1. Introduction
The type of reactor included gas cooled reactor (GCR), light water reactor (LWGR), heavy water reactors (PWR), and fast reactors (FRs) [
1]. The space reactor included thermal neutron reactor and fast neutron reactor. There were Romaska, Topaz-I, Topaz-II and Topaz* in Russia’s research on space reactor, and the life of them were from Several months to 7 years. American research on space reactor included SNAP-10A and SNAP-100, and the life of them were 43 days and 7–10 years [
2,
3].
Because of its high thermal stability, high hydrogen density [
4], low neutron capture cross-section, and good thermal conductivity [
5,
6,
7], zirconium hydride became an ideal neutron moderator and was put into application in space nuclear reactor. However, hydrogen will escape from zirconium hydride in the working environment, leading to the loss of hydrogen and hence deteriorating the moderating effect of the moderator [
8]. The hydrogen loss can be prohibited or hindered by manufacturing a hydrogen permeation barrier film on the surface of zirconium hydride.
Substantial research has been conducted on the preparation of hydrogen permeation barrier on stainless steel, such as oxide films of Y
2O
3, Cr
2O
3, and Al
2O
3 [
9,
10,
11], carbide films of SiC [
12], and nitride films of AlN, Si
3N
4, BN, TiAlN [
13,
14,
15,
16,
17], and PRF values measured were from 40 to 20,000. Zirconium hydride was generally used in honeycomb structure, which made the preparation method of the hydrogen permeation barrier on stainless steel unsuitable for it. Therefore electroplating, micro arc oxidation, sol-gel, and in situ reaction methods were developed to form hydrogen permeation barrier on the zirconium hydride. Chen et al. [
18] studied the preparation of the films on zirconium hydride by in situ reaction method in O
2, CO
2, and CO
2 + P atmospheres, and the results showed the M-ZrO
2 and T-ZrO
2 film structure, and the films prepared in CO
2 + P atmosphere had the best hydrogen blocking effect. Wu et al. [
19,
20] prepared composite oxide films containing Si-Al, Si-Zr, and Si-P on the zirconium hydride by sol-gel method, obtaining a top sol-gel layer and a transition zirconium oxide bottom layer with excellent hydrogen barrier effect. Yan et al. [
21] fabricated ZrO
2 composite film by in situ reaction of urea with zirconium hydride at high temperature, resulting in a film composed of Zr, C, N, and O elements; and C-H, O-H, and N-H bonds were detected in the film, revealing that hydrogen was trapped by C, N, and O in the coating. Qi et al. [
22] prepared phosphating film on zirconium hydride, for which the hydrogen permeation rate of samples was measured by gas chromatography, and the hydrogen loss predicted showed a good performance.
Nitride film has also been an important hydrogen barrier category because of its good hydrogen permeation barrier effect, high hardness, and excellent corrosion resistance [
23,
24]. Methods for preparing nitride film include ion nitriding, carbonitriding and gas nitriding, in which gas nitriding is divided into ammonia nitriding and nitrogen nitriding [
24]. Zhang et al. [
25] utilized ultrasonic nanocrystalline surface modification (UNSM) technology and low-temperature ammonia nitriding to prepare nitriding layer on the surface of NiCr 718 alloy where the grain boundaries on the surface of the nanocrystals provided the diffusion channels of nitrogen, and this technology reduced the nitriding temperature and time, and improved the hardness, wear resistance, and corrosion resistance of the nitride films. Yilbas et al. [
26] prepared nitriding layers on the surface of the tungsten using high-pressure nitrogen with laser assistant, which made the surface of the tungsten nanocrystalline free of microcracks and large-size holes, and the surface hardness was significantly improved, and this nitriding process overcame the binding force of the metal atoms on the metal surface by producing active nitrogen atoms, thus penetrated into the matrix [
24]. However, thus far, no research has been published on forming nitride film on zirconium hydride.
In this study, zirconium nitride film was prepared on zirconium hydride by nitrogen nitriding. The phase structure, morphology, element and valence distribution of the film were investigated. The hydrogen permeation performance of nitride film was studied, and was compared with oxide film manufactured by in situ reaction with CO
2 [
18]. The hydrogen permeation mechanism of the nitride film was revealed.
4. Conclusions
In this work, a composite nitride film was prepared on zirconium hydride by the in situ reaction method in a nitrogen atmosphere.
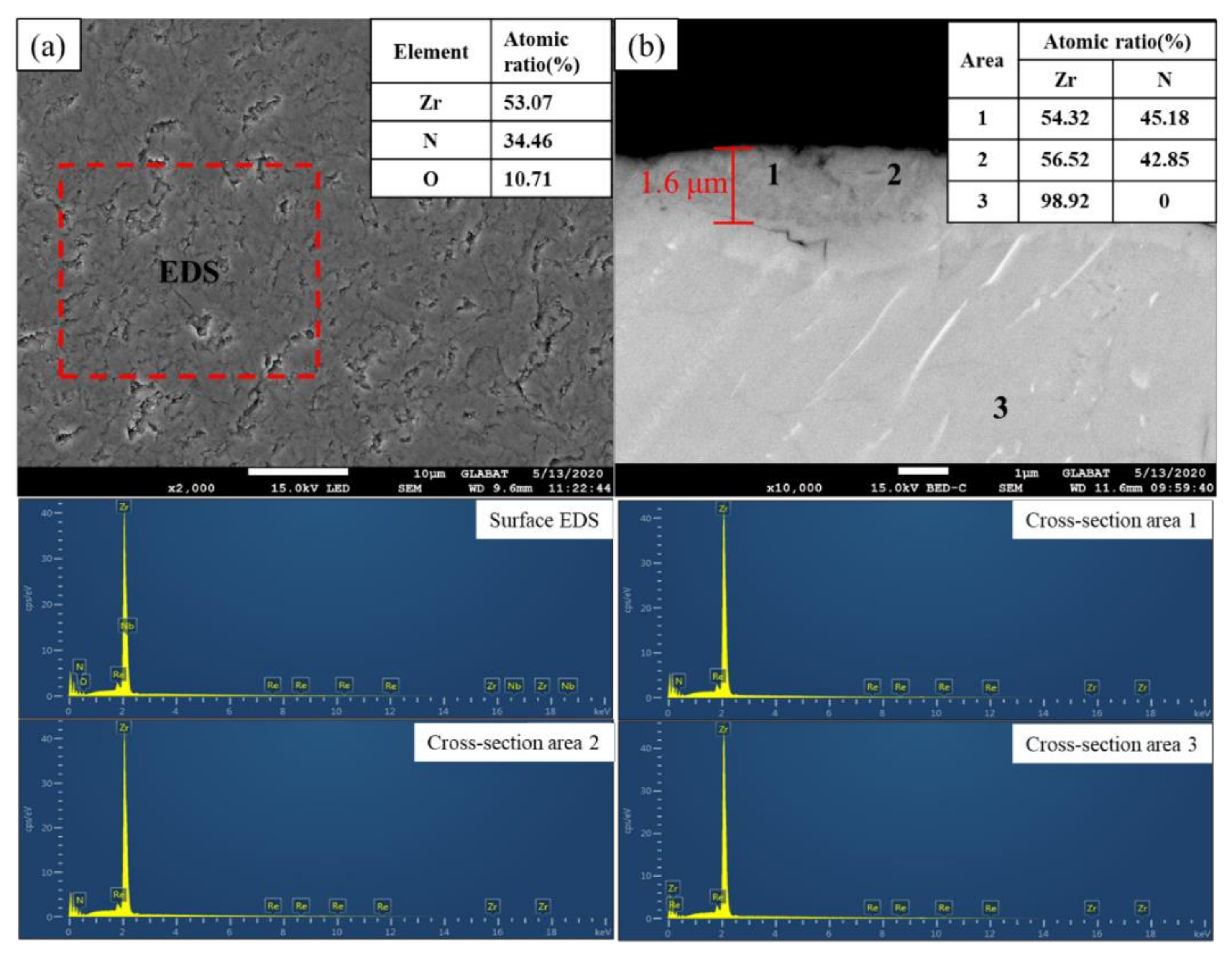
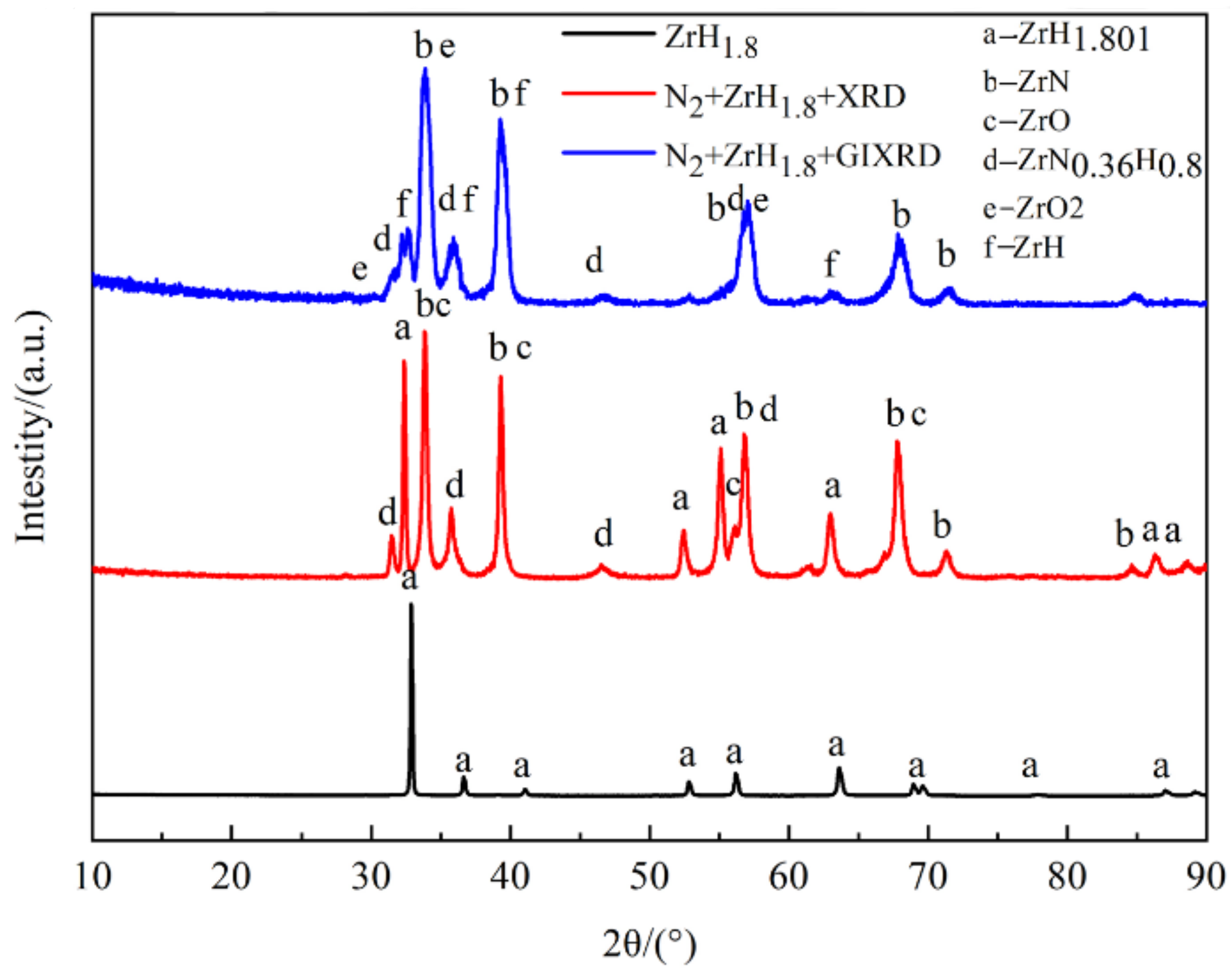
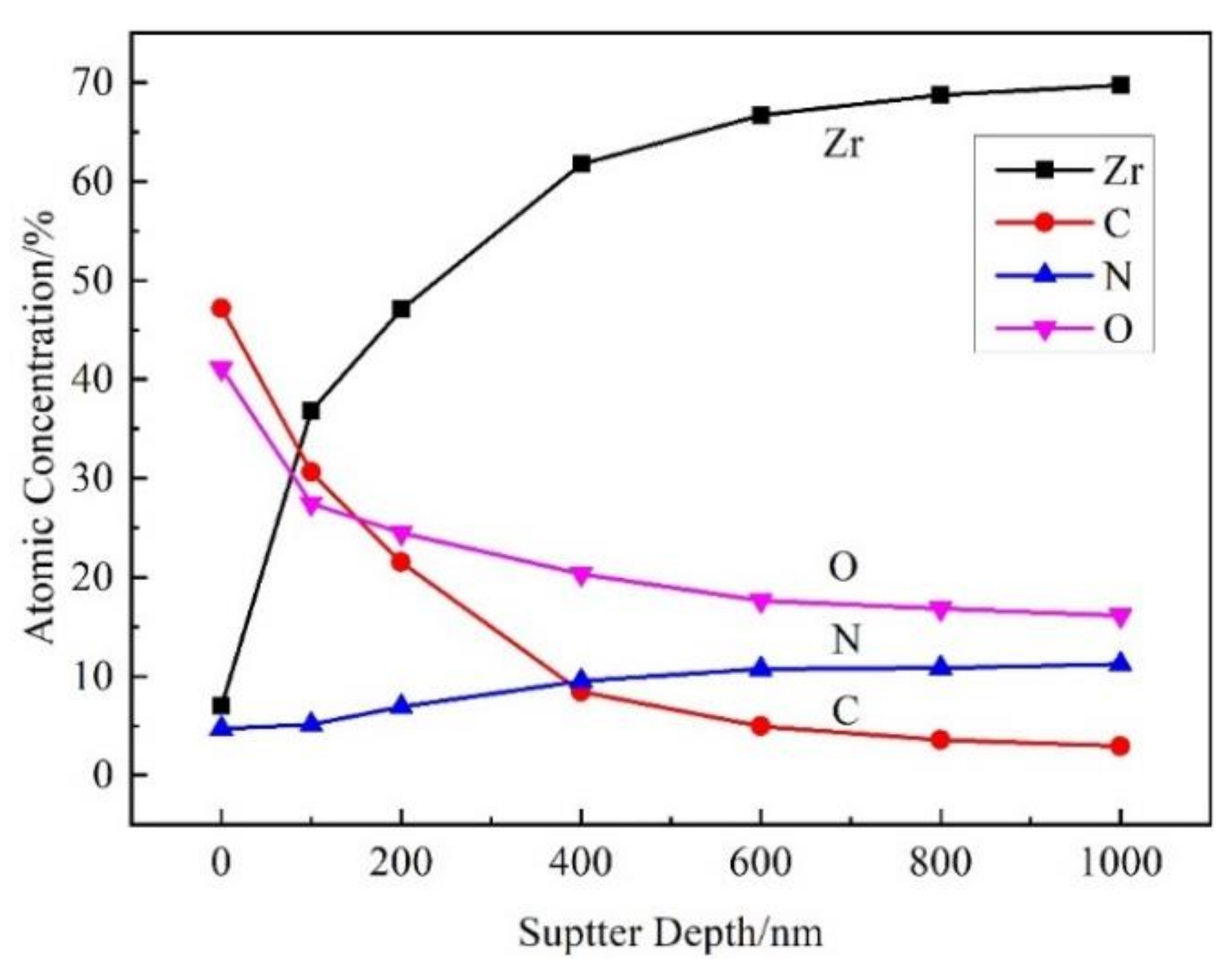
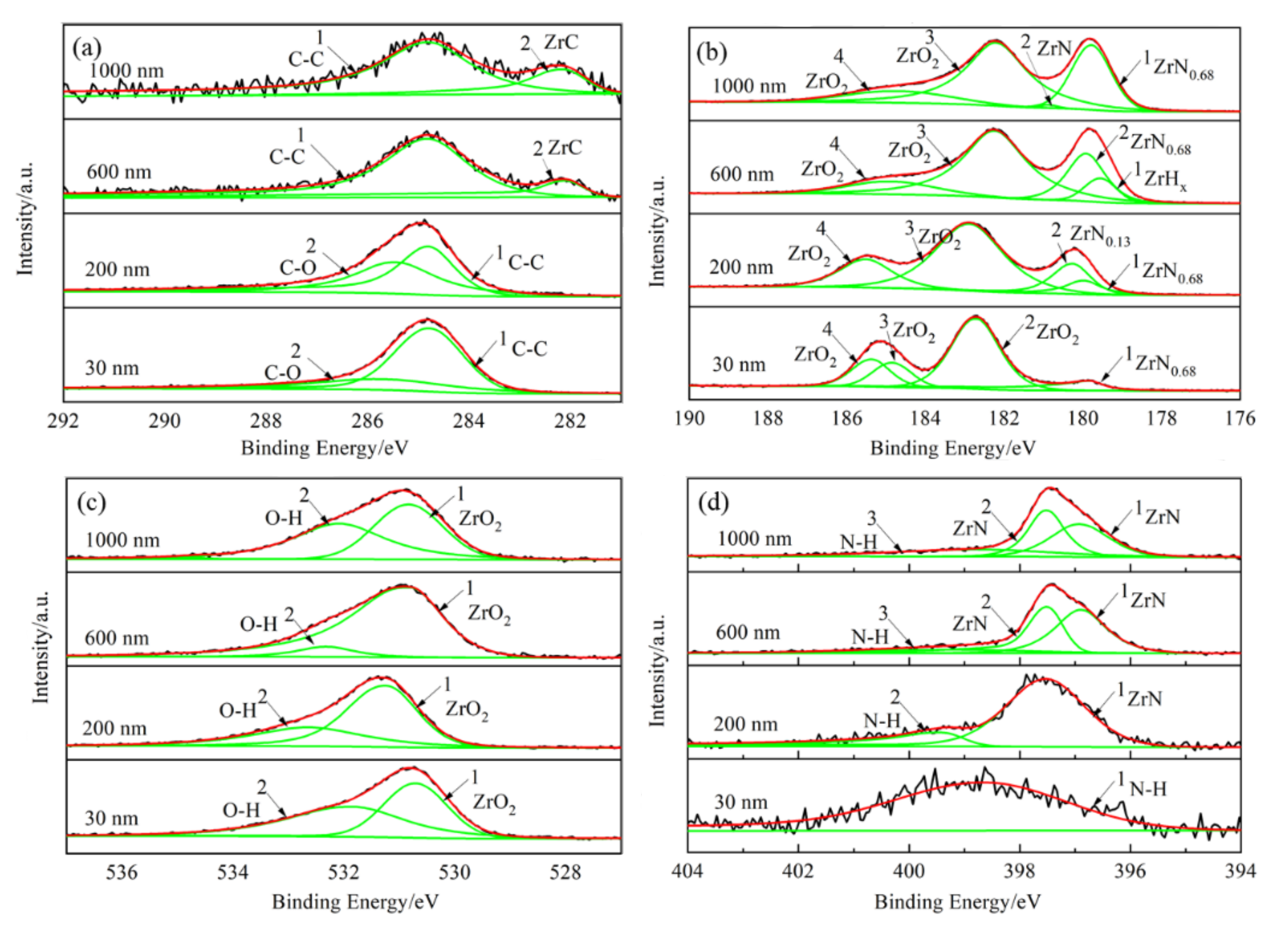
The film was golden color, smooth, continuous, and dense, with a thickness of about 1.6 μm. XRD analysis showed that the main phase of the film was ZrN, with coexistence of ZrO2, ZrO, and ZrN0.36H0.8. The element distribution of nitride film was compared with that of previously studied by in situ reaction with urea at 600 °C by AES analysis. Two types of films were composed of elements C, N, O, and Zr; the content of N in the film of this study was the main interstitial element, but O was the main interstitial element in the film of the previous study, and the content of C in the film of this study was obviously lower than that of the previously studied with urea as the reaction material. XPS analysis revealed that there were Zr-C, Zr-O, Zr-N, O-H, N-H bonds in the film, which further verified the results of AES analysis. Based on these characterization results, the hydrogen barrier mechanism could be attributed to the existence of the ZrN0.36H0.8 phase and the bonds of O-H and N-H, indicating that the nitrogen and oxygen in the film could capture the hydrogen from zirconium hydride matrix.
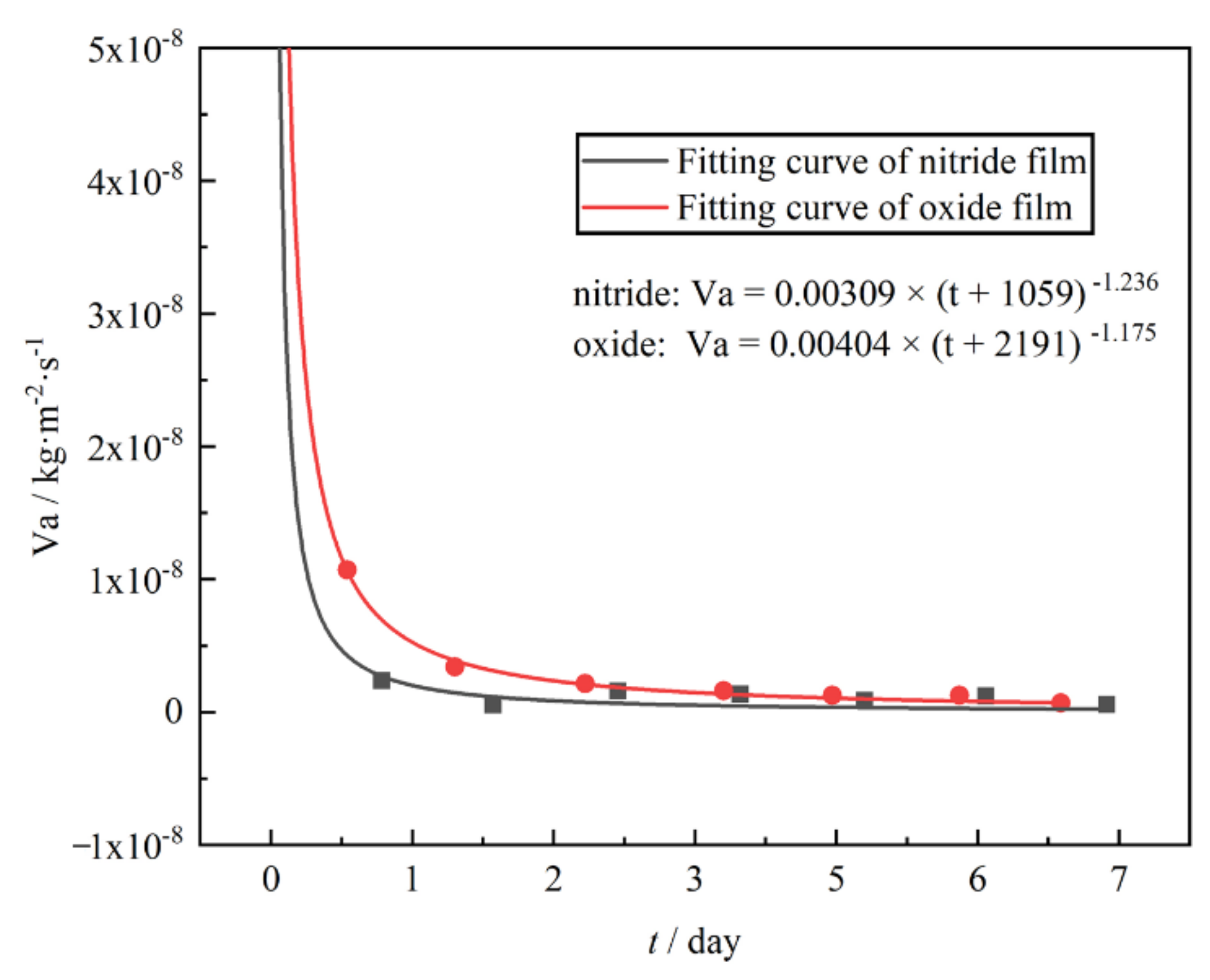
Finally, the hydrogen permeation resistance performance of the nitride film was compared with that of oxide film by PRF, VTD, and HPR methods, and the results showed that the hydrogen permeation barrier effects of nitride film was better than that of oxide film. This study investigated the preparation and hydrogen permeation behavior of zirconium nitride film on zirconium hydride and revealed the hydrogen barrier mechanism of nitride films.