3.1. Mechanochemical Synthesis of Ceramics Powders
Figure 1 illustrates the synthesis of the CaCu
3Ti
4O
12 perovskite compound and CaCu
3−xMg
xTi
4O
12 solid solutions by the mechanochemical method. The X-ray diffraction (XRD) patterns of the substrate powders after different times of high-energy ball milling are shown in
Figure 1a.
The gradual disappearance of the X-ray diffraction reflections corresponding from the substrates during the mechanochemical treatment, and the appearance of the reflections that indicate the formation of a crystalline product—CCTO—are clearly visible. After two hours of the high-energy milling of the powder, only the perovskite phase is noticeable. The synthesis of CaCu
3−xMg
xTi
4O
12 solid solutions with different modifier concentrations of 0.1 ≤ x ≤ 0.5 was performed in an analogous manner. The XRD patterns of the Mg
2+ ion-doped CCTO ceramics are shown in
Figure 1b. They confirm the presence of a primary CaCu
3Ti
4O
12 phase (JCPDS 75-2188) in all of the prepared ceramics. A small amount of a secondary TiO
2 phase was observed in the samples in which the concentration of magnesium oxide was high. Singh et al. [
30] previously reported the benefits of using mechanochemical synthesis to produce magnesium-doped CaCu
3Ti
4O
12. It has been shown that the grinding of the substrates in ethanol led to a decrease in the synthesis temperature of the compound (800 °C), and positively affected the dielectric properties of the ceramics. In this study, a significantly higher milling energy was used, which led to the formation of the crystalline product only due to the mechanochemical treatment of solids. The downside of using such high-energy milling is the presence of impurities in the product from abrasive milling equipment components. Hence, milling balls and a vessel made of ZrO
2 were used, which has a minor influence on the dielectric properties of the ceramic [
29]. In this case, no X-ray diffraction reflections from zirconia were observed on the diffraction patterns (
Figure 1), which indicates a low amount of this pollution, i.e., below the detection limit of the XRD method. The elemental analysis of the samples performed by EDS showed the presence of zirconium at around 2 wt %.
Figure 2 presents the results of the elemental analysis studies for the selected samples, i.e., the pure CaCu
3Ti
4O
12 compound and two solid solutions: CaCu
2.8Mg
0.2Ti
4O
12 and CaCu
2.6Mg
0.4Ti
4O
12.
All of the EDS peaks from individual elements (Ca, Cu, Mg, Ti, O) and from the above-mentioned zirconia impurity were detected. The percentage of various elements in the tested samples was determined from the whole surface of the samples. The values of the individual elements for each sample, presented in the tables (
Figure 2), are consistent with the expected stoichiometry of compounds.
The ceramic samples, after the mechanochemical treatment, were subjected to sintering at 1075 °C for different lengths of time—2 and 10 h—in order to increase the density of the ceramics. The length of calcination time significantly affected the morphology of the ceramic powders, in particular the size of the crystallites. The analysis of the chemical composition of the tested materials before and after the high-temperature treatment process showed no changes in the content of individual elements. None of the tested samples showed any deviations from the stoichiometric composition, regardless of the sintering time (2 or 10 h). The surface morphologies of the selected samples—CaCu
3Ti
4O
12, CaCu
2.8Mg
0.2Ti
4O
12 and CaCu
2.6Mg
0.4Ti
4O
12, sintered at 1075 °C for 2 h—are shown in
Figure 3.
The effect of the addition of magnesium ions on the shape and size of the crystallites is clearly visible; the higher concentration of the modifier and the larger grains with irregular shapes are observed. Mechanochemically-prepared CCTO powder is characterized by fine-grained microstructure with the smallest particle size, good homogeneity, and similar shape. The average crystallite size is around 150–200 nm. The ceramics containing the modifier are characterized by a different surface morphology; they consist of two types of crystallites: large irregular grains and very small grains with various shapes and sizes. The presence of the Mg
2+ dopant seems to promote grain growth. The size of the crystallites varies from 2 µm for the sample of CaCu
2.9Mg
0.1Ti
4O
12 to 10 µm for the sample of CaCu
2.5Mg
0.5Ti
4O
12 (not shown). Additionally, the higher concentration of the modifier reduces the proportion of small grains in the material. Extending the heat treatment time significantly effects on the morphology of the ceramics.
Figure 4 shows the SEM image for the surface morphologies of the undoped CCTO sample and the magnesium-doped ceramics sintered at 1075 °C for 10 h. The differences in comparison with the corresponding materials calcined for 2 h are clearly visible. The microstructure of all of the investigated ceramics consisted of very large grains in the size range of 20–40 µm, which is a consequence of long-term high-temperature treatment. Additionally, a large number of sinters and pores are observed in both the undoped and doped samples. The similar observations in the microstructure of such ceramics have already been reported by previous researchers for samples prepared by the conventional high-temperature synthesis method [
22,
23]. The mechanism for the grain growth of CCTO in high-temperatures is complex, and is based on the CuO liquid sintering phase. The Cu-rich phases caused from the deoxidization of the liquid Cu
2O, which is initially reduced from CuO at a temperature of around 900 °C. These phases segregate at the grooved grain boundaries, and lead to the abnormal grain grow and porous structure. Additionally, in samples containing the modifier in the form of magnesium ions, CuO-MgO solid solutions may form. Such a solid solution with a higher melting temperature can limit the mobility of the ions (diffusion) in the solid phase, and can distort the crystal lattice of the matrix.
Figure 5a shows the XRD patterns of mechanochemically-prepared CaCu
3−xMg
xTi
4O
12 powders with different Mg
2+ ions concentrations subjected to subsequent sintering at 1075 °C for 2h. The analogous samples, sintered at the same temperature for 10 h, are not shown in the paper due to their high similarity. In all of the diffraction patterns, regardless of the amount of modifier and sintering time, only one CaCu
3Ti
4O
12 phase is visible (JCPDS 75-2188). Comparing these results to the diffraction patterns of the powders after mechanochemical treatment (
Figure 1), the differences are clearly visible. The subsequent thermal treatment of the powders led to the reaction of the residual substrates; there are no visible diffraction reflections corresponding to the secondary phases (e.g., TiO
2) or impurities from the grinding media (ZrO
2). Additionally, the degree of the crystallinity of the ceramics has improved, as evidenced by the lower half-widths and higher intensities of the CCTO diffraction reflections.
The diffraction peaks in the XRD patterns for the CaCu
3Ti
4O
12 and all of the CaCu
3−xMg
xTi
4O
12 solid solution (0.1 ≤ x ≤ 0.5) powders, regardless of the sintering time, are perfectly indexed to the bcc structure with the space group
Im3. The lattice parameter values of all of the investigated samples were calculated, and are summarized in
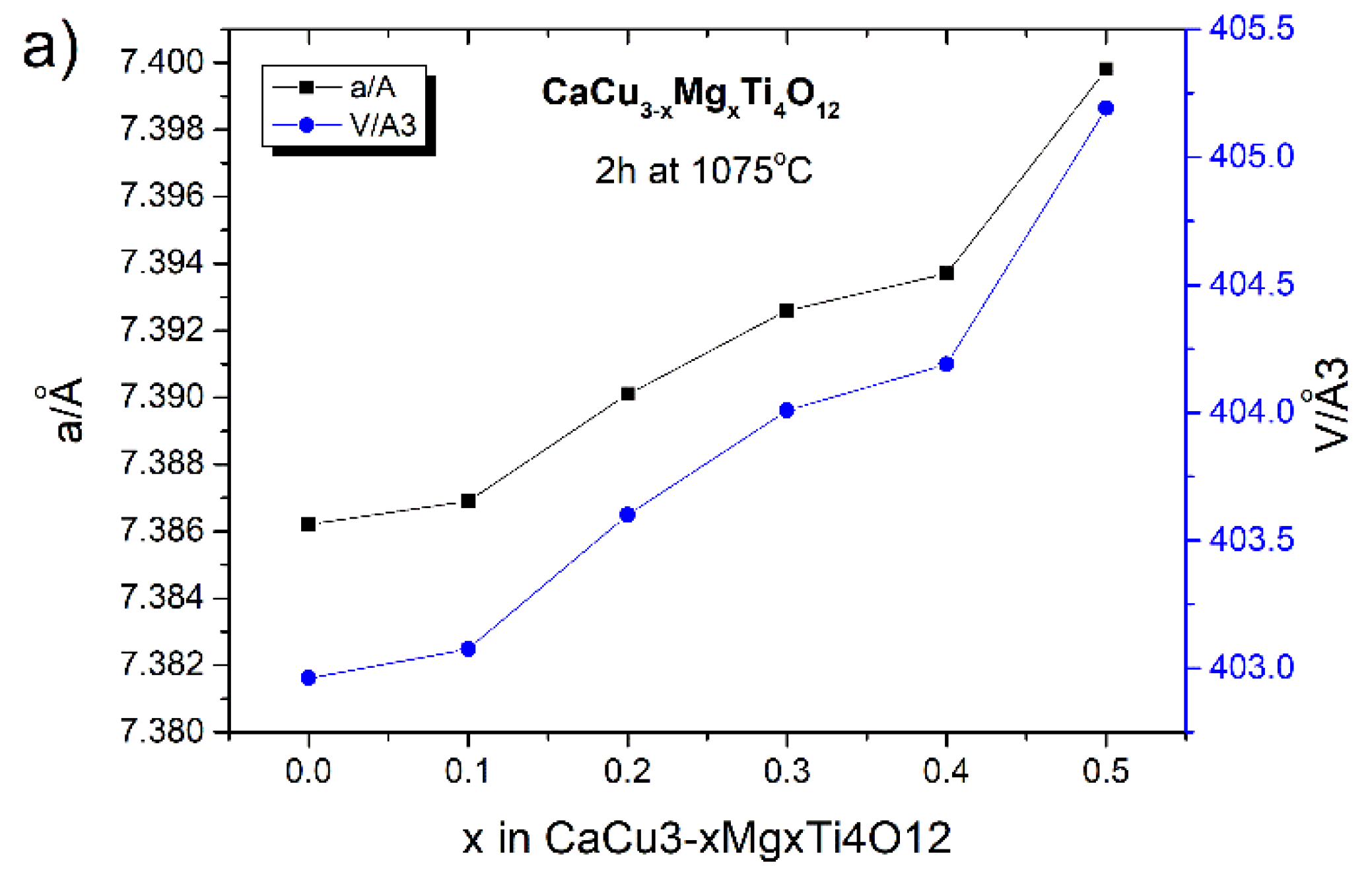
Figure 6. A detailed analysis of the diffraction data showed slight differences in the values of the CCTO unit cell parameters due to the doping and the length of the sintering time.
In the case of the mechanochemically-produced solid solutions sintered for 2 h at 1075 °C, the value of the unit cell parameter (a) increases with the amount of modifier added. The calculated lattice parameters of these samples were 7.3862(4), 7.3869(5), 7.3901(3), 7.3926(1), 7.3937(2), and 7.3998(6) Å for the samples with x = 0.0, 0.1, 0.2, 0.3, 0.4, and 0.5, respectively. The unit cell volumes [Å
3] for these samples are as follows: 402.9612; 403.0757; 403.5998; 404.0095; 404.1899; and 405.1911. This is also visible in a slight shift of the CCTO diffraction reflections towards lower values of two Theta angles (
Figure 5b). Despite the similar ion radii in the same coordination, the partial substitution of magnesium ions (Mg
2+ = 0.57 Å) for copper ions (Cu
2+ = 0.57 Å) in the CCTO crystal lattice causes a slight change of unit cell parameter (a) because of the increase in the surface energy of the crystal. This is attributed to the chemical bond strength, defined as the ratio of the valence over the internuclear distance; the chemical bond of Mg–O (1.09) is larger than that of Cu–O (1.01). The obtained results are in accordance with the results reported by other authors [
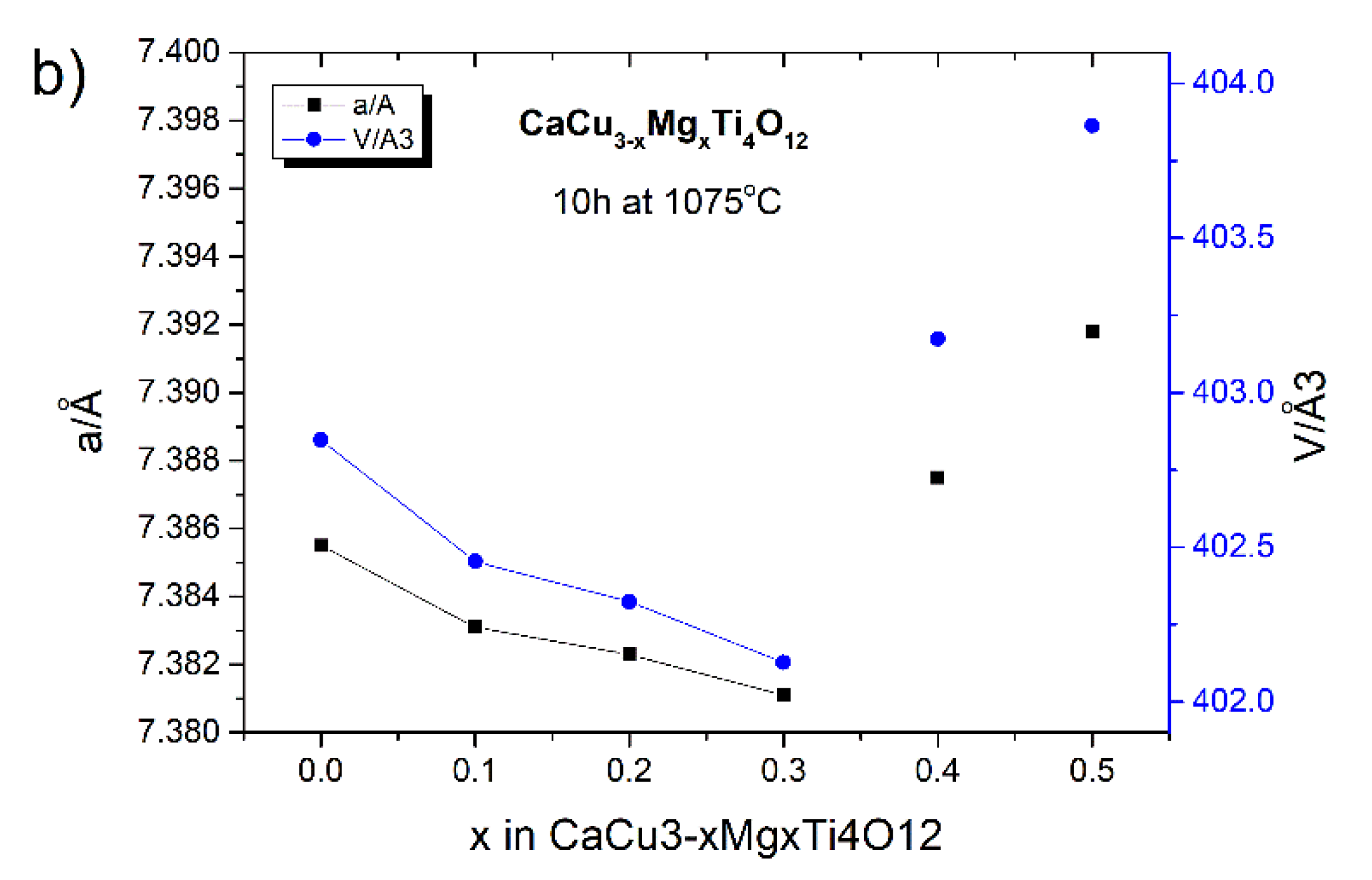
24]. Extending the time of the high-temperature treatment to 10 h also changes lattice parameter (a) of the CCTO unit cell. In this case, for smaller amounts of the modifier (x ≤ 0.3), this value decreases, while for larger amounts of Mg
2+ (x ≥ 0.4) the (a) value increases (see
Figure 5c and
Figure 6. The sequence of lattice parameters of these samples was calculated: 7.3855(4), 7.3831(7), 7.3823(8), 7.3811(1), 7.3875(3), and 7.3898(7) Å. The unit cell volumes [Å
3] for these samples are as follows: 402.8466, 402.4540, 402.3232, 402.1270, 403.1740, and 404.0570. The significant differences of the lattice parameters were observed. These results, in comparison with the previous ones, indicate that—in this case—other substitutional types of solid solutions may be formed. In addition, the long-term heat treatment of ceramics at a high temperature in an atmosphere containing oxygen (air) may also damage the crystal structure through the formation of various types of structural defects, including oxygen vacancies, etc. [
27]. Moreover, the long-term high-temperature treatment of the ceramics could stimulate diffusion processes in the solid, and could thus lead to the substitution of larger zirconium ions (Zr
4+ = 0.59 Å), from the grinding media, into the CCTO crystal lattice.
3.2. Dielectric Properties
The temperature dependences of the real part of the dielectric permittivity (ε’) for the chosen frequency (1 kHz) are shown in
Figure 7a,b for the solid solutions sintered at 1075 °C for 2 h and 10 h, respectively. The values of the function ε’(T) slightly increase below room temperature (RT) for all of the investigated samples. Below room temperature (RT), the values of the ε’(T) function do not change significantly for all of the investigated samples. The ε’ values at RT in
Figure 7a for the CaCu
3−xMg
xTi
4O
12 ceramics with the lower Mg
2+ ions content (x ≤ 0.2) are approximately 18,000 and 24,000 for x = 0 and x = 0.2, respectively. One can see that the ε’ for the samples with higher amounts of Mg
2+ ions (x ≥ 0.3) shows ten-times lower values (~2000). In the temperature range above RT, the ε’ values for CaCu
3−xMg
xTi
4O
12 ceramics with x ≥ 0.3 increase rapidly. The high temperature treatment (10 h at 1075 °C) of the samples increases the values of the ε’ (
Figure 7b). For the chosen ceramics at RT, the ε’ values are approximately 19,000, 34,000, 42,000 and 5600 for x = 0, 0.2, 0.3 and 0.5, respectively. These results are usually observed in the literature [
8,
9,
26,
30]. The high ε’ values may be interpreted as an internal barrier layer capacitor (IBLC) effect [
31]. According to this model, CaCu
3−xMg
xTi
4O
12 ceramics are constituted of semiconducting grains and insulating grain boundary layers. The studies of the ceramics using impedance spectroscopy allowed us to separate the grain and the grain boundary contributions [
26]. All of the parameters of the equivalent circuit model show a variation in the resistance of the grain and the grain boundary with the amount of MgO used during the ceramic synthesis. According to the literature [
32] data, it can be concluded that the grain boundary resistance of the investigated ceramics is closely related to the concentration of oxygen vacancies at the grain boundaries, the charge of which contributes to the polarization mechanism. The value of this resistance depends on the thermal treatment (the sintering time and sintering temperature) of the CaCu
3−xMg
xTi
4O
12 ceramics. As the content of Mg
2+ ions in the samples increases, the grain boundary resistance increases. This is due to the ability of the Mg
2+ doping ions to inhibit oxygen loss in the samples. Therefore, the increase of ε’ with Mg
2+ ion substitution may be attributed to the formation of the insulating grain boundary layers with an Mg-rich phase. The results reported in the literature [
8,
15,
26,
27,
30] show that the high ε’ values of CCTO ceramics are caused by the electrical response of the grain boundaries. In all probability, higher amounts of Mg
2+ ions in CaCu
3−xMg
xTi
4O
12 ceramics cause an increase in the grain boundary capacitance, which leads to an increase in the values of ε’. On the other hand, according to the results of the microstructure analysis (
Section 3.1), it was found that the ε’ values depend on the grain size, their shape, and their number of pores. Thus, the properties of the microstructure (e.g., the number of pores) may result in decreased ε’ values. The highest values of ε’ in the whole temperature range were observed for CaCu
3−xMg
xTi
4O
12 ceramics with an Mg
2+ ion content of x = 0.3, sintered at 1075 °C for 10 h (
Figure 7b).
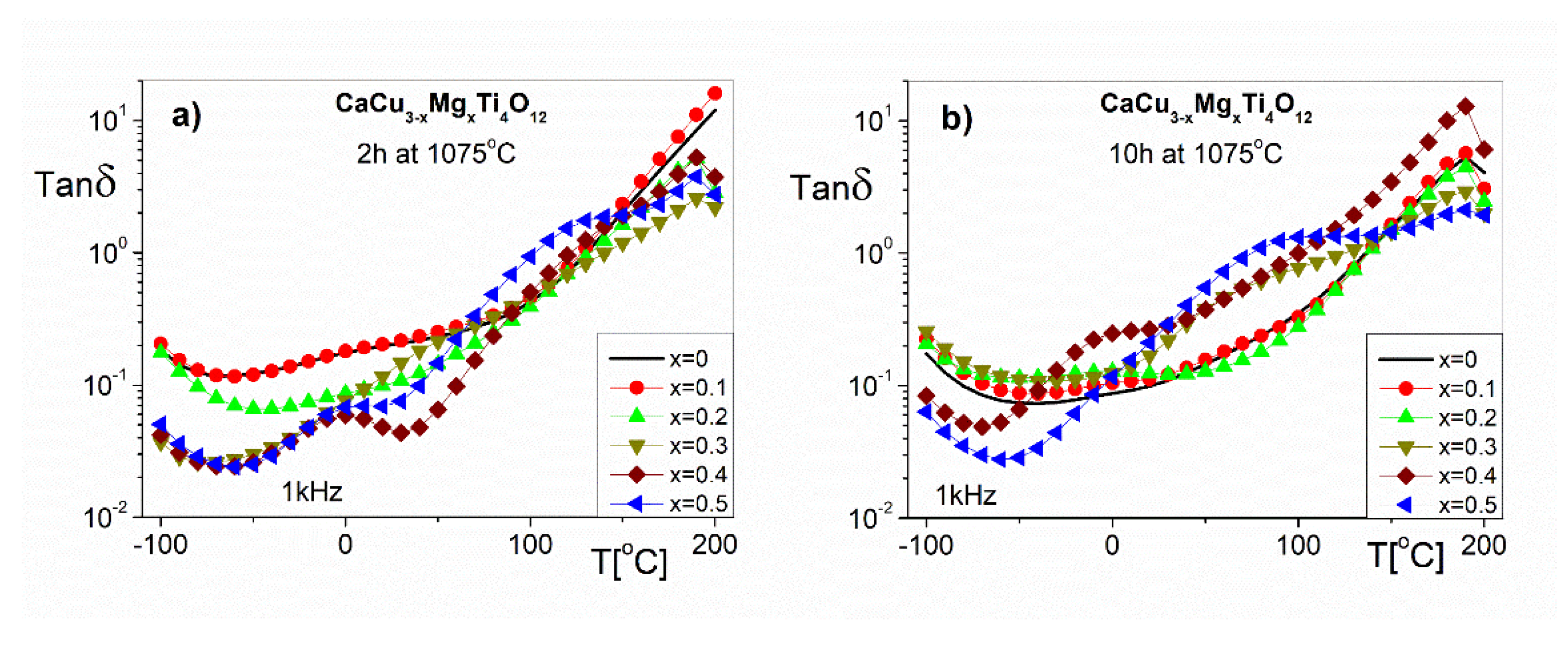
Complementary to the polarization phenomenon represented by the ε’ are the dielectric losses expressed by the imaginary part of the dielectric permittivity or Tanδ. The temperature dependences of the Tanδ at 1 kHz are shown in
Figure 8. These dependencies describe the dielectric properties related to the energy loss processes of the electric field in the investigation samples. The values of Tanδ at RT (
Figure 8a) for CaCu
3−xMg
xTi
4O
12 ceramics in the entirety of the doping concentration range (x = 0–0.5) are approximately (0.2–0.05), respectively. As can be seen in
Figure 8b, the Tanδ values of the ceramics sintered at 1075 °C for 10 h are larger than 0.1, which excludes them in applications as capacitors [
1]. The Tan δ (T) functions for all of the investigated samples in the temperature range below RT show a local minimum caused by the existence of dielectric relaxation phenomena (in the range of the higher frequencies). We can also see that, at temperatures higher than RT, the Tan δ increases monotonically to 190 °C, which is caused by the increase in the dc conductivity in the bulk samples, and low frequency relaxation behavior.
In order to explore the Mg
2+ ion doping effects on the dielectric properties of the grains and the grain boundaries, we used the complex electric modulus formalism (M* = M’ + iM”). The M* formalism describes the phenomena of electric charge transport and dielectric relaxation in these types of materials [
32]. The electric modulus studies in the frequency domain correspond to the relaxation of the electric field in the material when the electric displacement remains constant [
33]. The formalism of the electrical modulus is very sensitive to small changes in the value of the local polarization (capacitance), so it is—among others—the most suitable for the description of the electrical properties of grains and grain boundaries in ceramic (or inhomogeneous) materials. The frequency dependences of the M” (in a log–log scale) are shown in three separate figures, in order to represent the different temperature ranges and different samples.
Figure 9a shows the M”(ν) functions at the chosen temperatures of 100 °C and 150 °C for the CCTO samples with different sintering times. Both samples show the maximum, which shifts systematically in the temperature range from RT to 200°C, with increasing frequency. The shift of the M” maximum corresponds to the so-called ‘conductivity relaxation process’ [
34]. We attribute the conduction process to the electrical properties of grain boundaries. In the temperature range from −100 °C to 50 °C can be seen another low-frequency dielectric relaxation process (
Figure 9b). In the case of the CCTO sample, the changes in the maximum value of M” are slight, which corresponds to relatively large values of the sample capacitance. On the other hand, CaCu
3−xMg
xTi
4O
12 ceramics with an Mg
2+ ion content of x = 0.5 have lower capacitance values compared to CCTO, which results in lowered ε’ values. These low-frequency relaxation processes may be related to the movement of free charges through the sample towards the opposite electrode in the presence of an external ac electric field. This creates a macrodipole of which the oscillations give rise to relaxation and conduction processes [
35,
36]. We attribute this process to Maxwell–Wagner relaxation because the samples consist of heterogeneous dielectric components (grains and grain boundaries) that have different conductivities. Moreover, we notice that the conductivity relaxation process (maximum M”) in CaCu
3−xMg
xTi
4O
12 solid solutions with higher amounts of Mg
2+ ions (x ≥ 0.3) appears at a lower temperature than that of CCTO (
Figure 9b). This means that Mg
2+ ion doping decreases the activation energy of the conduction process.
Figure 9c show an example the frequency dependences of the M” with a temperature range of −100 °C to −70 °C for CaCu
3−xMg
xTi
4O
12 ceramics with an Mg
2+ ion content of x = 0 and x = 0.5. The values of M” at the maximum confirm the presence of a small capacitance, which is responsible for the dielectric properties of the grains. These maxima, in the case of the grains, are about 100 times greater than those corresponding to the grain boundaries. This indicates that the grain capacitance is approximately 100 times smaller than that of grain boundaries.
This maximum (
Figure 9a–c) indicates a transition from the short range to long range mobility of the charge carriers, with decreasing frequency. The characteristic relaxation time, τ = (2πν
m)
–1, is the most probable conductivity relaxation time determined from the maximum frequency (ν
m) of the M”(ν) functions. The relaxation time, τ, exhibits a thermally-activated dependence. This generally follows the Arrhenius law: τ = τ
oexp(E
a/k
BT), where τ
o is the pre-exponential factor and E
a denotes the activation energy for dielectric relaxation. In
Figure 9d, we show the temperature dependence of the relaxation times from the M”(ν) spectra for the CCTO (sintered at 1075 °C for 2 h and 10 h) samples. Based on the Arrhenius plot, it can be determined that the grain response maximum M”(ν) is observed at a low temperature and high frequency range (above 1 MHz), whereas for the grain-boundary, these responses of M”(ν) correspond to higher temperatures (above RT) and a low frequency range (below 100 kHz).
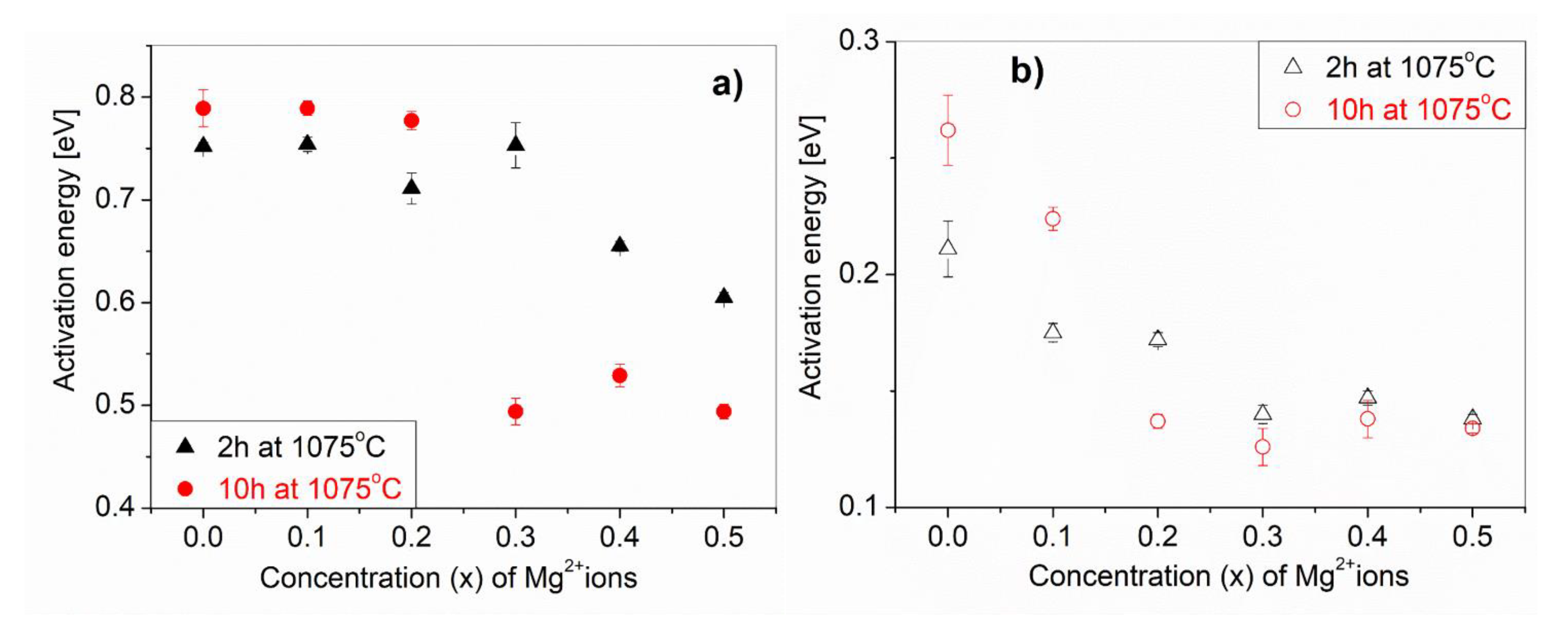
Figure 10 shows the dependence of the activation energy of the conduction process (a) and dielectric relaxation (b) from the amounts of Mg
2+ ions (x) for CaCu
3−xMg
xTi
4O
12 solid solutions. We can clearly see that Mg
2+ ion doping decreases the activation energy of both processes. The activation energy of the conductivity relaxation process corresponding to the grain boundaries for the CCTO sample was found to be approximately 0.8eV. Sinclair et al. [
31] determined an activation energy of the CCTO grain boundaries equal to 0.60 eV. For the CaCu
3−xMg
xTi
4O
12 solid solutions with higher amounts of Mg
2+ ions (x ≥ 0.3) and a high temperature treatment (10 h at 1075 °C), this activation energy decreased to about 0.5eV (
Figure 10a). The grain activation energy 0.140eV of our CCTO sample is different from the 0.08eV of the CCTO grains [
32]. From the slopes of the fitted straight lines, we obtain the activation energies (
Figure 10b) for the Maxwell–Wagner relaxation, which generally refers to interfacial polarization occurring in electrically-inhomogeneous systems.