A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present
Abstract
:1. Introduction
2. Materials and Methods
3. Biomaterials for Orthopaedic Surgery and Traumatology
3.1. Metallic Biomaterials
3.1.1. Steel
3.1.2. Titanium
3.1.3. Cobalt–Chromium-Molybdenum (CoCrMo) Alloys
3.2. Ceramic Biomaterials
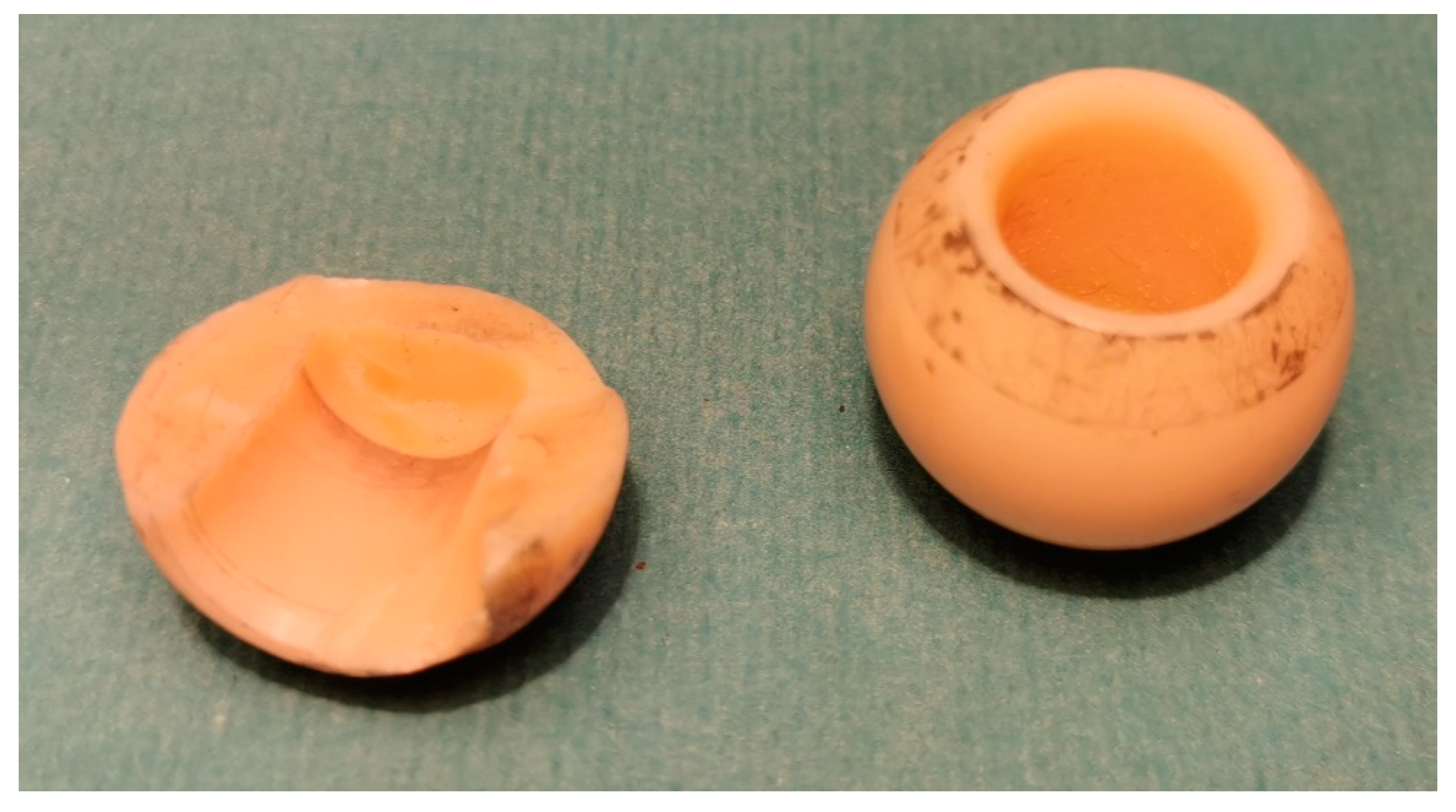
3.3. Polymeric Biomaterials
3.3.1. Teflon
3.3.2. Polyethylene
3.3.3. Polimethylmetacrylate
4. Surface Modifications
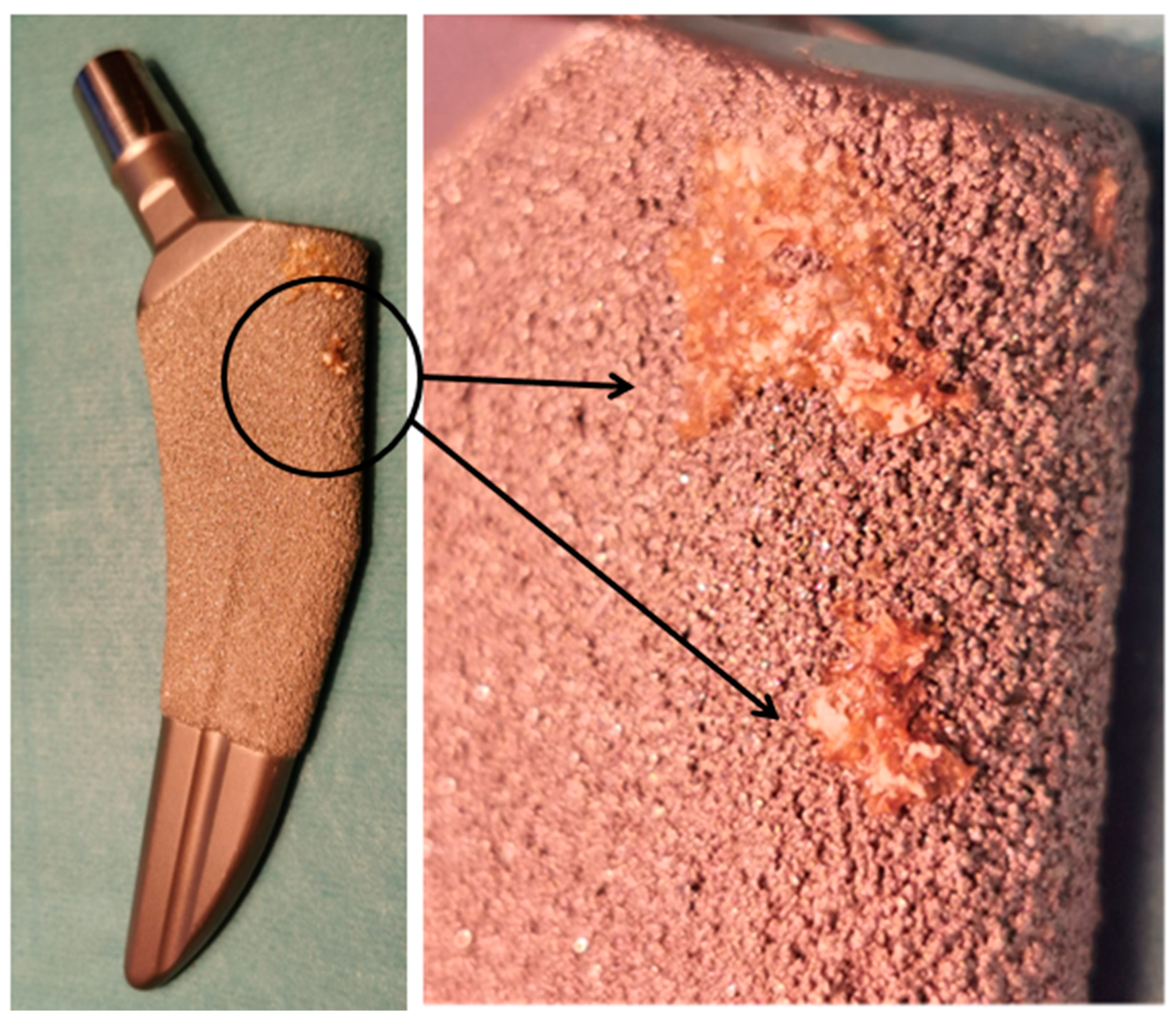
5. The Limitations in Implant Manufacturing Technologies and Applications




6. Summary and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marin, E.; Boschetto, F.; Pezzotti, G. Biomaterials and biocompatibility: An historical overview. J. Biomed. Mater. Res. Part A 2020, 108, 1617–1633. [Google Scholar] [CrossRef]
- Bartoníček, J. Early history of operative treatment of fractures. Arch. Orthop. Trauma. Surg. 2010, 130, 1385–1396. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Mol. Clin. Environ. Toxicol. 2012, 101, 133–164. [Google Scholar] [CrossRef] [Green Version]
- Abdalla, S.S.I.; Katas, H.; Azmi, F.; Busra, M.F.M. Antibacterial and Anti-Biofilm Biosynthesised Silver and Gold Nanoparticles for Medical Applications: Mechanism of Action, Toxicity and Current Status. Curr. Drug Deliv. 2020, 17, 88–100. [Google Scholar] [CrossRef]
- Sherman, W.O. Vanadium steel plates and screws. Surg Gynecol. Obstet. 1912, 14, 629. [Google Scholar]
- Punj, S.; Singh, J.; Singh, K. Ceramic biomaterials: Properties, state of the art and future prospectives. Ceram. Int. 2021, 47, 28059–28074. [Google Scholar] [CrossRef]
- Ambrose, C.G.; Hartline, B.E.; Clanton, T.O.; Lowe, W.R.; McGarvey, W.C. Polymers in Orthopaedic Surgery. In Advanced Polymers in Medicine; Puoci, F., Ed.; Springer: Cham, Switzerland, 2014; pp. 129–145. [Google Scholar]
- Beaupré, G.S.; Csongradi, J.J. Refracture Risk After Plate Removal in the Forearm. J. Orthop. Trauma 1996, 10, 87–92. [Google Scholar] [CrossRef]
- Fransway, A.F.; Zug, K.A.; Belsito, D.V.; DeLeo, V.A.; Fowler, J.F.; Maibach, H.I.; Marks, J.G.; Mathias, C.T.; Pratt, M.D.; Rietschel, R.L.; et al. North American Contact Dermatitis Group Patch Test Results for 2007–2008. Dermatitis 2013, 24, 10–21. [Google Scholar] [CrossRef]
- Wu, Y.; Kong, L. Advance on toxicity of metal nickel nanoparticles. Environ. Geochem. Health 2020, 42, 2277–2286. [Google Scholar] [CrossRef]
- Bradberry, S.M.; Wilkinson, J.M.; Ferner, R.E. Systemic toxicity related to metal hip prostheses. Clin. Toxicol. 2014, 52, 837–847. [Google Scholar] [CrossRef]
- Yatera, K.; Morimoto, Y.; Ueno, S.; Noguchi, S.; Kawaguchi, T.; Tanaka, F.; Suzuki, H.; Higashi, T. Cancer Risks of Hexavalent Chromium in the Respiratory Tract. J. UOEH 2018, 40, 157–172. [Google Scholar] [CrossRef] [Green Version]
- Kaluarachchi, H.; Chung, K.C.C.; Zamble, D.B. Microbial nickel proteins. Nat. Prod. Rep. 2010, 27, 681–694. [Google Scholar] [CrossRef]
- Lautenschlager, E.P.; Sarker, N.K.; Acharya, A.; Galante, J.O.; Rostoker, W. Anodic polarization of porous fiber metals. J. Biomed. Mater. Res. 1974, 8, 189–191. [Google Scholar] [CrossRef]
- Pauwels, F. Überraschende Erfolge durch die Anwendung einer Zuggurtung bei der Patellarfraktur. Langenbeck’s Arch. Chir. 1966, 316, 221–224. [Google Scholar] [CrossRef]
- Levanthal, G.C. Titanium: A metal for surgery. J. Bone Jt. Surg. 1951, 33-A, 473–474. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Su, K.; Su, L.; Liang, P.; Ji, P.; Wang, C. Comparison of 3D-printed porous tantalum and titanium scaffolds on osteointegration and osteogenesis. Mater. Sci. Eng. C 2019, 104, 109908. [Google Scholar] [CrossRef]
- Neuman, G.; Spångberg, L.; Langeland, S. Methodology and criteria in the evaluation of dental implants. J. Endod. 1975, 1, 193–202. [Google Scholar] [CrossRef]
- Golberg, S.Z.; Vainer, E.l.; Vasiljeva, G.S. Study of the possibility of using titanium in medical technology. Med. Prom. SSSR 1963, 17, 30–35. [Google Scholar]
- Galante, J.; Rostoker, W. Corrosion-related Failures in Metallic Implants. Clin. Orthop. Relat. Res. 1972, 86, 237–244. [Google Scholar] [CrossRef]
- Williams, D.F. Corrosion and corrosion prevention in orthopaedic implants. Proc. R. Soc. Med. 1972, 65, 1027. [Google Scholar] [CrossRef] [Green Version]
- Woo, S.L.-Y.; Lothringer, K.S.; Akeson, W.H.; Coutts, R.D.; Woo, Y.K.; Simon, B.R.; Gomez, M.A. Less rigid internal fixation plates: Historical perspectives and new concepts. J. Orthop. Res. 1983, 1, 431–449. [Google Scholar] [CrossRef]
- Semlitsch, M. Concept and material properties of a cementless hip prosthesis system with Al2O3 ceramic ball heads and wrought Ti-6Al-4V Stems. Arch. Orthop. Trauma. Surg. 1982, 100, 229–236. [Google Scholar] [CrossRef]
- Henk, C.B.; Brodner, W.; Grampp, S.; Breitenseher, M.; Thurnher, M.; Mostbeck, G.H.; Imhof, H. The Postoperative Spine. Top. Magn. Reson. Imaging 1999, 10, 247–264. [Google Scholar] [CrossRef]
- Li, Q.; Min, X.; Bai, P.; Wang, W.; Tao, X.; Zhong, G.; Bai, S.; Zhao, J. Microstructure, mechanical properties and springback behaviour of Ti-6Al-4V alloy connection rod for spinal fixation device. Mater. Sci. Eng. C 2018, 94, 811–820. [Google Scholar] [CrossRef]
- García-Gareta, E.; Hua, J.; Orera, A.; Kohli, N.; Knowles, J.C.; Blunn, G.W. Biomimetic surface functionalization of clinically relevant metals used as orthopaedic and dental implants. Biomed. Mater. 2017, 13, 015008. [Google Scholar] [CrossRef]
- Head, W.C.; Bauk, D.J.; Emerson, R.H. Titanium as the material of choice for cementless femoral components in total hip arthroplasty. Clin. Orthop. Relat. Res. 1995, 311, 85–90. [Google Scholar]
- Radley, G.; Pieper, I.L.; Thomas, B.; Hawkins, K.; Thornton, C.A. Artificial shear stress effects on leukocytes at a biomaterial interface. Artif. Organs 2018, 43, E139–E151. [Google Scholar] [CrossRef]
- Perren, S.M.; Regazzoni, P.; Fernandez, A.A. How to Choose between the Implant Materials Steel and Titanium in Orthopaedic Trauma Surgery: Part 2—Biological Aspects. Acta Chir. Orthop. Traumatol. Cech. 2017, 84, 85–90. [Google Scholar]
- Shah, F.A.; Trobos, M.; Thomsen, P.; Palmquist, A. Commercially pure titanium (cp-Ti) versus titanium alloy (Ti6Al4V) materials as bone anchored implants—Is one truly better than the other? Mater. Sci. Eng. C 2016, 62, 960–966. [Google Scholar] [CrossRef]
- Wypych, A.; Siwak, P.; Andrzejewski, D.; Jakubowicz, J. Titanium Plasma-Sprayed Coatings on Polymers for Hard Tissue Applications. Materials 2018, 11, 2536. [Google Scholar] [CrossRef] [Green Version]
- Kopova, I.; Stráský, J.; Harcuba, P.; Landa, M.; Janeček, M.; Bačákova, L. Newly developed Ti–Nb–Zr–Ta–Si–Fe biomedical beta titanium alloys with increased strength and enhanced biocompatibility. Mater. Sci. Eng. C 2016, 60, 230–238. [Google Scholar] [CrossRef]
- Abdelrhman, Y.; Gepreel, M.A.-H.; Kobayashi, S.; Okano, S.; Okamoto, T. Biocompatibility of new low-cost (α + β)-type Ti-Mo-Fe alloys for long-term implantation. Mater. Sci. Eng. C 2019, 99, 552–562. [Google Scholar] [CrossRef]
- Nnamchi, P.; Obayi, C.; Todd, I.; Rainforth, M. Mechanical and electrochemical characterisation of new Ti–Mo–Nb–Zr alloys for biomedical applications. J. Mech. Behav. Biomed. Mater. 2016, 60, 68–77. [Google Scholar] [CrossRef]
- Ghosh, S.; Abanteriba, S.; Wong, S.; Houshyar, S. Selective laser melted titanium alloys for hip implant applications: Surface modification with new method of polymer grafting. J. Mech. Behav. Biomed. Mater. 2018, 87, 312–324. [Google Scholar] [CrossRef]
- Charnley, J. ARTHROPLASTY OF THE HIP A New Operation. Lancet 1961, 277, 1129–1132. [Google Scholar] [CrossRef]
- Available online: https://www.guinnessworldrecords.com/news/2019/11/the-inspiring-story-of-the-man-whose-pioneering-hip-replacement-has-lasted-a-reco-597819 (accessed on 1 March 2022).
- Bardos, D.I. Metallurgy of orthopaedic implants. In Materials Sciences and Implant Orthopaedic Surgery; Kossowsky, R., Kossowsky, N., Eds.; Springer: Dordrecht, The Netherlands, 1986. [Google Scholar]
- Gaillard, M.D.; Gross, T.P. Metal-on-metal hip resurfacing in patients younger than 50 years: A retrospective analysis. J. Orthop. Surg. Res. 2017, 12, 79. [Google Scholar] [CrossRef]
- Munemoto, M.; Grammatopoulos, G.; Tanaka, Y.; Gibbons, M.; Athanasou, N.A. The pathology of failed McKee-Farrar implants: Correlation with modern metal-on-metal-implant failure. J. Mater. Sci. Mater. Electron. 2017, 28, 66. [Google Scholar] [CrossRef] [Green Version]
- Ho, J.H.; Leikin, J.B.; Dargan, P.I.; Archer, J.R.H.; Wood, D.M.; Brent, J. Metal-on-Metal Hip Joint Prostheses: A Retrospective Case Series Investigating the Association of Systemic Toxicity with Serum Cobalt and Chromium Concentrations. J. Med. Toxicol. 2017, 13, 321–328. [Google Scholar] [CrossRef]
- Morgan, E.F.; Unnikrisnan, G.U.; Hussein, A.I. Bone Mechanical Properties in Healthy and Diseased States. Annu. Rev. Biomed. Eng. 2018, 20, 119–143. [Google Scholar] [CrossRef]
- Lakes, R. Elastic Anisotropy of Bone. Available online: http://silver.neep.wisc.edu/~lakes/BoneAniso.html (accessed on 1 March 2022).
- Navarro, M.; Michiardi, A.; Castaño, O.; Planell, J.A. Biomaterials in orthopaedics. J. R. Soc. Interface 2008, 5, 1137–1158. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.-C.; Chen, L.-Y. A Review on Biomedical Titanium Alloys: Recent Progress and Prospect. Adv. Eng. Mater. 2019, 21, 1801215. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Klemm, D.; Eckert, J.; Hao, Y.; Sercombe, T. Manufacture by selective laser melting and mechanical behavior of a biomedical Ti–24Nb–4Zr–8Sn alloy. Scr. Mater. 2011, 65, 21–24. [Google Scholar] [CrossRef]
- Niinomi, M. Mechanical properties of biomedical titanium alloys. Mater. Sci. Eng. A 1998, 243, 231–236. [Google Scholar] [CrossRef]
- Davis, J. Handbook of Materials for Medical Devices; ASTM International: Novelty, OH, USA, 2003. [Google Scholar]
- Chen, Q.; Thouas, G.A. Metallic implant biomaterials. Mater. Sci. Eng. R Rep. 2015, 87, 1–57. [Google Scholar] [CrossRef]
- Biesiekierski, A.; Lin, J.; Li, Y.; Ping, D.; Yamabe-Mitarai, Y.; Wen, C. Investigations into Ti–(Nb,Ta)–Fe alloys for biomedical applications. Acta Biomater. 2016, 32, 336–347. [Google Scholar] [CrossRef]
- Santos, P.F.; Niinomi, M.; Liu, H.; Cho, K.; Nakai, M.; Trenggono, A.; Champagne, S.; Hermawan, H.; Narushima, T. Improvement of microstructure, mechanical and corrosion properties of biomedical Ti-Mn alloys by Mo addition. Mater. Des. 2016, 110, 414–424. [Google Scholar] [CrossRef]
- Gepreel, M.A.-H. Improved Elasticity of New Ti-Alloys for Biomedical Applications. Mater. Today Proc. 2015, 2, S979–S982. [Google Scholar] [CrossRef]
- Kusano, Y.; Inamura, T.; Kanetaka, H.; Miyazaki, S.; Hosoda, H. Phase Constitution and Mechanical Properties of Ti-(Cr, Mn)-Sn Biomedical Alloys. Mater. Sci. Forum 2010, 654-656, 2118–2121. [Google Scholar] [CrossRef]
- Talling, R.; Dashwood, R.; Jackson, M.; Dye, D. Compositional variability in gum metal. Scr. Mater. 2009, 60, 1000–1003. [Google Scholar] [CrossRef]
- Ijaz, M.F.; Héraud, L.; Castany, P.; Thibon, I.; Gloriant, T. Superelastic Behavior of Biomedical Metallic Alloys. Met. Mater. Trans. A 2020, 51, 3733–3741. [Google Scholar] [CrossRef]
- ASTM F75-18; Standard Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Castings and Casting Alloy for Surgical Implants (UNS R30075). ASTM International: West Conshohocken, PA, USA, 2018. Available online: www.astm.org (accessed on 1 March 2022).
- ASTM F1537-20; Standard Specification for Wrought Cobalt-28Chromium-6Molybdenum Alloys for Surgical Implants (UNS R31537, UNS R31538, and UNS R31539). ASTM International: West Conshohocken, PA, USA, 2020.
- ASTM F799-19; Standard Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Forgings for Surgical Implants (UNS R31537, R31538, R31539). ASTM International: West Conshohocken, PA, USA, 2019. Available online: www.astm.org (accessed on 1 March 2022).
- Catanese, J., III; Cooke, D.; Maas, C.; Pruitt, L. Mechanical properties of medical grade expanded polytetrafluoroethylene: The effects of internodal distance, density, and displacement rate. J. Biomed. Mater. Res. 2002, 48, 187–192. [Google Scholar] [CrossRef]
- Kocadal, O.; Ozler, T.; Bolukbasi, A.E.T.; Altintas, F. Non-traumatic Ceramic Head Fracture in Total Hip Arthroplasty with Ceramic-on-Ceramic Articulation at Postoperative 16th Years. Hip Pelvis 2019, 31, 124–127. [Google Scholar] [CrossRef]
- Sentuerk, U.; Von Roth, P.; Perka, C.; Surgeon, O. Ceramic on ceramic arthroplasty of the hip. Bone Jt. J. 2016, 98-B, 14–17. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Lawn, B. Novel Zirconia Materials in Dentistry. J. Dent. Res. 2017, 97, 140–147. [Google Scholar] [CrossRef]
- D’Addazio, G.; Santilli, M.; Rollo, M.L.; Cardelli, P.; Rexhepi, I.; Murmura, G.; Husain, N.A.-H.; Sinjari, B.; Traini, T.; Özcan, M.; et al. Fracture Resistance of Zirconia-Reinforced Lithium Silicate Ceramic Crowns Cemented with Conventional or Adhesive Systems: An In Vitro Study. Materials 2020, 13, 2012. [Google Scholar] [CrossRef]
- Mussano, F.; Genova, T.; Munaron, L.; Faga, M.G.; Carossa, S. Ceramic Biomaterials for Dental Implants: Current Use and Future Perspectives. In Dental Implantology and Biomaterial; Jawad, M.A., Almasri, A., Eds.; Intech: London, UK, 2016; Available online: https://www.intechopen.com/books/dental-implantology-and-biomaterial/ceramic-biomaterials-for-dental-implants-current-use-and-future-perspectives (accessed on 1 March 2022).
- De Freitas, B.X.; Alves, M.F.R.P.; Santos, C.; Ramos, A.S.; Ramos, E.C.T.; Strecker, K. Mechanical properties of biocompatible Y-TZP/Al2O3 composites obtained from mechanically alloyed powders. J. Braz. Soc. Mech. Sci. Eng. 2020, 42, 353. [Google Scholar] [CrossRef]
- Duraccio, D.; Mussano, F.; Faga, M.G. Biomaterials for dental implants: Current and future trends. J. Mater. Sci. 2015, 50, 4779–4812. [Google Scholar] [CrossRef]
- Faga, M.; Vallée, A.; Bellosi, A.; Mazzocchi, M.; Thinh, N.N.; Martra, G.; Coluccia, S. Chemical treatment on alumina–zirconia composites inducing apatite formation with maintained mechanical properties. J. Eur. Ceram. Soc. 2012, 32, 2113–2120. [Google Scholar] [CrossRef]
- Spies, B.C.; Sauter, C.; Wolkewitz, M.; Kohal, R.-J. Alumina reinforced zirconia implants: Effects of cyclic loading and abutment modification on fracture resistance. Dent. Mater. 2015, 31, 262–272. [Google Scholar] [CrossRef]
- Harrison, J.H.; Swanson, D.S.; Lincoln, A.F. A Comparison of the Tissue Reactions to Plastic Materials. AMA Arch. Surg. 1957, 74, 139–144. [Google Scholar] [CrossRef]
- Ludington, L.G.; Woodward, E.R. Use of teflon mesh in the repair of musculofascial defects. Surgery 1959, 46, 364–373. [Google Scholar]
- Butler, H.C. Teflon as a prosthetic ligament in repair of ruptured anterior cruciate ligaments. Am. J. Vet. Res. 1964, 25, 55–60. [Google Scholar]
- Williams, R.D.; August, S.F. Experimental evaluation of a Teflon tendon prosthesis. Am. J. Surg. 1964, 107, 913–916. [Google Scholar] [CrossRef]
- Charnley, J. Using Teflon in arthroplasty of the hip-joint. J. Bone Jt. Surg. Am. 1966, 48, 819. [Google Scholar] [CrossRef]
- Trubnikov, V.F. Primenenie plastmassy tetraftoretilena (ftoroplast-4) s tseliu osteosinteza. Ortop. Travmatol. Protez. 1963, 24, 25–30. [Google Scholar]
- Burns, K.M. The “Fluon” arthroplasty: Treatment note. Physiotherapy 1963, 49, 182. [Google Scholar]
- Buechel, F.F.; Pappas, M.J. Properties of Materials Used in Orthopaedic Implant Systems. In Principles of Human Joint Replacement; Springer: Cham, Swizterland, 2015. [Google Scholar] [CrossRef]
- Von Pechmann, H. Über Diazomethan und Nitrosoacylamine. Berichte der Deutschen Chemischen Gesellschaft zu Berlin 1898, 31, 2640–2646. [Google Scholar] [CrossRef] [Green Version]
- Kalia, S.; Avérous, L. Biopolymers: Biomedical and Environmental Applications; Wiley: Hoboken, NJ, USA, 2011; ISBN 978-0-470-63923-8. [Google Scholar]
- Ruso, J.M.; Messina, P.V. Biopolymers for Medical Applications; CRC Press: Boca Raton, FL, USA, 2021; ISBN 9780367782702. [Google Scholar]
- Li, S.; Burstein, A.H. Ultra-high molecular weight polyethylene. The material and its use in total joint implants. J. Bone Jt. Surg. 1994, 76, 1080–1090. [Google Scholar] [CrossRef]
- Kurtz, S.M. (Ed.) The UHMWPE Handbook: Ultra-High Molecular Weight Polyethylene in Total Joint Replacement; Elsevier: San Diego, CA, USA, 2004; ISBN 978-0-12-429851-4. [Google Scholar]
- Veruva, S.Y.; Lanman, T.H.; Isaza, J.E.; MacDonald, D.W.; Kurtz, S.M.; Steinbeck, M.J. UHMWPE Wear Debris and Tissue Reactions Are Reduced for Contemporary Designs of Lumbar Total Disc Replacements. Clin. Orthop. Relat. Res. 2015, 473, 987–998. [Google Scholar] [CrossRef] [Green Version]
- Dowling, J.; Atkinson, J.R.; Dowson, D.; Charnley, J. The characteristics of acetabular cups worn in the human body. J. Bone Jt. Sur. 1978, 60-B, 375–382. [Google Scholar] [CrossRef]
- Geerdink, C.H.; Grimm, B.; Vencken, W.; Heyligers, I.C.; Tonino, A.J. Cross-linked Compared with Historical Polyethylene in THA: An 8-year Clinical Study. Clin. Orthop. Relat. Res. 2008, 467, 979–984. [Google Scholar] [CrossRef] [Green Version]
- Deng, M.; Latour, R.A.; Ogale, A.A.; Shalaby, S.W. Study of creep behavior of ultra-high-molecular-weight polyethylene systems. J. Biomed. Mater. Res. 1998, 40, 214–223. [Google Scholar] [CrossRef]
- Judet, R. Hip Reconstruction by Acrylic Prosthesis. BMJ 1951, 2, 1401. [Google Scholar] [CrossRef] [Green Version]
- Ali, U.; Karim, K.J.B.A.; Buang, N.A. A Review of the Properties and Applications of Poly (Methyl Methacrylate) (PMMA). Polym. Rev. 2015, 55, 678–705. [Google Scholar] [CrossRef]
- Kutz, M. Handbook of Materials Selection; Wiley: Hoboken, NJ, USA, 2002; 341p. [Google Scholar]
- Frazer, R.Q.; Byron, R.T.; Osborne, P.B.; West, K.P. PMMA: An Essential Material in Medicine and Dentistry. J. Long-term Eff. Med Implant. 2005, 15, 629–639. [Google Scholar] [CrossRef] [Green Version]
- İzmir, M.; Ercan, B. Anodization of titanium alloys for orthopedic applications. Front. Chem. Sci. Eng. 2019, 13, 28–45. [Google Scholar] [CrossRef]
- Łapaj, Ł.; Rozwalka, J. Retrieval analysis of TiN (titanium nitride) coated knee replacements: Coating wear and degradation in vivo. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 108, 1251–1261. [Google Scholar] [CrossRef]
- Zhang, X.; Lv, Y.; Fu, S.; Wu, Y.; Lu, X.; Yang, L.; Liu, H.; Dong, Z. Synthesis, microstructure, anti-corrosion property and biological performances of Mn-incorporated Ca-P/TiO2 composite coating fabricated via micro-arc oxidation. Mater. Sci. Eng. C 2020, 117, 111321. [Google Scholar] [CrossRef]
- Milan, P.B.; Khamseh, S.; Zarintaj, P.; Ramezanzadeh, B.; Badawi, M.; Morisset, S.; Vahabi, H.; Saeb, M.R.; Mozafari, M. Copper-enriched diamond-like carbon coatings promote regeneration at the bone–implant interface. Heliyon 2020, 6, e03798. [Google Scholar] [CrossRef]
- Aviles, T.; Hsu, S.-M.; Clark, A.; Ren, F.; Fares, C.; Carey, P.H., IV; Esquivel-Upshaw, J.F. Hydroxyapatite Formation on Coated Titanium Implants Submerged in Simulated Body Fluid. Materials 2020, 13, 5593. [Google Scholar] [CrossRef]
- Ong, J.; Chan, D.C.N. A Review of Hydroxapatite and its use as a Coating in Dental Implants. Crit. Rev. Biomed. Eng. 2017, 45, 411–451. [Google Scholar] [CrossRef]
- Van Hove, R.P.; Sierevelt, I.N.; van Royen, B.J.; Nolte, P.A. Titanium-Nitride Coating of Orthopaedic Implants: A Review of the Literature. BioMed Res. Int. 2015, 2015, 485975. [Google Scholar] [CrossRef] [Green Version]
- Zielinski, J.; Lacy, T.A.; Phillips, J.H. Carbon Coated Implants as a New Solution for Metal Allergy in Early-Onset Scoliosis: A Case Report and Review of the Literature. Spine Deform. 2014, 2, 76–80. [Google Scholar] [CrossRef]
- Zhang, M.; Xie, T.; Qian, X.; Zhu, Y.; Liu, X. Mechanical Properties and Biocompatibility of Ti-doped Diamond-like Carbon Films. ACS Omega 2020, 5, 22772–22777. [Google Scholar] [CrossRef]
- Döring, J.; Crackau, M.; Nestler, C.; Welzel, F.; Bertrand, J.; Lohmann, C.H. Characteristics of different cathodic arc deposition coatings on CoCrMo for biomedical applications. J. Mech. Behav. Biomed. Mater. 2019, 97, 212–221. [Google Scholar] [CrossRef]
- Gallo, J.; Holinka, M.; Moucha, C.S. Antibacterial Surface Treatment for Orthopaedic Implants. Int. J. Mol. Sci. 2014, 15, 13849–13880. [Google Scholar] [CrossRef] [Green Version]
- Bhusari, S.A.; Sharma, V.; Bose, S.; Basu, B. HDPE/UHMWPE hybrid nanocomposites with surface functionalized graphene oxide towards improved strength and cytocompatibility. J. R. Soc. Interface 2019, 16, 20180273. [Google Scholar] [CrossRef] [Green Version]
- Harrasser, N.; Jüssen, S.; Obermeier, A.; Kmeth, R.; Stritzker, B.; Gollwitzer, H.; Burgkart, R. Antibacterial potency of different deposition methods of silver and copper containing diamond-like carbon coated polyethylene. Biomater. Res. 2016, 20, 17. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://www.daido.co.jp/en/products/titanium/medical/index.html (accessed on 1 March 2022).
- Wang, X.; Dong, H.; Liu, J.; Qin, G.; Chen, D.; Zhang, E. In vivo antibacterial property of Ti-Cu sintered alloy implant. Mater. Sci. Eng. C 2019, 100, 38–47. [Google Scholar] [CrossRef]
- Shi, A.; Zhu, C.; Fu, S.; Wang, R.; Qin, G.; Chen, D.; Zhang, E. What controls the antibacterial activity of Ti-Ag alloy, Ag ion or Ti2Ag particles? Mater. Sci. Eng. C 2019, 109, 110548. [Google Scholar] [CrossRef]
- Alt, V. Antimicrobial coated implants in trauma and orthopaedics–A clinical review and risk-benefit analysis. Injury 2017, 48, 599–607. [Google Scholar] [CrossRef]
- Al-Zain, Y.; Yamamoto, A.; AlAjlouni, J.M.; Al-Abbadi, M.A.; Al-Sayyed, M.R.; Aloweidi, A.S.; Kim, H.Y.; Miyazaki, S. Corrosion behavior, in vitro and in vivo biocompatibility of a newly developed Ti–16Nb–3Mo–1Sn superelastic alloy. Mater. Sci. Eng. C 2019, 104, 109906. [Google Scholar] [CrossRef]
- Xue, P.; Li, Y.; Li, K.; Zhang, D.; Zhou, C. Superelasticity, corrosion resistance and biocompatibility of the Ti–19Zr–10Nb–1Fe alloy. Mater. Sci. Eng. C 2015, 50, 179–186. [Google Scholar] [CrossRef]
- Nguyen, Q.P.; Deng, T.Z.; Witherden, D.A.; Goldrath, A.W. Origins of CD4+ circulating and tissue-resident memory T-cells. Immunology 2019, 157, 3–12. [Google Scholar] [CrossRef] [Green Version]
- Shin, H.; Iwasaki, A. Tissue-resident memory T cells. Immunol. Rev. 2013, 255, 165–181. [Google Scholar] [CrossRef] [Green Version]


| Implant Type | Yield Point [MPa] | Ultimate Tensile Strength [MPa] | Young’s Modulus [GPa] | Elongation [%] | References | |
|---|---|---|---|---|---|---|
| Bone | bone | 130–205 MPa | 17.9–18.2 | [42,43] | ||
| Steel | 316 L | 170–750 | 465–950 | 205–210 | 30–70 | [44] |
| Ti and alloys | CP-titanium | 170–480 | 240–550 | 105 | 15–24 | [45,46,47,48,49,50,51,52,53,54,55] |
| Ti6Al4V | 795–875 | 895–965 | 100–114 | 10 | ||
| Ti6Al7Nb | 795 | 860 | 105 | 10 | ||
| Ti5Al2.5Fe | 820 | 900 | 110 | 6 | ||
| Ti3Al2.5V | 585 | 690 | 100 | 15 | ||
| Ti13Nb13Zr | 836–908 | 937–1037 | 79–84 | 42–44 | ||
| Ti12Mo6Zr2Fe | 1000–1060 | 1060–1100 | 14–85 | 18–22 | ||
| Ti24Nb4Zr8Sn | 570–700 | 755–830 | 46–55 | 13–15 | ||
| CoCrMo alloys | Cast 28Co6CrMo | 450 | 655 | 210–250 | 8 | [56,57,58] |
| Wrought Co28Cr6Mo | 517–827 | 897–1192 | 220 | 12–20 | ||
| Co28Cr6Mo Forging | 827 | 1172 | 220–230 | 12 | ||
| PTFE | PTFE | 4.6–7.8 | 7.8–11.1 | 42–59 | 20–29 | [59] |
| Implant Type | Density [g/cm3] | Microhardness [HV] | Young’s Modulus [GPa] | Bending Strength [MPa] | Toughness K1C [MPa × m1/2] | References |
|---|---|---|---|---|---|---|
| Y-ZPT | 6 | 1000–1300 | 200 | 1200 | 9–10 | [66,67,68] |
| zirconia-toughened alumina (ZTA) | 1460–1620 | 236–254 | 500–760 | 7–7.2 | ||
| alumina-toughened zirconia (AZT) | 5.5 | 2000–2200 | 358–368 | 420–460 | 3.9 |
| Material | Advantages | Disadvantages |
|---|---|---|
| Steel | High material strength Good ductility | Corrosive Aseptic loosening Inadequate wear resistance |
| Titanium alloys | High biocompatibility Low density Corrosion resistance | Poor tribological properties Low wear resistance Toxic effect of aluminium and vanadium |
| CoCrMo alloys | High material strength High wear resistance Corrosion resistance | Allergy consideration with nickel, chrome and cobalt |
| Ceramics | High hardness Wear resistance Good wettability Good biocompatibility | Brittle High stiffness Low flexibility |
| Polymers | Low density Biodegradable Easy fabrication Flexible | Hard to sterilise Poor tribological properties Absorb water and proteins |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szczęsny, G.; Kopec, M.; Politis, D.J.; Kowalewski, Z.L.; Łazarski, A.; Szolc, T. A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present. Materials 2022, 15, 3622. https://doi.org/10.3390/ma15103622
Szczęsny G, Kopec M, Politis DJ, Kowalewski ZL, Łazarski A, Szolc T. A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present. Materials. 2022; 15(10):3622. https://doi.org/10.3390/ma15103622
Chicago/Turabian StyleSzczęsny, Grzegorz, Mateusz Kopec, Denis J. Politis, Zbigniew L. Kowalewski, Adam Łazarski, and Tomasz Szolc. 2022. "A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present" Materials 15, no. 10: 3622. https://doi.org/10.3390/ma15103622
APA StyleSzczęsny, G., Kopec, M., Politis, D. J., Kowalewski, Z. L., Łazarski, A., & Szolc, T. (2022). A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present. Materials, 15(10), 3622. https://doi.org/10.3390/ma15103622









