Optimization of In Situ Indentation Protocol to Map the Mechanical Properties of Articular Cartilage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Management
2.2. A Priori Estimate of the Articular Cartilage Thickness at the Indentation Point
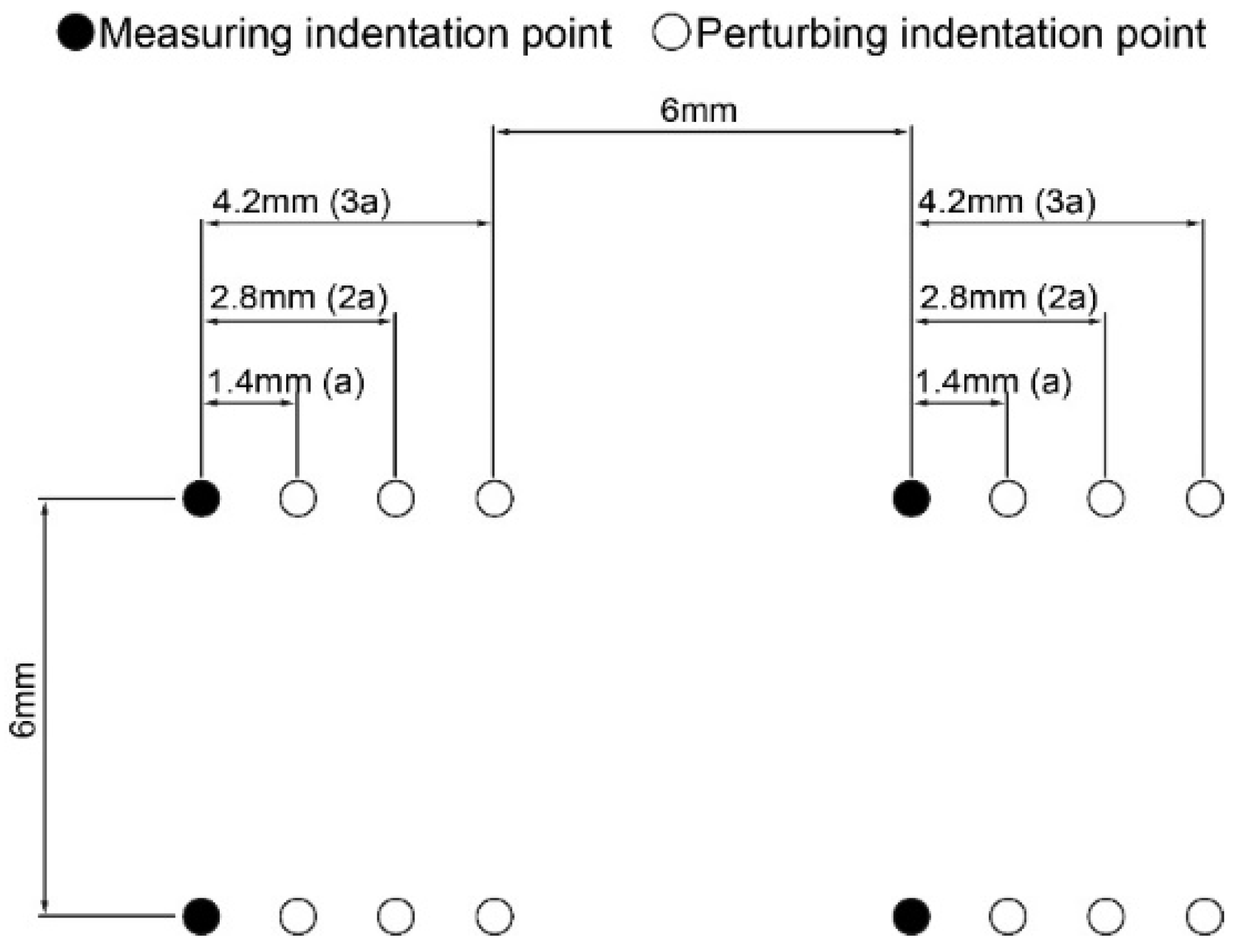
2.3. Optimization of Grid Spacing
2.4. Effect of Indenter Diameter on Articular Cartilage Response
2.5. Pilot Study on a Human Tibial Plateau
2.6. Data Analysis
3. Results
3.1. A Priori Estimate of the Articular Cartilage Thickness at the Indentation Point
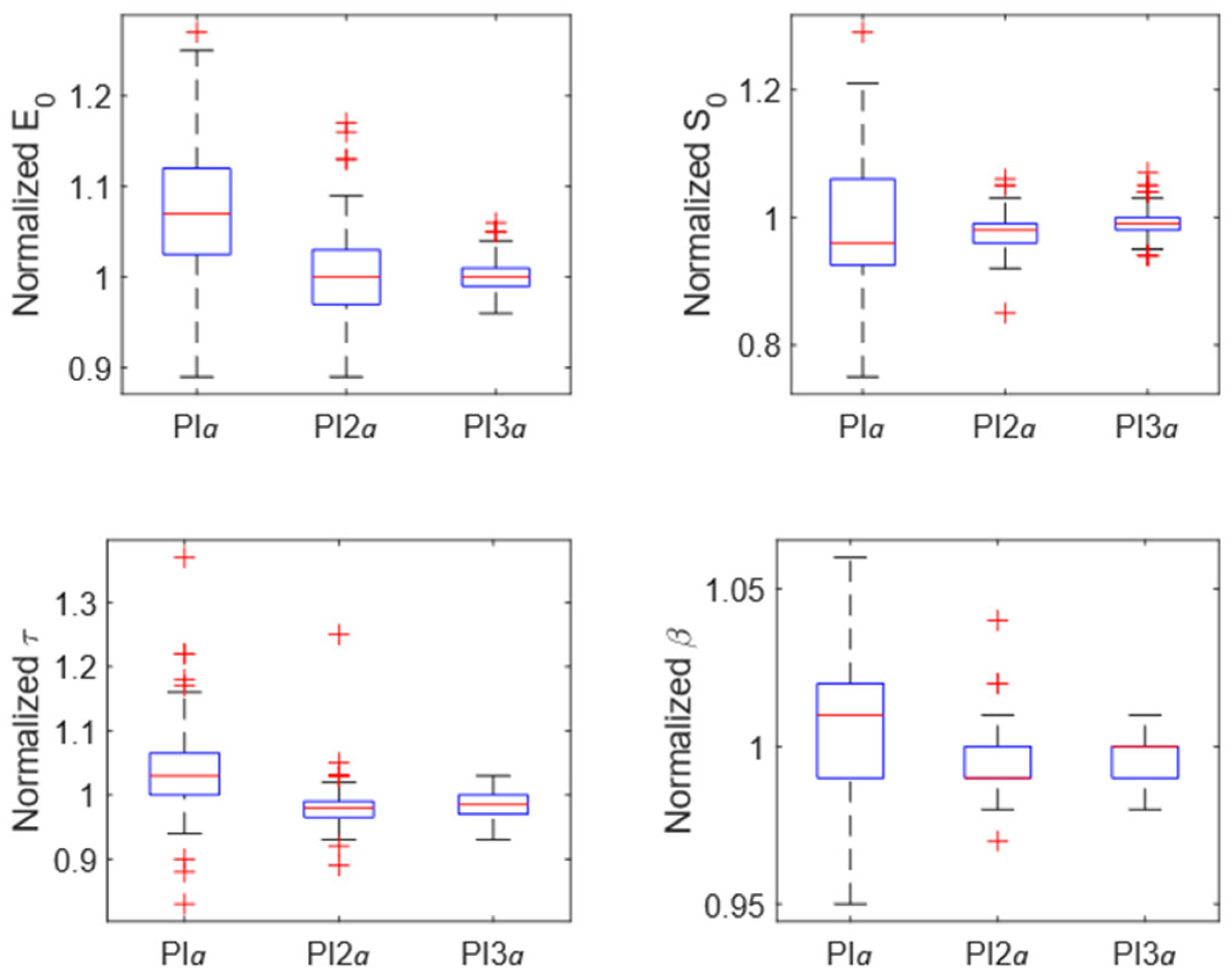
3.2. Optimization of Grid Spacing
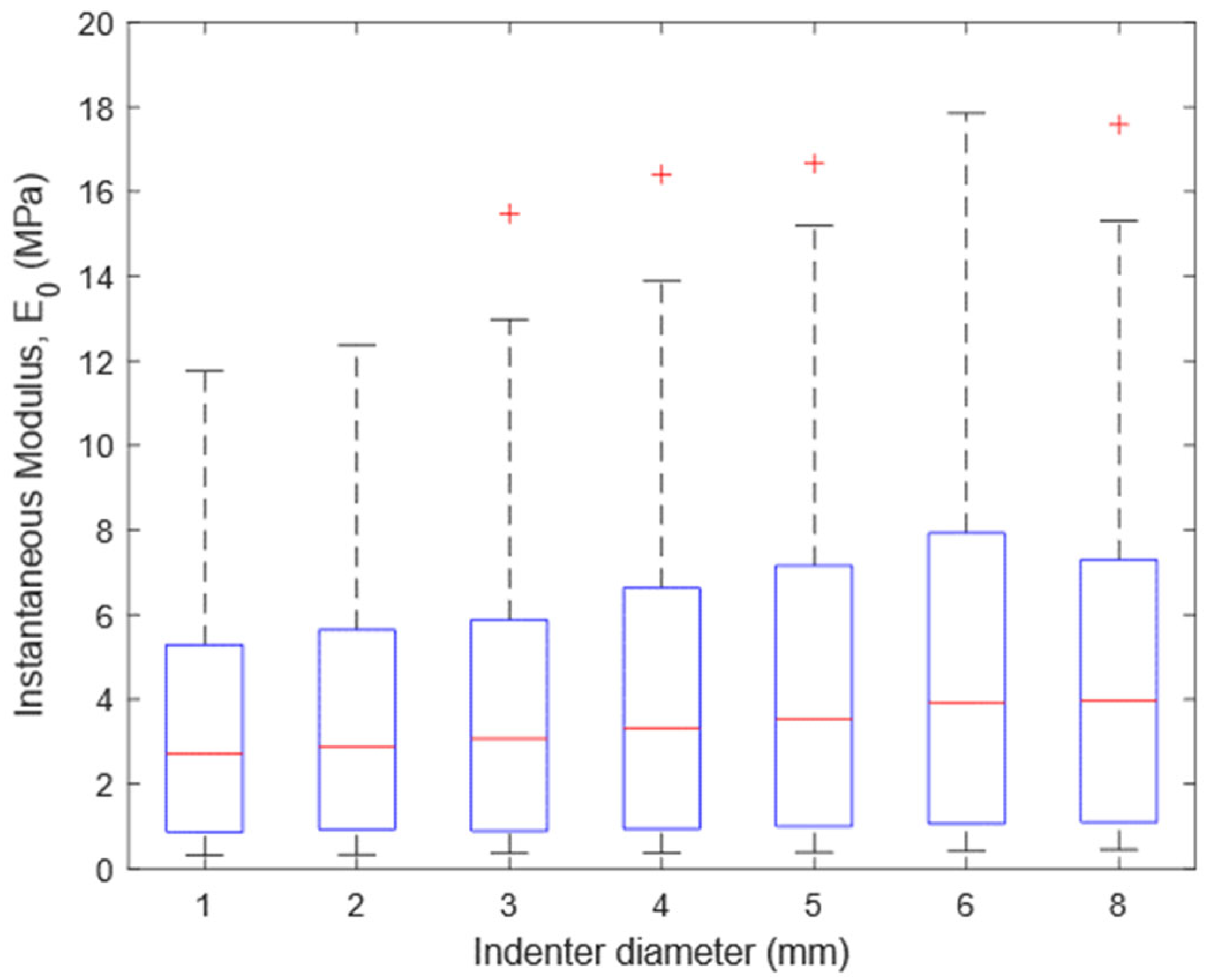
3.3. Effect of Indenter Diameter on Articular Cartilage Response
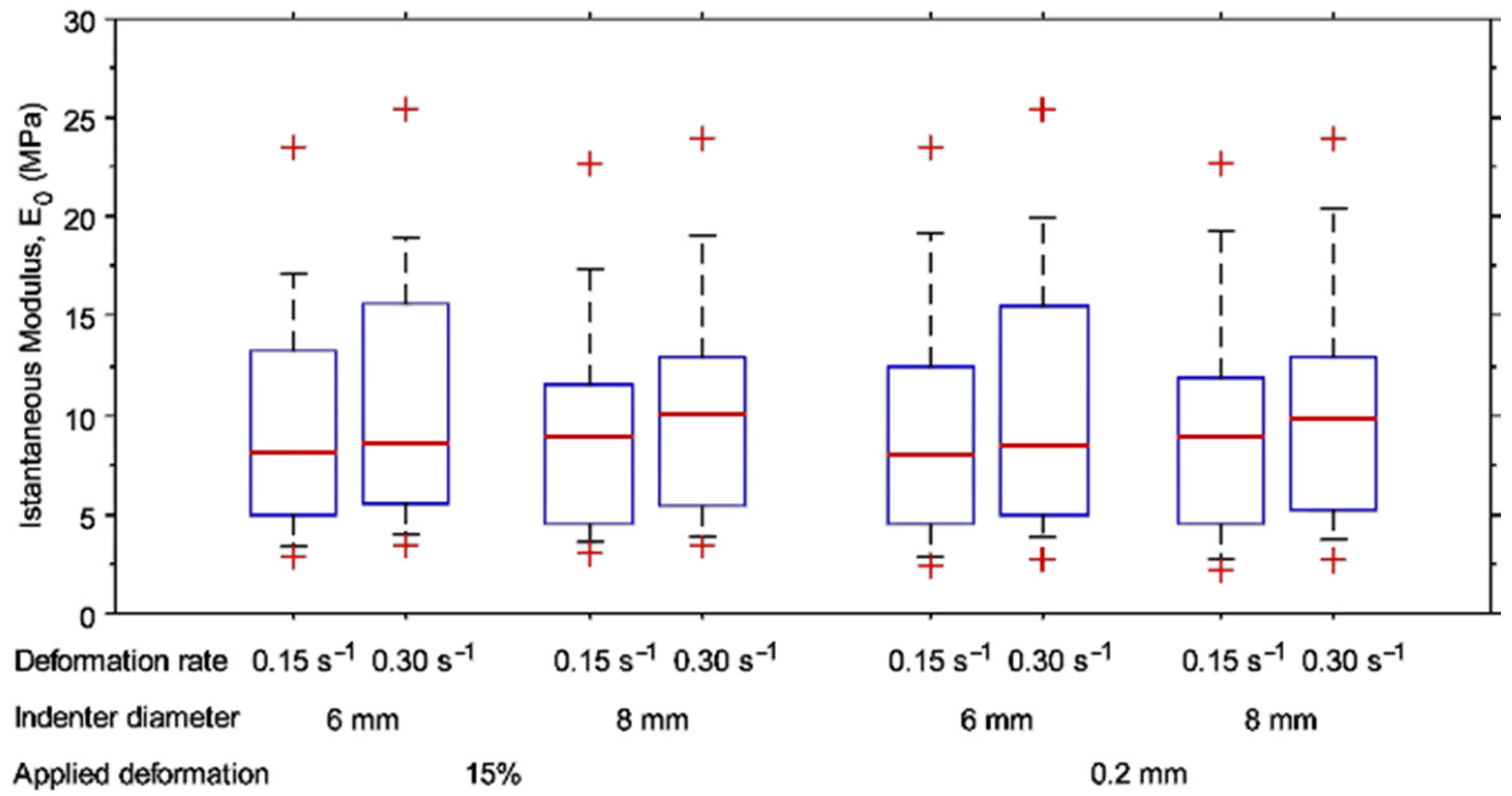
3.4. Pilot Study on a Human Tibial Plateau
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
References
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The Basic Science of Articular Cartilage: Structure, Composition, and Function. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, M.; Turunen, M.J.; Finnilä, M.A.; Joukainen, A.; Kröger, H.; Saarakkala, S.; Korhonen, R.K.; Tanska, P. Structure-Function Relationships of Healthy and Osteoarthritic Human Tibial Cartilage: Experimental and Numerical Investigation. Ann. Biomed. Eng. 2020, 48, 2887–2900. [Google Scholar] [CrossRef] [PubMed]
- Laasanen, M.S.; Töyräs, J.; Korhonen, R.K.; Rieppo, J.; Saarakkala, S.; Nieminen, M.T.; Hirvonen, J.; Jurvelin, J.S. Biomechanical properties of knee articular cartilage. Biorheology 2003, 40, 133–140. [Google Scholar] [PubMed]
- McMahon, L.A.; O’Brien, F.J.; Prendergast, P.J. Biomechanics and mechanobiology in osteochondral tissues. Regen. Med. 2008, 3, 743–759. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, M.G.; Otarola, G.A.; Hu, J.C.; Athanasiou, K.A. Cartilage Assessment Requires a Surface Characterization Protocol: Roughness, Friction, and Function. Tissue Eng. Part C Methods 2021, 27, 276–286. [Google Scholar] [CrossRef]
- Marchiori, G.; Berni, M.; Boi, M.; Filardo, G. Cartilage mechanical tests: Evolution of current standards for cartilage repair and tissue engineering. A literature review. Clin. Biomech. 2019, 68, 58–72. [Google Scholar] [CrossRef]
- Seitz, A.M.; Osthaus, F.; Schwer, J.; Warnecke, D.; Faschingbauer, M.; Sgroi, M.; Ignatius, A.; Dürselen, L. Osteoarthritis-Related Degeneration Alters the Biomechanical Properties of Human Menisci Before the Articular Cartilage. Front. Bioeng. Biotechnol. 2021, 9, 659989. [Google Scholar] [CrossRef]
- Kabir, W.; Di Bella, C.; Choong, P.F.M.; O’Connell, C.D. Assessment of Native Human Articular Cartilage: A Biomechanical Protocol. Cartilage 2021, 13 (Suppl. 2), 427S–437S. [Google Scholar] [CrossRef]
- Warner, M.D.; Taylor, W.R.; Clift, S.E. Finite element biphasic indentation of cartilage: A comparison of experimental indenter and physiological contact geometries. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2001, 215, 487–496. [Google Scholar] [CrossRef]
- Swann, A.C.; Seedhom, B.B. Improved techniques for measuring the indentation and thickness of articular cartilage. Proc. Inst. Mech. Eng. Part H 1989, 203, 143–150. [Google Scholar] [CrossRef]
- Seidenstuecker, M.; Watrinet, J.; Bernstein, A. Viscoelasticity and histology of the human cartilage in healthy and degenerated conditions of the knee. J. Orthop. Surg. Res. 2019, 14, 256. [Google Scholar] [CrossRef] [PubMed]
- Antons, J.; Marascio, M.G.M.; Nohava, J.; Martin, R.; Applegate, L.A.; Bourban, P.E.; Pioletti, D.P. Zone-dependent mechanical properties of human articular cartilage obtained by indentation measurements. J. Mater. Sci. Mater. Med. 2018, 29, 57. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Chevrier, A.; Garon, M.; Quenneville, E.; Lavigne, P.; Yaroshinsky, A.; Hoemann, C.D.; Buschmann, M.D. Electromechanical probe and automated indentation maps are sensitive techniques in assessing early degenerated human articular cartilage. J. Orthop. Res. 2017, 35, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Bae, W.C.; Schumacher, B.L.; Sah, R.L. Indentation probing of human articular cartilage: Effect on chondrocyte viability. Osteoarthr. Cartil. 2007, 15, 9–18. [Google Scholar] [CrossRef]
- Kiviranta, P.; Lammentausta, E.; Töyräs, J.; Kiviranta, I.; Jurvelin, J.S. Indentation diagnostics of cartilage degeneration. Osteoarthr. Cartil. 2008, 16, 796–804. [Google Scholar] [CrossRef]
- Korhonen, R.K.; Laasanen, M.S.; Töyräs, J.; Rieppo, J.; Hirvonen, J.; Helminen, H.J.; Jurvelin, J.S. Comparison of the equilibrium response of articular cartilage in unconfined compression, confined compression and indentation. J. Biomech. 2002, 35, 903–909. [Google Scholar] [CrossRef]
- Simha, N.K.; Jin, H.; Hall, M.L.; Chiravarambath, S.; Lewis, J.L. Effect of indenter size on elastic modulus of cartilage measured by indentation. J. Biomech. Eng. 2007, 129, 767–775. [Google Scholar] [CrossRef]
- Moore, A.C.; Zimmerman, B.K.; Chen, X.; Lu, X.L.; Burris, D.L. Experimental characterization of biphasic materials using rate-controlled Hertzian indentation. Tribol. Int. 2015, 89, 2–8. [Google Scholar] [CrossRef]
- Chandwadkar, S.A. Indentation Protocol to Determine Viscoelastic Properties of Cartilage before and after Crosslinking. Ph.D. Thesis, Purdue University, West Lafayette, IN, USA, 2017. Available online: https://scholarworks.iupui.edu/bitstream/handle/1805/15092/Indentation%20Protocol%20to%20Determine%20Viscoelastic%20Properties%20of%20Cartilage%20Before%20and%20After%20Crosslinking.pdf;jsessionid=0455762F255B53B8074AC2952AD83A12?sequence=1 (accessed on 15 July 2022).
- Moshtagh, P.R.; Pouran, B.; Korthagen, N.M.; Zadpoor, A.A.; Weinans, H. Guidelines for an optimized indentation protocol for measurement of cartilage stiffness: The effects of spatial variation and indentation parameters. J. Biomech. 2016, 49, 3602–3607. [Google Scholar] [CrossRef]
- Campbell, S.E.; Ferguson, V.L.; Hurley, D.C. Nanomechanical mapping of the osteochondral interface with contact resonance force microscopy and nanoindentation. Acta Biomater. 2012, 8, 4389–4396. [Google Scholar] [CrossRef]
- Pordzik, J.; Bernstein, A.; Watrinet, J.; Mayr, H.O.; Latorre, S.H.; Schmal, H.; Seidenstuecker, M. Correlation of Biomechanical Alterations under Gonarthritis between Overlying Menisci and Articular Cartilage. Appl. Sci. 2020, 10, 8673. [Google Scholar] [CrossRef]
- Jin, H.; Lewis, J.L. Determination of Poisson’s ratio of articular cartilage by indentation using different-sized indenters. J. Biomech. Eng. 2004, 126, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Mäkelä, J.T.A.; Korhonen, R.K. Highly nonlinear stress-relaxation response of articular cartilage in indentation: Importance of collagen nonlinearity. J. Biomech. 2016, 49, 1734–1741. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, M.; Ojanen, S.; Mohammadi, A.; Finnilä, M.A.; Joukainen, A.; Kröger, H.; Saarakkala, S.; Korhonen, R.K.; Tanska, P. Elastic, Viscoelastic and Fibril-Reinforced Poroelastic Material Properties of Healthy and Osteoarthritic Human Tibial Cartilage. Ann. Biomed. Eng. 2019, 47, 953–966. [Google Scholar] [CrossRef]
- Wan, C.; Ge, L.; Souza, R.B.; Tang, S.Y.; Alliston, T.; Hao, Z.; Li, X. T1ρ-based fibril-reinforced poroviscoelastic constitutive relation of human articular cartilage using inverse finite element technology. Quant. Imaging Med. Surg. 2019, 9, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Nickmanesh, R.; Stewart, R.C.; Snyder, B.D.; Grinstaff, M.W.; Masri, B.A.; Wilson, D.R. Contrast-enhanced computed tomography (CECT) attenuation is associated with stiffness of intact knee cartilage. J. Orthop. Res. 2018, 36, 2641–2647. [Google Scholar] [CrossRef]
- Shepherd, D.E.; Seedhom, B.B. Thickness of human articular cartilage in joints of the lower limb. Ann. Rheum. Dis. 1999, 58, 27–34. [Google Scholar] [CrossRef]
- Steineman, B.D.; LaPrade, R.F.; Santangelo, K.S.; Warner, B.T.; Goodrich, L.R.; Haut Donahue, T.L. Early Osteoarthritis After Untreated Anterior Meniscal Root Tears: An In Vivo Animal Study. Orthop. J. Sports Med. 2017, 5, 2325967117702452. [Google Scholar] [CrossRef]
- Bushby, A.; Jennett, N. Determining the Area Function of Spherical Indenters for Nanoindentation. MRS Proc. 2000, 649, Q7.17. [Google Scholar] [CrossRef]
- Hayes, W.C.; Keer, L.M.; Herrmann, G.; Mockros, L.F. A mathematical analysis for indentation tests of articular cartilage. J. Biomech. 1972, 5, 541–551. [Google Scholar] [CrossRef]
- Li, G.; Lopez, O.; Rubash, H. Variability of a three-dimensional finite element model constructed using magnetic resonance images of a knee for joint contact stress analysis. J. Biomech. Eng. 2001, 123, 341–346. [Google Scholar] [CrossRef] [PubMed]
- June, R.K.; Fyhrie, D.P. A comparison of cartilage stress-relaxation models in unconfined compression: QLV and stretched exponential in combination with fluid flow. Comput. Methods Biomech. Biomed. Eng. 2013, 16, 565–576. [Google Scholar] [CrossRef]
- Brown, C.P.; Nguyen, T.C.; Moody, H.R.; Crawford, R.W.; Oloyede, A. Assessment of common hyperelastic constitutive equations for describing normal and osteoarthritic articular cartilage. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2009, 223, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Pierce, D.M.; Isaksen, V.; Davies, C.L.; Drogset, J.O.; Lilledahl, M.B. Comparison of Compressive Stress-Relaxation Behavior in Osteoarthritic (ICRS Graded) Human Articular Cartilage. Int. J. Mol. Sci. 2018, 19, 413. [Google Scholar] [CrossRef]
- Liu, F.; Kozanek, M.; Hosseini, A.; Van de Velde, S.K.; Gill, T.J.; Rubash, H.E.; Li, G. In vivo tibiofemoral cartilage deformation during the stance phase of gait. J. Biomech. 2010, 43, 658–665. [Google Scholar] [CrossRef]
- Liu, C.; Liu, C.; Ren, X. Quantitative evaluation of subchondral bone microarchitecture in knee osteoarthritis using 3T MRI. BMC Musculoskelet. Disord. 2017, 18, 496. [Google Scholar] [CrossRef]
- Alsaaod, M.; Luternauer, M.; Hausegger, T.; Kredel, R.; Steiner, A. The cow pedogram-Analysis of gait cycle variables allows the detection of lameness and foot pathologies. J. Dairy Sci. 2017, 100, 1417–1426. [Google Scholar] [CrossRef]
- Nilsson, J.; Thorstensson, A. Adaptability in frequency and amplitude of leg movements during human locomotion at different speeds. Acta Physiol. Scand. 1987, 129, 107–114. [Google Scholar] [CrossRef]
- Danion, F.; Varraine, E.; Bonnard, M.; Pailhous, J. Stride variability in human gait: The effect of stride frequency and stride length. Gait Posture 2003, 18, 69–77. [Google Scholar] [CrossRef]
- Jurvelin, J.S.; Räsänen, T.; Kolmonen, P.; Lyyra, T. Comparison of optical, needle probe and ultrasonic techniques for the measurement of articular cartilage thickness. J. Biomech. 1995, 28, 231–235. [Google Scholar] [CrossRef]
- Horbert, V.; Lange, M.; Reuter, T.; Hoffmann, M.; Bischoff, S.; Borowski, J.; Schubert, H.; Driesch, D.; Mika, J.; Hurschler, C.; et al. Comparison of Near-Infrared Spectroscopy with Needle Indentation and Histology for the Determination of Cartilage Thickness in the Large Animal Model Sheep. Cartilage 2019, 10, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Lin, A.S.; Levenston, M.E.; Guldberg, R.E. Quantitative assessment of articular cartilage morphology via EPIC-microCT. Osteoarthr. Cartil. 2009, 17, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Hadjab, I.; Sim, S.; Karhula, S.S.; Kauppinen, S.; Garon, M.; Quenneville, E.; Lavigne, P.; Lehenkari, P.P.; Saarakkala, S.; Buschmann, M.D. Electromechanical properties of human osteoarthritic and asymptomatic articular cartilage are sensitive and early detectors of degeneration. Osteoarthr. Cartil. 2018, 26, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Steppacher, S.D.; Hanke, M.S.; Zurmühle, C.A. Ultrasonic cartilage thickness measurement is accurate, reproducible, and reliable—validation study using contrast-enhanced micro-CT. J. Orthop. Surg. Res. 2019, 14, 67. [Google Scholar] [CrossRef]
- Veronesi, F.; Berni, M.; Marchiori., G.; Cassiolas, G.; Muttini, A.; Barboni, B.; Martini, L.; Fini, M.; Lopomo, N.F.; Marcacci, M.; et al. Evaluation of cartilage biomechanics and knee joint microenvironment after different cell-based treatments in a sheep model of early osteoarthritis. Int. Orthop. 2021, 45, 427–435. [Google Scholar] [CrossRef]
- Suh, J.K.; Youn, I.; Fu, F.H. An in situ calibration of an ultrasound transducer: A potential application for an ultrasonic indentation test of articular cartilage. J. Biomech. 2001, 34, 1347–1353. [Google Scholar] [CrossRef]
- Miller, G.J.; Morgan, E.F. Use of microindentation to characterize the mechanical properties of articular cartilage: Comparison of biphasic material properties across length scales. Osteoarthr. Cartil. 2010, 18, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Oyen, M.; Shean, T.; Strange, D.; Galli, M. Size effects in indentation of hydrated biological tissues. J. Mater. Res. 2012, 27, 245–255. [Google Scholar] [CrossRef]
- Shepherd, D.E.; Seedhom, B.B. The ‘instantaneous’ compressive modulus of human articular cartilage in joints of the lower limb. Rheumatology 1999, 38, 124–132. [Google Scholar] [CrossRef]
- Han, G.; Chowdhury, U.; Eriten, M.; Henak, C.R. Relaxation capacity of cartilage is a critical factor in rate- and integrity-dependent fracture. Sci. Rep. 2021, 11, 9527. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Yu, S.; Wu, C.; Zhang, W. The mechanical properties of tibiofemoral and patellofemoral articular cartilage in compression depend on anatomical regions. Sci. Rep. 2021, 11, 6128. [Google Scholar] [CrossRef] [PubMed]
- Wahlquist, J.A.; DelRio, F.W.; Randolph, M.A.; Aziz, A.H.; Heveran, C.M.; Bryant, S.J.; Neu, C.P.; Ferguson, V.L. Indentation mapping revealed poroelastic, but not viscoelastic, properties spanning native zonal articular cartilage. Acta Biomater. 2017, 64, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Nia, H.T.; Han, L.; Li, Y.; Ortiz, C.; Grodzinsky, A. Poroelasticity of cartilage at the nanoscale. Biophys. J. 2011, 101, 2304–2313. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.S.; Falcovitz, Y.H.; Schneiderman, R.; Maroudas, A.; Sah, R.L. Depth-dependent compressive properties of normal aged human femoral head articular cartilage: Relationship to fixed charge density. Osteoarthr. Cartil. 2001, 9, 561–569. [Google Scholar] [CrossRef]
- Thambyah, A.; Broom, N.D. Further insight into the depth-dependent microstructural response of cartilage to compression using a channel indentation technique. Comput. Math. Methods 2013, 2013, 358192. [Google Scholar] [CrossRef]
- Wilson, W.; Huyghe, J.M.; van Donkelaar, C.C. Depth-dependent Compressive Equilibrium Properties of Articular Cartilage Explained by its Composition. Biomech. Modeling Mechanobiol. 2007, 6, 43–53. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berni, M.; Erani, P.; Lopomo, N.F.; Baleani, M. Optimization of In Situ Indentation Protocol to Map the Mechanical Properties of Articular Cartilage. Materials 2022, 15, 6425. https://doi.org/10.3390/ma15186425
Berni M, Erani P, Lopomo NF, Baleani M. Optimization of In Situ Indentation Protocol to Map the Mechanical Properties of Articular Cartilage. Materials. 2022; 15(18):6425. https://doi.org/10.3390/ma15186425
Chicago/Turabian StyleBerni, Matteo, Paolo Erani, Nicola Francesco Lopomo, and Massimiliano Baleani. 2022. "Optimization of In Situ Indentation Protocol to Map the Mechanical Properties of Articular Cartilage" Materials 15, no. 18: 6425. https://doi.org/10.3390/ma15186425






