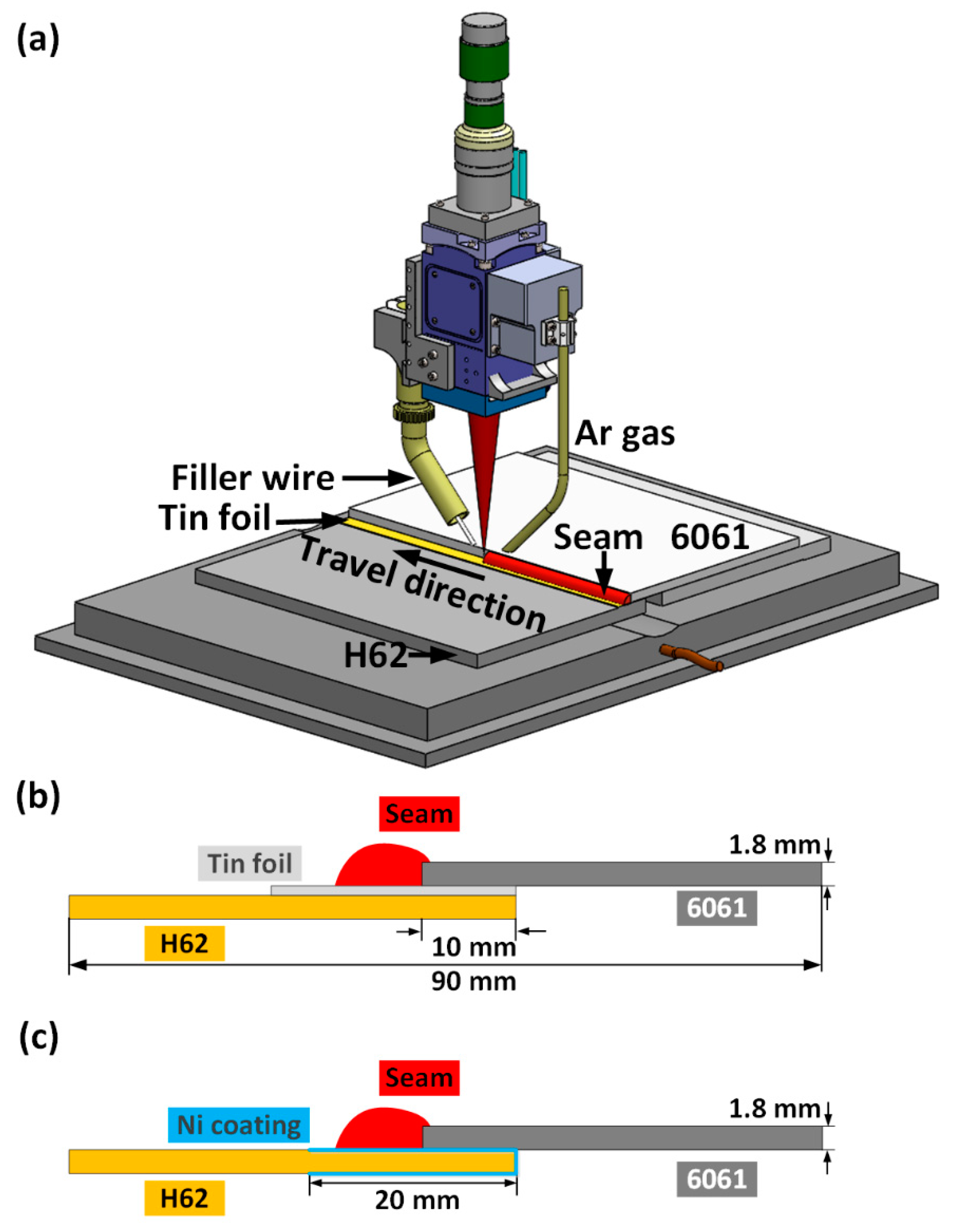
3.1. Joint Appearances and Cross-Sections
The melting point of tin foil is only 231 °C; because of this, it has good fluidity during the welding process, which leads to improved wetting/spreading ability of filler solders on the surface of the copper sheet. First, the effects of the addition of tin foil on the macroscopic morphology of the welding seam were investigated.
Figure 2a,b [
40], from our previous research shows the appearance and the cross sections of the joints, respectively, without tin foil. After bonding with tin foil, the joints had wide seams (
Figure 2c,e). When the thickness of tin foil was 0.3 mm, the seam was continuous and smooth and showed a good profile (
Figure 2c). For tin foil thickness of 0.5 mm, the seam showed a poor profile (
Figure 2e) with uneven width and defects due to incomplete fusion. Thus, large thicknesses of tin foil led to unstable spreading, which negatively affected the macro appearance. As seen in
Figure 2g, the joint with the Ni coating appeared continuous and stable, but it had a narrow seam.
Figure 2b,d,f,h [
40] shows the cross-sections of the joints. The contact angle, θ, and the spreading width of the seam were measured to examine the wetting/spreading ability. The contact angle was 46.74° and 54.53° for tin foil thickness of 0.3 mm and 0.5 mm, and the spreading widths were 8.54 mm and 7.88 mm, respectively. Compared with the Al/Cu joints without tin foil [
35,
40], with addition of the tin foil, the contact angle, the spreading width, and the wetting/spreading ability were enhanced. Thus, using tin foil can increase the spreading ability of a Zn–2Al filler.
When tin foil thickness increased from 0.3 mm to 0.5 mm, there was a slight decrease of the seam width (0.66 mm) and an increase in the contact angle (7.79°). Hence, the proper thickness of tin foil was 0.3 mm for the Al/Cu joint with the Zn–2Al weld filler.
For the Al/Ni-coated Cu joint, the contact angle was 75.22° and the spreading width was 5.18 mm, showing a slightly reduced wetting and spreading ability compared with the joint with only the Zn–2Al filler.
Overall, the findings of this study show that, compared with the joint with only a Zn-based filler, tin foil significantly enhanced the wetting ability of the filler, while the Ni coating had only a slight effect on it.
3.3. Mechanical Properties
Tensile strength tests were carried out on the samples using the Zn–2Al filler with the tin foil or the Ni coating.
Figure 9 shows the tensile strengths of the various joints. In our previous work, the tensile strength of the joint with only a Zn–2Al filler was 148 MPa [
40]. The average tensile strength of the joint with 0.3 mm tin foil was 151 MPa, which was not considerably higher than that without tin foil. A thicker tin foil caused a decrease in the strength, 111 MPa, which was lower than that of the joint without tin foil. As mentioned in the discussion on macroscopic morphology, the joint with a 0.5 mm thick tin foil had more defects such as incomplete fusion and depressions in the middle of the seam due to excessive penetration, which seriously affected the joint strength. The nonuniform morphology of the seam considerably affected the tensile strength, representing a larger error bar.
The joint with the Ni coating was significantly strengthened. The tensile strength increased to 171 MPa due to the enhanced interfacial bonding. Addition of the Ni coating was the most effective way to strengthen an Al/Cu joint with the Zn–2Al filler.
Figure 10 shows the fracture paths and the micromorphology of the fracture surfaces, in which the specimens with various interlayers had the highest strength.
Figure 10a,d shows the fracture paths of the joints with the tin foil and the Ni coating, respectively. Both joints fractured at the seam near the FZ/Al interface, indicating the enhancement of the strength at the FZ/Cu interface.
Figure 10b shows that the fracture morphology of the joint with the tin foil was fine depressions. Studies have reported that the CuZn IMC layer at the FZ/Cu interface is often the weak area in Al/Cu joints [
7,
35,
40]. The tin foil did not inhibit the formation of CuZn IMCs, but the spreading/wetting ability of the filler in the presence of the tin foil was considerably improved. Using a 0.3 mm tin foil, the weak area was transferred to the seam near the FZ/Al interface. According to the EDS line results in
Figure 4c, the tin foil melted and flowed to the region close to the Al base metal, and the low strength of the Sn solid solution weakened the joint at the Al side. Hence, the strength increased slightly with the tin foil.
With the Ni coating, the fracture surfaces shown in
Figure 10e,f exhibit shallow depressions and large smooth cracks, indicating more brittle characteristics.
Thus, addition of the tin foil and the Ni coating strengthened the joints, while the tensile strength was limited by the bonding near the Al sheet.
3.4. Thermodynamic Analysis
As mentioned above, addition of the tin foil did not improve the interfacial bonding. Instead, the increased thickness of the CuZn IMC layer was detrimental to the interfacial bonding. The main reason for the increase in joint strength was that the tin foil improved the spreading/wetting ability of the molten solder. The interfacial microstructure of the joint with the Ni coating was complex, and it replaced the Cu–Zn IMCs. The results of the mechanical tests showed that using the Ni coating enhanced the FZ/Cu interfacial bonding. Hence, only the effects of the Ni coating were examined in this study. The interfacial reaction mechanism was further analyzed using thermodynamic calculations.
It is difficult to measure the temperature field of the FZ/Cu interface directly. Researchers often use numerical simulations to discern the thermal cycle at the interface. In our previous research [
40], when a Zn–2Al weld filler was used with Al/Cu joints, the thermal cycle of three selected regions at the FZ/Cu interface was determined, and the average peak temperature of 838 °C was given as input to the Toop model to obtain the thermodynamic results. The Ni coating was very thin and the macromorphology of the joint was almost the same as that with only the Zn–2Al filler; therefore, its thermal effect was ignored in this study. Hence, the same interfacial peak temperature of 838 °C was used in the thermodynamic calculation even after the Ni coating was added.
Based on the Al–Zn–Cu ternary phase diagram [
51], CuZn
5 and CuZn could be produced at 598 °C and 468 °C, respectively. Ni
3Zn
14 and Al
3Ni could be produced at 872–490 °C [
52] and 850–643 °C, respectively [
53]. The τ
2 Zn–Ni–Al ternary phase could be formed at 850–340 °C [
50], and the α-Zn and α-Al + η-Zn solid solutions could be formed at 419.6 °C and 381 °C [
54]. All of the above phases can be produced at 838 °C.
In the Ni–Zn–Cu–Al quaternary system, to clearly describe the thermodynamic results, the data were divided into two stages. At the beginning of the welding process (stage I), the interface was assumed to be a 0.02Al–Zn–Ni–Cu quaternary system, with fixed Al content of 0.02 (same as the content of the Zn-based filler); this was used to analyze the diffusion tendency of Cu and Al atoms. Based on the distribution of Al and Cu, the subsequent reaction was analyzed, which was stage II. According to the EDS point results of the interfacial microstructure (
Table 5), the Cu content in the seam was very small, with the maximum value of 0.057, suggesting that Cu did not participate in the interfacial reaction. Hence, the Cu content was fixed at 0.05 for the analysis of the 0.05Cu–Zn–Ni–Cu pseudoternary system. The molar fractions of Zn and Ni in the calculations were 0–0.95 each and those of Al were 0.05–0.997.
Figure 11 shows the thermodynamic calculation results of the 0.02Al–(Ni–Zn–Cu) pseudoternary system at 838 °C in stage I.
Figure 11a shows the formation enthalpy in binary systems. Since the standard molar enthalpy is positive, Cu–Ni and Zn–Al systems cannot react with each other, but the metallurgical reaction can occur in the Ni–Al, Ni–Zn, Al–Cu, and Cu–Zn systems. The lowest formation enthalpy was observed for the Ni–Al system, implying that Ni–Al IMCs are easily produced. The Gibbs free energy of the 0.02Al–Cu–Zn–Ni system in stage I is shown in
Figure 11b. Ni–Zn–Cu ternary IMCs with the lowest Gibbs free energy are easily produced in the system. However, the corresponding ternary IMCs were not observed at the FZ/Cu interface of the joints with the Ni coatings, which might be attributed to the changed Cu and Al content. Therefore, further analysis of the diffusion tendency of Cu was carried out, as shown in
Figure 11c. According to the EDS results in
Table 5, Cu content was very small in the seam near the Ni coating and 0.055 in the Ni coating, which might be attributed to the unmelted Ni coating. To clearly describe the diffusion tendency of Cu, the chemical potential gradient from P3 (
Figure 11) to P4 (
Figure 11) was calculated. Initial Cu content was set to 0.055 in the Ni coating (
Table 5 P1), and the final Cu content was set to 0.036 in the α-Zn solid solution (
Table 5 P4). The chemical potential gradient of Cu from the Ni coating to the α-Zn solid solution was −4.159 kJ/mol, as shown in
Table 6, which suggested that the diffusion of Cu atoms was much more difficult when the Cu sheet was covered with the Ni coating.
Figure 11d shows the chemical potential of Al atoms during stage I. With low Cu content, the Al atoms tended to diffuse from the molten filler with high Zn content to the Ni-enriched region. This led to the aggregation of Al atoms adjacent to the interface, which promoted the formation of Al–Ni IMCs. The final content of Al at the interface was much greater than 0.02, indicating that the previous assumption about the Al content (0.02) was incorrect. Hence, further analysis of stage II was carried out.
Figure 12 shows the thermodynamic calculation results of stage II. To determine the preferentially generated phases at the interface, the Gibbs free energy of the 0.05Cu–Zn–Ni–Cu quaternary system was calculated, as shown in
Figure 12a. The lowest standard Gibbs free energy of the system was observed in the blue area, implying that the Al–Ni–Zn ternary compounds might be the preferable phase produced. However, the actual phase generated at the FZ/Cu interface was not consistent with the inference, which resulted from the changing Al content. In the beginning, the Al content adjacent to the Ni-coated Cu sheet was only 2%. Then, Al atoms gradually gathered near the interface. Ni
3Zn
14 was produced preferentially when the Al content was low. According to the EDS results in
Table 5, Al content in the Ni
3Zn
14 phase was almost zero. Hence, the preferable formation of Ni
3Zn
14 further resulted in the increase in the Al content at the interface. Thus, after the preferable formation of Ni
3Zn
14, the Al
3Ni phase and the τ
2 Zn–Al–Ni ternary phase containing 15.8 at.% Al were generated.
Figure 12b shows the chemical potential of Ni in the 0.05Cu–Zn–Ni–Cu pseudoternary system. Ni tended to diffuse to the gathering area of Al and Zn, illustrating the mutual diffusion between Ni and Al. As mentioned above, the Al content increased gradually during the welding process. Hence, three chemical potential gradients of Ni were compared for the Al contents of 0.050, 0.260, and 0.524, and the Ni content was set to 0.317 based on the EDS point results in
Table 5 (P5). The specific values of Ni chemical potential at P3, P4, and P5 (shown in
Figure 12) were −70.14, −76.86, and −85.26 kJ/mol, as shown in
Table 7. The order of chemical potentials at these three points was P3 > P4 > P5, indicating that Ni tended to diffuse to the region with a higher Al content. Thus, the unique diffusion phenomenon of Ni can be described. At the beginning of the welding, the content of Al was low, the Ni atoms gathered at the FZ/Cu interface, and the Ni
3Zn
14 phase was preferentially produced. Then, as the Al content increased, most of the Ni atoms diffused to the Al-aggregating region, and the Al–Ni IMCs formed.
The highest Al content near the interface was 0.469 (EDS results shown in
Table 5), which was much higher than the Al content of the Zn–2Al filler. To determine the diffusion tendency of Al, the chemical potential of Al in the 0.05Cu–Ni–Zn–Al system was calculated, as shown in
Figure 12c. Al contents at P6 and P7 were set to 0.051, and the Ni contents at P6 and P7 were set to 0.001 and 0.317, respectively. The chemical potential gradient of Al from P6 to P7 (−13.76 kJ/mol, as determined from
Table 7) in
Figure 12 indicates the diffusion tendency of Al from the molten filler to the FZ/Cu interface. Thus, Al diffused to the interfacial region with high Ni content. The chemical potential of P8 in
Figure 12c was −72.47 kJ/mol, which was nearly the same as that of P6, suggesting that the Al content increased until the Al content reached 0.155 when the highest Ni content was 0.317. Hence, the Al content was limited, which inhibited the formation of the brittle Al
3Ni phase. The final interfacial microstructure showed that the Al
3Ni IMC content was low, and the thin zigzag Al
3Ni IMC layer had little effect on the bonding of the interface.
3.5. Bonding Mechanism of the Al/Ni-Coated Cu Joint
Based on the microstructure analyses and thermodynamic calculations of the Al/Ni-coated Cu joint, the formation of phases and the diffusion tendency of atoms at different temperature intervals are illustrated in
Figure 13. Once the LWB process began, the Zn–2Al filler melted and rapidly spread on the surface of the Ni-coated Cu sheet, as shown in
Figure 13a. Then, the interfacial temperature increased rapidly due to the spreading of the molten filler. In the heating process, as shown in
Figure 13b, atoms from the Ni coating and the molten filler diffused toward each other due to the concentration gradient. As the temperature increased, the Ni atoms gathered at the region adjacent to the FZ/Cu interface, the Al atoms diffused from the molten filler to the Ni-gathering region, and few Cu atoms diffused to the seam, as shown in
Figure 13c. In this diffusion process, the diffusion direction and the driving force depended on the chemical potential of the system. According to the aforementioned analysis, when the temperature decreased to 872 °C, the Ni
3Zn
14 layer was formed adjacent to the Ni coating due to the gathering of large quantities of Ni and Zn atoms, as shown in
Figure 13d. Notably, the thickness of the Ni
3Zn
14 layer increased in a large temperature range of 872–490 °C. Due to the interdiffusion of Al and Ni atoms, the Al content gradually increased, and a gathering area of Ni and Al atoms formed close to the Ni
3Zn
14 layer, which resulted in the formation of black diamond-shaped Al
3Ni particles in a temperature range of 850–643 °C, which dispersed in the seam near the Ni
3Zn
14 layer, as shown in
Figure 13e. As the temperature decreased to 643–420 °C, a small amount of the τ
2 Zn–Ni–Al ternary phase formed adjacent to the Ni
3Zn
14 layer, as shown in
Figure 13f. Then, α-Zn solid solution formed when the temperature decreased to 420 °C, as shown in
Figure 13g. Finally, the thickness of the α-Zn solid solution increased to an average of 7.66 μm. Due to the force of the solid front of the α-Zn solid solution layer around the Ni
3Zn
14 layer, the diamond-shaped Al
3Ni phase aggregated, and finally, the zigzag Al
3Ni layer generated around the α-Al + η-Zn solid solution adjacent to the α-Zn layer. As shown in
Figure 13h, at room temperature, composite layers of the Ni
3Zn
14–τ2 Zn–Ni–Al ternary phase–α-Zn solid solution–Al
3Ni were produced at the interface instead of the CuZn–CuZn
5 layers without the Ni coating, which strengthened the interfacial bonding.