Fatigue and Fracture Resistance Testing of Polyether Ether Ketone (PEEK) Implant Abutments in an Ex Vivo Chewing Simulator Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Outcome Measurements
2.3. Statistics
3. Results
3.1. Dynamic Loading in the Chewing Simulator
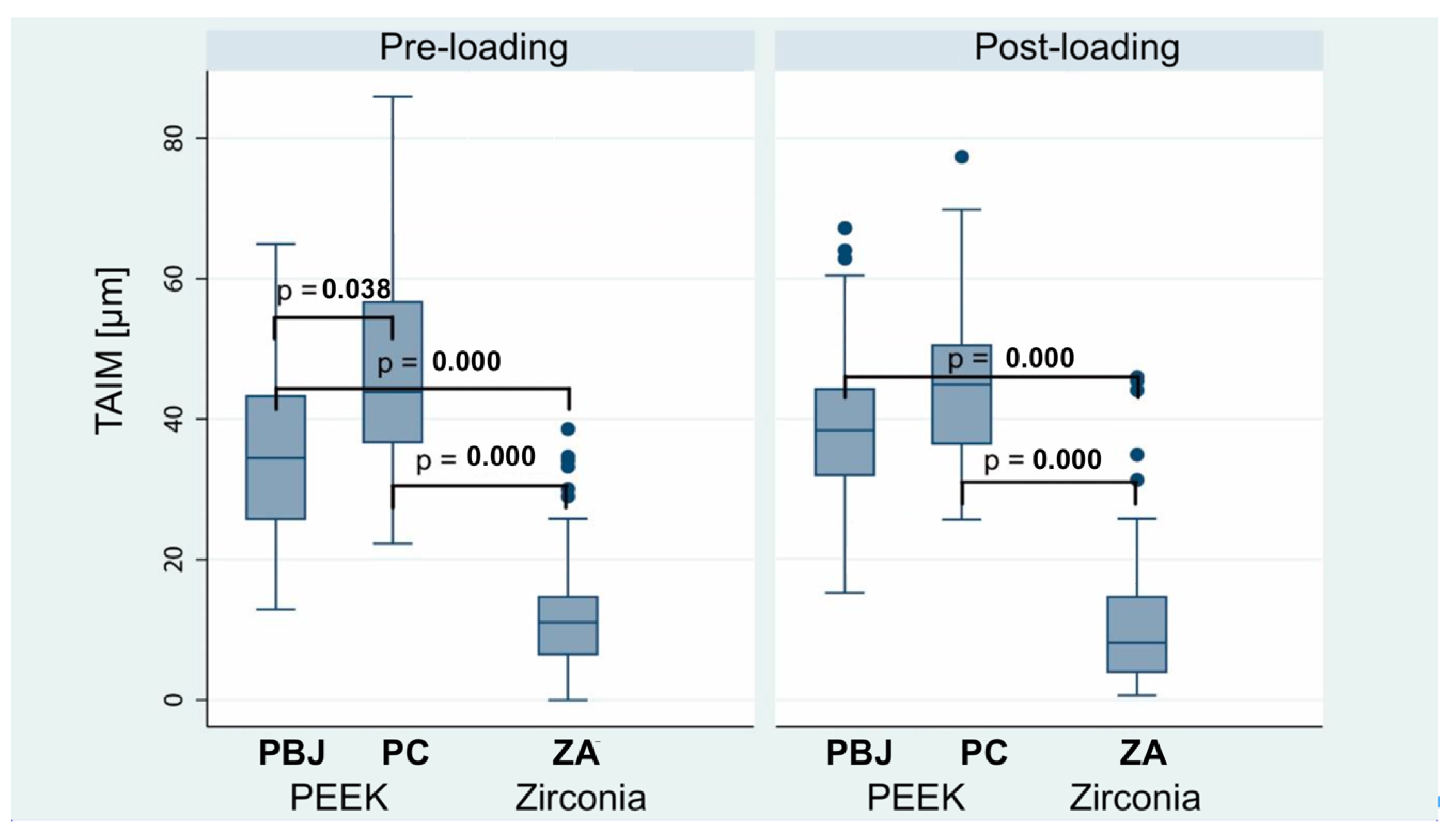
3.2. Assessment of Implant-Abutment Connection Microgaps (IACMs) and Titanium Base-Abutment Interface Microgaps (TAIMs)
3.3. Evaluation of Surface Characteristics
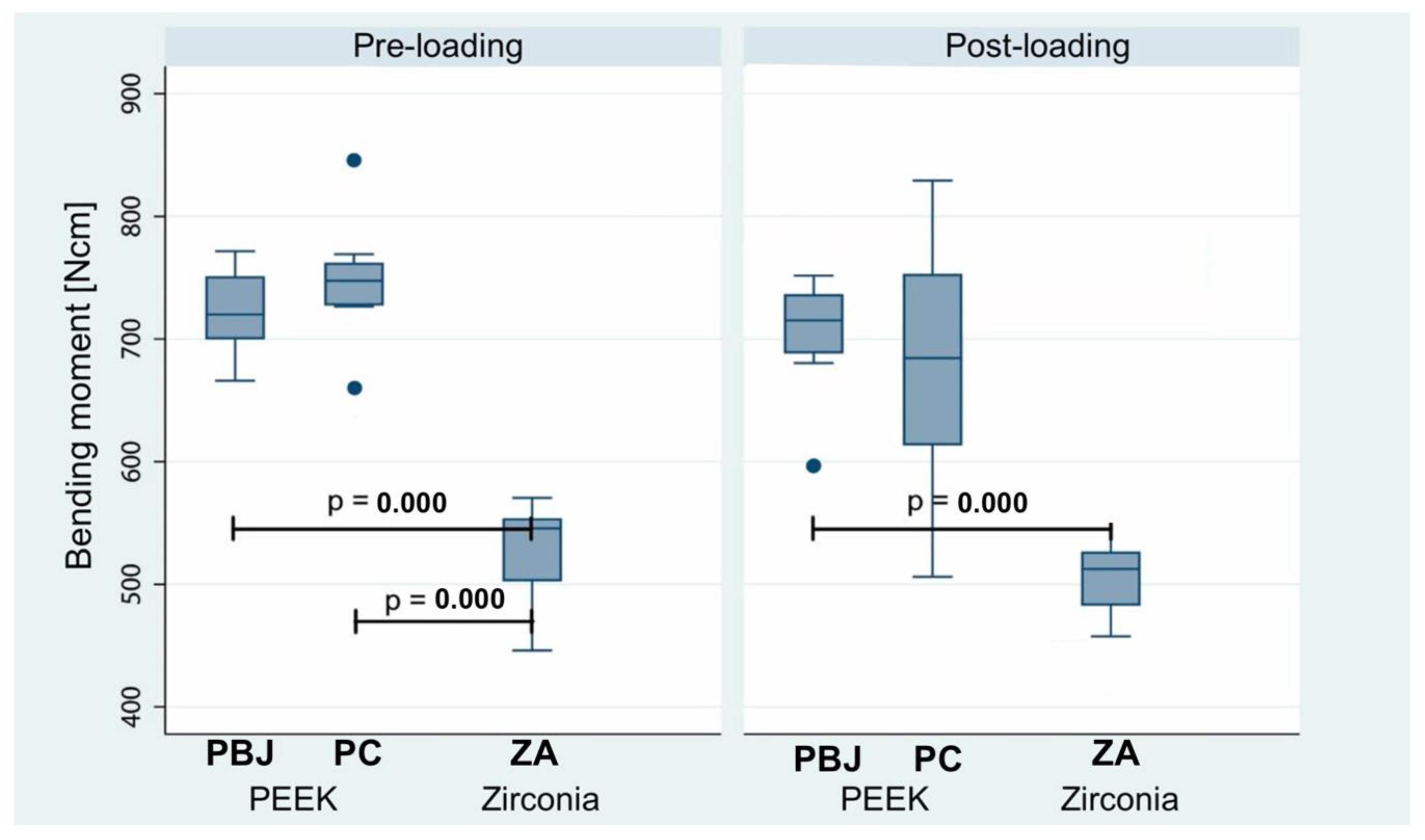
3.4. Static Loading Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sailer, I.; Karasan, D.; Todorovic, A.; Ligoutsikou, M.; Pjetursson, B.E. Prosthetic Failures in Dental Implant Therapy. Periodontol. 2000 2022, 88, 130–144. [Google Scholar] [CrossRef] [PubMed]
- Montero, J. A Review of the Major Prosthetic Factors Influencing the Prognosis of Implant Prosthodontics. J. Clin. Med. 2021, 10, 816. [Google Scholar] [CrossRef]
- Mayuri, S.; Irfan, A.K.; Raj, R.; Sen, A.; Malik, R.; Bandgar, S.; Rangari, P. Success of Dental Implant Influenced by Abutment Types and Loading Protocol. J. Pharm. Bioallied. Sci. 2022, 14, S1019–S1022. [Google Scholar] [CrossRef]
- Kohal, R.; Fross, A.; Adolfsson, E.; Bagegni, A.; Doerken, S.; Spies, B.C. Fatigue Resistance of a Two-Piece Zirconia Oral Implant. An Investigation in the Chewing Simulator. Clin. Oral Implant. Res. 2018, 29, 54. [Google Scholar] [CrossRef][Green Version]
- Bagegni, A.; Weihrauch, V.; Vach, K.; Kohal, R. The Mechanical Behavior of a Screwless Morse Taper Implant-Abutment Connection: An In Vitro Study. Materials 2022, 15, 3381. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, M.; Geramipanah, F.; Negahdari, R.; Rakhshan, V. Active Tactile Sensibility of Single-Tooth Implants versus Natural Dentition: A Split-Mouth Double-Blind Randomized Clinical Trial. Clin. Implant. Dent. Relat. Res. 2014, 16, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Totou, D.; Naka, O.; Mehta, S.B.; Banerji, S. Esthetic, Mechanical, and Biological Outcomes of Various Implant Abutments for Single-Tooth Replacement in the Anterior Region: A Systematic Review of the Literature. Int. J. Implant Dent. 2021, 7, 85. [Google Scholar] [CrossRef] [PubMed]
- Halim, F.C.; Pesce, P.; De Angelis, N.; Benedicenti, S.; Menini, M. Comparison of the Clinical Outcomes of Titanium and Zirconia Implant Abutments: A Systematic Review of Systematic Reviews. J. Clin. Med. 2022, 11, 5052. [Google Scholar] [CrossRef] [PubMed]
- de Holanda Cavalcanti Pereira, A.K.; de Oliveira Limirio, J.P.J.; Cavalcanti do Egito Vasconcelos, B.; Pellizzer, E.P.; Dantas de Moraes, S.L. Mechanical Behavior of Titanium and Zirconia Abutments at the Implant-Abutment Interface: A Systematic Review. J. Prosthet. Dent. 2022, in press. [Google Scholar] [CrossRef]
- Zembic, A.; Bösch, A.; Jung, R.E.; Hämmerle, C.H.F.; Sailer, I. Five-Year Results of a Randomized Controlled Clinical Trial Comparing Zirconia and Titanium Abutments Supporting Single-Implant Crowns in Canine and Posterior Regions. Clin. Oral Implants Res. 2013, 24, 384–390. [Google Scholar] [CrossRef]
- Alqutaibi, A.Y.; Ghulam, O.; Krsoum, M.; Binmahmoud, S.; Taher, H.; Elmalky, W.; Zafar, M.S. Revolution of Current Dental Zirconia: A Comprehensive Review. Molecules 2022, 27, 1699. [Google Scholar] [CrossRef] [PubMed]
- Shelar, P.; Abdolvand, H.; Butler, S. On the Behaviour of Zirconia-Based Dental Materials: A Review. J. Mech. Behav. Biomed. Mater. 2021, 124, 104861. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.A.; Hastings, R.S.; Mason, J.J.; Moet, A. Characterization of Short-Fibre Reinforced Thermoplastics for Fracture Fixation Devices. Biomaterials 1990, 11, 541–547. [Google Scholar] [CrossRef]
- Allan, J.M.; Wrana, J.S.; Linden, D.E.; Shalaby, S.W.; Farris, H.; Budsberg, S.; Dooley, R.L. Bone Formation into Surface Phosphonylated Polymeric Implants. Crit. Rev. Biomed. Eng. 2000, 28, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Tang, T. Current Strategies to Improve the Bioactivity of PEEK. Int. J. Mol. Sci. 2014, 15, 5426–5445. [Google Scholar] [CrossRef]
- Suphangul, S.; Rokaya, D.; Kanchanasobhana, C.; Rungsiyakull, P.; Chaijareenont, P. PEEK Biomaterial in Long-Term Provisional Implant Restorations: A Review. J. Funct. Biomater. 2022, 13, 33. [Google Scholar] [CrossRef]
- Knaus, J.; Schaffarczyk, D.; Cölfen, H. On the Future Design of Bio-Inspired Polyetheretherketone Dental Implants. Macromol. Biosci. 2020, 20, 1900239. [Google Scholar] [CrossRef]
- Koch, F.P.; Weng, D.; Krämer, S.; Biesterfeld, S.; Jahn-Eimermacher, A.; Wagner, W. Osseointegration of One-Piece Zirconia Implants Compared with a Titanium Implant of Identical Design: A Histomorphometric Study in the Dog. Clin. Oral Implants Res. 2010, 21, 350–356. [Google Scholar] [CrossRef]
- AlOtaibi, N.; Naudi, K.; Conway, D.; Ayoub, A. The Current State of PEEK Implant Osseointegration and Future Perspectives: A Systematic Review. eCM 2020, 40, 1–20. [Google Scholar] [CrossRef]
- Ghazal-Maghras, R.; Vilaplana-Vivo, J.; Camacho-Alonso, F.; Martínez-Beneyto, Y. Properties of Polyetheretheretherketone (PEEK) Implant Abutments: A Systematic Review. J. Clin. Exp. Dent. 2022, 14, e349–e358. [Google Scholar] [CrossRef]
- Tetelman, E.D.; Babbush, C.A. A New Transitional Abutment for Immediate Aesthetics and Function. Implant Dent. 2008, 17, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Hahnel, S.; Wieser, A.; Lang, R.; Rosentritt, M. Biofilm Formation on the Surface of Modern Implant Abutment Materials. Clin. Oral Implants Res. 2015, 26, 1297–1301. [Google Scholar] [CrossRef]
- Santing, H.J.; Meijer, H.J.A.; Raghoebar, G.M.; Özcan, M. Fracture Strength and Failure Mode of Maxillary Implant-Supported Provisional Single Crowns: A Comparison of Composite Resin Crowns Fabricated Directly over PEEK Abutments and Solid Titanium Abutments. Clin. Implant Dent. Relat. Res. 2012, 14, 882–889. [Google Scholar] [CrossRef] [PubMed]
- Rosentritt, M.; Rembs, A.; Behr, M.; Hahnel, S.; Preis, V. In Vitro Performance of Implant-Supported Monolithic Zirconia Crowns: Influence of Patient-Specific Tooth-Coloured Abutments with Titanium Adhesive Bases. J. Dent. 2015, 43, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Preis, V.; Hahnel, S.; Behr, M.; Bein, L.; Rosentritt, M. In-Vitro Fatigue and Fracture Testing of CAD/CAM-Materials in Implant-Supported Molar Crowns. Dent. Mater. 2017, 33, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Martínez, J.; Delgado, L.M.; Ortiz-Hernández, M.; Punset, M.; Cano-Batalla, J.; Cayon, M.R.; Cabratosa-Termes, J. In Vitro Assessment of PEEK and Titanium Implant Abutments: Screw Loosening and Microleakage Evaluations under Dynamic Mechanical Testing. J. Prosthet. Dent. 2022, 127, 470–476. [Google Scholar] [CrossRef]
- DeLong, R.; Douglas, W.H. An Artificial Oral Environment for Testing Dental Materials. IEEE Trans. Biomed. Eng. 1991, 38, 339–345. [Google Scholar] [CrossRef]
- Walia, T.; Brigi, C.; KhirAllah, A.R.M.M. Comparative Evaluation of Surface Roughness of Posterior Primary Zirconia Crowns. Eur. Arch. Paediatr. Dent. 2019, 20, 33–40. [Google Scholar] [CrossRef]
- Elshiyab, S.H.; Nawafleh, N.; Khan, U.; Öchsner, A.; George, R. Impact of Coping Veneering Techniques on the Survival of Implant-Supported Zirconia-Based-Crowns Cemented to Hybrid-Abutments: An in-Vitro Study. Bioengineering 2020, 2020, 117. [Google Scholar] [CrossRef]
- Mitsias, M.; Koutayas, S.O.; Wolfart, S.; Kern, M. Influence of Zirconia Abutment Preparation on the Fracture Strength of Single Implant Lithium Disilicate Crowns after Chewing Simulation. Clin. Oral Implants Res. 2014, 25, 675–682. [Google Scholar] [CrossRef]
- Baladhandayutham, B.; Lawson, N.C.; Burgess, J.O. Fracture Load of Ceramic Restorations after Fatigue Loading. J. Prosthet. Dent. 2015, 114, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gao, S.; Han, Y.; Yang, Q.; Arola, D.; Zhang, D. Bearing Capacity of Ceramic Crowns before and after Cyclic Loading: An in Vitro Study. J. Mech. Behav. Biomed. Mater. 2018, 87, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, M.; Kleven, E.; Gotfredsen, K. Fracture Mode during Cyclic Loading of Implant-Supported Single-Tooth Restorations. J. Prosthet. Dent. 2012, 108, 74–83. [Google Scholar] [CrossRef]
- Bankoğlu Güngör, M.; Karakoca Nemli, S. Fracture Resistance of CAD-CAM Monolithic Ceramic and Veneered Zirconia Molar Crowns after Aging in a Mastication Simulator. J. Prosthet. Dent. 2018, 119, 473–480. [Google Scholar] [CrossRef]
- Johansson, C.; Kmet, G.; Rivera, J.; Larsson, C.; Vult Von Steyern, P. Fracture Strength of Monolithic All-Ceramic Crowns Made of High Translucent Yttrium Oxide-Stabilized Zirconium Dioxide Compared to Porcelain-Veneered Crowns and Lithium Disilicate Crowns. Acta Odontol. Scand. 2014, 72, 145–153. [Google Scholar] [CrossRef]
- Nawafleh, N.; Hatamleh, M.M.; Öchsner, A.; Mack, F. The Impact of Core/Veneer Thickness Ratio and Cyclic Loading on Fracture Resistance of Lithium Disilicate Crown. J. Prosthodont. 2018, 27, 75–82. [Google Scholar] [CrossRef]
- Homaei, E.; Farhangdoost, K.; Pow, E.H.N. Fatigue Resistance of Monolithic CAD/CAM Ceramic Crowns on Human Premolars. Ceram. Int. 2016, 42, 15709–15717. [Google Scholar] [CrossRef]
- Obermeier, M.; Ristow, O.; Erdelt, K.; Beuer, F. Mechanical Performance of Cement- and Screw-Retained All-Ceramic Single Crowns on Dental Implants. Clin Oral Investig. 2018, 22, 981–991. [Google Scholar] [CrossRef]
- Garoushi, S.; Säilynoja, E.; Vallittu, P.K.; Lassila, L. Fracture-Behavior of CAD/CAM Ceramic Crowns before and after Cyclic Fatigue Aging. Int. J. Prosthodont. 2021. online ahead of print. [Google Scholar] [CrossRef]
- Wang, R.-R.; Lu, C.-L.; Wang, G.; Zhang, D.-S. Influence of Cyclic Loading on the Fracture Toughness and Load Bearing Capacities of All-Ceramic Crowns. Int. J. Oral Sci. 2014, 6, 99–104. [Google Scholar] [CrossRef]
- Nawafleh, N.A.; Hatamleh, M.M.; Öchsner, A.; Mack, F. Fracture Load and Survival of Anatomically Representative Monolithic Lithium Disilicate Crowns with Reduced Tooth Preparation and Ceramic Thickness. J. Adv. Prosthodont. 2017, 9, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Kessler, A.; Reymus, M.; Hickel, R.; Kunzelmann, K.-H. Three-Body Wear of 3D Printed Temporary Materials. Dent. Mater. 2019, 35, 1805–1812. [Google Scholar] [CrossRef] [PubMed]
- Myagmar, G.; Lee, J.-H.; Ahn, J.-S.; Yeo, I.-S.L.; Yoon, H.-I.; Han, J.-S. Wear of 3D Printed and CAD/CAM Milled Interim Resin Materials after Chewing Simulation. J. Adv. Prosthodont. 2021, 13, 144. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis. Curr. Dir. Psychol. Sci. 1992, 1, 98–101. [Google Scholar] [CrossRef]
- Agustín-Panadero, R.; Serra-Pastor, B.; Roig-Vanaclocha, A.; Román-Rodriguez, J.-L.; Fons-Font, A. Mechanical Behavior of Provisional Implant Prosthetic Abutments. Med. Oral Patol. Oral Cir. Bucal. 2015, 20, e94–e102. [Google Scholar] [CrossRef]
- Al-Omiri, M.K.; Sghaireen, M.G.; Alhijawi, M.M.; Alzoubi, I.A.; Lynch, C.D.; Lynch, E. Maximum Bite Force Following Unilateral Implant-Supported Prosthetic Treatment: Within-Subject Comparison to Opposite Dentate Side. J. Oral Rehabil. 2014, 41, 624–629. [Google Scholar] [CrossRef]
- Gehrke, P.; Sing, T.; Fischer, C.; Spintzyk, S.; Geis-Gerstorfer, J. Marginal and Internal Adaptation of Hybrid Abutment Assemblies After Central and Local Manufacturing, Respectively. Int. J. Oral Maxillofac. Implants 2018, 33, 808–814. [Google Scholar] [CrossRef]
- Baj, A.; Beltramini, G.A.; Bolzoni, A.; Cura, F.; Palmieri, A.; Scarano, A.; Ottria, L.; Giannì, A.B. Bacterial Colonization of the Implant-Abutment Interface of Conical Connection with an Internal Octagon: An in Vitro Study Using Real-Time PCR. J. Biol. Regul. Homeost. Agents 2017, 31, 163–168. [Google Scholar]
- Mencio, F.; De Angelis, F.; Papi, P.; Rosella, D.; Pompa, G.; Di Carlo, S. A Randomized Clinical Trial about Presence of Pathogenic Microflora and Risk of Peri-Implantitis: Comparison of Two Different Types of Implant-Abutment Connections. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1443–1451. [Google Scholar]
- Bollen, C.M.; Lambrechts, P.; Quirynen, M. Comparison of Surface Roughness of Oral Hard Materials to the Threshold Surface Roughness for Bacterial Plaque Retention: A Review of the Literature. Dent. Mater. 1997, 13, 258–269. [Google Scholar] [CrossRef]
- Rashid, H. The Effect of Surface Roughness on Ceramics Used in Dentistry: A Review of Literature. Eur. J. Dent. 2014, 08, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Coray, R.; Zeltner, M.; Özcan, M. Fracture Strength of Implant Abutments after Fatigue Testing: A Systematic Review and a Meta-Analysis. J. Mech. Behav. Biomed. Mater. 2016, 62, 333–346. [Google Scholar] [CrossRef]
- Keyak, J.H.; Kaneko, T.S.; Tehranzadeh, J.; Skinner, H.B. Predicting Proximal Femoral Strength Using Structural Engineering Models. Clin. Orthop. Relat. Res. 2005, 437, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; van Staden, R.; Loo, Y.-C.; Johnson, N.; Ivanovski, S.; Meredith, N. Influence of Bone and Dental Implant Parameters on Stress Distribution in the Mandible: A Finite Element Study. Int. J. Oral Maxillofac. Implants 2009, 24, 866–876. [Google Scholar]
- Saravi, B.E.; Putz, M.; Patzelt, S.; Alkalak, A.; Uelkuemen, S.; Boeker, M. Marginal Bone Loss around Oral Implants Supporting Fixed versus Removable Prostheses: A Systematic Review. Int. J. Implant Dent. 2020, 6, 20. [Google Scholar] [CrossRef]
- Korioth, T.W.; Waldron, T.W.; Versluis, A.; Schulte, J.K. Forces and Moments Generated at the Dental Incisors during Forceful Biting in Humans. J. Biomech. 1997, 30, 631–633. [Google Scholar] [CrossRef]
- Fontijn-Tekamp, F.A.; Slagter, A.P.; Van Der Bilt, A.; Van ’T Hof, M.A.; Witter, D.J.; Kalk, W.; Jansen, J.A. Biting and Chewing in Overdentures, Full Dentures, and Natural Dentitions. J. Dent. Res. 2000, 79, 1519–1524. [Google Scholar] [CrossRef]
- Geminiani, A.; Lee, H.; Feng, C.; Ercoli, C. The Influence of Incisal Veneering Porcelain Thickness of Two Metal Ceramic Crown Systems on Failure Resistance after Cyclic Loading. J Prosthet Dent 2010, 103, 275–282. [Google Scholar] [CrossRef]
- Kohal, R.-J.; Finke, H.C.; Klaus, G. Stability of Prototype Two-Piece Zirconia and Titanium Implants after Artificial Aging: An in Vitro Pilot Study. Clin. Implant Dent. Relat. Res. 2009, 11, 323–329. [Google Scholar] [CrossRef]
- Heintze, S.D.; Eser, A.; Monreal, D.; Rousson, V. Using a Chewing Simulator for Fatigue Testing of Metal Ceramic Crowns. J. Mech. Behav. Biomed. Mater. 2017, 65, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, M.; Buciumeanu, M.; Henriques, B.; Silva, F.S.; Souza, J.C.M.; Gomes, J.R. Comparison between PEEK and Ti6Al4V Concerning Micro-Scale Abrasion Wear on Dental Applications. J. Mech. Behav. Biomed. Mater. 2016, 60, 212–219. [Google Scholar] [CrossRef] [PubMed]







| Pre-Loading | |||||
|---|---|---|---|---|---|
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 6.9 | 6.8 | 1.5 | 3.0 | 10.9 |
| PBJ | 15.7 | 15.9 | 4.5 | 6.9 | 32.3 |
| ZA | 1.8 | 2.0 | 1.1 | 0 | 5.9 |
| Post-Loading | |||||
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 9.2 | 9.3 | 1.7 | 5.3 | 13.2 |
| PBJ | 10.1 | 10.5 | 5.1 | 2.4 | 21.1 |
| ZA | 1.7 | 2.0 | 1.4 | 0 | 7.6 |
| Pre-Loading | |||||
|---|---|---|---|---|---|
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 43.9 | 47.2 | 14.2 | 22.3 | 85.9 |
| PBJ | 34.5 | 36.3 | 11.8 | 12.9 | 64.9 |
| ZA | 11.1 | 11.9 | 8.0 | 0 | 38.6 |
| Post-Loading | |||||
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 44.9 | 44.8 | 10.3 | 25.7 | 77.3 |
| PBJ | 38.4 | 38.8 | 10.2 | 15.3 | 67.2 |
| ZA | 8.2 | 11.0 | 9.7 | 0.7 | 46.0 |
| Pre-Loading Ra (in µm) | |||||
|---|---|---|---|---|---|
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 1.2 | 1.2 | 0.4 | 0.5 | 2.7 |
| PBJ | 0.9 | 1.0 | 0.2 | 0.6 | 1.9 |
| ZA | 1.5 | 1.5 | 0.4 | 0.6 | 3.1 |
| Post-Loading Ra (in µm) | |||||
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 1.0 | 1.0 | 0.2 | 0.6 | 2.3 |
| PBJ | 1.0 | 1.0 | 0.2 | 0.7 | 1.5 |
| ZA | 1.5 | 1.5 | 0.4 | 0.7 | 2.7 |
| Pre-Loading Rz (in µm) | |||||
|---|---|---|---|---|---|
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 11.3 | 12.9 | 7.2 | 5.7 | 68.1 |
| PBJ | 8.6 | 9.0 | 2.1 | 6.1 | 19.0 |
| ZA | 16.3 | 16.6 | 4.6 | 6.1 | 36.9 |
| Post-Loading Rz (in µm) | |||||
| Group | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC | 8.9 | 9.3 | 2.4 | 5.5 | 25.2 |
| PBJ | 8.6 | 8.8 | 1.3 | 6.1 | 13.1 |
| ZA | 15.7 | 16.3 | 4.6 | 6.9 | 33.6 |
| Unloaded Control Groups | ||||||
|---|---|---|---|---|---|---|
| Group | n | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC-NL | 8 | 1100 | 1101 0,a | 86 | 965 | 1270 |
| PBJ-NL | 8 | 1055 | 1056 0,a | 48 | 997 | 1150 |
| ZA-NL | 8 | 797 | 772 1*,a | 58 | 652 | 822 |
| Loaded Experimental Groups | ||||||
| Group | n | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC-L | 8 | 944 | 851 0,a | 354 | 49 | 1200 |
| PBJ-L | 8 | 1045 | 1018 0,a | 68 | 871 | 1080 |
| ZA-L | 8 | 748 | 738 1*,a | 42 | 665 | 805 |
| Unloaded Control Groups | ||||||
|---|---|---|---|---|---|---|
| Group | n | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC-NL | 8 | 747 | 747 0,a | 52 | 659 | 846 |
| PBJ-NL | 8 | 720 | 722 0,a | 35 | 666 | 772 |
| ZA-NL | 8 | 546 | 528 1*,a | 41 | 446 | 570 |
| Loaded Experimental Groups | ||||||
| Group | n | Median | Mean | Standard Deviation | Minimum Value | Maximum Value |
| PC-L | 8 | 661 | 596 0,1,a | 246 | 36 | 829 |
| PBJ-L | 8 | 715 | 704 0,a | 49 | 597 | 752 |
| ZA-L | 8 | 512 | 508 1*,a | 33 | 458 | 561 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saravi, B.; Flohr, A.; Patzelt, S.B.; Spies, B.C.; Hazard, D.; Kohal, R.J. Fatigue and Fracture Resistance Testing of Polyether Ether Ketone (PEEK) Implant Abutments in an Ex Vivo Chewing Simulator Model. Materials 2022, 15, 6927. https://doi.org/10.3390/ma15196927
Saravi B, Flohr A, Patzelt SB, Spies BC, Hazard D, Kohal RJ. Fatigue and Fracture Resistance Testing of Polyether Ether Ketone (PEEK) Implant Abutments in an Ex Vivo Chewing Simulator Model. Materials. 2022; 15(19):6927. https://doi.org/10.3390/ma15196927
Chicago/Turabian StyleSaravi, Babak, Anselm Flohr, Sebastian B. Patzelt, Benedikt C. Spies, Derek Hazard, and Ralf J. Kohal. 2022. "Fatigue and Fracture Resistance Testing of Polyether Ether Ketone (PEEK) Implant Abutments in an Ex Vivo Chewing Simulator Model" Materials 15, no. 19: 6927. https://doi.org/10.3390/ma15196927
APA StyleSaravi, B., Flohr, A., Patzelt, S. B., Spies, B. C., Hazard, D., & Kohal, R. J. (2022). Fatigue and Fracture Resistance Testing of Polyether Ether Ketone (PEEK) Implant Abutments in an Ex Vivo Chewing Simulator Model. Materials, 15(19), 6927. https://doi.org/10.3390/ma15196927












