Biointeraction of Erythrocyte Ghost Membranes with Gold Nanoparticles Fluorescents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Gold Nanoparticles
Preparation of Fluorescein Isothiocyanate-Coated Gold Nanoparticles (AuNPs-FI)
2.2. Nanoparticle Characterization
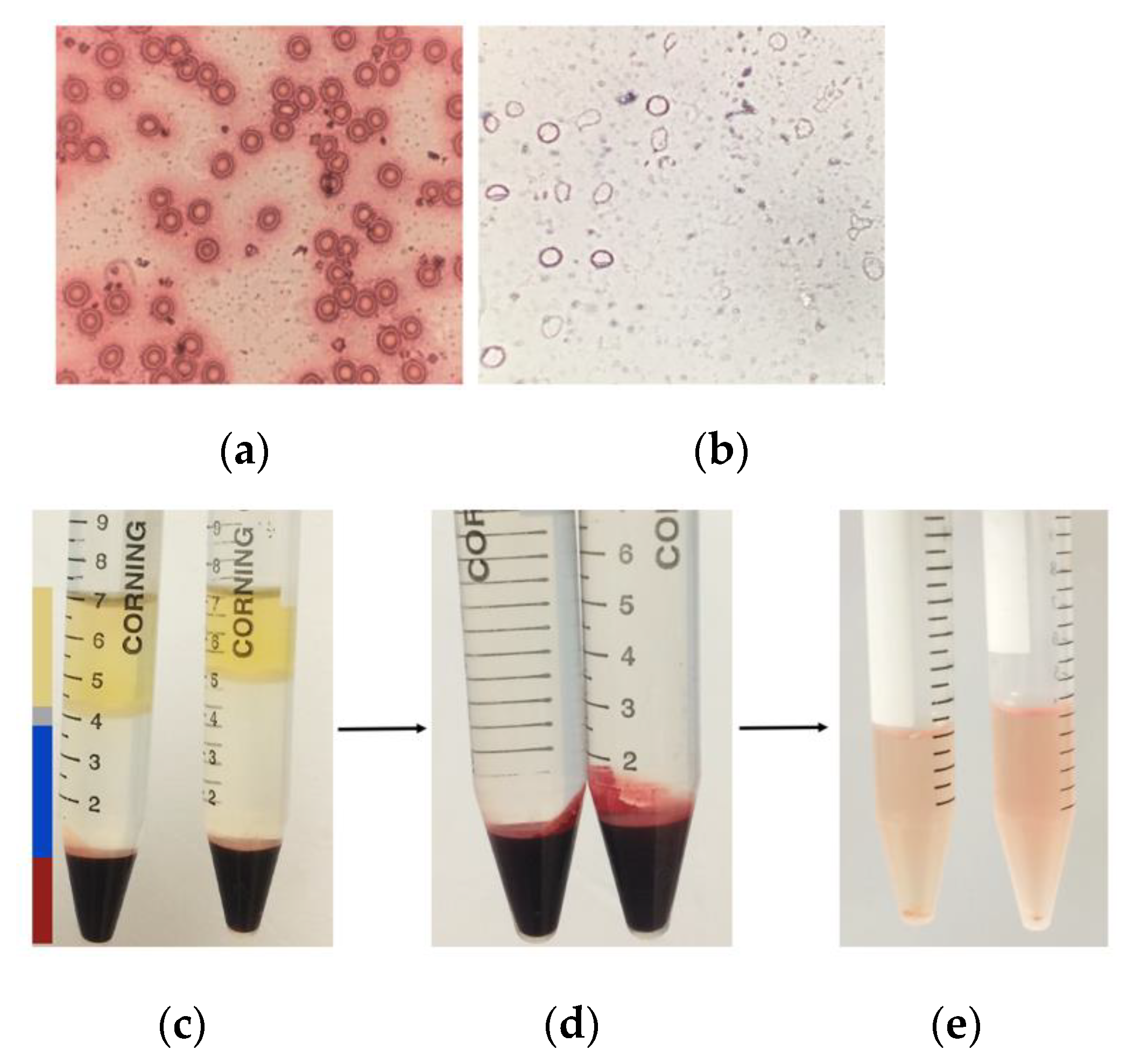
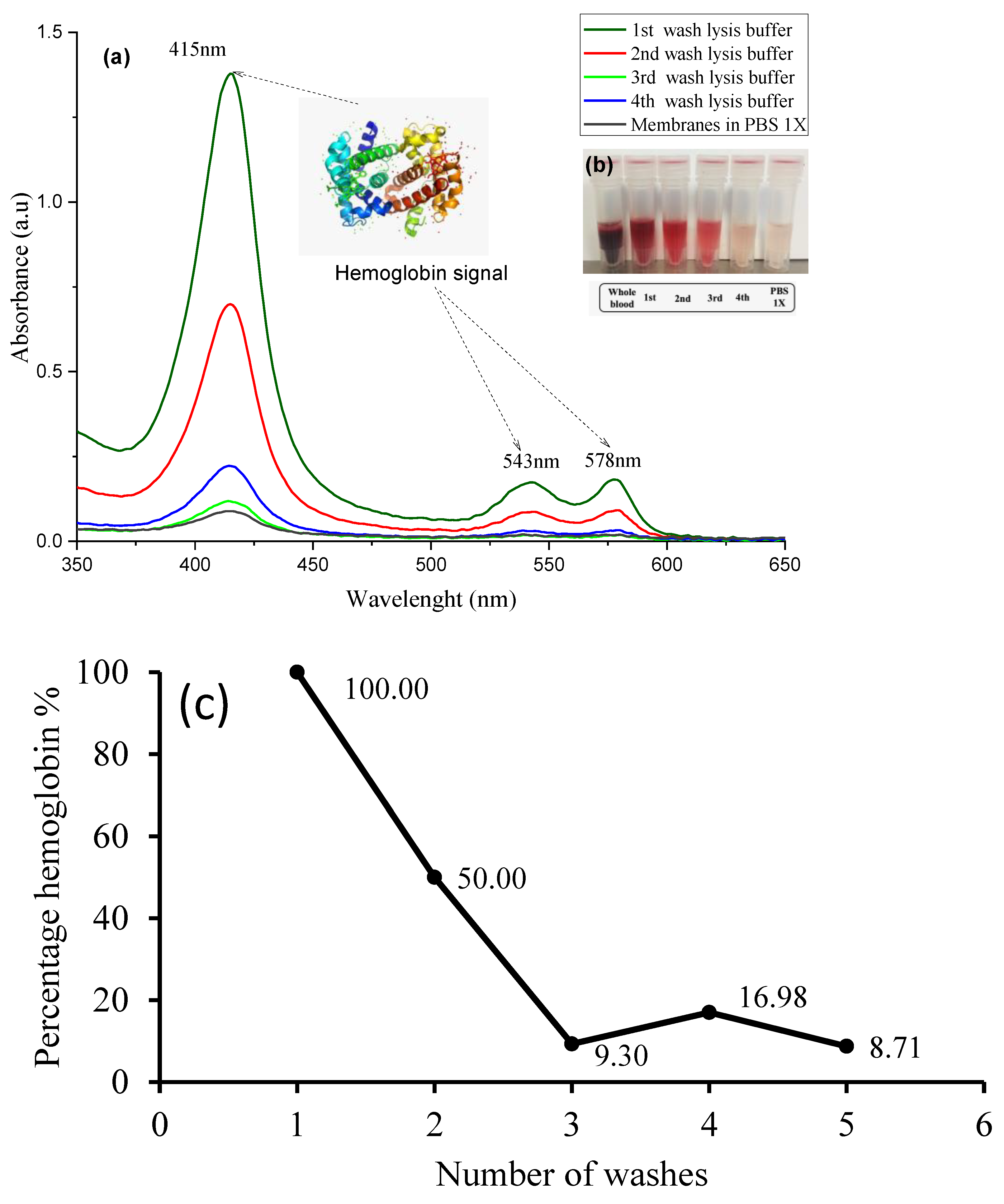
2.3. Isolation of Erythrocyte Ghost Membranes
2.4. Bio-Interaction
3. Results
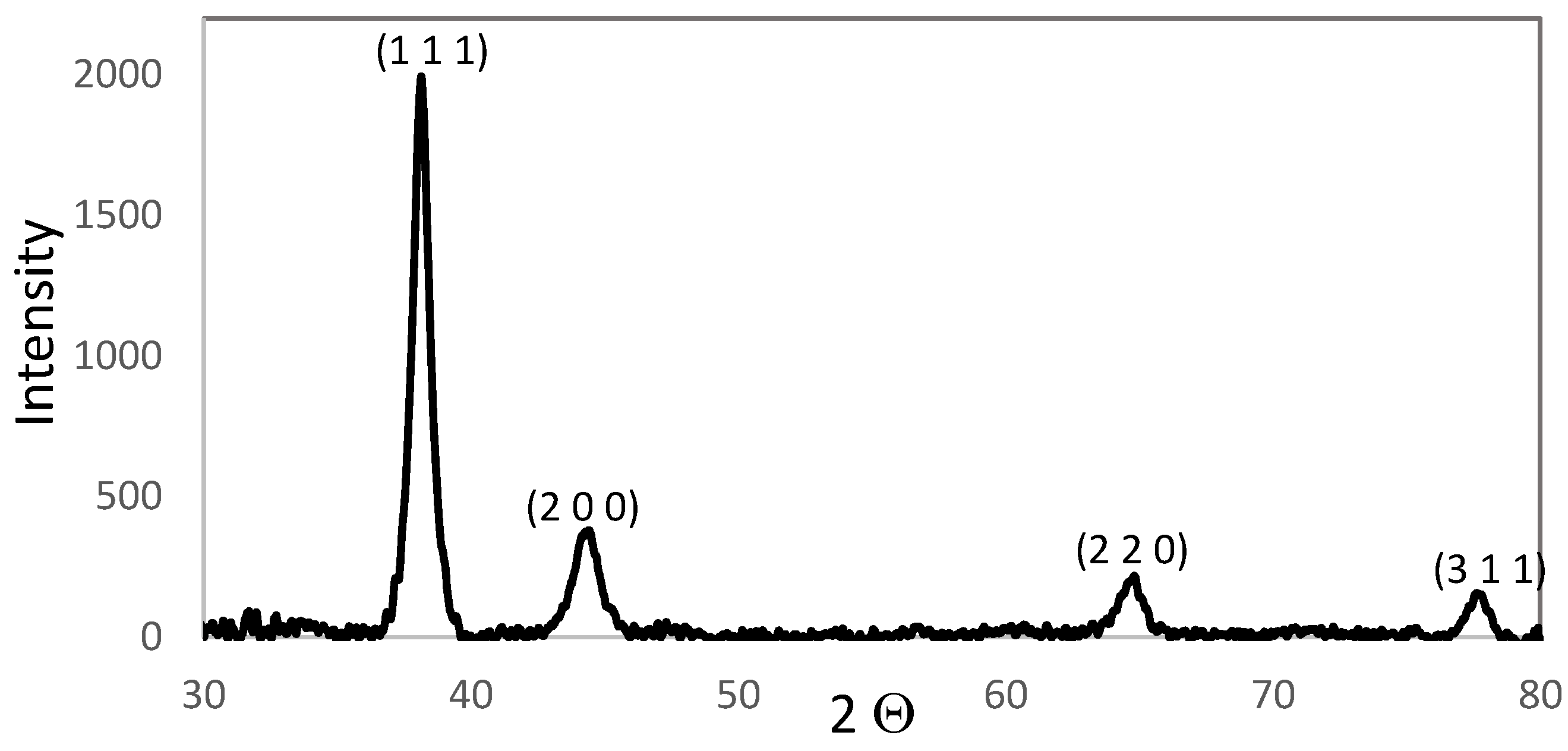
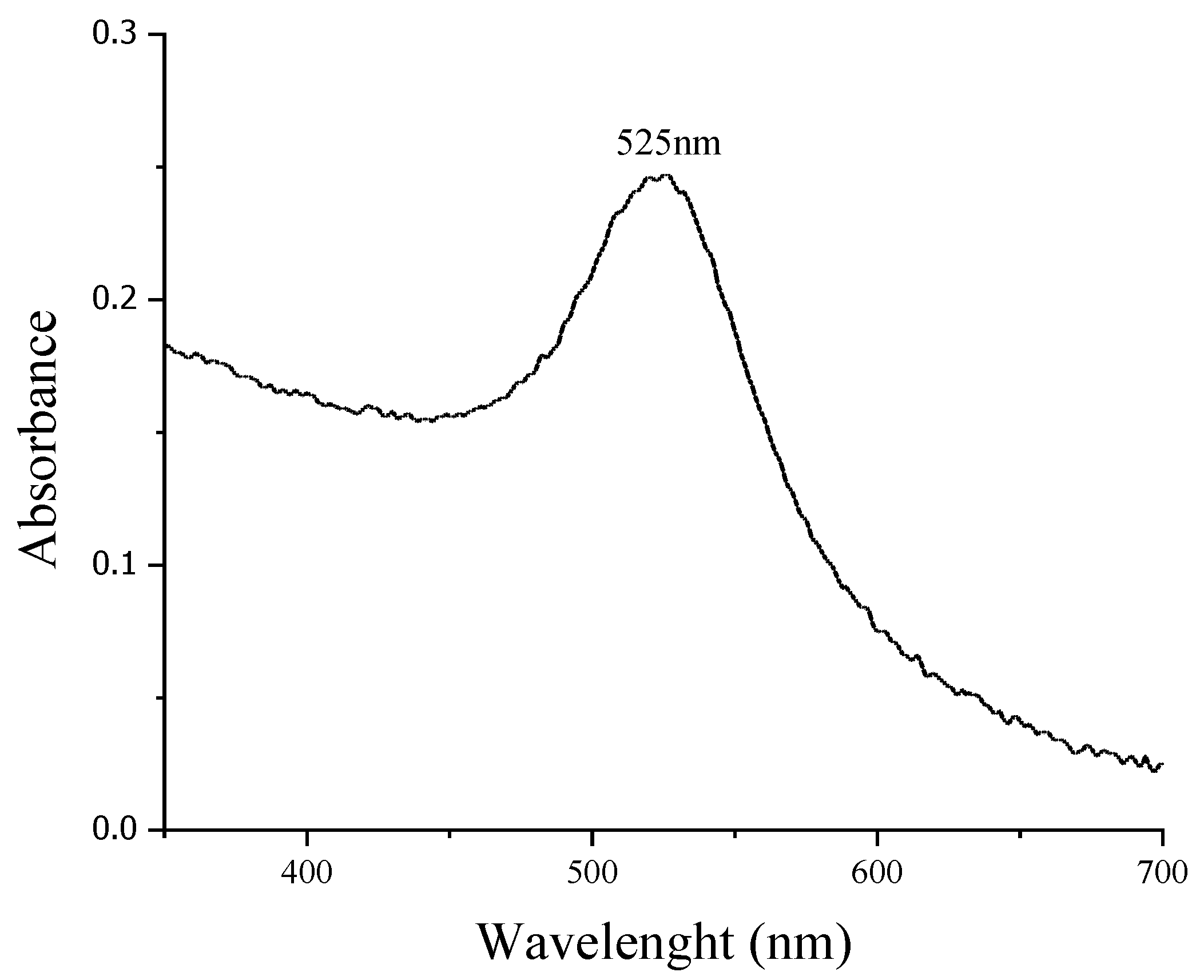
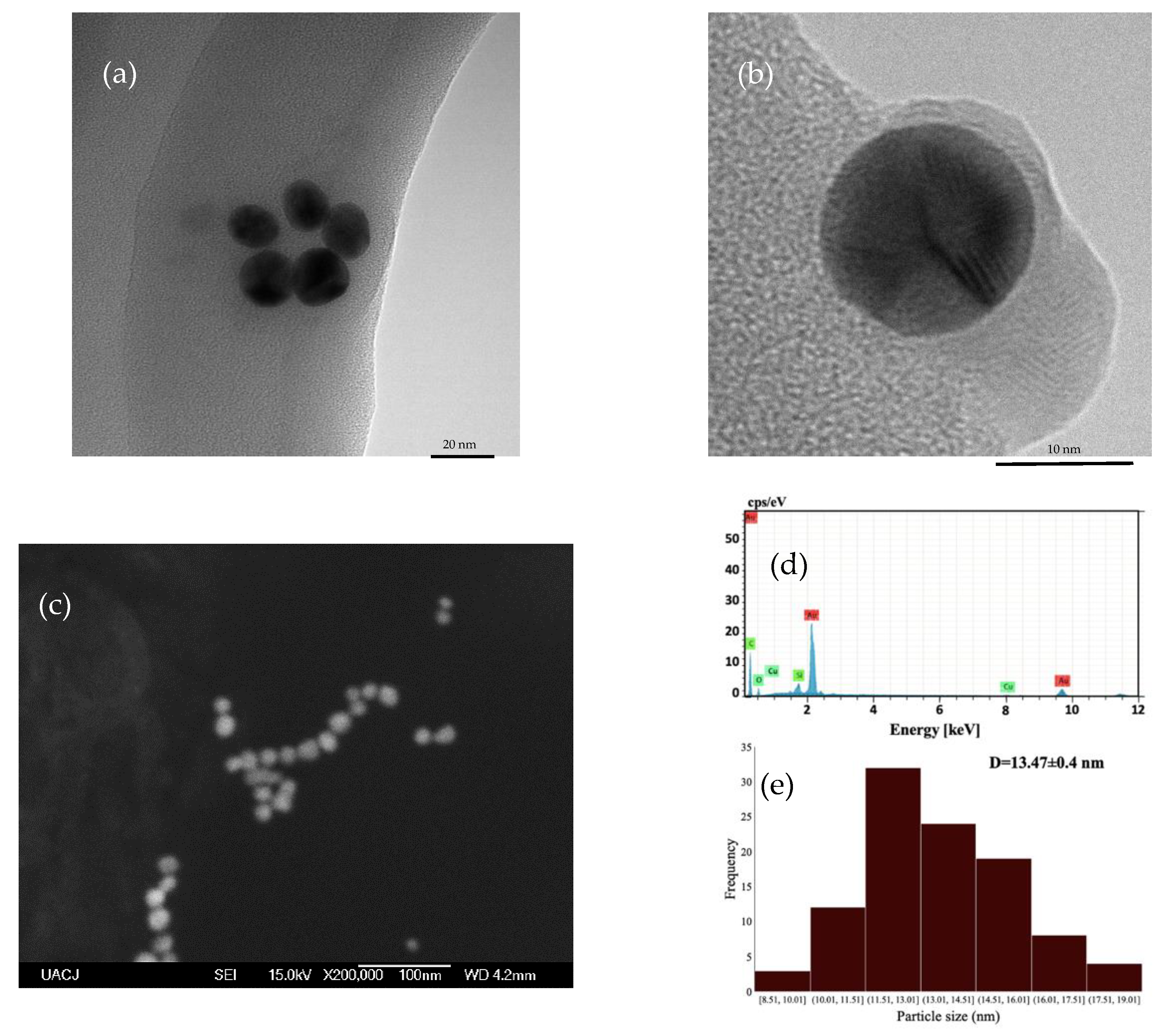
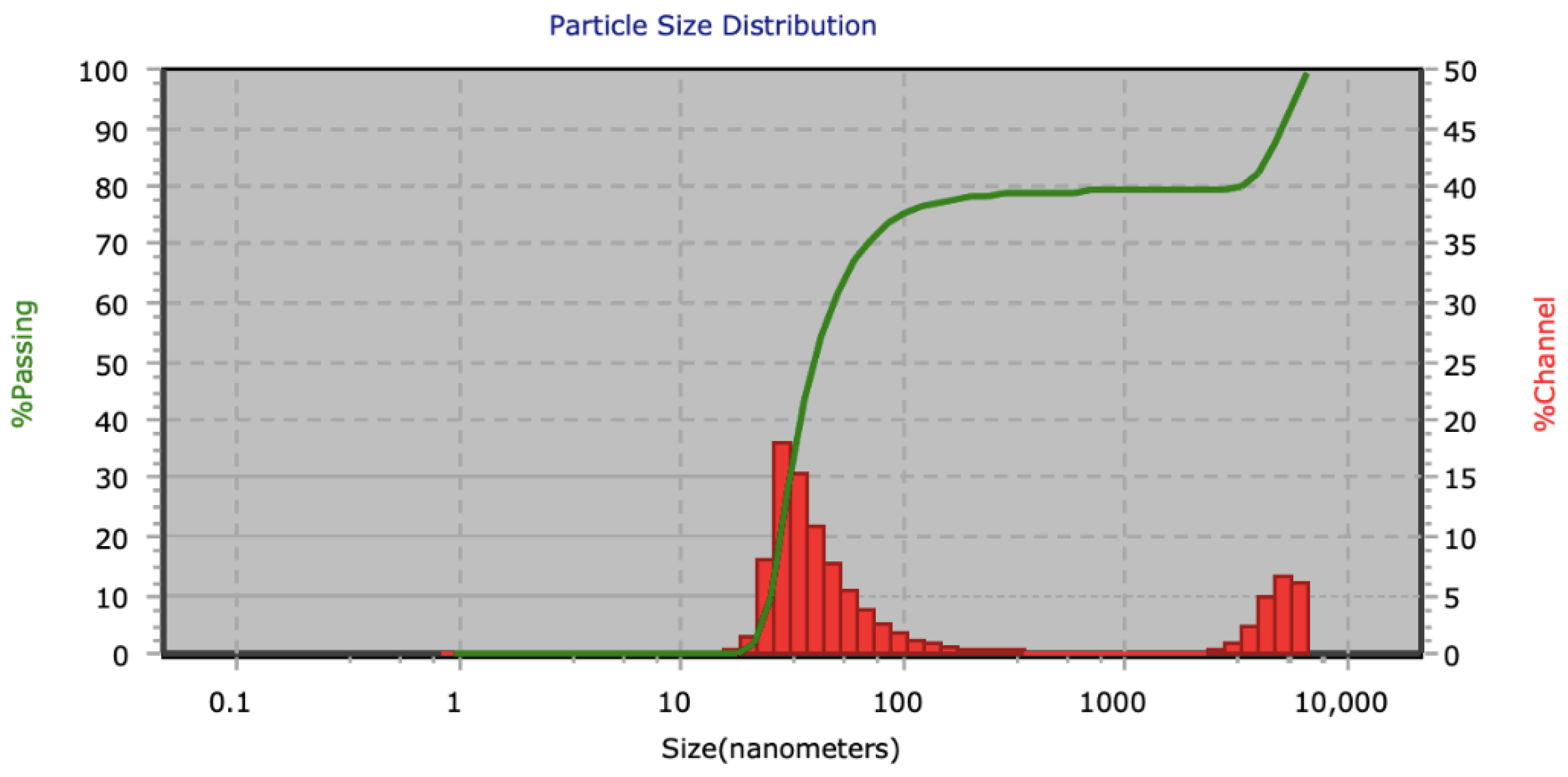
3.1. AuNPs
3.2. AuNPs-FI
Hemolytic Assays
3.3. Erythrocyte Ghost
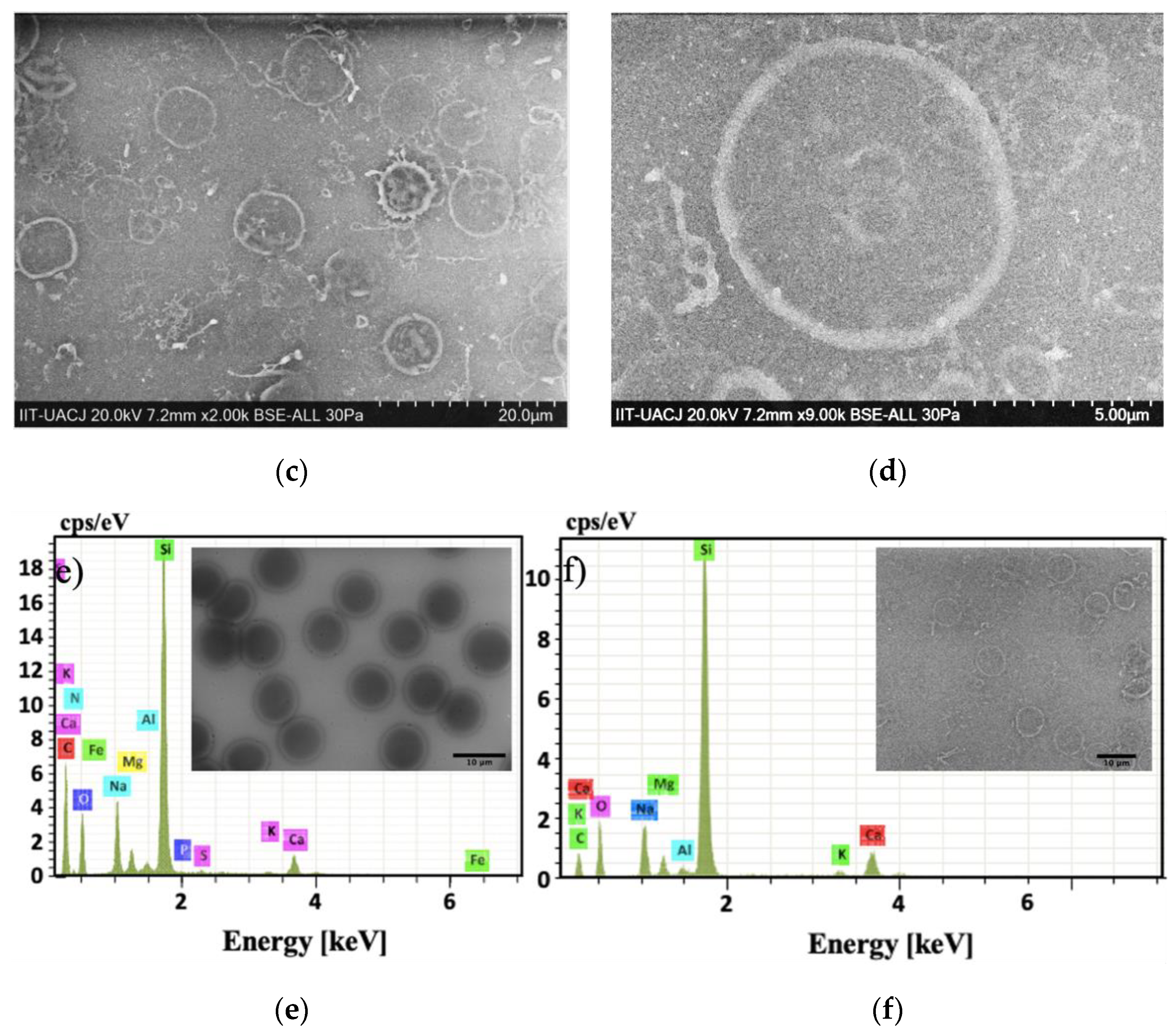
3.4. Biointeraction/Bioconjugation (AuNPs–Membrane Erythrocyte Ghost)
Biointeraction/Bioconjugation (AuNP-Fl-Membrane Erythrocyte Ghost)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schroeder, V.; Savagatrup, S.; He, M.; Lin, S.; Swager, T. Carbon Nanotube Chemical Sensors. Chem. Revn. 2019, 9, 599–663. [Google Scholar] [CrossRef]
- Yuan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, Y.; Yang, X.; Zhang, P.; et al. Stable Metal–Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 30, 1–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qu, A.; Sun, M.; Kim, J.-Y.; Xu, L.; Hao, C.; Ma, W.; Wu, X.; Liu, X.; Kuang, H.; Kotov, N.A.; et al. Stimulation of Neural Stem Cell Differentiation by Circularly Polarized Light Transduced by Chiral Nanoassemblies. Nat. Biomed. Eng. 2021, 5, 103–113. [Google Scholar] [CrossRef]
- Flores, A.M.; Hosseini-Nassab, N.; Jarr, K.-U.; Ye, J.; Zhu, X.; Wirka, R.; Koh, A.L.; Tsantilas, P.; Wang, Y.; Nanda, V.; et al. Pro-Efferocytic Nanoparticles Are Specifically Taken up by Lesional Macrophages and Prevent Atherosclerosis. Nat. Nanotechnol. 2020, 15, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Zhang, Y.S.; Pang, B.; Hyun, D.C.; Yang, M.; Xia, Y. Engineered Nanoparticles for Drug Delivery in Cancer Therapy. Angew. Chem. Int. Ed. 2014, 53, 12320–12364. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Chen, Q.; Qi, H.; Ruan, L.; Ren, Y. Experimental Comparison of Photothermal Conversion Efficiency of Gold Nanotriangle and Nanorod in Laser Induced Thermal Therapy. Nanomaterials 2017, 7, 416. [Google Scholar] [CrossRef] [Green Version]
- Gan, R.; Fan, H.; Wei, Z.; Liu, H.; Lan, S.; Dai, Q. Photothermal Response of Hollow Gold Nanorods under Femtosecond Laser Irradiation. Nanomaterials 2019, 9, 711. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; El-Sayed, M.A. Gold Nanoparticles: Optical Properties and Implementations in Cancer Diagnosis and Photothermal Therapy. J. Adv. Res. 2010, 1, 13–28. [Google Scholar] [CrossRef] [Green Version]
- Moon, H.J.; Ku, M.; Lee, H.; Yoon, N.; Yang, J.; Bong, K.W. Implantable Photothermal Agents Based on Gold Nanorods-Encapsulated Microcube. Sci. Rep. 2018, 8, 13683. [Google Scholar] [CrossRef]
- Nair, L.V.; Nazeer, S.S.; Jayasree, R.S.; Ajayaghosh, A. Fluorescence Imaging Assisted Photodynamic Therapy Using Photosensitizer-Linked Gold Quantum Clusters. ACS Nano 2015, 9, 5825–5832. [Google Scholar] [CrossRef]
- Xing, S.; Zhang, X.; Luo, L.; Cao, W.; Li, L.; He, Y.; An, J.; Gao, D. Doxorubicin/Gold Nanoparticles Coated with Liposomes for Chemo-Photothermal Synergetic Antitumor Therapy. Nanotechnology 2018, 29, 405101. [Google Scholar] [CrossRef]
- Laksee, S.; Puthong, S.; Kongkavitoon, P.; Palaga, T.; Muangsin, N. Facile and Green Synthesis of Pullulan Derivative-Stabilized Au Nanoparticles as Drug Carriers for Enhancing Anticancer Activity. Carbohydr. Polym. 2018, 198, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Loynachan, C.N.; Soleimany, A.P.; Dudani, J.S.; Lin, Y.; Najer, A.; Bekdemir, A.; Chen, Q.; Bhatia, S.N.; Stevens, M.M. Renal Clearable Catalytic Gold Nanoclusters for in Vivo Disease Monitoring. Nat. Nanotechnol. 2019, 14, 883–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, F.; Setyawati, M.I.; Tee, J.K.; Ding, X.; Wang, J.; Nga, M.E.; Ho, H.K.; Leong, D.T. Nanoparticles Promote in Vivo Breast Cancer Cell Intravasation and Extravasation by Inducing Endothelial Leakiness. Nat. Nanotechnol. 2019, 14, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.M.; Fouad, S.A.; Elshoky, H.A.; Mohammed, G.M.; Salaheldin, T.A. Antibacterial Effect of Gold Nanoparticles against Corynebacterium Pseudotuberculosis. Int. J. Vet. Sci. Med. 2017, 5, 23–29. [Google Scholar] [CrossRef] [Green Version]
- Lynn, G.M.; Sedlik, C.; Baharom, F.; Zhu, Y.; Ramirez-Valdez, R.A.; Coble, V.L.; Tobin, K.; Nichols, S.R.; Itzkowitz, Y.; Zaidi, N.; et al. Peptide–TLR-7/8a Conjugate Vaccines Chemically Programmed for Nanoparticle Self-Assembly Enhance CD8 T-Cell Immunity to Tumor Antigens. Nat. Biotechnol. 2020, 38, 320–332. [Google Scholar] [CrossRef]
- Shilo, M.; Berenstein, P.; Dreifuss, T.; Nash, Y.; Goldsmith, G.; Kazimirsky, G.; Motiei, M.; Frenkel, D.; Brodie, C.; Popovtzer, R. Insulin-Coated Gold Nanoparticles as a New Concept for Personalized and Adjustable Glucose Regulation. Nanoscale 2015, 7, 20489–20496. [Google Scholar] [CrossRef]
- Rosi, N.L. Oligonucleotide-Modified Gold Nanoparticles for Intracellular Gene Regulation. Science 2006, 312, 1027–1030. [Google Scholar] [CrossRef] [PubMed]
- Alaqad, K.; Saleh, T.A. Gold and Silver Nanoparticles: Synthesis Methods, Characterization Routes and Applications towards Drugs. J. Environ. Anal. Toxicol. 2016, 6, 525–2161. [Google Scholar] [CrossRef]
- Ogarev, V.A.; Rudoi, V.M.; Dement’eva, O.V. Gold Nanoparticles: Synthesis, Optical Properties, and Application. Inorg. Mater. Appl. Res. 2018, 9, 134–140. [Google Scholar] [CrossRef]
- Aslan, K.; Gryczynski, I.; Malicka, J.; Matveeva, E.; Lakowicz, J.; Geddes, C. Metal-enhanced fluorescence: An emerging tool in biotechnology. Curr. Opin. Biotech. 2005, 16, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Delcea, M.; Sternberg, N.; Yashchenok, A.M.; Georgieva, R.; Bäumler, H.; Möhwald, H.; Skirtach, A.G. Nanoplasmonics for Dual-Molecule Release through Nanopores in the Membrane of Red Blood Cells. ACS Nano 2012, 6, 4169–4180. [Google Scholar] [CrossRef]
- Hanley, T.; Vankayala, R.; Lee, C.; Tang, J.; Burns, J.; Anvari, B. Phototheranostics Using Erythrocyte-Based Particles. Biomology 2021, 11, 729. [Google Scholar] [CrossRef]
- Himbert, S.; Blacker, M.J.; Kihm, A.; Pauli, Q.; Khondker, A.; Yang, K.; Sinjari, S.; Johnson, M.; Juhasz, J.; Wagner, C.; et al. Hybrid Erythrocyte Liposomes: Functionalized Red Blood Cell Membranes for Molecule Encapsulation. Adv. Biosyst. 2020, 4, 1900185. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Ukidve, A.; Krishnan, V.; Fehnel, A.; Pan, D.C.; Gao, Y.; Kim, J.; Evans, M.A.; Mandal, A.; Guo, J.; et al. Systemic Tumour Suppression via the Preferential Accumulation of Erythrocyte-Anchored Chemokine-Encapsulating Nanoparticles in Lung Metastases. Nat. Biomed. Eng. 2020, 5, 441–454. [Google Scholar] [CrossRef] [PubMed]
- Rho, J.; Chung, J.; Im, H.; Liong, M.; Shao, H.; Castro, C.M.; Weissleder, R.; Lee, H. Magnetic Nanosensor for Detection and Profiling of Erythrocyte-Derived Microvesicles. ACS Nano 2013, 7, 11227–11233. [Google Scholar] [CrossRef] [Green Version]
- Koleva, L.; Bovt, E.; Ataullakhanov, F.; Sinauridze, E. Erythrocytes as Carriers: From Drug Delivery to Biosensors. Pharmaceutics 2020, 12, 276. [Google Scholar] [CrossRef] [Green Version]
- Hoffman, J.F. Biconcave Shape of Human Red-Blood-Cell Ghosts Relies on Density Differences between the Rim and Dimple of the Ghost’s Plasma Membrane. Proc. Natl. Acad. Sci. USA 2016, 113, 14847–14851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, F.M.; Smith, S.K.; Bell, J.D. Physical Properties of Erythrocyte Ghosts That Determine Susceptibility to Secretory Phospholipase A2. J. Biol. Chem. 2001, 276, 22722–22731. [Google Scholar] [CrossRef] [Green Version]
- Gupta, N.; Patel, B.; Ahsan, F. Nano-Engineered Erythrocyte Ghosts as Inhalational Carriers for Delivery of Fasudil: Preparation and Characterization. Pharm. Res. 2014, 31, 1553–1565. [Google Scholar] [CrossRef] [Green Version]
- Brenner, J.S.; Pan, D.C.; Myerson, J.W.; Marcos-Contreras, O.A.; Villa, C.H.; Patel, P.; Hekierski, H.; Chatterjee, S.; Tao, J.-Q.; Parhiz, H.; et al. Red Blood Cell-Hitchhiking Boosts Delivery of Nanocarriers to Chosen Organs by Orders of Magnitude. Nat. Commun. 2018, 9, 2684. [Google Scholar] [CrossRef]
- Hu, C.-M.J.; Zhang, L.; Aryal, S.; Cheung, C.; Fang, R.H.; Zhang, L. Erythrocyte Membrane-Camouflaged Polymeric Nanoparticles as a Biomimetic Delivery Platform. Proc. Natl. Acad. Sci. USA 2011, 108, 10980–10985. [Google Scholar] [CrossRef] [Green Version]
- Gray, M.; Woulfe, M. Striatal Blood-brain barrier permeability in Parkinson´s disease. J. Cereb. Blood Flow Metab. 2015, 35, 747–750. [Google Scholar] [CrossRef] [Green Version]
- Meléndez-Martínez, D.; Macias-Rodríguez, E.; Vargas-Caraveo, A.; Martínez-Martínez, A.; Gatica-Colima, A.; Plenge-Tellechea, L. Capillary damage in the area postrema by venom of the northern black-tailed rattlesnake (Crotalus molossus molossus). J. Venom. Res. 2014, 5, 1–5. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4102126/pdf/JVR-05-01.pdf (accessed on 5 July 2021).
- Chen, Y.; Liu, Z.; Gao, S.W.; Hu, C.-M.J.; Fang, R.H.; Luk, B.T.; Su, J.; Zhang, L. Surface Functionalization of Gold Nanoparticles with Red Blood Cell Membranes. Adv. Mater. 2013, 25, 3549–3553. [Google Scholar] [CrossRef] [Green Version]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A Study of the Nucleation and Growth Processes in the Synthesis of Colloidal Gold. Discuss. Faraday Soc. 1951, 11, 55. [Google Scholar] [CrossRef]
- Elfeky, A.S.; Al-Sherbini, A.-S. Synthesis and Spectral Characteristics of Gold Nanoparticles Labelled with Fluorescein isothiocyanate Sodium. Curr. Nanosci. 2011, 7, 1028–1033. [Google Scholar] [CrossRef]
- F04 Committee. Practice for Assessment of Hemolytic Properties of Materials; ASTM International: West Conshohocken PA, USA, 2013. [Google Scholar] [CrossRef]
- Grigoruţă, M.; Vargas-Caraveo, A.; Vázquez-Mayorga, E.; Castillo-Michel, H.A.; Díaz-Sánchez, Á.G.; Reyes-Herrera, J.; Martínez-Martínez, A. Blood Mononuclear Cells as Speculum of Emotional Stress Analyzed by Synchrotron Infrared Spectroscopy and a Nootropic Drug. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 204, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Niggli, V.; Adunyah, E.S.; Penniston, J.T.; Carafoli, E. Purified (Ca2+-Mg2+)-ATPase of the Erythrocyte Membrane. Reconstitution and Effect of Calmodulin and Phospholipids. J. Biol. Chem. 1981, 256, 395–401. [Google Scholar] [CrossRef]
- International Centre for Diffraction Data Database Search–ICDD. Available online: https://www.icdd.com (accessed on 5 July 2021).
- Shi, C.; Zhu, N.; Cao, Y.; Wu, P. Biosynthesis of Gold Nanoparticles Assisted by the Intracellular Protein Extract of Pycnoporus Sanguineus and Its Catalysis in Degradation of 4-Nitroaniline. Nanoscale Res. Lett. 2015, 10, 147. [Google Scholar] [CrossRef] [Green Version]
- Aswathy Aromal, S.; Dinesh Babu, K.V.; Philip, D. Characterization and Catalytic Activity of Gold Nanoparticles Synthesized Using Ayurvedic Arishtams. Spectrochim Acta A Mol. Biomol. Spectrosc. 2012, 96, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.K.; Lee, K.S.; El-Sayed, I.H.; El-Sayed, M.A. Calculated Absorption and Scattering Properties of Gold Nanoparticles of Different Size, Shape, and Composition: Applications in Biological Imaging and Biomedicine. J. Phys. Chem. B 2006, 110, 7238–7248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suchomel, P.; Kvitek, L.; Prucek, R.; Panacek, A.; Halder, A.; Vajda, S.; Zboril, R. Simple Size-Controlled Synthesis of Au Nanoparticles and Their Size-Dependent Catalytic Activity. Sci. Rep. 2018, 8, 4589. [Google Scholar] [CrossRef] [Green Version]
- Kapadia, C.H.; Melamed, J.R.; Day, E.S. Quantification of Gold Nanoparticles Using the NanoDrop One Microvolume UV-Vis Spectrophotometer; Thermo Fisher Scientific: Waltham, MA, USA, 2019. [Google Scholar]
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Determination of Size and Concentration of Gold Nanoparticles from UV/vis Spectra; University of Liverpool: Liverpool, UK, 2007. [Google Scholar]
- Olejnik, M.; Bujak, Ł.; Mackowski, S. Plasmonic Molecular Nanohybrids—Spectral Dependence of Fluorescence Quenching. Int. J. Mol. Sci. 2012, 13, 1018–1028. [Google Scholar] [CrossRef]
- Fratoddi, I.; Cartoni, A.; Venditti, I.; Catone, D.; O’Keeffe, P.; Paladini, A.; Toschi, F.; Turchini, S.; Sciubba, F.; Testa, G.; et al. Gold Nanoparticles Functionalized by Rhodamine B Isothiocyanate: A New Tool to Control Plasmonic Effects. J. Colloid Interface Sci. 2018, 513, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Pelaz, B.; Alexiou, C.; Alvarez-Puebla, R.A.; Alves, F.; Andrews, A.M.; Ashraf, S.; Balogh, L.P.; Ballerini, L.; Bestetti, A.; Brendel, C.; et al. Diverse Applications of Nanomedicine. ACS Nano 2017, 11, 2313–2381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luan, J.; Seth, A.; Gupta, R.; Wang, Z.; Rathi, P.; Cao, S.; Gholami Derami, H.; Tang, R.; Xu, B.; Achilefu, S.; et al. Ultrabright Fluorescent Nanoscale Labels for the Femtomolar Detection of Analytes with Standard Bioassays. Nat. Biomed. Eng. 2020, 4, 518–530. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-M.; Cheng, T.-L.; Tseng, W.-L. Fluorescence Turn-on Detection of Iodide, Iodate and Total Iodine Using Fluorescein isothiocyanate-5-Isothiocyanate-Modified Gold Nanoparticles. The Analyst 2009, 134, 2106. [Google Scholar] [CrossRef] [PubMed]
- Chekina, N.; Horak, D.; Jendelova, P.; Trchova, M.; Benes, M.; Hruby, M.; Herynek, V.; Turnovcov, K.; Sykov, E. Fluorescent magnetic nanoparticles for biomedical applications. J. Mater. Chem. 2011, 21, 7630. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, L.; Bae, S.C.; Granick, S. Nanoparticle-Induced Surface Reconstruction of Phospholipid Membranes. Proc. Natl. Acad. Sci. USA 2008, 105, 18171–18175. [Google Scholar] [CrossRef] [Green Version]
- Wißbrock, A.; Paul George, A.A.; Brewitz, H.H.; Kühl, T.; Imhof, D. The Molecular Basis of Transient Heme-Protein Interactions: Analysis, Concept and Implementation. Biosci. Rep. 2019, 39, BSR20181940. [Google Scholar] [CrossRef]
- Himbert, S.; Alsop, R.J.; Rose, M.; Hertz, L.; Dhaliwal, A.; Moran-Mirabal, J.M.; Verschoor, C.P.; Bowdish, D.M.E.; Kaestner, L.; Wagner, C.; et al. The Molecular Structure of Human Red Blood Cell Membranes from Highly Oriented, Solid Supported Multi-Lamellar Membranes. Sci. Rep. 2017, 7, 39661. [Google Scholar] [CrossRef] [Green Version]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Paoli, M.; Liddington, R.; Tame, J.; Wilkinson, A.; Dodson, G. Crystal Structure of T State Haemoglobin with Oxygen Bound At All Four Haems. J. Mol. Biol. 1996, 256, 775–792. [Google Scholar] [CrossRef] [PubMed]
- Oh, N.; Park, J.-H. Surface Chemistry of Gold Nanoparticles Mediates Their Exocytosis in Macrophages. ACS Nano 2014, 8, 6232–6241. [Google Scholar] [CrossRef]
- Kuhn, D.A.; Vanhecke, D.; Michen, B.; Blank, F.; Gehr, P.; Petri-Fink, A.; Rothen-Rutishauser, B. Different Endocytotic Uptake Mechanisms for Nanoparticles in Epithelial Cells and Macrophages. Beilstein J. Nanotechnol. 2014, 5, 1625–1636. [Google Scholar] [CrossRef] [Green Version]
- Nel, A.E.; Mädler, L.; Velegol, D.; Xia, T.; Hoek, E.M.V.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding Biophysicochemical Interactions at the Nano–Bio Interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, P.D.; Bracamonte, G. Fluorescencia Incrementada por el Metal y Plasmónica Exaltada en Base a Nanopartículas de Dimensiones Menores a las Longitudes de Onda del Espectro Electromagnético; Repositorio Institucional CONICET Digital: Córdoba, Argentina, 2019. [Google Scholar]
- Li, C.; Xie, Z.; Chen, Q.; Zhang, Y.; Chu, Y.; Guo, Q.; Zhou, W.; Zhang, Y.; Liu, P.; Chen, H.; et al. Supramolecular Hunter Stationed on Red Blood Cells for Detoxification Based on Specific Molecular Recognition. ACS Nano 2020, 14, 4950–4962. [Google Scholar] [CrossRef]
- Tang, H.; Ye, H.; Zhang, H.; Zheng, Y. Aggregation of Nanoparticles Regulated by Mechanical Properties of Nanoparticle–Membrane System. Nanotechnology 2018, 29, 405102. [Google Scholar] [CrossRef]
- Guo, J.; Agola, J.O.; Serda, R.; Franco, S.; Lei, Q.; Wang, L.; Minster, J.; Croissant, J.G.; Butler, K.S.; Zhu, W.; et al. Biomimetic Rebuilding of Multifunctional Red Blood Cells: Modular Design Using Functional Components. ACS Nano 2020, 14, 7847–7859. [Google Scholar] [CrossRef] [PubMed]
- Hamadani, C.M.; Goetz, M.J.; Mitragotri, S.; Tanner, E.E.L. Protein-Avoidant Ionic Liquid (PAIL)–Coated Nanoparticles to Increase Bloodstream Circulation and Drive Biodistribution. Sci. Adv. 2020, 6, eabd7563. [Google Scholar] [CrossRef] [PubMed]










Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez Flores, V.; Martínez-Martínez, A.; Roacho Pérez, J.A.; Acosta Bezada, J.; Aguirre-Tostado, F.S.; García Casillas, P.E. Biointeraction of Erythrocyte Ghost Membranes with Gold Nanoparticles Fluorescents. Materials 2021, 14, 6390. https://doi.org/10.3390/ma14216390
Gómez Flores V, Martínez-Martínez A, Roacho Pérez JA, Acosta Bezada J, Aguirre-Tostado FS, García Casillas PE. Biointeraction of Erythrocyte Ghost Membranes with Gold Nanoparticles Fluorescents. Materials. 2021; 14(21):6390. https://doi.org/10.3390/ma14216390
Chicago/Turabian StyleGómez Flores, Víctor, Alejandro Martínez-Martínez, Jorge A Roacho Pérez, Jazzely Acosta Bezada, Francisco S. Aguirre-Tostado, and Perla Elvia García Casillas. 2021. "Biointeraction of Erythrocyte Ghost Membranes with Gold Nanoparticles Fluorescents" Materials 14, no. 21: 6390. https://doi.org/10.3390/ma14216390
APA StyleGómez Flores, V., Martínez-Martínez, A., Roacho Pérez, J. A., Acosta Bezada, J., Aguirre-Tostado, F. S., & García Casillas, P. E. (2021). Biointeraction of Erythrocyte Ghost Membranes with Gold Nanoparticles Fluorescents. Materials, 14(21), 6390. https://doi.org/10.3390/ma14216390






