New Insights into the Application of 3D-Printing Technology in Hernia Repair
Abstract
:1. Introduction
2. General Features of Prosthetic Materials for Hernia Repair
3. Current Trends for Synthetic Meshes
4. Overview of 3D-Printing Technology for Biomedical Applications
5. Innovative 3D-Printed Meshes for Tissue Engineering Applications
| Device Developed | FDM Printing Parameters | Main Experimental Procedures | References |
|---|---|---|---|
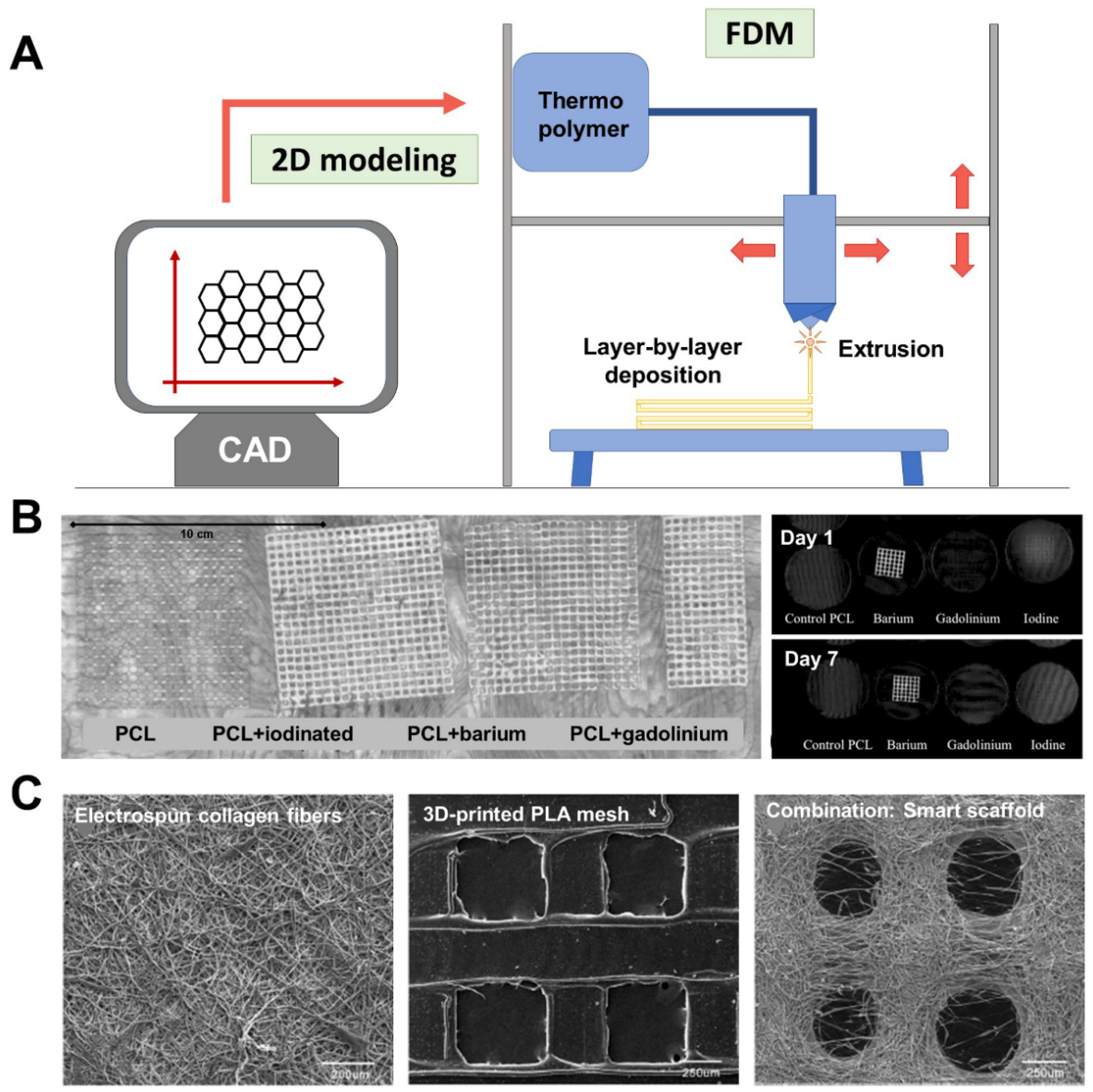
| PLA meshes containing contrast agents: iodine-based, gadolinium, or barium (1:10 w/w). | Extrusion temperature: 125–130 °C Speed: 7 mm/s Layer height: 0.2 mm Mesh pore size: NP | In vitro: CT imaging (120 kVp 120, 220 mAs). Stability of radio-opacity at body temperature: agar plate incubation (7 days, 37 °C). | [86] |
| Composite scaffolds made of printed PCL meshes and electrospun type-I collagen fibers. | Extrusion temperature: 190 °C Speed: 300 mm/min Layer height: 225 µm Mesh pore size: 800 µm Additional technology: electrospinning of type-I collagen (filament diameter 0.9 mm; flow 0.39 mL/min) alternating with PCL layers | In vitro: SEM visualization. Cytocompatibility (rat skin fibroblasts): proliferation studies (days 1, 3, 5, and 7 post-seeding). In vivo: Full-thickness abdominal wall defect repaired with scaffolds (2 × 3 cm) in male Sprague–Dawley rats (n = 54, three study groups). Clean surgery. Groups (3): PCL/PCL + electrospun collagen/Marketed collagen-based biomesh. Macroscopic, biomechanical and histopathological assessment at postoperative weeks 2, 4, and 14. | [87] |
| Inflammation modulating bioscaffold made of phosphate-crosslinked PVA. | Extrusion temperature: NP Speed: 10 mm/s Layer height: NP Mesh pore size: NP | In vitro: Mechanical testing. Crosslink reaction (3 phosphate concentrations: 15, 10, 7.5%). Swelling capacity over time (144 h). Material degradation. Cytocompatibility (human skin fibroblasts and microvascular endothelial cells): proliferation studies (72 h post-seeding). Trapping of proinflammatory cytokines (bead-based bioassay). In vivo: Ventral hernia defect (2 cm) repaired by the intraperitoneal implantation of scaffolds (2 × 2 cm) in male Sprague–Dawley rats (n = 36, three study groups). Additional implantation of bioscaffolds with modified surface charge (positive, neutral, negative; n = 6 each). Clean surgery. Groups (3): PLA bioscaffold/Marketed PP/Composite of marketed PP + PLA bioscaffold. Macroscopic, histopathological, and mRNA determination of cytokines at postoperative weeks 2 and 4. | [89] |
6. 3D-Printed Bioactive Meshes for Hernia Repair
7. Biomechanical Considerations for Developing 3D-Printed Meshes
8. Future Perspectives for 3D-Printed Meshes
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brown, S.H.; Ward, S.R.; Cook, M.S.; Lieber, R.L. Architectural analysis of human abdominal wall muscles: Implications for mechanical function. Spine 2011, 36, 355–362. [Google Scholar] [CrossRef] [Green Version]
- Hope, W.W.; Abdul, W.; Winters, R. Abdominal Wall Reconstruction; StatPearls Publishing (Internet): Treasure Island, FL, USA, 2021; Available online: https://www.ncbi.nlm.nih.gov/books/NBK431108/ (accessed on 27 September 2021).
- Ger, R. The clinical anatomy of the anterolateral abdominal wall musculature. Clin. Anat. 2009, 22, 392–397. [Google Scholar] [CrossRef]
- Patel, N.G.; Ratanshi, I.; Buchel, E.W. The best of abdominal wall reconstruction. Plast. Reconstr. Surg. 2018, 141, 113e–136e. [Google Scholar] [CrossRef]
- Deeken, C.R.; Lake, S.P. Mechanical properties of the abdominal wall and biomaterials utilized for hernia repair. J. Mech Behav. Biomed. Mater. 2017, 74, 411–427. [Google Scholar] [CrossRef]
- Brown, S.H.; McGill, S.M. A comparison of ultrasound and electromyography measures of force and activation to examine the mechanics of abdominal wall contraction. Clin. Biomech. 2010, 25, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Ozdogan, M.; Yildiz, F.; Gurer, A.; Orhun, S.; Kulacoglu, H.; Aydin, R. Changes in collagen and elastic fiber contents of the skin, rectus sheath, transversalis fascia and peritoneum in primary inguinal hernia patients. Bratisl. Lek. Listy 2006, 107, 235–238. [Google Scholar] [PubMed]
- Hernández-Gascón, B.; Mena, A.; Peña, E.; Pascual, G.; Bellón, J.M.; Calvo, B. Understanding the passive mechanical behavior of the human abdominal wall. Ann. Biomed. Eng. 2013, 41, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Förstemann, T.; Trzewik, J.; Holste, J.; Batke, B.; Konerding, M.A.; Wolloscheck, T.; Hartung, C. Forces and deformations of the abdominal wall—A mechanical and geometrical approach to the linea alba. J. Biomech. 2011, 44, 600–666. [Google Scholar] [CrossRef]
- Song, C.; Alijani, A.; Frank, T.; Hanna, G.; Cuschieri, A. Elasticity of the living abdominal wall in laparoscopic surgery. J. Biomech. 2006, 39, 587–591. [Google Scholar] [CrossRef]
- Kalaba, S.; Gerhard, E.; Winder, J.S.; Pauli, E.M.; Haluck, R.S.; Yang, J. Design strategies and applications of biomaterials and devices for hernia repair. Bioact. Mater. 2016, 1, 2–17. [Google Scholar] [CrossRef] [Green Version]
- Muysoms, F.E.; Miserez, M.; Berrevoet, F.; Campanelli, G.; Champault, G.G.; Chelala, E.; Dietz, U.A.; Eker, H.H.; El Nakadi, I.; Hauters, P.; et al. Classification of primary and incisional abdominal wall hernias. Hernia 2009, 13, 407–414. [Google Scholar] [CrossRef] [Green Version]
- Bellón, J.M.; Bajo, A.; García-Honduvilla, N.; Gimeno, M.J.; Pascual, G.; Guerrero, A.; Buján, J. Fibroblasts from the transversalis fascia of young patients with direct inguinal hernias show constitutive MMP-2 overexpression. Ann. Surg. 2001, 233, 287–291. [Google Scholar] [CrossRef]
- Franz, M.G. The biology of hernias and the abdominal wall. Hernia 2006, 10, 462–471. [Google Scholar] [CrossRef]
- Pascual, G.; Rodríguez, M.; Gómez-Gil, V.; Trejo, C.; Buján, J.; Bellón, J.M. Active matrix metalloproteinase-2 upregulation in the abdominal skin of patients with direct inguinal hernia. Eur. J. Clin. Investig. 2010, 40, 1113–1121. [Google Scholar] [CrossRef]
- Burcharth, J.; Pommergaard, H.C.; Rosenberg, J. The inheritance of groin hernia: A systematic review. Hernia 2013, 17, 183–189. [Google Scholar] [CrossRef]
- Öberg, S.; Andresen, K.; Rosenberg, J. Etiology of inguinal hernias: A comprehensive review. Front. Surg. 2017, 4, 52. [Google Scholar] [CrossRef] [Green Version]
- Thankam, F.G.; Larsen, N.K.; Varghese, A.; Bui, T.N.; Reilly, M.; Fitzgibbons, R.J.; Agrawal, D.K. Biomarkers and heterogeneous fibroblast phenotype associated with incisional hernia. Mol. Cell Biochem. 2021, 476, 3353–3363. [Google Scholar] [CrossRef] [PubMed]
- Kingsnorth, A.; LeBlanc, K. Hernias: Inguinal and incisional. Lancet 2003, 362, 1561–1571. [Google Scholar] [CrossRef]
- HerniaSurge Group. International guidelines for groin hernia management. Hernia 2018, 22, 1–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandey, S.; Sumant, O. Hernia Repair Devices and Consumables Market by Product (Fixation Devices and Consumables), Surgery Type (Open Tension-Free Repair Surgery and Laparoscopic Surgery), and Hernia Type (Incisional Hernia, Umbilical, Inguinal Hernia, and Femoral Hernia): Global Opportunity Analysis and Industry Forecast, 2020–2027; Allied Market Research: Portland, OR, USA, 2020; Available online: https://www.alliedmarketresearch.com/hernia-repair-devices-market (accessed on 15 September 2021).
- Rastegarpour, A.; Cheung, M.; Vardhan, M.; Ibrahim, M.M.; Butler, C.E.; Levinson, H. Surgical mesh for ventral incisional hernia repairs: Understanding mesh design. Plast. Surg. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Luijendijk, R.W.; Hop, W.C.; van den Tol, M.P.; de Lange, D.C.; Braaksma, M.M.; IJzermans, J.N.; Boelhouwer, R.U.; de Vries, B.C.; Salu, M.K.; Wereldsma, J.C.; et al. A comparison of suture repair with mesh repair for incisional hernia. N. Engl. J. Med. 2000, 343, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Bilsel, Y.; Abci, I. The search for ideal hernia repair; mesh materials and types. Int. J. Surg. 2012, 10, 317–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bellón, J.M. Classification of prosthetic materials used in hernia repair according to their structure and behavior with regard to tissue integration. Rev. Hisp. Hernia 2014, 2, 49–57. [Google Scholar] [CrossRef] [Green Version]
- Cevasco, M.; Itani, K.M. Ventral hernia repair with synthetic, composite, and biologic mesh: Characteristics, indications, and infection profile. Surg. Infect. 2012, 13, 209–215. [Google Scholar] [CrossRef]
- Le, D.; Deveney, C.W.; Reaven, N.L.; Funk, S.E.; McGaughey, K.J.; Martindale, R.G. Mesh choice in ventral hernia repair: So many choices, so little time. Am. J. Surg. 2013, 205, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Franklin, M.E., Jr.; Gonzalez, J.J., Jr.; Michaelson, R.P.; Glass, J.L.; Chock, D.A. Preliminary experience with new bioactive prosthetic material for repair of hernias in infected fields. Hernia 2002, 6, 171–174. [Google Scholar] [CrossRef]
- Miserez, M.; Jairam, A.P.; Boersema, G.S.A.; Bayon, Y.; Jeekel, J.; Lange, J.F. Resorbable synthetic meshes for abdominal wall defects in preclinical setting: A literature review. J. Surg Res. 2019, 237, 67–75. [Google Scholar] [CrossRef]
- Mori da Cunha, M.G.M.C.; Hympanova, L.; Rynkevic, R.; Mes, T.; Bosman, A.W.; Deprest, J. Biomechanical behaviour and biocompatibility of ureidopyrimidinone-polycarbonate electrospun and polypropylene meshes in a hernia repair in rabbits. Materials 2019, 12, 1174. [Google Scholar] [CrossRef] [Green Version]
- Sanders, D.L.; Kingsnorth, A.N. Prosthetic mesh materials used in hernia surgery. Expert Rev. Med. Devices 2012, 9, 159–179. [Google Scholar] [CrossRef]
- Harris, H.W.; Primus, F.; Young, C.; Carter, J.T.; Lin, M.; Mukhtar, R.A.; Yeh, B.; Allen, E.G.; Freise, C.; Kim, E.; et al. Preventing recurrence in clean and contaminated hernias using biologic versus synthetic mesh in ventral hernia repair: The PRICE randomized clinical trial. Ann. Surg. 2021, 273, 648–655. [Google Scholar] [CrossRef]
- Shankaran, V.; Weber, D.J.; Reed, R.L.; Luchette, F.A. A review of available prosthetics for ventral hernia repair. Ann. Surg. 2011, 253, 16–26. [Google Scholar] [CrossRef]
- FitzGerald, J.F.; Kumar, A.S. Biologic versus synthetic mesh reinforcement: What are the pros and cons? Clin. Colon Rectal Surg. 2014, 27, 140–148. [Google Scholar] [PubMed] [Green Version]
- Patel, H.; Ostergard, D.R.; Sternschuss, G. Polypropylene mesh and the host response. Int. Urogynecol. J. 2012, 23, 669–679. [Google Scholar] [CrossRef]
- Totten, C.; Becker, P.; Lourd, M.; Roth, J.S. Polyester vs polypropylene, do mesh materials matter? A meta-analysis and systematic review. Med. Devices 2019, 12, 369–378. [Google Scholar] [CrossRef] [Green Version]
- Alarcón, I.; Balla, A.; Soler-Frías, J.R.; Barranco, A.; Bellido-Luque, J.; Morales-Conde, S. Polytetrafluoroethylene versus polypropylene mesh during laparoscopic totally extraperitoneal (TEP) repair of inguinal hernia: Short- and long-term results of a double-blind clinical randomized controlled trial. Hernia 2020, 24, 1011–1018. [Google Scholar] [CrossRef]
- Verbo, A.; Pafundi, P.; Manno, A.; Baccaro, R.; Veneziani, A.; Colli, R.; Coco, C. Polyvinylidene fluoride mesh (PVDF, DynaMesh®-IPOM) in the laparoscopic treatment of incisional hernia: A prospective comparative trial versus Gore® ePTFE DUALMESH® Plus. Surg. Technol. Int. 2016, 28, 147–151. [Google Scholar] [PubMed]
- Lambertz, A.; van den Hil, L.C.L.; Schöb, D.S.; Binnebösel, M.; Kroh, A.; Klinge, U.; Neumann, U.P.; Klink, C.D. Analysis of adhesion formation of a new elastic thermoplastic polyurethane (TPU) mesh in comparison to polypropylene (PP) meshes in IPOM position. J. Mech. Behav. Biomed. Mater. 2016, 53, 366–372. [Google Scholar] [CrossRef]
- Levy, A.S.; Bernstein, J.L.; Premaratne, I.D.; Rohde, C.H.; Otterburn, D.M.; Morrison, K.A.; Lieberman, M.; Pomp, A.; Spector, J.A. Poly-4-hydroxybutyrate (Phasix™) mesh onlay in complex abdominal wall repair. Surg. Endosc. 2021, 35, 2049–2058. [Google Scholar] [CrossRef] [PubMed]
- Renard, Y.; de Mestier, L.; Henriques, J.; de Boissieu, P.; de Mestier, P.; Fingerhut, A.; Palot, J.P.; Kianmanesh, R. Absorbable polyglactin vs. non-cross-linked porcine biological mesh for the surgical treatment of infected incisional hernia. J. Gastrointest. Surg. 2020, 24, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Han, X.; Wei, B.; Huang, J.; Yang, X.; Tang, X.; Zheng, Z.; Luo, L.; Zhan, Z.; Wei, H. Ventral hernia repair in rat using nanofibrous polylactic acid/polypropylene meshes. Nanomedicine 2018, 13, 2187–2199. [Google Scholar]
- Fatkhudinov, T.; Tsedik, L.; Arutyunyan, I.; Lokhonina, A.; Makarov, A.; Korshunov, A.; Elchaninov, A.; Kananykhina, E.; Vasyukova, O.; Usman, N.; et al. Evaluation of resorbable polydioxanone and polyglycolic acid meshes in a rat model of ventral hernia repair. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Borman, D.A.; Sunshein, K.E.; Stigall, K.S.; Madabhushi, V.V.; Davenport, D.L.; Plymale, M.A.; Roth, J.S. Clinical and quality of life assessment of patients undergoing laparoscopic hiatal hernia repair. Am. Surg. 2019, 85, 1269–1275. [Google Scholar] [CrossRef]
- Sezer, U.A.; Sanko, V.; Gulmez, M.; Aru, B.; Sayman, E.; Aktekin, A.; Vardar Aker, F.; Demirel, G.Y.; Sezer, S. Polypropylene composite hernia mesh with anti-adhesion layer composed of polycaprolactone and oxidized regenerated cellulose. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 1141–1152. [Google Scholar] [CrossRef] [PubMed]
- Shokrollahi, M.; Bahrami, S.H.; Nazarpak, M.H.; Solouk, A. Biomimetic double-sided polypropylene mesh modified by DOPA and ofloxacin loaded carboxyethyl chitosan/polyvinyl alcohol-polycaprolactone nanofibers for potential hernia repair applications. Int. J. Biol. Macromol. 2020, 165, 902–917. [Google Scholar] [CrossRef]
- Usher, F.C. Further observations the use of Marlex mesh: A new technique for the repair of inguinal hernias. Am. J. Surg. 1959, 25, 792–795. [Google Scholar]
- Usher, F.C.; Cogan, J.E.; Lowry, T. A new technique for the repair of inguinal and incisional hernias. Arch. Surg. 1960, 81, 847–850. [Google Scholar] [CrossRef] [PubMed]
- Antonopoulos, I.M.; Nhas, W.C.; Mazzuchi, E.; Piovesan, A.C.; Birolini, C.; Lucon, A.M. Is polypropylene mesh safe and effective for repairing infected incisional hernia in renal transplant recipients? Urology 2005, 66, 874–877. [Google Scholar] [CrossRef]
- Alaedeen, D.I.; Lipman, J.; Medalie, D.; Rosen, M.J. The single-staged approach to the surgical management of abdominal wall hernias in contaminated fields. Hernia 2007, 11, 41–45. [Google Scholar] [CrossRef]
- Rodríguez, M.; Gómez-Gil, V.; Pérez-Köhler, B.; Pascual, G.; Bellón, J.M. Polymer hernia repair materials: Adapting to patient needs and surgical techniques. Materials 2021, 14, 2790. [Google Scholar] [CrossRef]
- Manzini, G.; Henne-Bruns, D.; Kremer, M. Severe complications after mesh migration following abdominal hernial repair: Report of two cases and review of literature. GMS Interdiscip. Plast. Reconstr. Surg. DGPW 2019, 8, Doc09. [Google Scholar]
- Schmidt, A.; Taylor, D. Erosion of soft tissue by polypropylene mesh products. J. Mech. Behav. Biomed. Mater. 2021, 115, 104281. [Google Scholar] [CrossRef]
- Nienhuijs, S.; Staal, E.; Strobbe, L.; Rosman, C.; Groenewoud, H.; Bleichrodt, R. Chronic pain after mesh repair of inguinal hernia: A systematic review. Am. J. Surg. 2007, 194, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Bittner, R.; Schmedt, C.G.; Leibl, B.J.; Schwarz, J. Early postoperative and one year results of a randomized controlled trial comparing the impact of extralight titanized polypropylene mesh and traditional heavyweight polypropylene mesh on pain and seroma production in laparoscopic hernia repair (TAPP). World J. Surg. 2011, 35, 1791–1797. [Google Scholar] [CrossRef]
- Felemovicius, I.; Bonsack, M.E.; Hagerman, G.; Delaney, J.P. Prevention of adhesions to polypropylene mesh. J. Am. Coll. Surg. 2004, 198, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Di Muria, A.; Formisano, V.; Di Carlo, F.; Aveta, A.; Giglio, D. Small bowel obstruction by mesh migration after umbilical hernia repair. Ann. Ital. Chir. 2007, 78, 59–60. [Google Scholar] [PubMed]
- Basoglu, M.; Yildirgan, M.I.; Yilmaz, I.; Balik, A.; Celebi, F.; Atamanalp, S.S.; Polat, K.Y.; Oren, D. Late complications of incisional hernias following prosthetic mesh repair. Acta Chir. Belg. 2004, 104, 425–428. [Google Scholar] [CrossRef]
- Carbonell, A.M.; Kercher, K.W.; Sin, R.F.; Heniford, B.T. Susceptibility of prosthetic biomaterials to infection. Surg. Endosc. 2005, 19, 1670. [Google Scholar] [CrossRef]
- Russo Serafini, M.; Medeiros Savi, F.; Ren, J.; Bas, O.; O’Rourke, N.; Maher, C.; Hutmacher, D.W. The patenting and technological trends in hernia mesh implants. Tissue Eng. Part B Rev. 2021, 27, 48–73. [Google Scholar] [CrossRef]
- Wales, E.; Holloway, S. The use of prosthetic mesh for abdominal wall repairs: A semi-systematic-literature review. Int. Wound J. 2019, 16, 30–40. [Google Scholar] [CrossRef] [Green Version]
- Liaw, C.Y.; Guvendiren, M. Current and emerging applications of 3D printing in medicine. Biofabrication 2017, 9, 024102. [Google Scholar] [CrossRef]
- Ravia, T.; Ranganathanb, R.; Pugalendhic, A.; Arumugamd, S. 3D printed patient specific models from medical imaging—A general workflow. Mater. Today Proc. 2020, 22, 1237–1243. [Google Scholar] [CrossRef]
- Mitsouras, D.; Liacouras, P.; Imanzadeh, A.; Giannopoulos, A.A.; Cai, T.; Kumamaru, K.K.; George, E.; Wake, N.; Caterson, E.J.; Pomahac, B.; et al. Medical 3D printing for the radiologist. Radiographics 2015, 35, 1965–1988. [Google Scholar] [CrossRef] [PubMed]
- Liacourras, P.C.; Sahajwalla, D.; Beachler, M.D.; Sleeman, T.; Ho, V.B.; Lichtenberger, J.P., 3rd. Using computed tomography and 3D printing to construct custom prosthetics attachments and devices. 3D Print. Med. 2017, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Hull, C.W. The birth of 3D printing. Res. Technol. Manag. 2015, 58, 25–30. [Google Scholar]
- Jain, A.; Bansal, K.K.; Tiwari, A.; Rosling, A.; Rosenholm, J.M. Role of polymers in 3D printing technology for drug delivery—An overview. Curr. Pharm. Des. 2018, 24, 4979–4990. [Google Scholar] [CrossRef] [PubMed]
- Pravin, S.; Sudhir, A. Integration of 3D printing with dosage forms: A new perspective for modern healthcare. Biomed. Pharm. 2018, 107, 146–154. [Google Scholar] [CrossRef]
- Calignano, F.; Galati, M.; Iuliano, L.; Minetola, P. Design of additively manufactured structures for biomedical applications: A review of the additive manufacturing processes applied to the biomedical sector. J. Healthc. Eng. 2019, 2019, 9748212. [Google Scholar] [CrossRef]
- Beg, S.; Almalki, W.H.; Malik, A.; Farhan, M.; Aatif, M.; Rahman, Z.; Alruwaili, N.K.; Alrobaian, M.; Tarique, M.; Rahman, M. 3D printing for drug delivery and biomedical applications. Drug Discov. Today 2020, 25, 1668–1681. [Google Scholar] [CrossRef]
- Hodgdon, T.; Danrad, R.; Patel, M.J.; Smith, S.E.; Richardson, M.L.; Ballard, D.H.; Ali, S.; Trace, A.P.; DeBenedectis, C.A.; Zygmont, M.E.; et al. Logistics of Three-dimensional Printing: Primer for Radiologists. Acad. Radiol. 2018, 25, 40–51. [Google Scholar] [CrossRef]
- Pantermehl, S.; Emmert, S.; Foth, A.; Grabow, N.; Alkildani, S.; Bader, R.; Barbeck, M.; Jung, O. 3D printing for soft tissue regeneration and applications in medicine. Biomedicines 2021, 9, 336. [Google Scholar] [CrossRef]
- Ventola, C.L. Medical applications for 3D printing: Current and projected uses. Pharm. Ther. 2014, 39, 704–711. [Google Scholar]
- Ballard, D.H.; Trace, A.P.; Ali, S.; Hodgdon, T.; Zygmont, M.E.; DeBenedectis, C.A.; Smith, S.E.; Richardson, M.L.; Patel, M.J.; Decker, S.J.; et al. Clinical applications of 3D printing: Primer for radiologists. Acad. Radiol. 2018, 25, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Dutra, G.V.S.; Neto, W.S.; Dutra, J.P.S.; Machado, F. Implantable medical devices and tissue engineering: An overview of manufacturing processes and the use of polymeric matrices for manufacturing and coating their surfaces. Curr. Med. Chem. 2020, 27, 1580–1599. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.; Shukla, R.; Yadav, A.; Ujjwal, R.R.; Flora, S.J.S. 3D printing in development of nanomedicines. Nanomaterials 2021, 11, 420. [Google Scholar] [CrossRef]
- Awad, A.; Fina, F.; Goyanes, A.; Gaisford, S.; Basit, A.W. Advances in powder bed fusion 3D printing in drug delivery and healthcare. Adv. Drug Deliv. Rev. 2021, 174, 406–424. [Google Scholar] [CrossRef]
- Guvendiren, M.; Molde, J.; Soares, R.M.; Kohn, J. Designing Biomaterials for 3D Printing. ACS Biomater. Sci. Eng. 2016, 2, 1679–1693. [Google Scholar] [CrossRef]
- Sta Agueda, J.R.H.; Chen, Q.; Maalihan, R.D.; Ren, J.; da Silva, I.G.M.; Dugos, N.P.; Caldona, E.B.; Advincula, R.C. 3D printing of biomedically relevant polymer materials and biocompatibility. MRS Commun. 2021, 26, 1–16. [Google Scholar] [CrossRef]
- Casanellas, I.; García-Lizarribar, A.; Lagunas, A.; Samitier, J. Producing 3D biomimetic nanomaterials for musculoskeletal system regeneration. Front. Bioeng. Biotechnol. 2018, 6, 128. [Google Scholar] [CrossRef]
- Derakhshanfar, S.; Mbeleck, R.; Xu, K.; Zhang, X.; Zhong, W.; Xing, M. 3D bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact. Mater. 2018, 3, 144–156. [Google Scholar] [CrossRef]
- Zhang, J.; Wehrle, E.; Rubert, M.; Müller, R. 3D Bioprinting of human tissues: Biofabrication, bioinks, and bioreactors. Int. J. Mol. Sci. 2021, 22, 3971. [Google Scholar] [CrossRef]
- Di Prima, M.; Coburn, J.; Hwang, D.; Kelly, J.; Khairuzzaman, A.; Ricles, L. Additively manufactured medical products—The FDA perspective. 3D Print. Med. 2016, 2, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirza, M.A.; Iqbal, Z. 3D printing in pharmaceuticals: Regulatory perspective. Curr. Pharm. Des. 2018, 24, 5081–5083. [Google Scholar] [CrossRef] [PubMed]
- Eisenstein, M. First 3D-printed pill. Nat. Biotechnol. 2015, 33, 1014. [Google Scholar]
- Ballard, D.H.; Jammalamadaka, U.; Tappa, K.; Weisman, J.A.; Boyer, C.J.; Alexander, J.S.; Woodard, P.K. 3D printing of surgical hernia meshes impregnated with contrast agents: In vitro proof of concept with imaging characteristics on computed tomography. 3D Print. Med. 2018, 4, 13. [Google Scholar] [CrossRef]
- Yang, Z.; Song, Z.; Nie, X.; Guo, K.; Gu, Y. A smart scaffold composed of three-dimensional printing and electrospinning techniques and its application in rat abdominal wall defects. Stem. Cell Res. 2020, 11, 533. [Google Scholar] [CrossRef]
- Pensa, N.W.; Curry, A.S.; Bonvallet, P.P.; Bellis, N.F.; Rettig, K.M.; Reddy, M.S.; Eberhardt, A.W.; Bellis, S.L. 3D printed mesh reinforcements enhance the mechanical properties of electrospun scaffolds. Biomater. Res. 2019, 23, 22. [Google Scholar] [CrossRef]
- Shin, C.S.; Cabrera, F.J.; Lee, R.; Kim, J.; Veettil, R.A.; Zaheer, M.; Adumbumkulath, A.; Mhatre, K.; Ajayan, P.M.; Curley, S.A.; et al. 3D-bioprinted inflammation modulating polymer scaffolds for soft tissue repair. Adv. Mater. 2021, 33, e2003778. [Google Scholar] [CrossRef]
- Chung, J.J.; Im, H.; Kim, S.H.; Park, J.W.; Jung, Y. Toward biomimetic scaffolds for tissue engineering: 3D printing techniques in regenerative medicine. Front. Bioeng. Biotechnol. 2020, 8, 586406. [Google Scholar] [CrossRef]
- Mir, M.; Ansari, U.; Najabat Ali, M. Macro-scale model study of a tunable drug dispensation mechanism for controlled drug delivery in potential wound-healing applications. J. Appl. Biomater. Funct. Mater. 2017, 15, e63–e69. [Google Scholar] [CrossRef] [Green Version]
- Do, A.V.; Worthington, K.S.; Tucker, B.A.; Salem, A.K. Controlled drug delivery from 3D printed two-photon polymerized poly(ethylene glycol) dimethacrylate devices. Int. J. Pharm. 2018, 552, 217–224. [Google Scholar] [CrossRef]
- Ballard, D.H.; Tappa, K.; Boyer, C.J.; Jammalamadaka, U.; Hemmanur, K.; Weisman, J.A.; Alexander, J.S.; Mills, D.K.; Woodard, P.K. Antibiotics in 3D-printed implants, instruments and materials: Benefits, challenges and future directions. J. 3D Print. Med. 2019, 3, 83–93. [Google Scholar] [CrossRef]
- Weisman, J.A.; Nicholson, J.C.; Tappa, K.; Jammalamadaka, U.; Wilson, C.G.; Mills, D.K. Antibiotic and chemotherapeutic enhanced three-dimensional printer filaments and constructs for biomedical applications. Int. J. Nanomed. 2015, 10, 357–370. [Google Scholar]
- Huang, Y.; Zhang, X.F.; Gao, G.; Yonezawa, T.; Cui, X. 3D bioprinting and the current applications in tissue engineering. Biotechnol. J. 2017, 12, 1600734. [Google Scholar] [CrossRef] [PubMed]
- Tappa, K.; Jammalamadaka, U.; Ballard, D.H.; Bruno, T.; Israel, M.R.; Vemula, H.; Meacham, J.M.; Mills, D.K.; Woodard, P.K.; Weisman, J.A. Medication eluting devices for the field of OBGYN (MEDOBGYN): 3D printed biodegradable hormone eluting constructs, a proof of concept study. PLoS ONE 2017, 12, e0182929. [Google Scholar] [CrossRef]
- Farmer, Z.L.; Utomo, E.; Domínguez-Robles, J.; Mancinelli, C.; Mathew, E.; Larrañeta, E.; Lamprou, D.A. 3D printed estradiol-eluting urogynecological mesh implants: Influence of material and mesh geometry on their mechanical properties. Int. J. Pharm. 2021, 593, 120145. [Google Scholar] [CrossRef] [PubMed]
- Koons, G.L.; Mikos, A.G. Progress in three-dimensional printing with growth factors. J. Control. Release 2019, 295, 50–59. [Google Scholar] [CrossRef]
- Ballard, D.H.; Weisman, J.A.; Jammalamadaka, U.; Tappa, K.; Alexander, J.S.; Griffen, F.D. Three-dimensional printing of bioactive hernia meshes: In vitro proof of principle. Surgery 2017, 161, 1479–1481. [Google Scholar] [CrossRef]
- Calero Castro, F.J.; Yuste, Y.; Pereira, S.; Garvín, M.D.; López García, M.A.; Padillo, F.J.; de la Portilla, F. Proof of concept, design, and manufacture via 3-D printing of a mesh with bactericidal capacity: Behaviour in vitro and in vivo. J. Tissue Eng. Regen. Med. 2019, 13, 1955–1964. [Google Scholar] [CrossRef]
- Qamar, N.; Abbas, N.; Irfan, M.; Hussain, A.; Arshad, M.S.; Latif, S.; Mehmood, F.; Ghori, M.U. Personalized 3D printed ciprofloxacin impregnated meshes for the management of hernia. J. Drug Deliv. Sci. Technol. 2019, 53, 101164. [Google Scholar] [CrossRef]
- Bose, S.; Robertson, S.F.; Bandyopadhyay, A. Surface modification of biomaterials and biomedical devices using additive manufacturing. Acta Biomater. 2018, 66, 6–22. [Google Scholar] [CrossRef]
- Durfee, W.K.; Iaizzo, P.A. Medical applications of 3D printing. In Engineering in Medicine. Advances and Challenges; Iaizzo, P.A., Ed.; Elsevier Inc.: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 2019; pp. 527–543. [Google Scholar]
- Anurov, M.V.; Titkova, S.M.; Oettinger, A.P. Biomechanical compatibility of surgical mesh and fascia being reinforced: Dependence of experimental hernia defect repair results on anisotropic surgical mesh positioning. Hernia 2012, 16, 199–210. [Google Scholar] [CrossRef]
- Astruc, L.; De Meulaere, M.; Witz, J.F.; Novácek, V.; Turquier, F.; Hoc, T.; Brieu, M. Characterization of the anisotropic mechanical behavior of human abdominal wall connective tissues. J. Mech. Behav. Biomed. Mater. 2018, 82, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Gascón, B.; Peña, E.; Pascual, G.; Rodríguez, M.; Bellón, J.M.; Calvo, B. Long-term anisotropic mechanical response of surgical meshes used to repair abdominal wall defects. J. Mech. Behav. Biomed. Mater. 2012, 5, 257–271. [Google Scholar] [CrossRef] [PubMed]
- Lambertz, A.; Vogels, R.R.; Binnebösel, M.; Schöb, D.S.; Kossel, K.; Klinge, U.; Neumann, U.P.; Klink, C.D. Elastic mesh with thermoplastic polyurethane filaments preserves effective porosity of textile implants. J. Biomed. Mater. Res. Part A 2015, 103, 2654–2660. [Google Scholar] [CrossRef]
- Pott, P.P.; Schwarz, M.L.; Gundling, R.; Nowak, K.; Hohenberger, P.; Roessner, E.D. Mechanical properties of mesh materials used for hernia repair and soft tissue augmentation. PLoS ONE 2012, 7, e46978. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Gascón, B.; Espés, N.; Peña, E.; Pascual, G.; Bellón, J.M.; Calvo, B. Computational framework to model and design surgical meshes for hernia repair. Comput. Methods Biomech. Biomed. Eng. 2014, 17, 1071–1085. [Google Scholar] [CrossRef] [PubMed]
- Cobb, W.S.; Peindl, R.M.; Zerey, M.; Carbonell, A.M.; Heniford, B.T. Mesh terminology 101. Hernia 2009, 13, 1–6. [Google Scholar] [CrossRef]
- Cobb, W.S.; Burns, J.M.; Peindl, R.D.; Carbonell, A.M.; Matthews, B.D.; Kercher, K.W.; Heniford, B.T. Textile analysis of heavy weight, mid-weight, and light weight polypropylene mesh in a porcine ventral hernia model. J. Surg. Res. 2006, 136, 1–7. [Google Scholar] [CrossRef]
- Bellón, J.M.; Moreno-Egea, A. Clinical guide for the use of prosthetic material in the repair of incisional hernia. Rev. Hisp. Hernia 2021, 9, 137–142. [Google Scholar]
- Bellón, J.M.; Rodríguez, M.; García-Honduvilla, N.; Gómez-Gil, V.; Pascual, G.; Buján, J. Comparing the behavior of different polypropylene meshes (heavy and lightweight) in an experimental model of ventral hernia repair. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 448–455. [Google Scholar] [CrossRef]
- Tanikella, N.G.; Wittbrodt, B.; Pearce, J.M. Tensile strength of commercial polymer materials for fused filament fabrication 3-D printing. Addit. Manuf. 2017, 15, 40–47. [Google Scholar] [CrossRef] [Green Version]
- Mellert, L.T.; Cheung, M.E.; Zografakis, J.G.; Dan, A.G. Laparoscopic inguinal hernia repair using progrip self-fixating mesh: Technical learning curve and mid-term outcomes. Surg. Technol. Int. 2019, 34, 235–240. [Google Scholar]
- Thölix, A.M.; Kössi, J.; Remes, V.; Scheinin, T.; Harju, J. Lower incidence of postoperative pain after open inguinal hernia surgery with the usage of synthetic glue-coated mesh (Adhesix®). Am. Surg. 2018, 84, 1932–1937. [Google Scholar] [CrossRef] [PubMed]
- Turza, K.C.; Butler, C.E. Adhesions and meshes: Synthetic versus bioprosthetic. Plast. Reconstr. Surg. 2012, 130, 206S–213S. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Kim, T.G.; Jeong, J.; Yi, H.G.; Park, J.W.; Hwang, W.; Cho, D.W. 3D cell printing of functional skeletal muscle constructs using skeletal muscle-derived bioink. Adv. Healthc. Mater. 2016, 5, 2636–2645. [Google Scholar] [CrossRef]
- Lui, Y.S.; Sow, W.T.; Tan, L.P.; Wu, Y.; Lai, Y.; Li, H. 4D printing and stimuli-responsive materials in biomedical aspects. Acta Biomater. 2019, 92, 19–36. [Google Scholar] [CrossRef] [PubMed]

| Device Developed | FDM Printing Parameters | Main Experimental Procedures | References |
|---|---|---|---|
| PLA filaments containing drugs: gentamicin or methotrexate (two concentrations: 1% and 2.5% w/w). | Extrusion temperature: 175 °C for gentamicin-loaded filaments; 160 °C for methotrexate-loaded filaments Speed: NP Layer height: NP | In vitro: SEM visualization. Thermal stability testing of drugs (220 °C and 190 °C for gentamicin and methotrexate, respectively). Antibacterial activity of gentamicin-loaded filaments (Escherichia coli): agar diffusion and liquid nutrient tests (24 h, 37 °C). Cytocompatibility of methotrexate-loaded filaments (CRL2836 osteosarcoma cells). | [94] |
| PLA meshes and filaments containing 1% (w/w) gentamicin. | Extrusion temperature: 175 °C Speed: NP Layer height: NP Mesh pore size: NP | In vitro: SEM visualization. Antibacterial activity (Escherichia coli; Staphylococcus aureus): agar diffusion tests (24 h, 37 °C). | [99] |
| PCL meshes containing gentamicin, with or without sodium alginate encapsulation (in vitro: ~10 µg; in vivo: ~1.25 mg. Deposited at six random points). | Extrusion temperature: NP Speed: 10 mm/s, flow 1 mm/s Layer height: NP Mesh pore size: two designs, macroporous (1.25 × 1.25 mm) and microporous (0.75 × 0.75 mm) | In vitro: Antibacterial activity (Escherichia coli): agar diffusion tests (24 h, 37 °C). In vivo: Supra-muscular implant (2 × 2 cm) in abdominal wall of female Wistar rats (n = 40, four study groups). Clean surgery. Groups (4): PCL/PCL + alginate/PCL + gentamicin/PCL + gentamicin + alginate Histopathological assessment at 7 days postimplant. | [100] |
| PP and PVA meshes loaded with ciprofloxacin (3 ± 1% w/w in PP; 5 ± 1% w/w in PVA). | Extrusion temperature: 190 °C for PP; 200 °C for PVA Speed: 90 mm/s for PP; 150 mm/s for PVA Layer height: 0.2 mm Mesh pore size: two designs of diameters 2 or 3 mm. 32 mesh configurations (variables: chemical composition; drug; pore size; thread width; pore shape; no. pores/100 cm2) | In vitro: Drug-release assay (Type V USP dissolution method). Differential scanning calorimetry. Mechanical response (tensile strength assessment). In vivo: Incisional abdominal wall defect (1 cm) repaired by the implant of meshes (1.5 × 1.5 cm) facing the abdominal cavity in rabbits (n = 20; five groups). Clean surgery. Configuration chosen for all the printed meshes: pore size 3 mm; mesh width 1.2 mm. Groups (5): PP/PVA/PP + ciprofloxacin/PVA + ciprofloxacin/Marketed PP. Macroscopic and histopathological assessment at postoperative week 6. | [101] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Köhler, B.; Benito-Martínez, S.; Gómez-Gil, V.; Rodríguez, M.; Pascual, G.; Bellón, J.M. New Insights into the Application of 3D-Printing Technology in Hernia Repair. Materials 2021, 14, 7092. https://doi.org/10.3390/ma14227092
Pérez-Köhler B, Benito-Martínez S, Gómez-Gil V, Rodríguez M, Pascual G, Bellón JM. New Insights into the Application of 3D-Printing Technology in Hernia Repair. Materials. 2021; 14(22):7092. https://doi.org/10.3390/ma14227092
Chicago/Turabian StylePérez-Köhler, Bárbara, Selma Benito-Martínez, Verónica Gómez-Gil, Marta Rodríguez, Gemma Pascual, and Juan Manuel Bellón. 2021. "New Insights into the Application of 3D-Printing Technology in Hernia Repair" Materials 14, no. 22: 7092. https://doi.org/10.3390/ma14227092
APA StylePérez-Köhler, B., Benito-Martínez, S., Gómez-Gil, V., Rodríguez, M., Pascual, G., & Bellón, J. M. (2021). New Insights into the Application of 3D-Printing Technology in Hernia Repair. Materials, 14(22), 7092. https://doi.org/10.3390/ma14227092







