Estimating a Stoichiometric Solid’s Gibbs Free Energy Model by Means of a Constrained Evolutionary Strategy
Abstract
1. Introduction
- data density,
- dispersion and abundance or lack of data sets,
- automation of the fitting procedure to avoid subjective data selection,
- correct thermodynamic model and its constraints,
- finding global minima considering the above.
2. Materials and Methods
2.1. Dealing with Different Data Densities: Weight Factors
- Experimental: Data points collected from experimental devices as they are most likely to be precise and reproducible.
- Mixed: A combination of experimental and theoretical approaches to obtain a given curve. For example, a fitted curve from a few experimental points. This is especially suitable when experimental points cannot be found but an equation from supposed experimental data is given.
- Theoretical/Table: Data obtained through simulations or theoretical formulations derived from other kind of properties. Table means that the value is from known data collection tables.
- Singular data: Singular points whose origin is unclear.
- Point: Just data points, they are often associated with experimental data and the combination of both gives the best weight possible.
- Quadratic: Quadratic (or higher order) equation or curve used to fit the data.
- Linear: the data comes in form of a linear temperature dependent fit.
- Constant: a constant value given for a range of temperatures.
- Not used: This extended flag has a weight of 0 and it is used to deactivate certain values for the fitting procedure without losing them entirely.
2.2. Evolutionary Strategy Method
- 1.
- The principle of maximum probability: The average distribution value is updated to maximize the likelihood that the previous most successful individuals are closer to the final solution. This is the selection and recombination step. The mean of the search distribution comes from the selection of the most relevant μ selected points from the sample. Writing the weight (not the same as the weight factors from the previous subpart) as ωi with the condition that:
- 2.
- Two types of temporal evolution of the average statistical distribution of the strategy are recorded. These paths contain significant information about the correlation between successive steps. In particular, the algorithm is effective if there is a large positive evolution in successive steps in the same direction. To do that, the covariance matrix, within the weighted selection mechanism is calculated with the following expression:
2.3. Definition of Constraints: Debye Model
if d cpfitted(l)/dT ≤ 0, a value pty1,l is added for 1 ≤ l ≤ 2000
if d2 cpfitted(l)/dT2 ≤ 0, a value pty2,l is added for 50 ≤ l ≤ 2000
if d2 cpfitted(l)/dT2 ≤ 0, a value pty3,l is added for 1 ≤ l ≤ 5
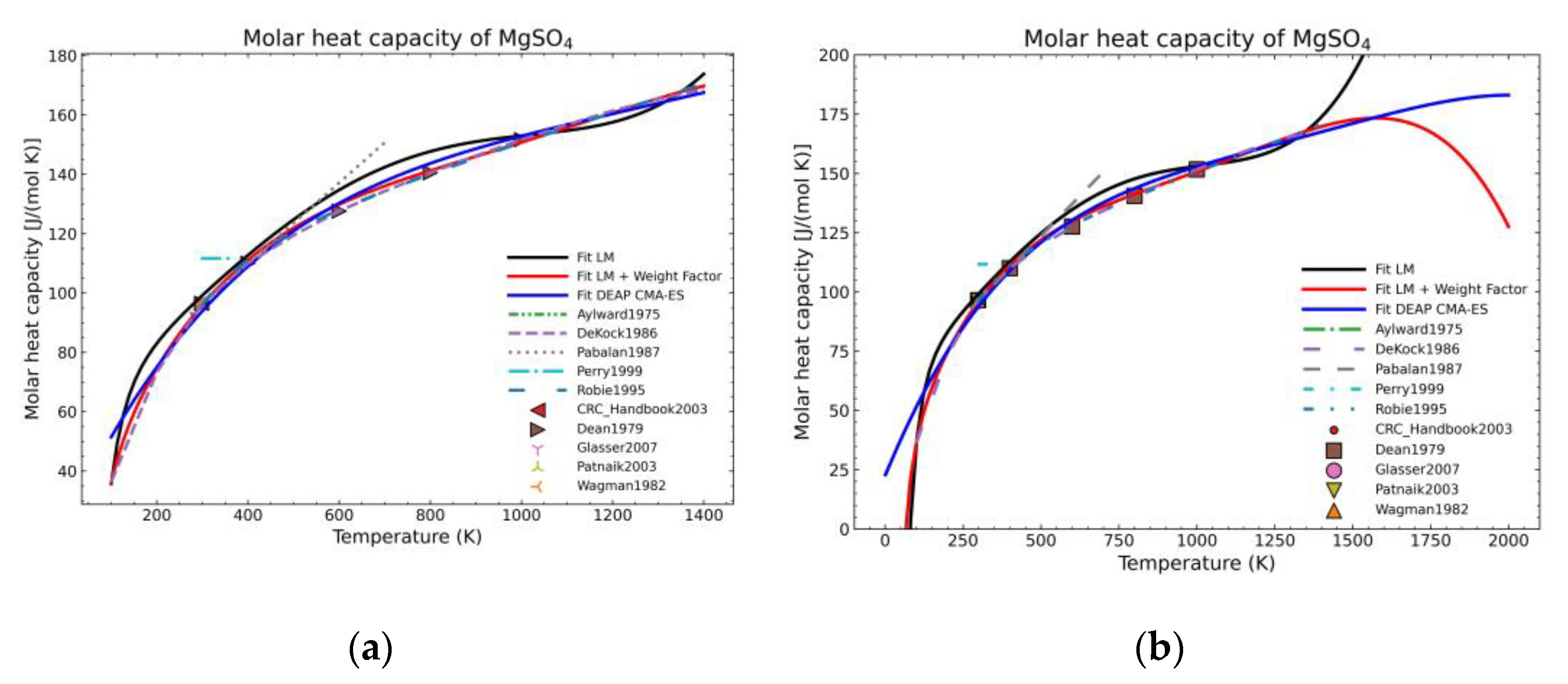
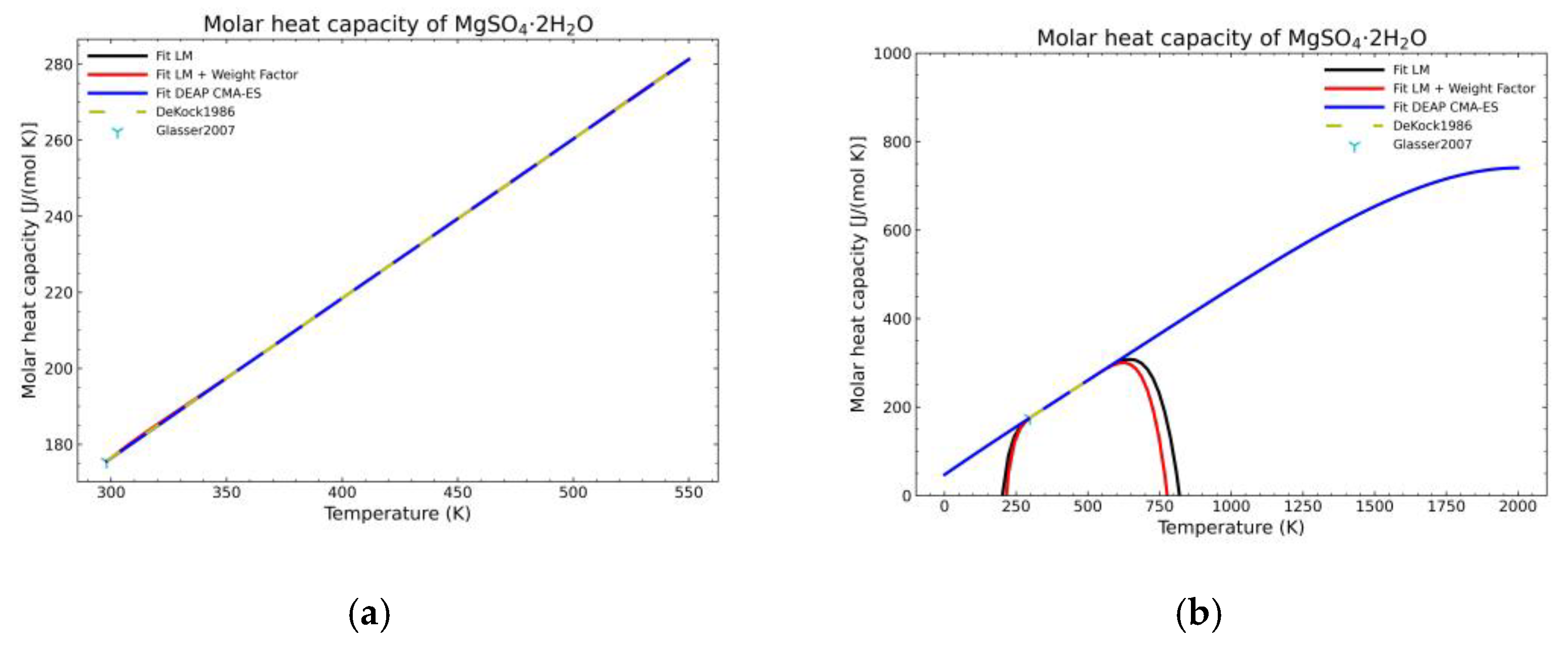
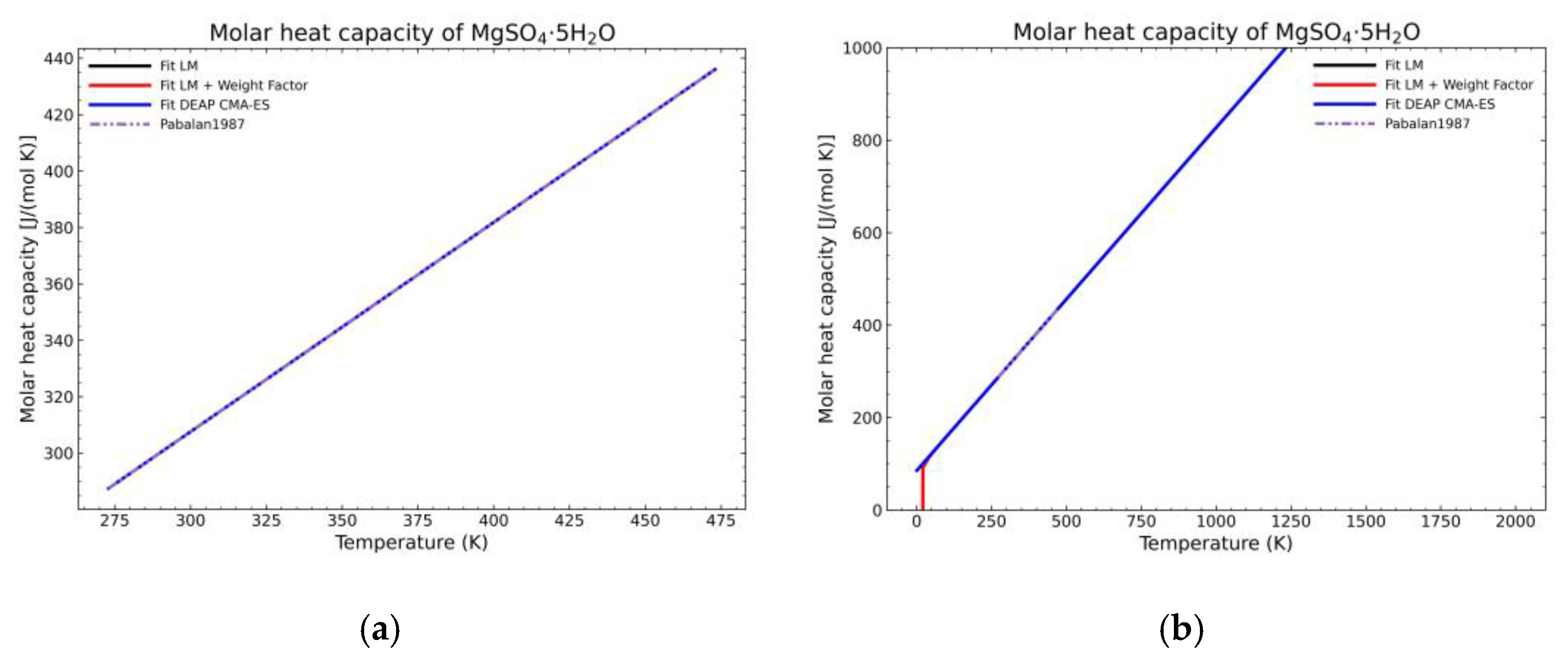
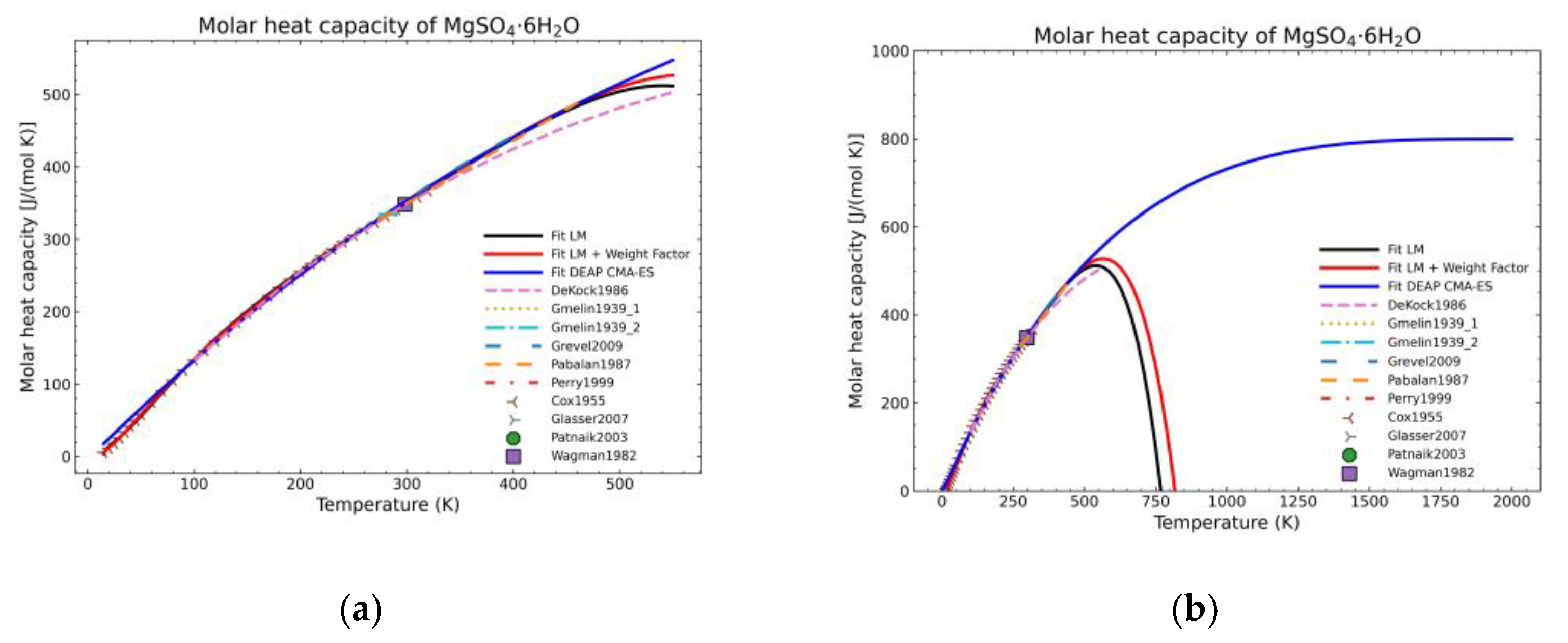
3. Results and Discussion
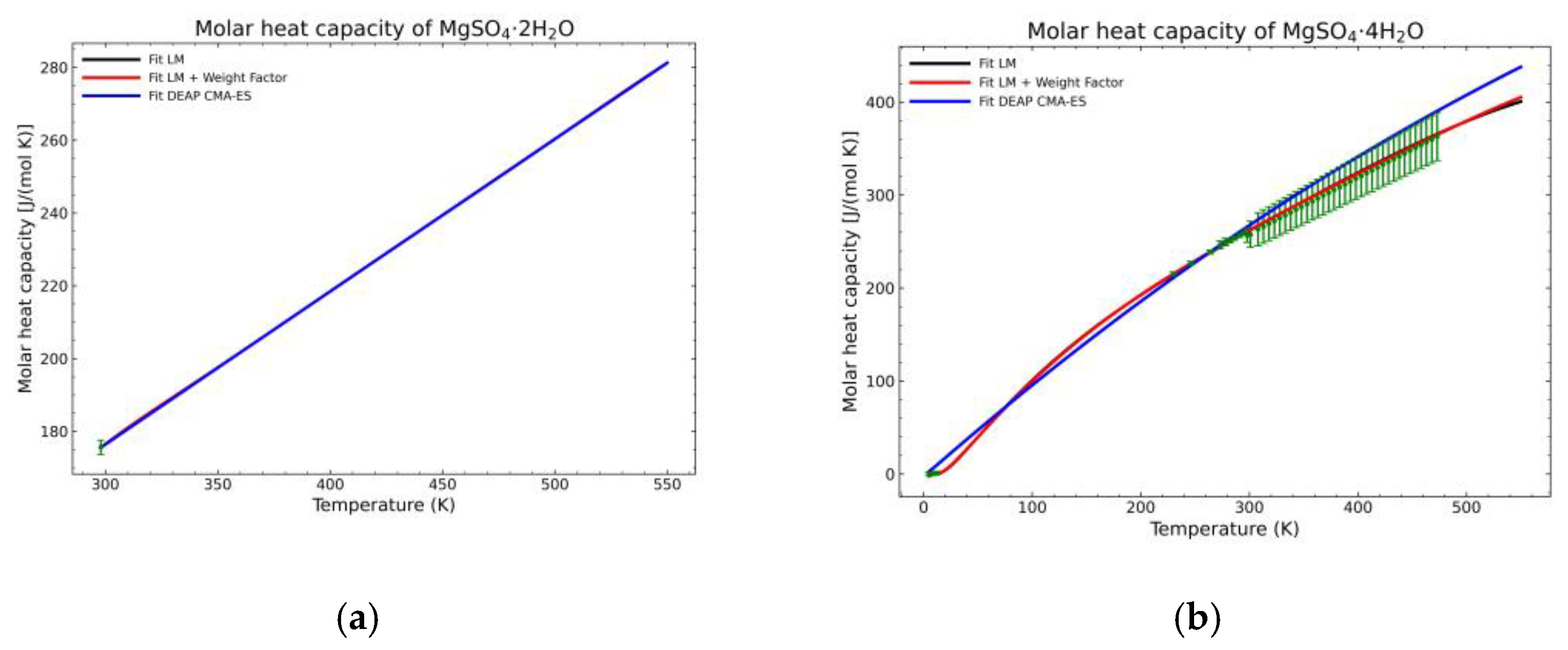
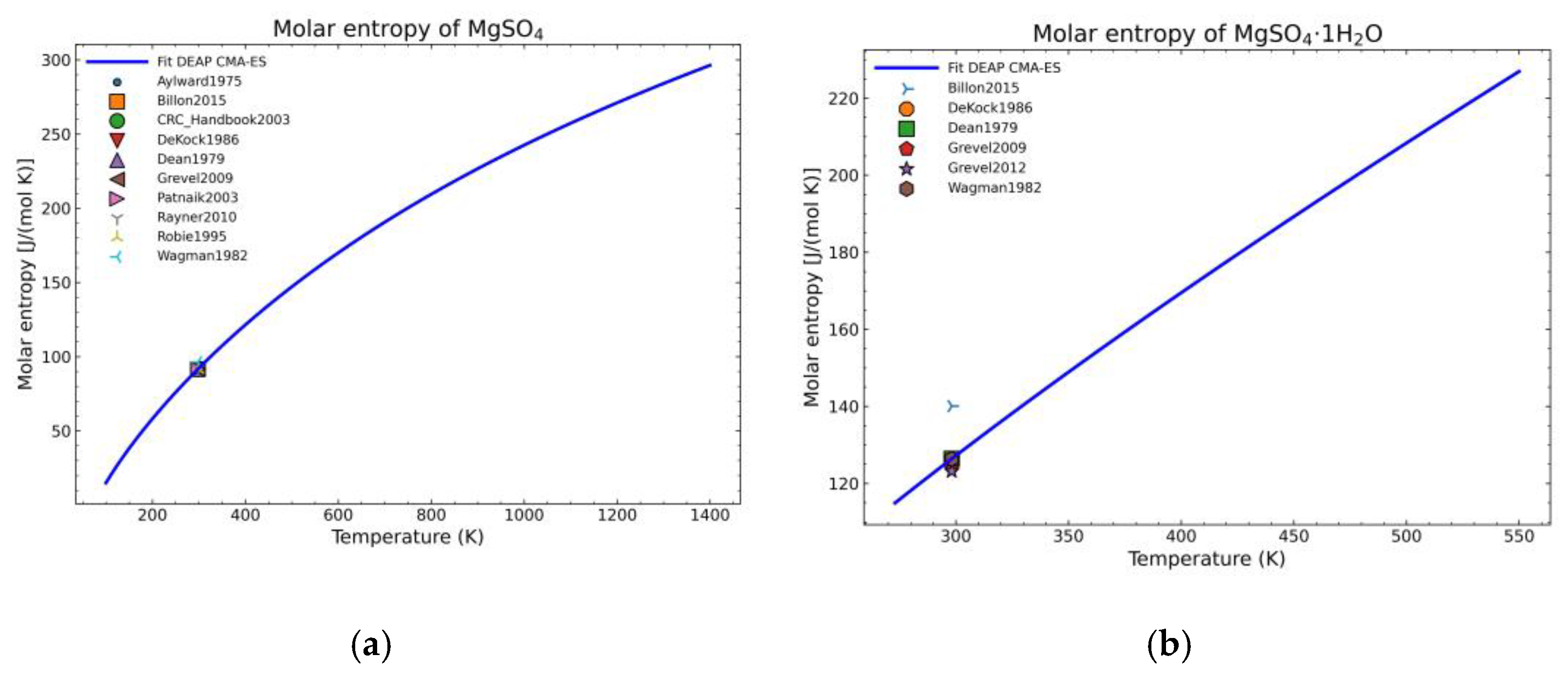
3.1. Computation of NASA9 Polynomial for Different Hydrates of Magnesium Sulfate
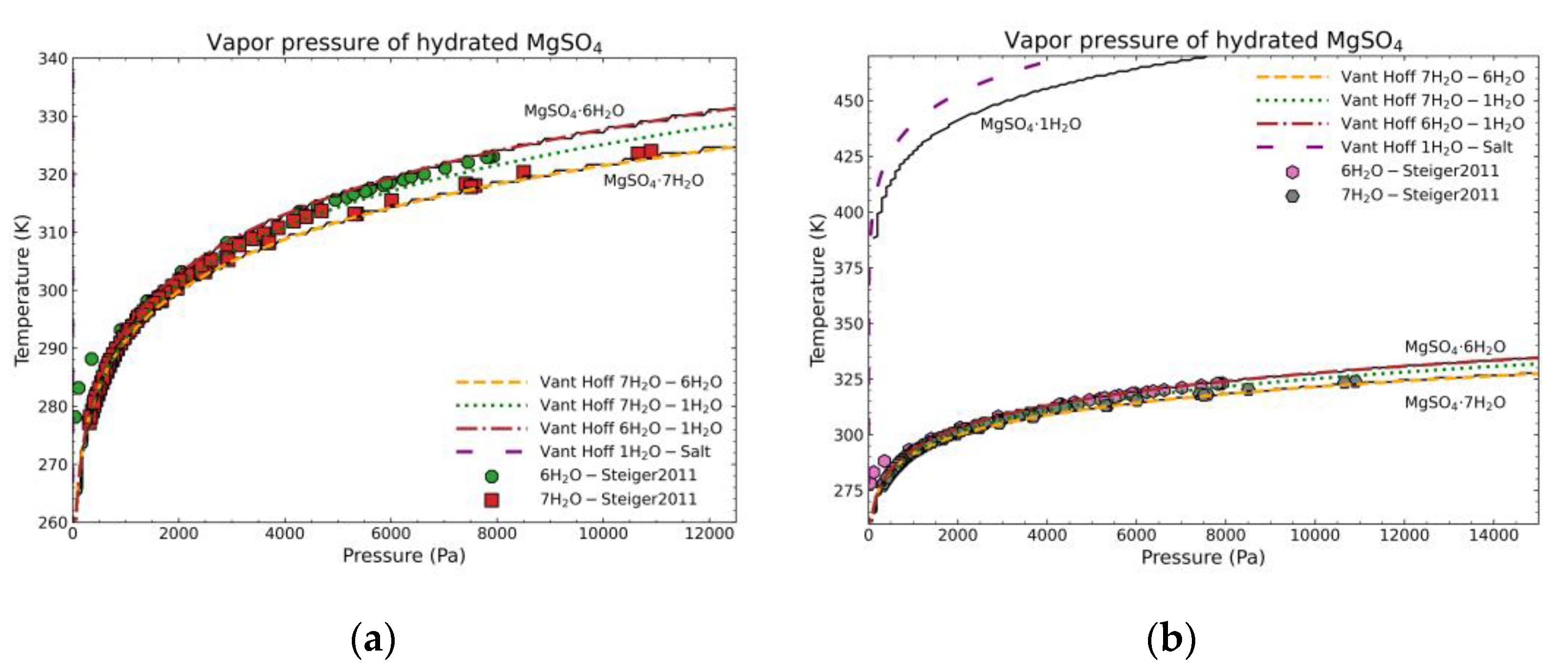
3.2. Validation of the Model: Heat Capacity Comparison and Vapor-Solid Equilibrium Curves
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moré, J.J. Numerical Analysis; Springer: Berlin/Heidelberg, Germany, 1978; pp. 105–116. [Google Scholar]
- Stephan, P.; Schaber, K.; Stephan, K.; Mayinger, F. Thermodynamik: Grundlagen und Technische Anwendungen-Band 2: Mehrstoffsysteme und Chemische Reaktionen; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Sharma, A.; Tyagi, V.V.; Chen, C.R.; Buddhi, D. Review on Thermal Energy Storage with Phase Change Materials and Applications. Renew. Sustain. Energy Rev. 2009, 13, 318–345. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Castell, A.; Barreneche, C.; de Gracia, A.; Fernández, A.I. Materials Used as PCM in Thermal Energy Storage in Buildings: A Review. Renew. Sustain. Energy Rev. 2011, 15, 1675–1695. [Google Scholar] [CrossRef]
- Donkers, P.A.J.; Sögütoglu, L.C.; Huinink, H.P.; Fischer, H.R.; Adan, O.C.G. A Review of Salt Hydrates for Seasonal Heat Storage in Domestic Applications. Appl. Energy 2017, 199, 45–68. [Google Scholar] [CrossRef]
- Glasser, L.; Jenkins, H.D.B. The Thermodynamic Solvate Difference Rule: Solvation Parameters and Their Use in Interpretation of the Role of Bound Solvent in Condensed-Phase Solvates. Inorg. Chem. 2007, 46, 9768–9778. [Google Scholar] [CrossRef] [PubMed]
- DeKock, C.W. Thermodynamic Properties of Selected Metal Sulfates and Their Hydrates; Information Circular/United States Department of the Interior, Bureau of Mines: Washington, DC, USA, 1986. [Google Scholar]
- Pabalan, R.; Pitzer, K.S. Thermodynamics of Concentrated Electrolyte Mixtures and the Prediction of Mineral Solubilities to High Temperatures for Mixtures in the System Na-K-Mg-Cl-SO-OH-H20. Geochim. Cosmochim. Acta 1987, 51, 15. [Google Scholar] [CrossRef]
- Frost, G.B.; Breck, W.G.; Clayton, R.N.; Reddoch, A.H.; Miller, C.G. The Heat Capacities of the Crystalline and Vacuum Dehydrated Form of Magnesium Sulphate Monohydrate. Can. J. Chem. 1957, 35, 1446–1453. [Google Scholar] [CrossRef]
- Gilbert, B. Gmelin-Handbuch Der Anorganischen Chemie-System-Nummer 27Mg, Magnesium Die Verbindungen Des Magnesiums; Verl. Chemie: Berlin, Germany, 1939. [Google Scholar]
- Perry, R.H.; Green, D.W.; Maloney, J.O. Perry’s Chemical Engineers’ Handbook; McGraw-Hill: New York, NY, USA, 1999. [Google Scholar]
- Grevel, K.-D.; Majzlan, J. Internally Consistent Thermodynamic Data for Magnesium Sulfate Hydrates. Geochim. Cosmochim. Acta 2009, 73, 6805–6815. [Google Scholar] [CrossRef]
- Wagman, D.D.; Evans, W.H.; Parker, V.B.; Schumm, R.H.; Halow, I.; Bailey, S.M.; Churney, K.L.; Nuttall, R.L. The NBS Tables of Chemical Thermodynamic Properties: Selected Values for Inorganic and C1 and C2 Organic Substances in SI Units; American Chemical Society and the American Institute of Physics for the National Bureau of Standards: Washington, DC, USA, 1982. [Google Scholar]
- Grevel, K.-D.; Majzlan, J.; Benisek, A.; Dachs, E.; Steiger, M.; Fortes, A.D.; Marler, B. Experimentally Determined Standard Thermodynamic Properties of Synthetic MgSO4 4H2O (Starkeyite) and MgSO4 3H2O: A Revised Internally Consistent Thermodynamic Data Set for Magnesium Sulfate Hydrates. Astrobiology 2012, 12, 1042–1054. [Google Scholar] [CrossRef] [PubMed]
- Dean, J.A.; Lange, N.A. (Eds.) Lange’s Handbook of Chemistry, 15th ed.; McGraw-Hill: New York, NY, USA, 1999. [Google Scholar]
- Billon, S.; Vieillard, P. Prediction of Enthalpies of Formation of Hydrous Sulfates. Am. Mineral. 2015, 100, 615–627. [Google Scholar] [CrossRef]
- Chase, M.W. NIST-JANAF Thermochemical Tables; American Chemical Society; American Institute of Physics for the National Institute of Standards and Technology: Washington, DC, USA; Woodbury, NY, USA, 1998. [Google Scholar]
- McBride, B.J.; Zehe, M.J.; Gordon, S. NASA Glenn Coefficients for Calculating Thermodynamic Properties of Individual Species; National Aeronautics and Space Administration: Cleveland, OH, USA, 2002; p. 297. [Google Scholar]
- Marzouk, O.A. Assessment of Three Databases for the NASA Seven-Coefficient Polynomial Fits for Calculating Thermodynamic Properties of Individual Species. IJASAR 2018, 150–163. [Google Scholar] [CrossRef]
- Aylward, G.H.; Aylward, G.H.; Findlay, T.J.V.; Findlay, T.J.V.; Zipprich, M. Datensammlung Chemie in SI-Einheiten; Chemie: Weinheim, Germany, 1975. [Google Scholar]
- Robie, R.A.; Hemingway, B.S. Thermodynamic Properties of Minerals and Related Substances at 298.15 K and 1 Bar (10^5 Pascals) Pressure and at Higher Temperatures; United States Government Printing Office: Washington, DC, USA, 1995.
- Lide, D.R. CRC Handbook of Chemistry and Physics, 84th ed.; Taylor & Francis: Milton Park, UK, 2003. [Google Scholar]
- Patnaik, P. Handbook of Inorganic Chemicals; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Rayner-Canham, G.; Overton, T. Descriptive Inorganic Chemistry; W.H. Freeman: New York, NY, USA, 2010. [Google Scholar]
- Hansen, N.; Ostermeier, A. Completely Derandomized Self-Adaptation in Evolution Strategies. Evol. Comput. 2001, 9, 159–195. [Google Scholar] [CrossRef] [PubMed]
- Hansen, N. Towards a New Evolutionary Computation. In Studies in Fuzziness and Soft Computing; Springer-Verlag: Heidelberg, Germany, 2007; Volume 192, pp. 75–102. [Google Scholar]
- De Rainville, F.-M.; Fortin, F.-A.; Gardner, M.-A.; Parizeau, M.; Gagné, C. DEAP: A Python Framework for Evolutionary Algorithms. In Proceedings of the 14th International Conference on Genetic and Evolutionary Computation Conference Companion-GECCO Companion ’12, Philadelphia, PA, USA, 7–11 July 2012; p. 85. [Google Scholar]
- Debye, P. Zur Theorie der spezifischen Wärmen. Ann. Phys. 1912, 344, 789–839. [Google Scholar] [CrossRef]
- Kittel, C. Introduction to Solid State Physics, 7th ed.; Wiley: New York, NY, USA, 1996. [Google Scholar]
- Misra, P. Physics of Condensed Matter; Elsevier Science: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Petit, A.T.; Dulong, P.L. Recherches Sur Quelques Points Importans de La Theorie de La Chaleur. Ann. Chim. Phys. 1819, 10, 395–413. [Google Scholar]
- Cox, W.P.; Hornun, E.W.; Giauque, W.F. The Spontaneous Transformation from Macrocrystalline to Microcrystalline Phases at Low Temperatures. The Heat Capacity of MgSO4·6H2O. J. Am. Chem. Soc. 1955, 77, 3935–3938. [Google Scholar] [CrossRef]
- Gurevich, V.M.; Kuskov, O.L.; Gavrichev, K.S.; Tyurin, A.V. Heat Capacity and Thermodynamic Functions of Epsomite MgSO4 7H2O at 0–303 K. Geochem. Int. 2007, 45, 206–209. [Google Scholar] [CrossRef]
- Denson, J.; Chevrier, V.; Sears, D.; Ivey, M. Sulfate Reduction: A Model for Subsurface Martian Life. Astrobiology 2009, 420, 213. [Google Scholar]
- Smith, W.R.; Missen, R. Chemical Reaction Analysis: Theory and Algorithms; John Wiley and Sons: Hoboken, NJ, USA, 1982. [Google Scholar]
- Goodwin, D.G.; Moffat, H.K.; Speth, R.L. Cantera: An Object-Oriented Software Toolkit for Chemical Kinetics, Thermodynamics, and Transport Processes; Caltech: Pasadena, CA, USA, 2017. [Google Scholar]
- Steiger, M.; Linnow, K.; Ehrhardt, D.; Rohde, M. Decomposition Reactions of Magnesium Sulfate Hydrates and Phase Equilibria in the MgSO4–H2O and Na+–Mg2+–Cl−–SO42−–H2O Systems with Implications for Mars. Geochim. Cosmochim. Acta 2011, 75, 3600–3626. [Google Scholar] [CrossRef]
- Wagner, W.; Pruß, A. The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use. J. Phys. Chem. Ref. Data 2002, 31, 387–535. [Google Scholar] [CrossRef]
- Okhrimenko, L.; Favergeon, L.; Johannes, K.; Kuznik, F.; Pijolat, M. Thermodynamic Study of MgSO4–H2O System Dehydration at Low Pressure in View of Heat Storage. Thermochim. Acta 2017, 656, 135–143. [Google Scholar] [CrossRef]
- Atkins, P.W.; De Paula, J. Atkins’ Physical Chemistry; Oxford University Press: Oxford, UK, 2006. [Google Scholar]
- van’t Hoff, J.H.; Dunsch, L. Studien zur Chemischen Dynamik: (Etudes de Dynamique Chimique) 1884; Geest & Portig: Leipzig, Germany, 1985. [Google Scholar]












| Type of Thermodynamic Data | Reference |
|---|---|
| Cp | Glasser, 2007 [6] |
| Cp, H0f, S0f | DeKock, 1986 [7] |
| Cp | Pabalan, 1987 [8] |
| Cp | Frost, 1957 [9] |
| Cp | Gmelin, 1939 [10] |
| Cp | Perry, 1999 [11] |
| H0f, S0f | Grevel, 2009 [12] |
| H0f, S0f | Wagman, 1982 [13] |
| H0f, S0f | Grevel, 2012 [14] |
| H0f, S0f | Dean, 1979 [15] |
| H0f, S0f | Billon, 2015 [16] |
| Group | Level Description | Assigned Weight Factor |
|---|---|---|
| Data source | Level 0—Experimental | 20 |
| Level 1—Mixed | 2−1 | |
| Level 2—Table/Theoretical | 2−2 | |
| Level 3—Singular data | 2−3 | |
| Equation type | Level 0—Point | 20 |
| Level 1—Quadratic | 2−1 | |
| Level 2—Linear | 2−2 | |
| Level 3—Constant | 2−3 |
| Type of Thermodynamic Data | Reference |
|---|---|
| Cp | Glasser, 2007 [6] |
| Cp, H0f, S0f | DeKock, 1986 [7] |
| Cp | Pabalan, 1987 [8] |
| Cp, H0f, S0f | Perry, 1999 [11] |
| H0f, S0f | Grevel, 2009 [12] |
| Cp, H0f, S0f | Wagman, 1982 [13] |
| Cp, H0f, S0f | Dean, 1979 [15] |
| H0f, S0f | Billon, 2015 [16] |
| Cp, H0f, S0f | Aylward, 1975 [20] |
| Cp, H0f, S0f | Robie, 1995 [21] |
| Cp, H0f, S0f | CRC_Handbook, 2003 [22] |
| Cp, H0f, S0f | Patnaik, 2003 [23] |
| H0f, S0f | Rayner, 2010 [24] |
| Type of Thermodynamic Data | Reference | Substance |
|---|---|---|
| Cp | Glasser, 2007 [6] | MgSO4·2H2O, MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| Cp, H0f, S0f | DeKock, 1986 [7] | MgSO4·2H2O, MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| Cp, H0f, S0f | Pabalan, 1987 [8] | MgSO4·5H2O, MgSO4·6H2O, MgSO4·7H2O |
| Cp | Gmelin, 1939 [10] | MgSO4·6H2O, MgSO4·7H2O |
| Cp | Perry, 1999 [11] | MgSO4·6H2O, MgSO4·7H2O |
| Cp, H0f, S0f | Grevel, 2009 [12] | MgSO4·2H2O, MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| Cp, H0f, S0f | Wagman, 1982 [13] | MgSO4·2H2O, MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| H0f, S0f | Grevel, 2012 [14] | MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| H0f, S0f | Dean, 1979 [15] | MgSO4·7H2O |
| H0f, S0f | Billon, 2015 [16] | MgSO4·4H2O, MgSO4·5H2O, MgSO4·6H2O, MgSO4·7H2O |
| H0f, S0f | Aylward, 1975 [20] | MgSO4·7H2O |
| H0f, S0f | Robie, 1995 [21] | MgSO4·7H2O |
| Cp, H0f, S0f | Patnaik, 2003 [23] | MgSO4·2H2O, MgSO4·4H2O, MgSO4·6H2O, MgSO4·7H2O |
| H0f, S0f | Rayner, 2010 [24] | MgSO4·7H2O |
| Cp, S0f | Cox, 1955 [32] | MgSO4·6H2O |
| Cp, S0f | Gurevich, 2007 [33] | MgSO4·7H2O |
| Minimum Initial Residual (No pty) | Maximum Final Residual (No pty) | pty0 | pty1 | pty2 | pty3 |
|---|---|---|---|---|---|
| 1016 | 102 | 106 | 1012 | 104 | 109 |
| Substance | Iterations | Population (λ) | Initial Distribution (σ) | Residual |
|---|---|---|---|---|
| MgSO4 | 290 | 315 | 3 | 1.72 × 102 |
| MgSO4·1H2O | 280 | 315 | 3 | 3.17 × 101 |
| MgSO4·2H2O | 350 | 315 | 3 | 2.75 × 10−4 |
| MgSO4·4H2O | 350 | 315 | 3 | 4.26 × 103 |
| MgSO4·5H2O | 350 | 315 | 3 | 6.11 × 10−26 |
| MgSO4·6H2O | 350 | 315 | 3 | 1.50 × 103 |
| MgSO4·7H2O | 350 | 315 | 3 | 5.79 × 103 |
| MRE | NRMSD | |||||||
|---|---|---|---|---|---|---|---|---|
| Substance | LM | LM + Weight Factor | CMA-ES | LM | LM + Weight Factor | CMA-ES | n | Number of Sources |
| MgSO4 | 3.02% | 2.98% | 3.92% | 3.66% | 4.73% | 5.11% | 694 | 10 |
| MgSO4·1H2O | 2.49% | 1.94% | 2.06% | 3.55% | 4.09% | 4.34% | 438 | 7 |
| MgSO4·2H2O | 0.02% | 0.02% | 0.02% | 0.02% | 0.03% | 0.03% | 13 | 2 |
| MgSO4·4H2O | 70.85% | 51.21% | 278.4% | 2.15% | 2.19% | 6.63% | 134 | 4 |
| MgSO4·5H2O | <0.01% | <0.01% | <0.01% | <0.01% | <0.01% | <0.01% | 201 | 1 |
| MgSO4·6H2O | 1.02% | 0.87% | 2.21% | 1.06% | 1.18% | 1.39% | 421 | 10 |
| MgSO4·7H2O | 246.79% | 468.28% | 24.93% | 2.88% | 3.04% | 3.09% | 650 | 9 |
| Substance | Iterations | Population (λ) | Initial Distribution (σ) |
|---|---|---|---|
| All | 100 | 55 | 5 |
| Enthalpy | Entropy | |||||
|---|---|---|---|---|---|---|
| Substance | MRE | NRMSD | n | MRE | NRMSD | n |
| MgSO4 | 0.81% | 1.85% | 15 | 0.54% | 1.41% | 14 |
| MgSO4·1H2O | 0.21% | 0.29% | 10 | 1.80% | 3.93% | 8 |
| MgSO4·2H2O | 0.03% | 0.04% | 4 | 10.22% | 11.17% | 2 |
| MgSO4·4H2O | 0.03% | 0.04% | 9 | 1.71% | 1.88% | 6 |
| MgSO4·5H2O | 0.03% | 0.03% | 2 | 1.65% | 1.65% | 2 |
| MgSO4·6H2O | 0.03% | 0.04% | 10 | 0.12% | 0.28% | 9 |
| MgSO4·7H2O | 0.02% | 0.03% | 14 | 0.60% | 1.28% | 14 |
| Substance | a0 | a1 | a2 | a3 | a4 | a5 | a6 | a7 | a8 |
|---|---|---|---|---|---|---|---|---|---|
| MgSO4 | 2.41 × 10−2 | −9.67 × 10−3 | 2.69 × 100 | 3.86 × 10−2 | −3.86 × 10−5 | 1.93 × 10−8 | −3.62 × 10−12 | −1.57 × 105 | −1.43 × 101 |
| MgSO4·1H2O | 2.37 × 10−2 | −1.38 × 10−2 | 6.22 × 100 | 3.36 × 10−2 | −3.19 × 10−6 | 4.44 × 10−9 | −2.31 × 10−12 | −1.97 × 105 | −3.01 × 101 |
| MgSO4·2H2O | 3.77 × 10−2 | −2.15 × 10−2 | 5.61 × 100 | 5.39 × 10−2 | −9.19 × 10−6 | 1.04 × 10−8 | −4.45 × 10−2 | −2.32 × 105 | −2.83 × 101 |
| MgSO4·4H2O | −1.24 × 10−1 | 3.71 × 10−1 | −3.71 × 10−1 | 1.24 × 10−1 | −5.20 × 10−5 | 3.78 × 10−9 | 1.22 × 10−12 | −3.05 × 105 | −2.21 × 100 |
| MgSO4·5H2O | 3.51 × 10−9 | −1.18 × 10−9 | 1.02 × 101 | 8.93 × 10−2 | −2.09 × 10−17 | 1.37 × 10−19 | −1.34 × 10−22 | −3.43 × 105 | −4.84 × 101 |
| MgSO4·6H2O | −1.76 × 10−1 | 5.27 × 10−1 | −5.28 × 10−1 | 1.76 × 10−1 | −1.19 × 10−4 | 3.54 × 10−8 | −3.88 × 10−12 | −3.78 × 105 | −2.64 × 100 |
| MgSO4·7H2O | −1.84 × 10−1 | 5.51 × 10−1 | −5.51 × 10−1 | 1.84 × 10−1 | −9.80 × 10−5 | 1.93 × 10−8 | −7.53 × 10−13 | −4.15 × 105 | −2.71 × 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grau Turuelo, C.; Pinnau, S.; Breitkopf, C. Estimating a Stoichiometric Solid’s Gibbs Free Energy Model by Means of a Constrained Evolutionary Strategy. Materials 2021, 14, 471. https://doi.org/10.3390/ma14020471
Grau Turuelo C, Pinnau S, Breitkopf C. Estimating a Stoichiometric Solid’s Gibbs Free Energy Model by Means of a Constrained Evolutionary Strategy. Materials. 2021; 14(2):471. https://doi.org/10.3390/ma14020471
Chicago/Turabian StyleGrau Turuelo, Constantino, Sebastian Pinnau, and Cornelia Breitkopf. 2021. "Estimating a Stoichiometric Solid’s Gibbs Free Energy Model by Means of a Constrained Evolutionary Strategy" Materials 14, no. 2: 471. https://doi.org/10.3390/ma14020471
APA StyleGrau Turuelo, C., Pinnau, S., & Breitkopf, C. (2021). Estimating a Stoichiometric Solid’s Gibbs Free Energy Model by Means of a Constrained Evolutionary Strategy. Materials, 14(2), 471. https://doi.org/10.3390/ma14020471






