Effect of Cu Modified Textile Structures on Antibacterial and Antiviral Protection
Abstract
:1. Introduction
2. Materials and Methods
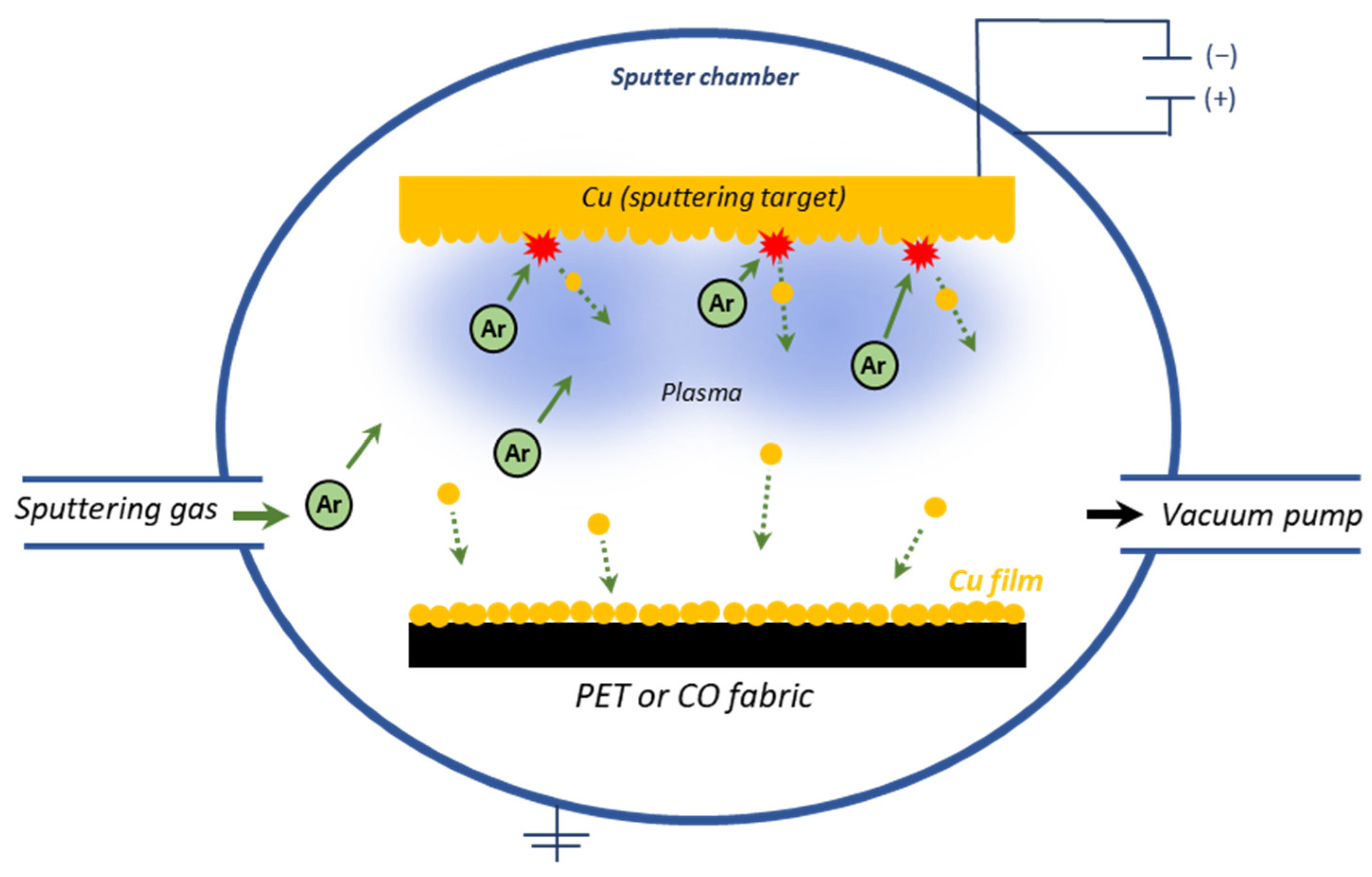
2.1. Modification of Fabrics
2.2. Microscopic Analysis
2.3. Determination of Cu Content
2.4. Antibacterial Test
2.5. Antiviral and Cytotoxicity Tests
2.6. Determination of Wettability and Surface Free Energy
2.7. Air Permeability Measurement
2.8. Water Vapour Permeability Measurement
2.9. Testing of Comfort Parameter
3. Results and Discussion
3.1. Analysis of Textile Structures and Cu Sputtering Effect
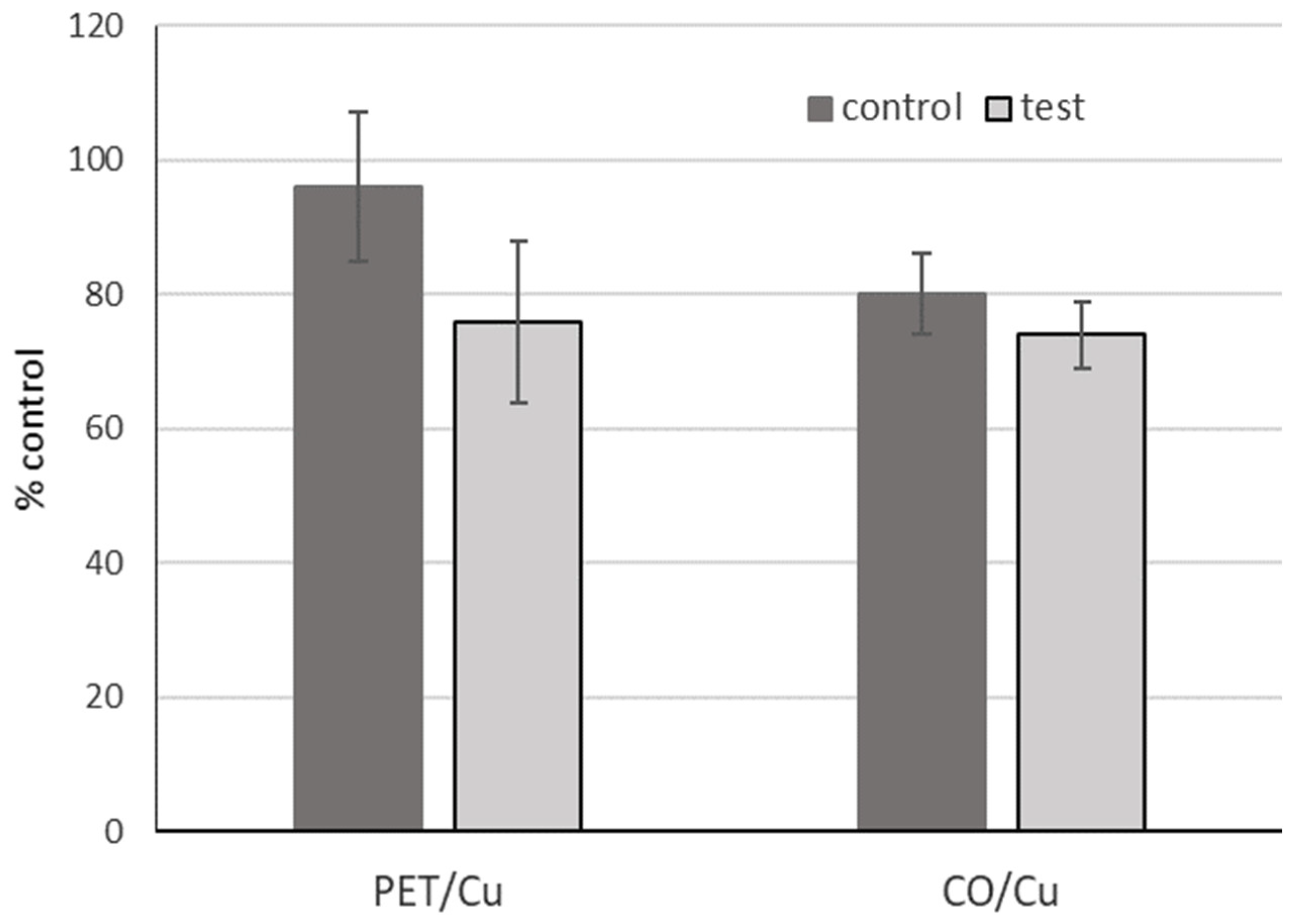
3.2. Antiviral Activity and Toxicity
3.3. Antibacterial Activity
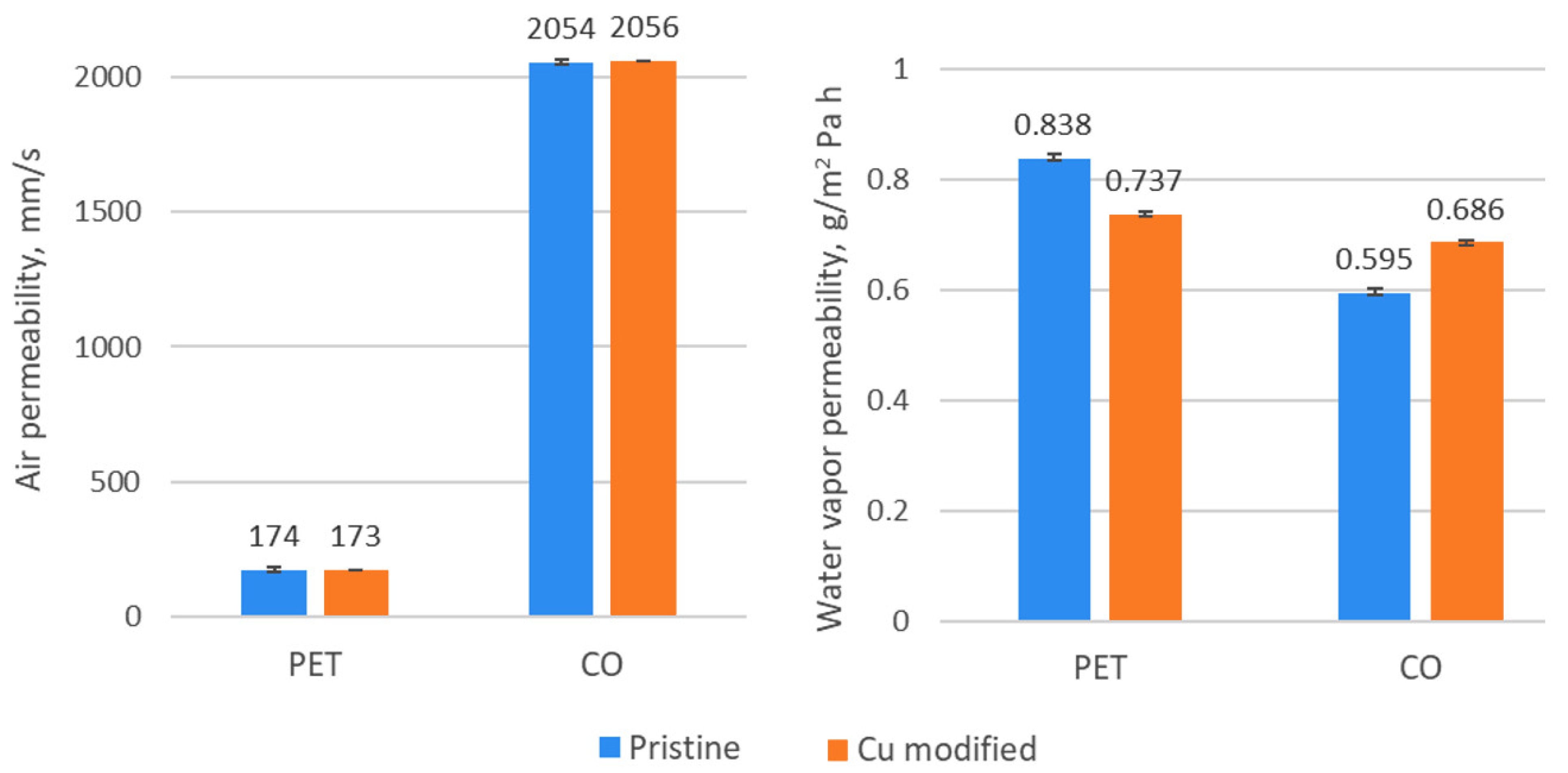
3.4. Surface Properties
3.5. Comfort Properties
4. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giesz, P.; Mackiewicz, E.; Grobelny, J.; Celichowski, G.; Cieślak, M. Multifunctional Hybrid Functionalization of Cellulose Fabrics with AgNWs and TiO2. Carbohydr. Polym. 2017, 177, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Giesz, P.; Mackiewicz, E.; Nejman, A.; Celichowski, G.; Cieślak, M. Investigation on Functionalization of Cotton and Viscose Fabrics with AgNWs. Cellulose 2017, 24, 409–422. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; Eid, B.M.; El-Aziz, E.A.; Elmaaty, T.M.A.; Ramadan, S.M. Loading of Chitosan—Nano Metal Oxide Hybrids onto Cotton/Polyester Fabrics to Impart Permanent and Effective Multifunctions. Int. J. Biol. Macromol. 2017, 105, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Pandimurugan, R.; Thambidurai, S. UV Protection and Antibacterial Properties of Seaweed Capped ZnO Nanoparticles Coated Cotton Fabrics. Int. J. Biol. Macromol. 2017, 105, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Granados, A.; Pleixats, R.; Vallribera, A. Recent Advances on Antimicrobial and Anti-Inflammatory Cotton Fabrics Containing Nanostructures. Molecules 2021, 26, 3008. [Google Scholar] [CrossRef]
- Eremenko, A.M.; Petrik, I.S.; Smirnova, N.P.; Rudenko, A.V.; Marikvas, Y.S. Antibacterial and Antimycotic Activity of Cotton Fabrics, Impregnated with Silver and Binary Silver/Copper Nanoparticles. Nanoscale Res. Lett. 2016, 11, 28. [Google Scholar] [CrossRef]
- El-Nahhal, I.M.; Elmanama, A.A.; Amara, N.; Qodih, F.S.; Selmane, M.; Chehimi, M.M. The Efficacy of Surfactants in Stabilizing Coating of Nano-Structured CuO Particles onto the Surface of Cotton Fibers and Their Antimicrobial Activity. Mater. Chem. Phys. 2018, 215, 221–228. [Google Scholar] [CrossRef]
- Marković, D.; Vasiljević, J.; Ašanin, J.; Ilic-Tomic, T.; Tomšič, B.; Jokić, B.; Mitrić, M.; Simončič, B.; Mišić, D.; Radetić, M. The Influence of Coating with Aminopropyl Triethoxysilane and CuO/Cu2O Nanoparticles on Antimicrobial Activity of Cotton Fabrics under Dark Conditions. J. Appl. Polym. Sci. 2020, 137, 49194. [Google Scholar] [CrossRef]
- Ohsumi, Y.; Kitamoto, K.; Anraku, Y. Changes Induced in the Permeability Barrier of the Yeast Plasma Membrane by Cupric Ion. J. Bacteriol. 1988, 170, 2676–2682. [Google Scholar] [CrossRef]
- Borkow, G.; Gabbay, J. Copper as a Biocidal Tool. Curr. Med. Chem. 2005, 12, 2163–2175. [Google Scholar] [CrossRef] [Green Version]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal Nanoparticles: Understanding the Mechanisms behind Antibacterial Activity. J. Nanobiotechnol. 2017, 15, 65. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T. Antiviral Activities of Cu2+ Ions in Viral Prevention, Replication, RNA Degradation, and for Antiviral Efficacies of Lytic Virus, ROS-Mediated Virus, Copper Chelation. World Sci. News 2018, 99, 149–168. [Google Scholar]
- Tavakoli, A.; Hashemzadeh, M.S. Inhibition of Herpes Simplex Virus Type 1 by Copper Oxide Nanoparticles. J. Virol. Methods 2020, 275, 113688. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.; Gaikwad, S.; Nimse, M.; Rajbhoj, A. Copper Oxide Nanoparticles: Synthesis, Characterization and Their Antibacterial Activity. J. Clust. Sci. 2011, 22, 121–129. [Google Scholar] [CrossRef]
- Borkow, G. Using Copper to Fight Microorganisms. Curr. Chem. Biol. 2012, 6, 93–103. [Google Scholar] [CrossRef]
- Ingle, A.P.; Duran, N.; Rai, M. Bioactivity, Mechanism of Action, and Cytotoxicity of Copper-Based Nanoparticles: A Review. Appl. Microbiol. Biotechnol. 2014, 98, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Bleichert, P.; Santo, C.E.; Hanczaruk, M.; Meyer, H.; Grass, G. Inactivation of Bacterial and Viral Biothreat Agents on Metallic Copper Surfaces. BioMetals 2014, 27, 1179–1189. [Google Scholar] [CrossRef]
- Hang, X.; Peng, H.; Song, H.; Qi, Z.; Miao, X.; Xu, W. Antiviral Activity of Cuprous Oxide Nanoparticles against Hepatitis C Virus in Vitro. J. Virol. Methods 2015, 222, 150–157. [Google Scholar] [CrossRef]
- Minoshima, M.; Lu, Y.; Kimura, T.; Nakano, R.; Ishiguro, H.; Kubota, Y.; Hashimoto, K.; Sunada, K. Comparison of the Antiviral Effect of Solid-State Copper and Silver Compounds. J. Hazard. Mater. 2016, 312, 1–7. [Google Scholar] [CrossRef]
- Hodek, J.; Zajícová, V.; Lovetinská-Šlamborová, I.; Stibor, I.; Müllerová, J.; Weber, J. Protective Hybrid Coating Containing Silver, Copper and Zinc Cations Effective against Human Immunodeficiency Virus and Other Enveloped Viruses. BMC Microbiol. 2016, 16, 56. [Google Scholar] [CrossRef]
- Vincent, M.; Duval, R.E.; Hartemann, P.; Engels-Deutsch, M. Contact Killing and Antimicrobial Properties of Copper. J. Appl. Microbiol. 2018, 124, 1032–1046. [Google Scholar] [CrossRef] [Green Version]
- Borkow, G.; Gabbay, J. Putting Copper into Action: Copper-impregnated Products with Potent Biocidal Activities. FASEB J. 2004, 18, 1728–1730. [Google Scholar] [CrossRef]
- Borkow, G.; Zhou, S.S.; Page, T.; Gabbay, J. A Novel Anti-Influenza Copper Oxide Containing Respiratory Face Mask. PLoS ONE 2010, 5, e11295. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ye, J.; Rauf, A.; Zhang, S.; Wang, G.; Shi, S.; Ning, G. A Flexible Fibrous Membrane Based on Copper(II) Metal-Organic Framework/Poly(Lactic Acid) Composites with Superior Antibacterial Performance. Biomater. Sci. 2021, 9, 3851–3859. [Google Scholar] [CrossRef] [PubMed]
- Kharaghani, D.; Khan, M.Q.; Tamada, Y.; Ogasawara, H.; Inoue, Y.; Saito, Y.; Hashmi, M.; Kim, I.S. Fabrication of Electrospun Antibacterial PVA/Cs Nanofibers Loaded with CuNPs and AgNPs by an in-Situ Method. Polym. Test. 2018, 72, 315–321. [Google Scholar] [CrossRef]
- Gurianov, Y.; Nakonechny, F.; Albo, Y.; Nisnevitch, M. Antibacterial Composites of Cuprous Oxide Nanoparticles and Polyethylene. Int. J. Mol. Sci. 2019, 20, 439. [Google Scholar] [CrossRef]
- Hashmi, M.; Ullah, S.; Kim, I.S. Copper Oxide (CuO) Loaded Polyacrylonitrile (PAN) Nanofiber Membranes for Antimicrobial Breath Mask Applications. Curr. Res. Biotechnol. 2019, 1, 1–10. [Google Scholar] [CrossRef]
- Ali, A.; Baheti, V.; Militky, J.; Khan, Z.; Tunakova, V.; Naeem, S. Copper Coated Multifunctional Cotton Fabrics. J. Ind. Text. 2018, 48, 448–464. [Google Scholar] [CrossRef]
- Champagne, V.; Sundberg, K.; Helfritch, D. Kinetically Deposited Copper Antimicrobial Surfaces. Coatings 2019, 9, 223. [Google Scholar] [CrossRef]
- Imani, S.M.; Ladouceur, L.; Marshall, T.; Maclachlan, R.; Soleymani, L.; Didar, T.F. Antimicrobial Nanomaterials and Coatings: Current Mechanisms and Future Perspectives to Control the Spread of Viruses Including SARS-CoV-2. ACS Nano 2020, 14, 12341–12369. [Google Scholar] [CrossRef]
- Kudzin, M.; Mrozinska, Z.; Kaczmarek, A.; Lisiak-Kucinska, A. Deposition of Copper on Poly (Lactide) Non-Woven Fabrics by Magnetron Sputtering—Fabrication of New Multi-Functional, Antimicrobial Composite Materials. Materials 2020, 13, 3971. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, B.; Prateek; Ranjan, S.; Saraf, M.; Kar, P.; Singh, S.P.; Thakur, V.K.; Singh, A.; Gupta, R.K. Antibacterial and Antiviral Functional Materials: Chemistry and Biological Activity toward Tackling COVID-19-like Pandemics. ACS Pharmacol. Transl. Sci. 2021, 4, 8–54. [Google Scholar] [CrossRef]
- Warnes, S.L.; Keevil, C.W. Inactivation of Norovirus on Dry Copper Alloy Surfaces. PLoS ONE 2013, 8, e75017. [Google Scholar] [CrossRef]
- Warnes, S.L.; Summersgill, E.N.; Keevil, C.W. Inactivation of Murine Norovirus on a Range of Copper Alloy Surfaces Is Accompanied by Loss of Capsid Integrity. Appl. Environ. Microbiol. 2015, 81, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.A.; Hanarp, P.; Sutherland, D.S.; Gold, J.; Mustin, C.; Rouxhet, P.G.; Dufrêne, Y.F. Protein Adsorption on Model Surfaces with Controlled Nanotopography and Chemistry. Langmuir 2002, 18, 819–828. [Google Scholar] [CrossRef]
- Rechendorff, K.; Hovgaard, M.B.; Foss, M.; Zhdanov, V.P.; Besenbacher, F. Enhancement of Protein Adsorption Induced by Surface Roughness. Langmuir 2006, 22, 10885–10888. [Google Scholar] [CrossRef]
- Anand, G.; Sharma, S.; Dutta, A.K.; Kumar, S.K.; Belfort, G. Conformational Transitions of Adsorbed Proteins on Surfaces of Varying Polarity. Langmuir 2010, 26, 10803–10811. [Google Scholar] [CrossRef]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding Protein Adsorption Phenomena at Solid Surfaces. Adv. Colloid Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef]
- Szymańska, E.; Orłowski, P.; Winnicka, K.; Tomaszewska, E.; Bąska, P.; Celichowski, G.; Grobelny, J.; Basa, A.; Krzyżowska, M. Multifunctional Tannic Acid/Silver Nanoparticle-Based Mucoadhesive Hydrogel for Improved Local Treatment of HSV Infection: In Vitro and In Vivo Studies. Int. J. Mol. Sci. 2018, 28, 387. [Google Scholar] [CrossRef]
- Kärber, G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Arch. Exp. Pathol. Pharmakol. 1931, 162, 480–483. [Google Scholar] [CrossRef]
- Miura, T.A.; Travanty, E.A.; Oko, L.; Bielefeldt-Ohmann, H.; Weiss, S.R.; Beauchemin, N.; Holmes, K.V. The Spike Glycoprotein of Murine Coronavirus MHV-JHM Mediates Receptor-Independent Infection and Spread in the Central Nervous Systems of Ceacam1a-/- Mice. J. Virol. 2008, 82, 755–763. [Google Scholar] [CrossRef] [Green Version]
- Cieslak, M.; Puchowicz, D.; Schmidt, H. Evaluation of the Possibility of Using Surface Free Energy Study to Design Protective Fabrics. Text. Res. J. 2012, 82, 1177–1189. [Google Scholar] [CrossRef]
- Hes, L. Non-Destructive Determination of Comfort Parameters during Marketing of Functional Garments and Clothing. Indian J. Fibre Text. Res. 2008, 33, 239–245. [Google Scholar]
- San, K.; Long, J.; Michels, C.A.; Gadura, N. Antimicrobial Copper Alloy Surfaces Are Effective against Vegetative but Not Sporulated Cells of Gram-Positive Bacillus Subtilis. Microbiologyopen 2015, 4, 753–763. [Google Scholar] [CrossRef]
- Vaidya, M.Y.; McBain, A.J.; Butler, J.A.; Banks, C.E.; Whitehead, K.A. Antimicrobial Efficacy and Synergy of Metal Ions against Enterococcus Faecium, Klebsiella Pneumoniae and Acinetobacter Baumannii in Planktonic and Biofilm Phenotypes. Sci. Rep. 2017, 7, 5911. [Google Scholar] [CrossRef]
- Souli, M.; Galani, I.; Plachouras, D.; Panagea, T.; Armaganidis, A.; Petrikkos, G.; Giamarellou, H. Antimicrobial Activity of Copper Surfaces against Carbapenemase-Producing Contemporary Gram-Negative Clinical Isolates. J. Antimicrob. Chemother. 2013, 68, 852–857. [Google Scholar] [CrossRef]
- Wenzel, R.W. Resistance of Solid Surfaces to Wetting by Water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of Porous Surface. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Wolansky, G.; Marmur, A. The Actual Contact Angle on a Heterogeneous Rough Surface in Three Dimensions. Langmuir 1988, 14, 5292–5297. [Google Scholar] [CrossRef]
- Marmur, A. Wetting on Hydrophobic Rough Surfaces: To Be Heterogeneous or Not To Be? Langmuir 2003, 19, 8343–8348. [Google Scholar] [CrossRef]
- Li, G.; Wang, B.; Liu, Y.; Tan, T.; Song, X.; Li, E.; Yan, H. Stable Superhydrophobic Surface: Fabrication of Interstitial Cottonlike Structure of Copper Nanocrystals by Magnetron Sputtering. Sci. Technol. Adv. Mater. 2008, 9, 025006. [Google Scholar] [CrossRef] [PubMed]
- Roach, P.; Farrar, D.; Perry, C.C. Surface Tailoring for Controlled Protein Adsorption: Effect of Topography at the Nanometer Scale and Chemistry. J. Am. Chem. Soc. 2006, 128, 3939–3945. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.D.; Hlady, V. Protein Adsorption and Materials Biocompatibility: A Tutorial Review and Suggested Hypoteheses. Adv. Polym. Sci. 1987, 79, 1–63. [Google Scholar] [CrossRef]
- Fields, B.N.; Knipe, D.M.; Howley, P.M. Fields Virology; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007. [Google Scholar]
- Mettenleiter, T.C.; Klupp, B.G.; Granzow, H. Herpesvirus assembly: A tale of two membranes Curr. Opin. Microbiol. 2006, 9, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.L. Encyclopedia of Virology, 3rd ed.; Mahy, B.W.J., Van Regenmortel, M.H.V., Eds.; Academic Press: Cambridge, MA, USA, 2008; pp. 243–250. [Google Scholar]
- Samji, T. Influenza A: Understanding the Viral Life Cycle. Yale J. Biol. Med. 2009, 82, 153–159. [Google Scholar]
- Homberger, F.R. Enterotropic Mouse Hepatitis Virus. Lab. Anim. 1997, 31, 97–115. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Liu, Y.; Yang, Y.; Zhang, P.; Zhong, W.; Wang, Y.; Wang, Q.; Xu, Y.; Li, M.; Li, X.; et al. Analysis of Therapeutic Targets for SARS-CoV-2 and Discovery of Potential Drugs by Computational Methods. Acta Pharm. Sin. B 2020, 10, 766–788. [Google Scholar] [CrossRef]
- Jamshidinia, N.; Mohammadipanah, F. Nanomaterial-Augmented Formulation of Disinfectants and Antiseptics in Controlling SARS CoV-2. Food Environ. Virol. 2022, 14, 105–119. [Google Scholar] [CrossRef]
- Favatela, M.F.; Otarola, J.; Ayala-Peña, V.B.; Dolcini, G.; Perez, S.; Torres Nicolini, A.; Alvarez, V.A.; Lassalle, V.L. Development and Characterization of Antimicrobial Textiles from Chitosan-Based Compounds: Possible Biomaterials Against SARS-CoV-2 Viruses. J. Inorg. Organomet. Polym. Mater. 2022, 32, 1473–1486. [Google Scholar] [CrossRef]
- Sampath, M.B.; Mani, S.; Nalankilli, G. Effect of Filament Fineness on Comfort Characteristics of Moisture Management Finished Polyester Knitted Fabrics. J. Ind. Text. 2011, 41, 160–173. [Google Scholar] [CrossRef]
- Hermans, P. Physics and Chemistry of Cellulose Fibres; Elsevier Publishing Company Inc.: Amsterdam, The Netherlands, 1949. [Google Scholar]
- Von Falkai, B. Synthesefasern; Verlag Chemie: Weinheim, Germany, 1981. [Google Scholar]
- Kawasaki, K.; Sekita, Y. Sorption and Diffusion of Water Vapor by Nylon 6. J. Polym. Sci. Part A Gen. Pap. 1964, 2, 2437–2443. [Google Scholar] [CrossRef]
- Cieślak, M.; Schmidt, H.; Świercz, R.; Wąsowicz, W. Fibers susceptibility to contamination by environmental tobacco smoke markers. Text. Res. J. 2014, 84, 840–853. [Google Scholar] [CrossRef]
- Román, L.E.; Gomez, E.D.; Solís, J.L.; Gómez, M.M. Antibacterial Cotton Fabric Functionalized with Copper Oxide Nanoparticles. Molecules 2020, 25, 5802. [Google Scholar] [CrossRef] [PubMed]



| Polyester Fabric (PET) | Cotton Fabric (CO) | ||
|---|---|---|---|
| Raw material | polyester (PET) 90% with polyamide (PA) 10% | cotton 100% | |
| Yarn | warp | PET DTY dtex 110 f 144 (textured microfiber), linear mass of filament 0.76 dtex | tex 8 |
| weft | PET dtex 167 f (75 × 8) (textured supermicrofiber, biocomponent 80% PET and 20% PA as a spacer in segmented-pie (orange cross section) filament), linear mass of filament 0.28 dtex | tex 8 | |
| Wave | ¼ (2)—five-thread satin | plain | |
| Treads number/10 cm | warp | 469 | 310 |
| weft | 332 | 300 | |
| Mass per unit area, g/m2 | 130 ± 1 | 55 ± 1 | |
| Thickness, mm | 0.41 ± 0.10 | 0.27 ± 0.10 | |
| Volume porosity *, % | 77.1 | 87.3 | |
| Pressure, Mbar | Effective Power, kWh | Circulating Power, kWh | Argon Content, % | Number of Passes | Speed, mm/s | |
|---|---|---|---|---|---|---|
| PET | 2.0 × 10−3 | 2.0–2.2 | 0.8–1.0 | 3 | 15 | 15 |
| CO | 2.0 × 10−3 | 2.0–2.2 | 0.8–1.0 | 3 | 15 | 15 |
| Elements, wt% | PET | CO | ||
|---|---|---|---|---|
| Pristine | Cu | Pristine | Cu | |
| C | 63.0 ± 0.08 | 35.7 ± 0.73 | 47.7 ± 0.25 | 26.6 ± 1.67 |
| O | 36.5 ± 0.09 | 25.3 ± 0.27 | 52.3 ± 0.25 | 30.0 ± 0.82 |
| Cu | - | 38.9 ± 0.47 | - | 43.4 ± 1.04 |
| Ti | 0.2 ± 0.01 | 0.2 ± 0.01 | - | - |
| Control Samples | Test Samples | Mv | |||
|---|---|---|---|---|---|
| Log Reduction PFU/mL VACV | Log Reduction PFU/mL HSV-1 | Log Reduction TCID50/mL IFV | Log Reduction TCID MHV | ||
| PET | PET/Cu | 0 | 1 | 0 | 1 |
| CO | CO/Cu | 2 | 2 | 2 | 1 |
| Staphylococcus aureus (ATCC 6538) | Klebsiella pneumoniae (ATCC 4352) | |||
|---|---|---|---|---|
| PET/Cu | CO/Cu | PET/Cu | CO/Cu | |
| Concentration of inoculum, CFU/mL | 2.7 × 105 | 2.8 × 105 | ||
| Growth value F—for the control sample (pristine) F = lg Ct − lg Co | 3.73 lg Ct: −8.32 lg Co: −4.59 | 4.95 lg Ct: −9.29 lg Co: –4.34 | 4.31 lg Ct: −9.56 lg Co: −5.25 | 4.42 lg Ct: −9.61 lg Co: −5.19 |
| Growth value G—for the test sample (Cu modified) G = lg Tt − lg To | −3.10 lg Tt: −1.30 lg To: −4.40 | 0.00 lg Tt: −1.30 lg To: –1.30 | −2.65 lg Tt: −1.30 lg To: −3.95 | 0.00 lg Tt: −1.30 lg To: −1.30 |
| Value of antimicrobial activity A A = (lg Ct − lg Co) − (lg Tt − lg Co) | 7.02 | 7.99 | 8.26 | 8.31 |
| Time and temperature of incubation | 22 h + 48 h (37 ± 2 °C) | |||
| Sample | Contact Angle Θ, Deg | Surface Free Energy γS and Dispersive γSd and Polar Components γSp, mJ/m2 | |||||
|---|---|---|---|---|---|---|---|
| Θw | ΘF | ΘH | γS | γSd | γSp | ||
| PET | pristine | 0.0 | 18.1 ± 2.1 | 0.0 | 72.04 | 17.21 | 54.82 |
| Cu | 131.2 ± 1.5 | 117.1 ± 1.2 | 0.0 | 16.43 | 15.38 | 1.05 | |
| CO | pristine | 0.0 | 22.8 ± 2.2 | 0.0 | 71.71 | 16.81 | 54.90 |
| Cu | 135.0 ± 1.9 | 128.6 ± 1.4 | 0.0 | 15.26 | 13.79 | 1.56 | |
| Sample | λ, Wm−1K−1 | a × 10−8, m2s−1 |
|---|---|---|
| PET | 0.041 ± 0.0008 | 5.07 ± 0.753 |
| PET/Cu | 0.044 ± 0.0012 | 7.11 ± 1.290 |
| CO | 0.038 ± 0.0007 | 7.90 ± 1.800 |
| CO/Cu | 0.039 ± 0.0008 | 8.10 ± 0.600 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieślak, M.; Kowalczyk, D.; Krzyżowska, M.; Janicka, M.; Witczak, E.; Kamińska, I. Effect of Cu Modified Textile Structures on Antibacterial and Antiviral Protection. Materials 2022, 15, 6164. https://doi.org/10.3390/ma15176164
Cieślak M, Kowalczyk D, Krzyżowska M, Janicka M, Witczak E, Kamińska I. Effect of Cu Modified Textile Structures on Antibacterial and Antiviral Protection. Materials. 2022; 15(17):6164. https://doi.org/10.3390/ma15176164
Chicago/Turabian StyleCieślak, Małgorzata, Dorota Kowalczyk, Małgorzata Krzyżowska, Martyna Janicka, Ewa Witczak, and Irena Kamińska. 2022. "Effect of Cu Modified Textile Structures on Antibacterial and Antiviral Protection" Materials 15, no. 17: 6164. https://doi.org/10.3390/ma15176164







