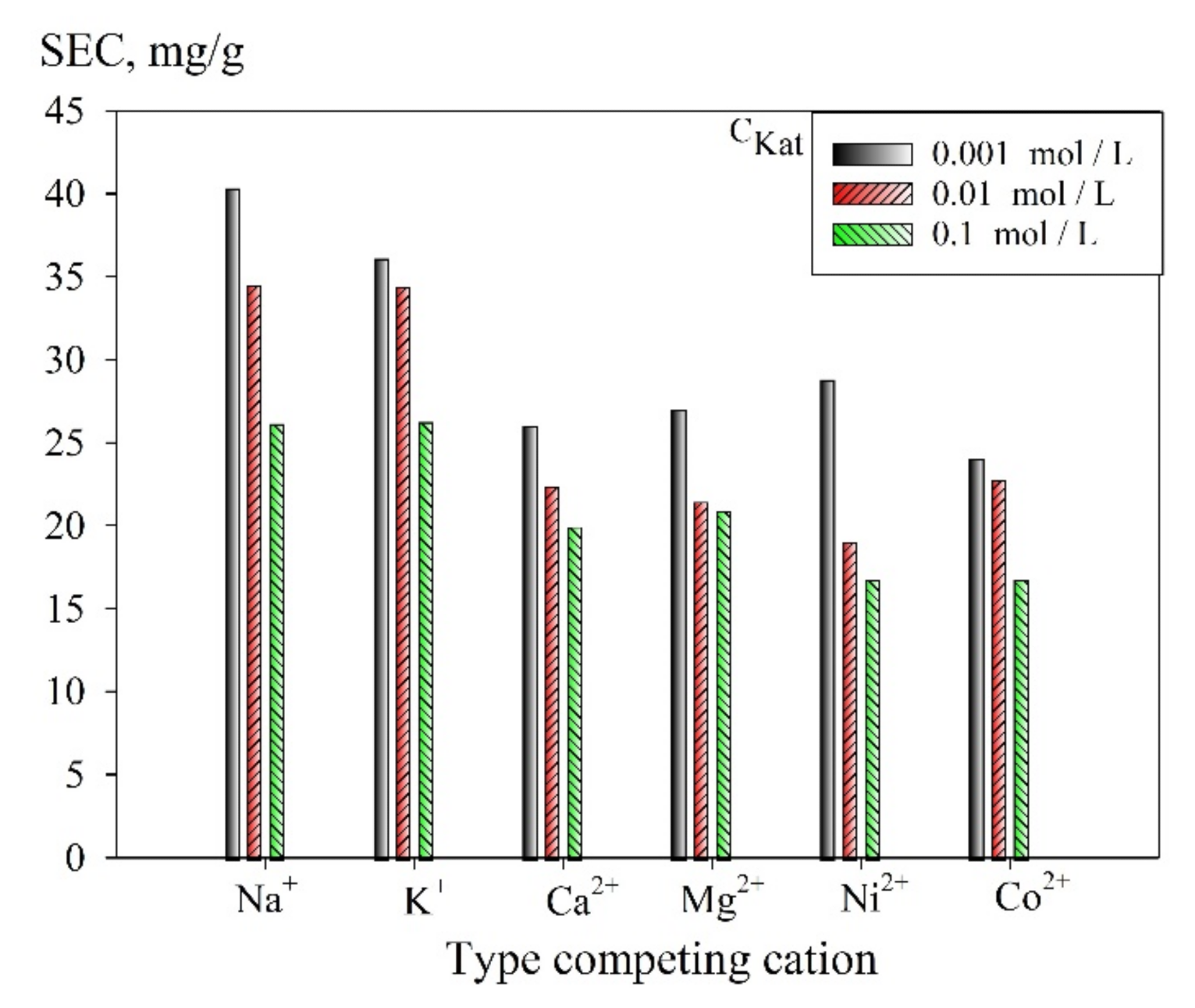
Figure 9 shows a diagram of the dependence of the SEC uranium values on the concentration of cations in the solution. It was found that double-charged cations reduce the SEC value to a greater extent than single-charged cations, which is explained by the electrostatic interaction, as well as due to the larger number of occupied adsorption sites, which is associated with an increase in the equivalence factor of multiply charged elements. The sequence of cations by the degree of increasing negative impact on the value of SEC is as follows: Na
+ ≤ K
+ < Mg
2+ ≤ Ni
2+ ≤ Ca
2+ ≤ Co
2+. However, it is worth mentioning that despite the decrease in SEC values at increasing the concentration of competing cations, the efficiency of the uranium recovery exceeds 75%, regardless of the competing ions.
The uranium extraction efficiency in the presence of other (competing) cations at pH 6 and 8 is generally comparable to each other. For this reason, this experiment was carried out only at pH6. The most negative effect on the uranium extraction efficiency is exerted by anions, the effect of which strongly depends on the solution pH.
Uranium undergoes hydrolysis and has the affinity to complexation: this effect is clearly expressed in neutral and slightly alkaline media, which significantly complicates its recovery by sorption. The presence of various anions in the solutions, such as bicarbonates or sulfates, can significantly reduce the efficiency of uranium extraction. Experiments were carried out to estimate the negative effect of anions.
Figure 10 shows the curves of the dependence of the SEC uranium values on the type and concentration of some anions that may be present in natural and technological liquid media.
It was found that the anions negatively affected the SEC value to varying degrees. The sequence of anions according to the degree of increasing negative impact on the value of SEC is as follows: NO
3− < PO
43− < Cl
− < HCO
3− < SO
42−. The greatest negative effect is provided by bicarbonate and sulfate ions, which is explained by the formation of stable anionic complexes of the type [UO
2(SO
4)
3]
4−, [(UO
2)
2CO
3(OH)
3]
−, [UO
2(CO
3)
3]
4−, [UO
2(CO
3)
2]
2− and [UO
2(CO
3)
2(H
2O)
2]
2− [
38], the probability of their formation increases with the pH growing. However, when the anion concentration is equal to 0.01 mol L
−1, one observes less than a two-fold decrease in the adsorption of uranium, which is also associated with the formation of complex ions such as [UO
2NO
3]
+, [UO
2PO
4]
–, [UO
2Cl]
+ UO
2Cl
20, etc. [
39]. The weakening of the sorption properties in alkaline solutions is related to the partial oxidation of the organic radical, accompanied by the destruction of the sorbent.
The Se-init after the sorption of uranyl ions was analyzed by the XPS spectroscopy: the results showed the presence of U 4f signals with binding energies of 392.8 and 382.0 eV, which indicated the binding of uranyl ions in the sorbent due to complexation and partial ion exchange.
Figure 11 illustrates the structure of the binding of uranyl ions by Se-init. After the deprotonation reaction of the –OH group, the binding of the radionuclide can proceed according to the M1 mechanism in acidic or neutral media, as well as by the M2 mechanism (
Figure 11) in slightly alkaline and alkaline media due to binding of the oxygen and nitrogen atoms at the oxime group.
The preservation of mechanical stability is an important requirement for sorption materials created for long-term use in sorption columns. This requirement can be fulfilled by developing composite materials by depositing them on a mechanically stable matrix.
In the present work, silica gel was used as such a matrix: it was modified with a polymer-based on the Se-derivative of amidoxime.
Table 3 shows the surface characteristics of the resulting materials: the data were obtained using nitrogen adsorption and subsequent calculation according to the BET equation. It was shown that, unlike the initial material (Se-init), the composite materials have a more developed surface, which increases proportionally to the increase in the weight content of silica gel.
Figure 12 shows the adsorption isotherms obtained in the solutions with pH 6 (
Figure 12a) and pH 8 (
Figure 12b), which can be attributed to the L-type [
40], thus indicating a high affinity of the adsorbents to uranium. Besides, the highest SEC is attained at pH 8, which is probably related to peculiarities of the ionic form of uranium in solution: in particular, at pH 8, the predominant forms are UO
2(OH)
3– and (UO2)
3(OH)
5+, while at pH 6—UO
2(OH)
+, (UO
2)
2(OH)
22+, UO
2(OH)
20, and (UO2)
3(OH)
5+, respectively [
37].
Table 4 shows the values of
Gmax and the adsorption equilibrium constant
Kl obtained by approximating the experimental points. The coefficient of determination exceeds 0.95, which indicates that the chosen model is correct. For the composite sorbents, the values of the adsorption equilibrium constant are quite close, which indicates the preservation of the adsorption mechanism. The adsorption equilibrium constants obtained for the composites have lower values in comparison with Se-init due to the reduced content of the sorption-active component (Se-derivative of 4-aminofurazane-3-carboxamidoxime). The highest values of the maximum adsorption were obtained for Se-50 and Se-65, which could be related to the greater availability of adsorption sites; however, we do not exclude the adsorption of uranyl ions on silica gel.
The highest values of the maximum adsorption were obtained for Se-50 and Se-65, which could be related to the greater availability of adsorption sites and also with partial adsorption of uranium on silica gel. In order to evaluate the silica gel effect on the composite’s sorption characteristics, a control experiment was carried out. In the control experiment, the efficiency of uranium adsorption with unmodified silica gel was evaluated.
Figure 13 shows the diagram of uranium sorption from model solutions with pH 6 and 8 on synthesized materials and pure silica gel used in the fabrication of composite materials. It is known that various types of silica gels are capable of adsorbing heavy metal ions from liquid media [
41,
42]. It should be noted that unmodified silica gel cannot efficiently extract uranium since ion-exchange hydroxyl groups in silica gel do not exhibit high selectivity to uranium in the presence of other metal ions and anions. In addition, the binding of uranium by embedding it in the crystal lattice of silica gel is significantly limited due to the low sorption capacity of the synthetic mineral. The efficiency of uranium extraction from the model solution does not exceed 50%; therefore, unmodified silica gel is not an effective sorbent. However, its use as a matrix for composite materials (along with Se-init) promotes the improvement of sorption properties, which is reflected in the increase in the efficiency of recovery (
Figure 13) and SEC (
Table 4), relatively to Se-init.
We evaluated the kinetic characteristics of the adsorption process on the composite sorbents.
Figure 14 shows the kinetic curves of the sorption of uranium from the model solution in semi-logarithmic coordinates (
Figure 14a). The kinetic curves indicate that the adsorption process on the composite sorbents proceeds at a higher rate, which is also associated with greater availability of the adsorption sites as compared with the non-composite sorbent. An increase in the specific surface area of composite materials leads to a decrease in the time to reach equilibrium adsorption, which is 10–20 min, while the adsorption efficiency exceeds 95%. A significant increase in the time to reach adsorption equilibrium on the Se-init sample is probably associated with the specific features of the adsorption process in the presence of uranium in various ionic forms. In addition, the diffusion process is important in the uranium-binding rate on a dense surface of a non-porous material, which in this case is likely to be limiting.
Figure 15 shows high-resolution SEM images for Se-init and the composite sorbents. Unlike Se-init, whose structure is represented by pronounced crystallites, the surface of the composite sorbents is relatively homogeneous and contains small particles of presumably the sorption-active component. The surface distribution of the sorption-active component indirectly corroborates the fact that the adsorption sites of the composite sorbent can have greater accessibility in comparison with the Se-init material, some of the sorption sites of which are located in the bulk of dense crystallites and therefore do not participate in the exchange.
Table 5 shows the parameters calculated for the pseudo-first and pseudo-second order equations (
Figure 14b). According to the determination coefficients, the kinetic adsorption curve of uranium for Se-init is better described by the pseudo-first-order equation, as opposed to the composite adsorbents. The increase in the reaction order for the composite sorbents could also indicate the increase in the availability of the adsorption sites and the predominance of the chemisorption process of uranium binding in comparison with the non-composite sorbent. It is also worth mentioning that the pseudo-second-order equation constant
k2 differs for different composite sorbents. For the sorbent Se-65, the calculated
k2 has the lowest value, which is associated with a reduced content of the sorption-active component affecting the kinetic characteristics.
Table 6 shows the results of testing the hydro-mechanical strength of the fabricated materials. One can note that at a transfer to Se-35, the material loses stability rather sharply, which is probably related to an excess of the sorption-active component washed off from the surface of the matrix during the stirring. The latter is corroborated by the fact that Se-init is mostly peptized in the model solutions, which complicates working with it.