Physicochemical Properties and Inductive Effect of Calcium Strontium Silicate on the Differentiation of Human Dental Pulp Stem Cells for Vital Pulp Therapies: An In Vitro Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis Procedures of the Experimental Cements
2.2. Characterization of the Synthesized Powder
2.3. Samples Preparation
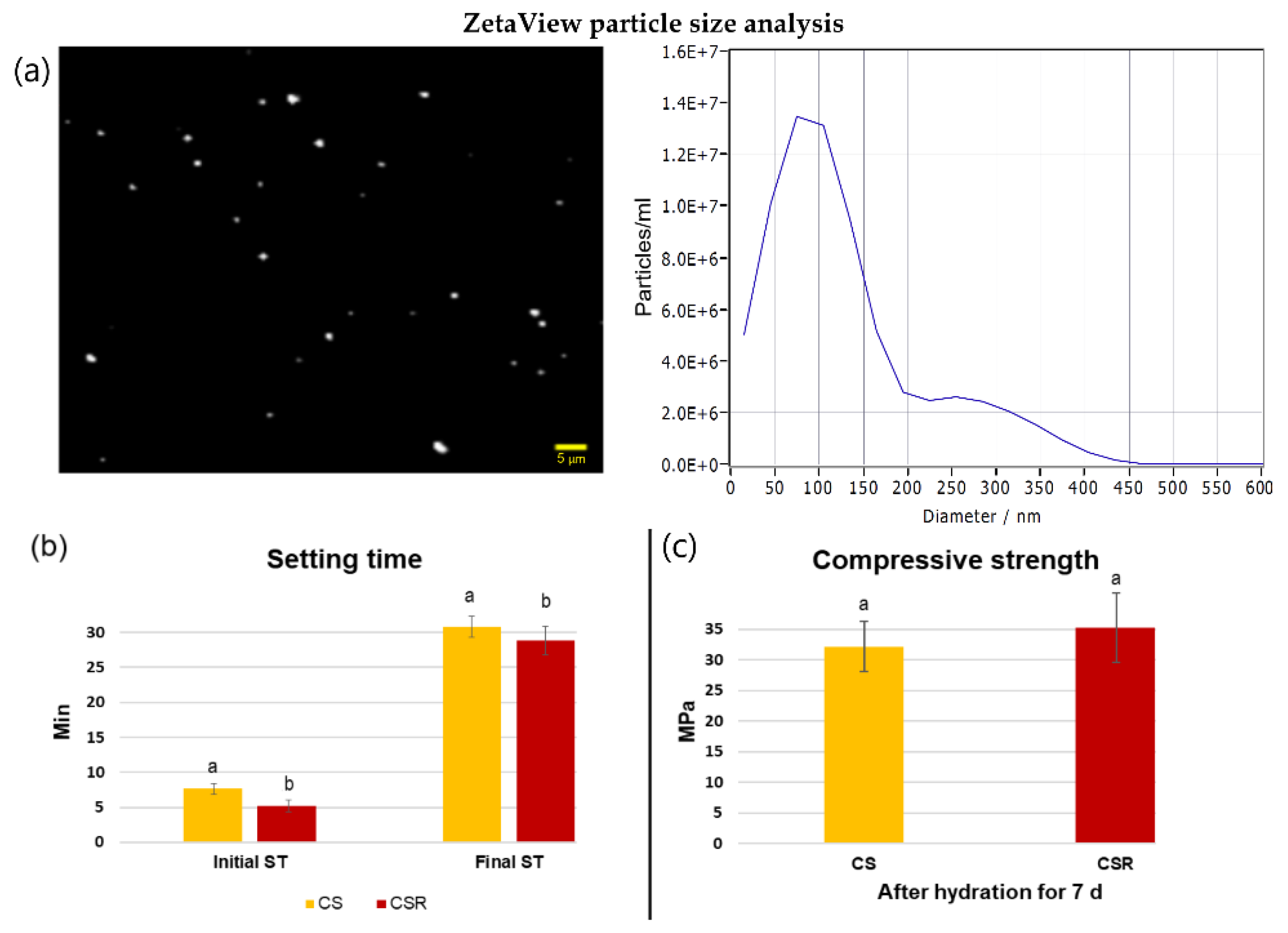
2.4. ZetaView Particle Size Analyzer
2.5. Setting Time
2.6. Compressive Strength Test
2.7. pH Measurement
2.8. In Vitro Apatite Formation and Characterization of the Formed Apatite Layer
2.9. Cell Viability and MTT Assay
2.10. Differentiation of HDPSC
2.10.1. Alkaline Phosphatase Activity
2.10.2. Alizarin Red Staining
2.11. Ion Release
2.12. Statistical Analysis
3. Results
3.1. Powder Characterization
3.2. Particle Size Analysis
3.3. Setting Time
3.4. Compressive Strength
3.5. pH Values
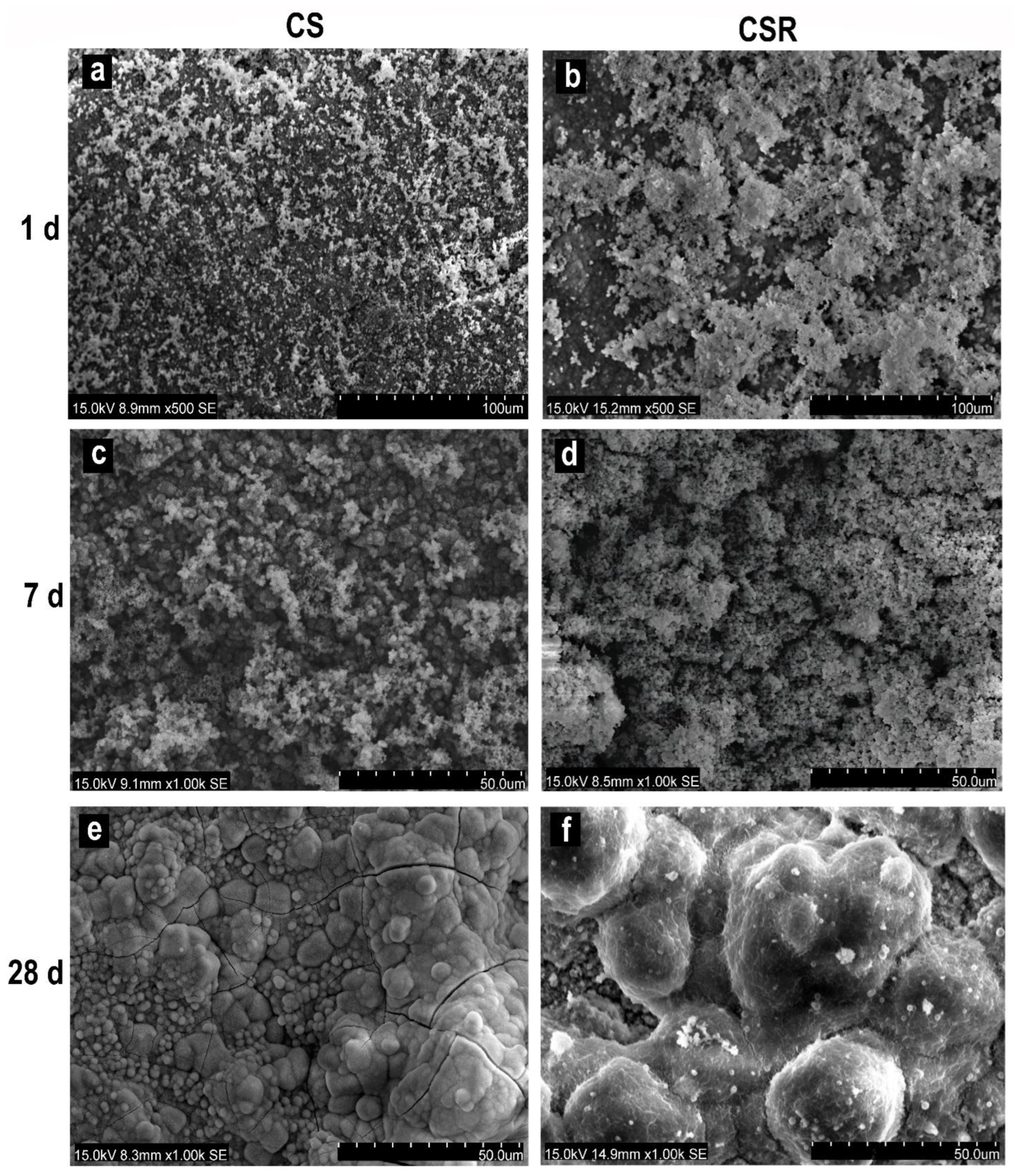
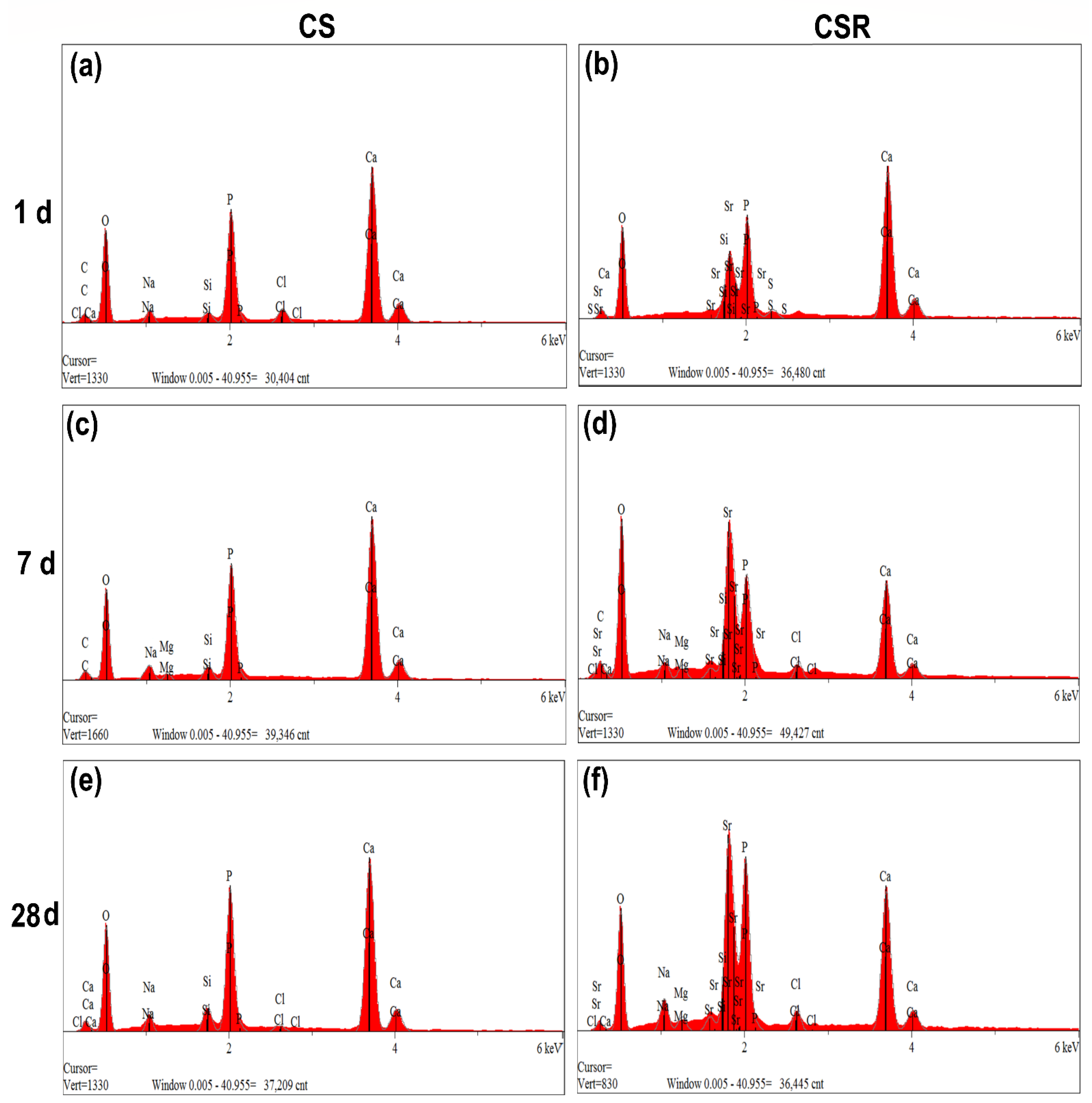
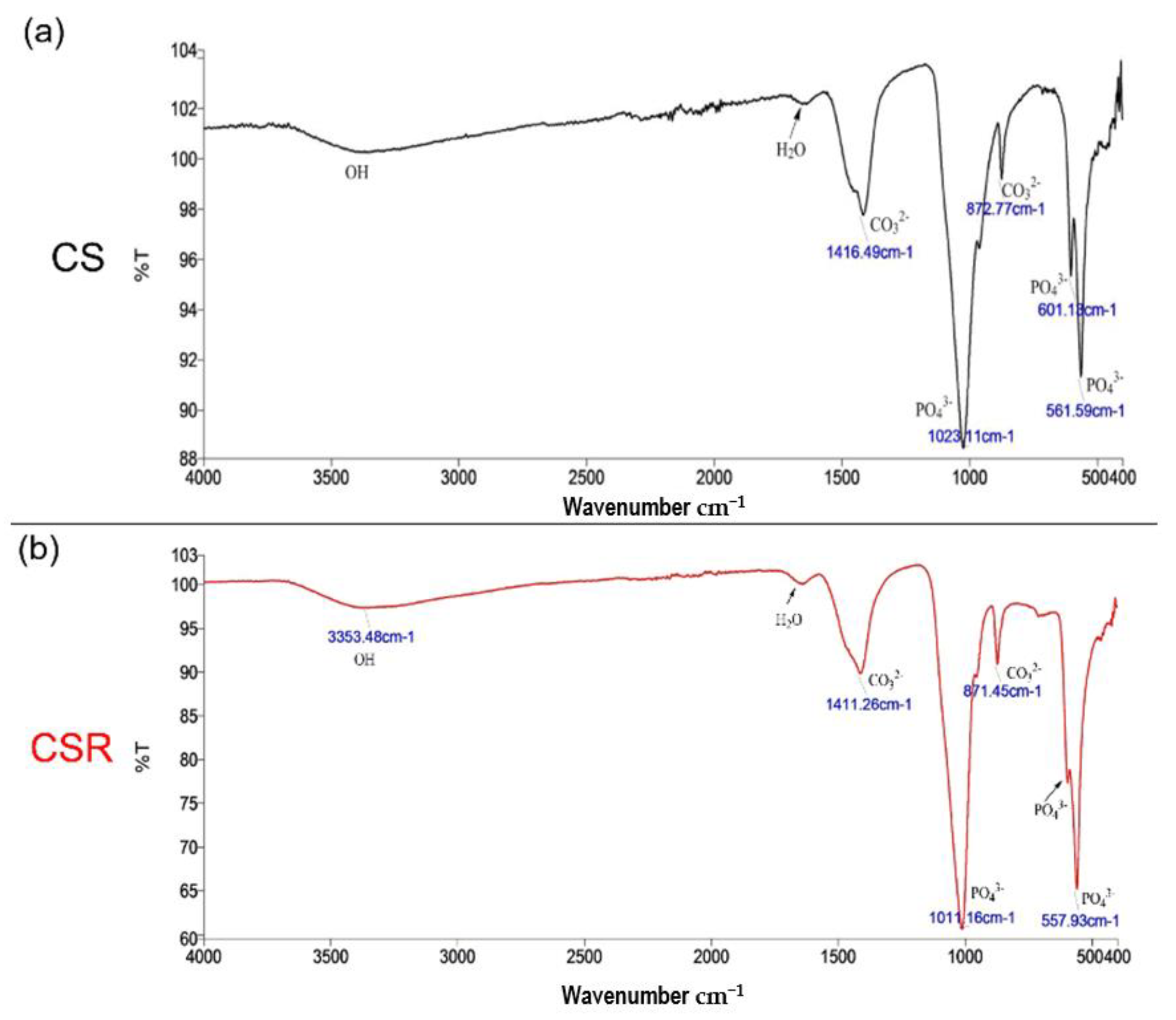
3.6. In Vitro Apatite Formation
3.6.1. SEM Observation
3.6.2. FTIR and EDX Analysis
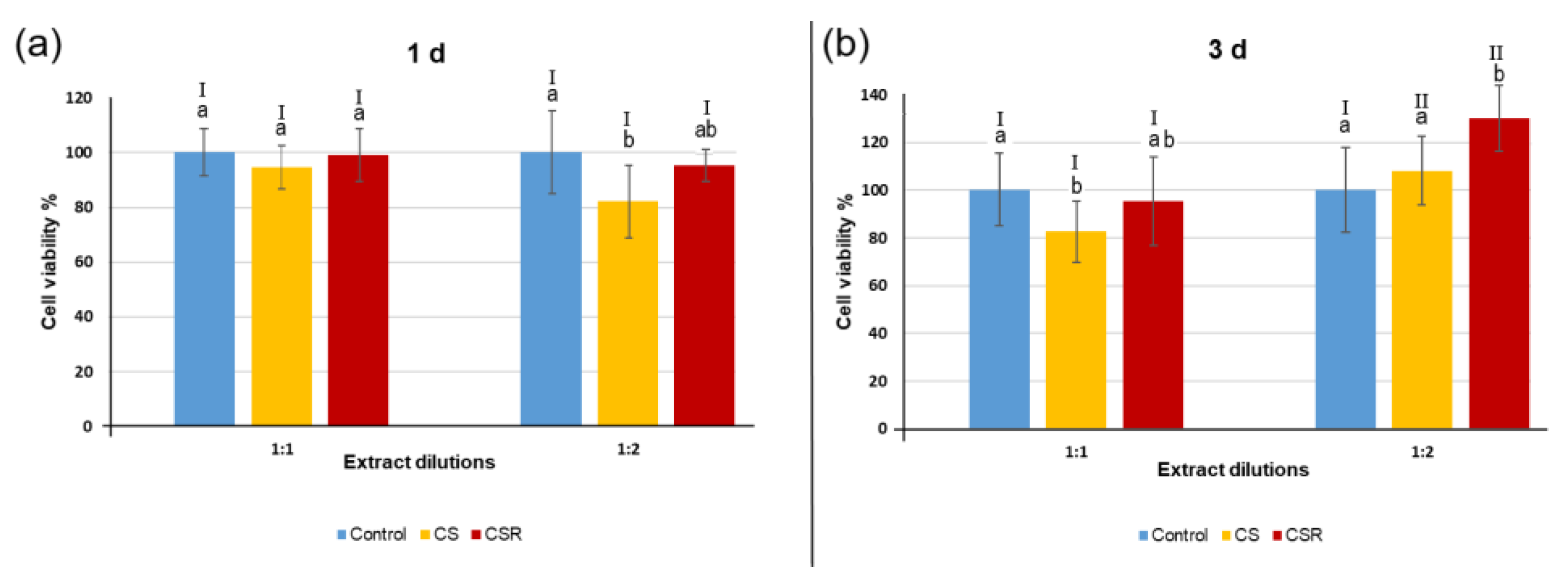
3.7. Cell Viability
3.8. HDPSC Differentiation Assays
3.8.1. Alkaline Phosphatase Activity
3.8.2. Alizarin Red Staining
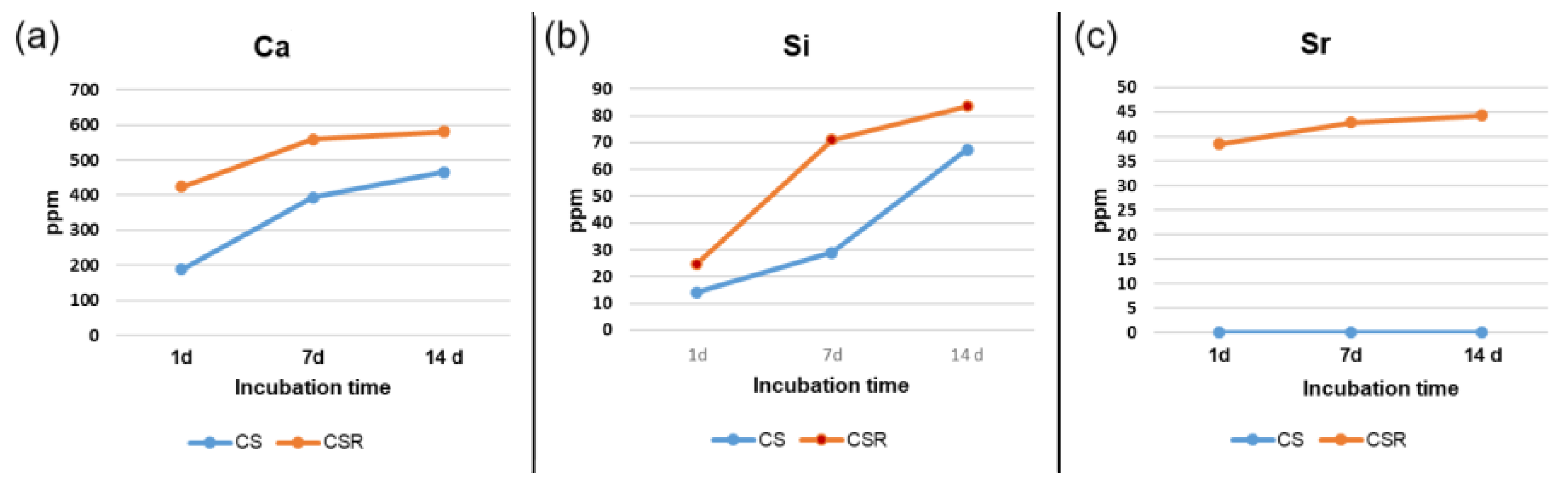
3.9. ICP-OES
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALP | Alkaline phosphatase |
| ANOVA | Analysis of variance |
| ARS | Alizarin red stain |
| CS | Calcium silicate |
| CSR | Calcium strontium silicate |
| DIW | Deionized water |
| DM | Differentiation medium |
| DMEM | Dulbecco Modified Eagle Medium |
| FTIR | Fourier-transformed infrared |
| HDPSC | Human dental pulp stem cells |
| ICP-OES | Inductively coupled plasma-optical emission spectrometry |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| OD | Optical density |
| RM | Regular medium |
| XRD | X-ray diffractometer |
| α-MEM | α-minimal essential medium |
References
- Amrollahi, B.P.; Shah, A.; Seifi, L. Tayebi, Recent advancements in regenerative dentistry: A review. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 69, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Vallittu, P.K.; Boccaccini, A.R.; Hupa, L.; Watts, D.C. Bioactive dental materials-Do they exist and what does bioactivity mean? Dent. Mater. 2018, 34, 693–694. [Google Scholar] [CrossRef]
- Saghiri, M.A.; Orangi, J.; Asatourian, A.; Gutmann, J.L.; Garcia-Godoy, F.; Lotfi, M.; Sheibani, N. Calcium silicate-based cements and functional impacts of various constituents. Dent. Mater. J. 2017, 36, 8–18. [Google Scholar] [CrossRef]
- De Oliveira, N.G.; de Souza Araújo, P.R.; Da Silveira, M.T.; Sobral, A.P.V.; de Vasconcelos Carvalho, M. Comparison of the biocompatibility of calcium silicate-based materials to mineral trioxide aggregate: Systematic review. Eur. J. Dent. 2018, 12, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Gronthos, S.; Shi, S. Dental pulp stem cells. Methods Enzym. 2006, 419, 99–113. [Google Scholar]
- Tatullo, M.; Marrelli, M.; Shakesheff, K.M.; White, L.J. Dental pulp stem cells: Function, isolation and applications in regenerative medicine. J. Tissue Eng. Regen. Med. 2015, 9, 1205–1216. [Google Scholar] [CrossRef]
- Raghavendra, S.S.; Jadhav, G.R.; Gathani, K.M.; Kotadia, P. Bioceramics in endodontics—A review. J. Istanb. Univ. Fac. Dent. 2017, 51, S128–S137. [Google Scholar] [CrossRef]
- Li, K.; Yu, J.; Xie, Y.; Huang, L.; Ye, X.; Zheng, X. Chemical stability and antimicrobial activity of plasma sprayed bioactive Ca2ZnSi2O7 coating. J. Mater. Sci. Mater. Electron. 2011, 22, 2781–2789. [Google Scholar] [CrossRef]
- Liu, W.C.; Wang, H.-Y.; Chen, L.-C.; Huang, S.-W.; Wu, C.; Chung, R.-J. Hydroxyapatite/tricalcium silicate composites cement derived from novel two-step sol-gel process with good biocompatibility and applications as bone cement and potential coating materials. Ceram. Int. 2019, 45, 5668–5679. [Google Scholar] [CrossRef]
- Abdalla, M.M.; Lung, C.Y.K.; Neelakantan, P.; Matinlinna, J.P. A novel, doped calcium silicate bioceramic synthesized by sol–gel method: Investigation of setting time and biological properties. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 56–66. [Google Scholar] [CrossRef]
- Kaur, G.; Pickrell, G.; Sriranganathan, N.; Kumar, V.; Homa, D. Review and the state of the art: Sol–gel and melt quenched bioactive glasses for tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 1248–1275. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; de Groot, K. Better bioactive ceramics through sol-gel process. J. Sol-Gel Sci. Technol. 1994, 2, 797–801. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Montalvo-González, E.; González-Silva, N.; Méndez-Robles, M.D.; Romero-Toledo, R.; Yahia, E.M.; Pérez-Larios, A. Synthesis and Characterization of TiO2-ZnO-MgO Mixed Oxide and Their Antibacterial Activity. Materials 2019, 12, 698. [Google Scholar] [CrossRef]
- Hu, D.; Li, K.; Xie, Y.; Pan, H.; Zhao, J.; Huang, L.; Zheng, X. Different response of osteoblastic cells to Mg2+, Zn2+ and Sr2+ doped calcium silicate coatings. J. Mater. Sci. Mater. Med. 2016, 27, 56. [Google Scholar] [CrossRef] [PubMed]
- No, Y.J.; Li, J.J.; Zreiqat, H. Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes. Materials 2017, 10, 153. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Hill, R.G.; Rawlinson, S.C. Strontium (Sr) elicits odontogenic differentiation of human dental pulp stem cells (hDPSCs): A therapeutic role for Sr in dentine repair? Acta Biomater. 2016, 38, 201–211. [Google Scholar] [CrossRef]
- Lin, K.; Xia, L.; Li, H.; Jiang, X.; Pan, H.; Xu, Y.; Lu, W.W.; Zhang, Z.; Chang, J. Enhanced osteoporotic bone regeneration by strontium-substituted calcium silicate bioactive ceramics. Biomaterials 2013, 34, 10028–10042. [Google Scholar] [CrossRef]
- Xuereb, M.; Sorrentino, F.; Damidot, D.; Camilleri, J. Development of novel tricalcium silicate-based endodontic cements with sintered radiopacifier phase. Clin. Oral Investig. 2016, 20, 967–982. [Google Scholar] [CrossRef]
- Olley, R.; Pilecki, P.; Austin, R.; Moazzez, R.; Bartlett, D. An in-situ study investigating dentine tubule occlusion. Br. Soc. Oral Dent. Res. 2012, 40, 585–593. [Google Scholar]
- Femiano, F.; Femiano, R.; Femiano, L.; Nucci, L.; Minervini, G.; Antonelli, A.; Bennardo, F.; Barone, S.; Scotti, N.; Sorice, V.; et al. A New Combined Protocol to Treat the Dentin Hypersensitivity Associated with Non-Carious Cervical Lesions: A Randomized Controlled Trial. Appl. Sci. 2021, 11, 187. [Google Scholar] [CrossRef]
- Guida, A.; Towler, M.R.; Wall, J.G.; Hill, R.; Eramo, S. Preliminary work on the antibacterial effect of strontium in glass ionomer cements. J. Mater. Sci. Lett. 2003, 22, 1401–1403. [Google Scholar] [CrossRef]
- Lippert, F.; Hara, A. Strontium and Caries: A Long and Complicated Relationship. Caries Res. 2013, 47, 34–49. [Google Scholar] [CrossRef] [PubMed]
- D’Onofrio, A.; Kent, N.; Shahdad, S.; Hill, R. Development of novel strontium containing bioactive glass-based calcium phos-phate cement. Dent. Mater. 2016, 32, 703–712. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, L.; Sun, X.; Chen, X.; Xie, K.; Lin, M.; Yang, G.; Xu, S.; Xia, W.; Gou, Z. Preparation and in vitro evaluation of strontium-doped calcium silicate/gypsum bioactive bone cement. Biomed. Mater. 2014, 9, 045002. [Google Scholar] [CrossRef]
- Huang, T.-H.; Kao, C.-T.; Shen, Y.-F.; Lin, Y.-T.; Liu, Y.-T.; Yen, S.-Y.; Ho, C.-C. Substitutions of strontium in bioactive calcium silicate bone cements stimulate osteogenic differentiation in human mesenchymal stem cells. J. Mater. Sci. Mater. Med. 2019, 30, 68. [Google Scholar] [CrossRef]
- Pelepenko, L.E.; Marciano, M.A.; Francati, T.M.; Bombarda, G.; Bessa Marconato Antunes, T.; Sorrentino, F.; Martin, R.A.; Boanini, E.; Cooper, P.R.; Shelton, R.M.; et al. Can strontium replace calcium in bioactive materials for dental applications? J. Biomed. Mater. Res. A 2022. [Google Scholar] [CrossRef]
- Yan, M.; Yu, Y.; Zhang, G.; Tang, C.; Yu, J. A Journey from Dental Pulp Stem Cells to a Bio-tooth. Stem Cell Rev. Rep. 2011, 7, 161–171. [Google Scholar] [CrossRef]
- Potdar, P.D.; Jethmalani, Y.D. Human dental pulp stem cells: Applications in future regenerative medicine. World J. Stem Cells 2015, 7, 839–851. [Google Scholar] [CrossRef]
- Do Carmo, S.; Néspoli, F.; Bachmann, L.; Miranda, C.; Castro-Raucci, L.; Oliveira, I.; Raucci-Neto, W. Influence of early mineral deposits of silicate-and aluminate-based cements on push-out bond strength to root dentine. Int. Endod. J. 2018, 51, 92–101. [Google Scholar] [CrossRef]
- Wang, C.-W.; Chiang, T.-Y.; Chang, H.-C.; Ding, S.-J. Physicochemical properties, and osteogenic activity of radiopaque calcium silicate–gelatin cements. J. Mater. Sci. Mater. Med. 2014, 25, 2193–2203. [Google Scholar] [CrossRef]
- Lin, M.-C.; Chen, C.-C.; Wu, I.-T.; Ding, S.-J. Enhanced antibacterial activity of calcium silicate-based hybrid cements for bone repair. Mater. Sci. Eng. C 2020, 110, 110727. [Google Scholar] [CrossRef] [PubMed]
- Dissanayaka, W.L.; Zhan, X.; Zhang, C.; Hargreaves, K.M.; Jin, L.; Tong, E.H. Coculture of dental pulp stem cells with endothelial cells enhances osteo-/odontogenic and angiogenic potential in vitro. J. Endod. 2012, 38, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Roi, A.; Ardelean, L.C.; Roi, C.I.; Boia, E.-R.; Boia, S.; Rusu, L.-C. Oral Bone Tissue Engineering: Advanced Biomaterials for Cell Adhesion, Proliferation and Differentiation. Materials 2019, 12, 2296. [Google Scholar] [CrossRef]
- Wei, S.; Ma, J.-X.; Xu, L.; Gu, X.-S.; Ma, X.-L. Biodegradable materials for bone defect repair. Mil. Med. Res. 2020, 7, 1–25. [Google Scholar] [CrossRef]
- Morotomi, T.; Washio, A.; Kitamura, C. Current and future options for dental pulp therapy. Jpn. Dent. Sci. Rev. 2018, 55, 5–11. [Google Scholar] [CrossRef]
- Primus, C.M.; Tay, F.R.; Niu, L.-N. Bioactive tri/dicalcium silicate cements for treatment of pulpal and periapical tissues. Acta Biomater. 2019, 96, 35–54. [Google Scholar] [CrossRef]
- Wongwatanasanti, N.; Jantarat, J.; Sritanaudomchai, H.; Hargreaves, K.M. Effect of Bioceramic Materials on Proliferation and Odontoblast Differentiation of Human Stem Cells from the Apical Papilla. J. Endod. 2018, 44, 1270–1275. [Google Scholar] [CrossRef]
- Liu, W.; Huan, Z.; Xing, M.; Tian, T.; Xia, W.; Wu, C.; Zhou, Z.; Chang, J. Strontium-Substituted Dicalcium Silicate Bone Cements with Enhanced Osteogenesis Potential for Orthopaedic Applications. Materials 2019, 12, 2276. [Google Scholar] [CrossRef]
- Wu, C.; Ramaswamy, Y.; Kwik, D.; Zreiqat, H. The effect of strontium incorporation into CaSiO3 ceramics on their physical and biological properties. Biomaterials 2007, 28, 3171–3181. [Google Scholar] [CrossRef]
- Grech, L.; Mallia, B.; Camilleri, J. Investigation of the physical properties of tricalcium silicate cement-based root-end filling materials. Dent. Mater. 2013, 29, e20–e28. [Google Scholar] [CrossRef]
- Chi, L.; Zhang, A.; Qiu, Z.; Zhang, L.; Wang, Z.; Lu, S.; Zhao, D. Hydration activity, crystal structural, and electronic properties studies of Ba-doped dicalcium silicate. Nanotechnol. Rev. 2020, 9, 1027–1033. [Google Scholar] [CrossRef]
- Liu, W.-C.; Hu, C.-C.; Tseng, Y.-Y.; Sakthivel, R.; Fan, K.-S.; Wang, A.-N.; Wang, Y.-M.; Chung, R.-J. Study on strontium doped tricalcium silicate synthesized through sol-gel process. Mater. Sci. Eng. C 2020, 108, 110431. [Google Scholar] [CrossRef]
- Xia, W.; Lindahl, C.; Lausmaa, J.; Borchardt, P.; Ballo, A.; Thomsen, P.; Engqvist, H. Biomineralized strontium-substituted apa-tite/titanium dioxide coating on titanium surfaces. Acta Biomater. 2010, 6, 1591–1600. [Google Scholar] [CrossRef]
- Fredholm, Y.C.; Karpukhina, N.; Brauer, D.S.; Jones, J.R.; Law, R.V.; Hill, R.G. Influence of strontium for calcium substitution in bioactive glasses on degradation, ion release and apatite formation. J. R. Soc. Interface 2012, 9, 880–889. [Google Scholar] [CrossRef]
- Al-Khafaji, T.J.; Wong, F.; Fleming, P.S.; Karpukhina, N.; Hill, R. Novel fluoride and strontium-containing bioactive glasses for dental varnishes-design and bioactivity in Tris buffer solution. J. Non-Cryst. Solids 2019, 503–504, 120–130. [Google Scholar] [CrossRef]
- Yu, Y.; Bacsik, Z.; Edén, M. Contrasting in vitro apatite growth from bioactive glass surfaces with that of spontaneous precip-itation. Materials 2018, 11, 1690. [Google Scholar] [CrossRef]
- Mestieri, L.B.; Gomes-Cornélio, A.L.; Rodrigues, E.; Salles, L.; Bosso-Martelo, R.; Guerreiro-Tanomaru, J.M.; Tanomaru-Filho, M. Biocompatibility and bioactivity of calcium silicate-based endodontic sealers in human dental pulp cells. J. Appl. Oral Sci. 2015, 23, 467–471. [Google Scholar] [CrossRef]
- Fernández, J.M.; Molinuevo, M.S.; McCarthy, A.D.; Cortizo, A.M. Strontium ranelate stimulates the activity of bone-specific alkaline phosphatase: Interaction with Zn2+ and Mg2+. BioMetals 2014, 27, 601–607. [Google Scholar] [CrossRef]
- Bizelli-Silveira, C.; Pullisaar, H.; Abildtrup, L.A.; Andersen, O.Z.; Spin-Neto, R.; Foss, M.; Kraft, D.C. Strontium enhances prolif-eration and osteogenic behavior of periodontal ligament cells in vitro. J. Periodontal Res. 2018, 53, 1020–1028. [Google Scholar] [CrossRef]
- Mandakhbayar, N.; El-Fiqi, A.; Lee, J.H.; Kim, H.W. Evaluation of Strontium-Doped Nanobioactive Glass Cement for Den-tin-Pulp Complex Regeneration Therapy. ACS Biomater. Sci. Eng. 2019, 5, 6117–6126. [Google Scholar] [CrossRef]


| Groups | Initial pH | 24 h | 72 h |
|---|---|---|---|
| Control | 6.9 ± 0.06 aI | 7 ± 0.11 aI | 7 ± 0.12 aI |
| CS | 11.1 ± 0.12 bI | 11.7 ± 0.21 bII | 11.8 ± 0.16 bII |
| CSR | 9.5 ± 0.15 cI | 11.4 ± 0.07 cII | 11.5 ± 0.14 cII |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdalla, M.M.; Lung, C.Y.K.; Bijle, M.N.; Yiu, C.K.Y. Physicochemical Properties and Inductive Effect of Calcium Strontium Silicate on the Differentiation of Human Dental Pulp Stem Cells for Vital Pulp Therapies: An In Vitro Study. Materials 2022, 15, 5854. https://doi.org/10.3390/ma15175854
Abdalla MM, Lung CYK, Bijle MN, Yiu CKY. Physicochemical Properties and Inductive Effect of Calcium Strontium Silicate on the Differentiation of Human Dental Pulp Stem Cells for Vital Pulp Therapies: An In Vitro Study. Materials. 2022; 15(17):5854. https://doi.org/10.3390/ma15175854
Chicago/Turabian StyleAbdalla, Mohamed Mahmoud, Christie Y. K. Lung, Mohammed Nadeem Bijle, and Cynthia Kar Yung Yiu. 2022. "Physicochemical Properties and Inductive Effect of Calcium Strontium Silicate on the Differentiation of Human Dental Pulp Stem Cells for Vital Pulp Therapies: An In Vitro Study" Materials 15, no. 17: 5854. https://doi.org/10.3390/ma15175854
APA StyleAbdalla, M. M., Lung, C. Y. K., Bijle, M. N., & Yiu, C. K. Y. (2022). Physicochemical Properties and Inductive Effect of Calcium Strontium Silicate on the Differentiation of Human Dental Pulp Stem Cells for Vital Pulp Therapies: An In Vitro Study. Materials, 15(17), 5854. https://doi.org/10.3390/ma15175854







