Study on Morphology and Chemical States of Surface Active Layer of Th-W Cathode
Abstract
:1. Introduction
2. Experiment Methods
3. Results and Discussion
3.1. Original Microstructure of Wire
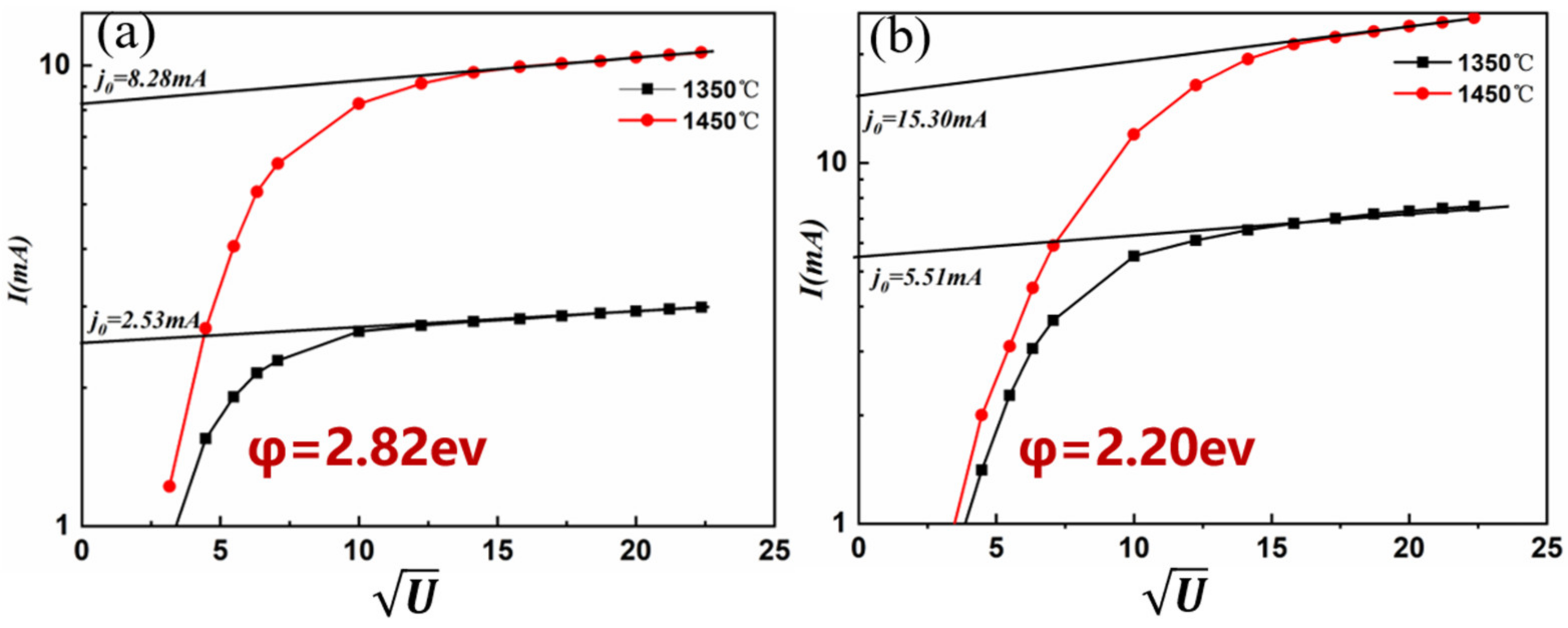
3.2. The Morphology and Structure of the Surface Active Layer
3.3. Study on the Valence State of Surface-Active Layer
4. Conclusions
- Under the action of vacuum and high temperature, the tungsten matrix recrystallises, the grain boundaries increase and the active atoms migrate outwards along the grain boundaries, which facilitates the formation of the active layer.
- For surfaces where an active layer has formed, the etching action of the Ar+ lance removes the active layer from the surface of the tungsten substrate. After the surface oxides and active elements have been removed, the surface is at this point almost composed of tungsten and such a surface does not have good electron emission capability.
- The active layer formed by the thorium-oxygen compound at high temperature would produce metal ultrafine particles, which increase the concentration of high-energy valence electrons, reduce the electron work function, and improve the emission capability.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Park, D.H.; Seo, E.R.; Kwon, M.K.; Lee, C.S. A study on thermal fluid flow of magnetron cooling for microwave oven. J. Mech. Sci. Technol. 2019, 33, 1915–1923. [Google Scholar] [CrossRef]
- Stolyarov, O.I. Matching the microwave-oven chamber to the magnetron. J. Commun. Technol. Electron. 2016, 61, 190–196. [Google Scholar] [CrossRef]
- Nie, Z.R.; Zhuo, M.L.; Zhang, J.X.; Chen, Y.; Zuo, T.Y. Rare earth tungsten electrode materials and the role of rare earth oxides. Rare Met. Mater. Eng. 1997, 26, 1–6. [Google Scholar]
- Jousten, K.; Boineau, F.; Bundaleski, N.; Illgen, C.; Setina, J.; Teodoro, O.M.N.D.; Vicar, M.; Wüest, M. A review on hot cathode ionisation gauges with focus on a suitable design for measurement accuracy and stability. Vacuum 2020, 179, 109545. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, P.; Yang, J.; Zhou, S.; Zhang, P.; Nie, Z. Study on morphology and chemical states of surface active layer of Ce-W cathode. Appl. Surf. Sci. 2019, 479, 815–821. [Google Scholar] [CrossRef]
- Li, S.-H.; Li, J.-Q. Studies of virtual cathode characteristics near thermionic emission cathodes in a vacuum. Vacuum 2021, 192, 110496. [Google Scholar] [CrossRef]
- Cui, Y.T.; Li, B.S.; Peng, Y.; Wang, L.Y.; Yang, J.C. Study on welding performance and fabrication of tungsten electrode doped with non-radiating rare earths oxide. J. Therm. Spray Technol. 2013, 5, 65–68. [Google Scholar]
- Liu, S.Y. Study on the Processing Technology of Ternary; Beijing University of Technology: Beijing, China, 2009. [Google Scholar]
- Ushakov, A.; Karpov, I.; Shaihadinov, A.; Fedorov, L.; Goncharova, E.; Demin, V. Ionization features in cathode spot region of vacuum arc. Vacuum 2020, 179, 109509. [Google Scholar] [CrossRef]
- Liu, S.H.; Liu, X.Y. Optimizing of addition of rare-earth elements in tungsten electrode. Rare Met. Cem. Carbides 2003, 31, 15–17. [Google Scholar]
- Lin, Z.L.; Wang, X.J. Cathode Electronics; National Defense Industry Press: Beijing, China, 2013. [Google Scholar]
- Wang, P.; Yang, J.; Zhang, T.; Gao, Z. Evolution of the Chemical Valence on the Surface of the Cerium—Tungsten Electrode. In Advances in Energy and Environmental Materials, Proceedings of Chinese Materials Conference; Springer: Singapore, 2018; pp. 727–737. [Google Scholar]
- Zhang, H.; Chen, X.; Yang, Z.; Ding, B. W–ThO2 thermionic cathode activated with nanoscale oxide. Mater. Lett. 1999, 38, 401–405. [Google Scholar] [CrossRef]
- Nie, Z.R.; Zuo, T.Y.; Zhou, M.L.; Wang, Y.M.; Wang, J.S.; Zhang, J.X. High temperature XPS/AES investigation of Mo–La2O3 Cathode-I. species and properties of oxygen on surface. J. Rare Earths 1999, 4, 267–270. [Google Scholar]
- Yang, J.; Nie, Z.; Wang, Y. Microstructure and emission ability of rare earth oxides doped molybdenum cathodes. Appl. Surf. Sci. 2003, 215, 87–95. [Google Scholar] [CrossRef]
- Böröczki, Á.; Gaál, I.; Gurbán, S.; Menyhárd, M.; Horváth, E.; Tóth, A.L.; Petrás, L.; Balázs, L. Electron stimulated thorium adatom enrichment on the surface of thoriated tungsten below 2300 K. Int. J. Refract. Met. Hard Mater. 2005, 24, 343–349. [Google Scholar] [CrossRef]
- Fait, M.J.G.; Radnik, J.; Lunk, H.J. Surface tungsten reduction during thermal decomposition of ammonium paratungstate tetrahydrate in oxidising atmosphere: A paradox? Thermochim. Acta 2016, 633, 77–81. [Google Scholar] [CrossRef]
- Bouvard, O.; Krammer, A.; Schüler, A. In situ core-level and valence-band photoelectron spectroscopy of reactively sputtered tungsten oxide films. Surf. Interface Anal. 2016, 48, 660–663. [Google Scholar] [CrossRef]
- Watts, J.F.; Wolstenholme, J. An Introduction to Surface Analysis by XPS and AES; Wiley: Hoboken, NJ, USA, 2003; Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1002/9781119417651.fmatter (accessed on 10 March 2022).
- Selverian, J.H. Finite difference model of thorium transport in tungsten. Acta Mater. 1997, 45, 5099–5110. [Google Scholar] [CrossRef]
- Xie, F.; Gong, L.; Liu, X.; Tao, Y.; Zhang, W.; Chen, S.; Meng, H.; Chen, J. XPS studies on surface reduction of tungsten oxide nanowire film by Ar+ bombardment. J. Electron Spectrosc. Relat. Phenom. 2012, 185, 112–118. [Google Scholar] [CrossRef]
- Paparazzo, E. On the number, binding energies, and mutual intensities of Ce3d peaks in the XPS analysis of cerium oxide systems: A response to Murugan et al., Superlatt. Microstruct. 85 (2015) 321. Superlattices Microstruct. 2017, 105, 216–220. [Google Scholar] [CrossRef]
- Vijayakumar, P.; Pandian, M.S.; Mukhopadhyay, S.; Ramasamy, P. Synthesis and characterizations of large surface tungsten oxide nanoparticles as a novel counter electrode for dye-sensitized solar cell. J. Sol-Gel Sci. Technol. 2015, 75, 487–494. [Google Scholar] [CrossRef]
- Zhao, L.Z. XPS studies of a series of Ce (IV) and Ce (III) compounds. Acta Phys. Sin. 1989, 38, 987–990. [Google Scholar] [CrossRef]
- Xue, Z.Q.; Liu, W.M. Nanoelectronics; Electronic Industry Press: Beijing, China, 2003. [Google Scholar]










| Element | Test Position | Cross-Sectional | Average Value | Longitudinal Section | Average Value |
|---|---|---|---|---|---|
| Th | 1 | 30.76 | 31.87 | 26.21 | 24.35 |
| 32 | 23.8 | ||||
| 33.35 | 23.04 | ||||
| 2 | 15.91 | 16.82 | 4.63 | 5.21 | |
| 17.1 | 6.1 | ||||
| 18.17 | 4.9 | ||||
| W | 1 | 54.15 | 53.26 | 45.47 | 46.26 |
| 53.8 | 47.1 | ||||
| 52.33 | 46.21 | ||||
| 2 | 76.13 | 75.31 | 60.06 | 59.66 | |
| 75.2 | 58.7 | ||||
| 74.6 | 60.22 | ||||
| O | 1 | 17.09 | 14.87 | 28.32 | 29.39 |
| 15.2 | 29.1 | ||||
| 13.32 | 30.75 | ||||
| 2 | 9.79 | 7.87 | 35.3 | 35.13 | |
| 7.7 | 35.2 | ||||
| 6.12 | 34.89 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Y.; Sun, Y.; Zhou, Y.; Wang, S.; Meng, J.; Cao, N.; Shi, W. Study on Morphology and Chemical States of Surface Active Layer of Th-W Cathode. Materials 2022, 15, 2726. https://doi.org/10.3390/ma15082726
Cheng Y, Sun Y, Zhou Y, Wang S, Meng J, Cao N, Shi W. Study on Morphology and Chemical States of Surface Active Layer of Th-W Cathode. Materials. 2022; 15(8):2726. https://doi.org/10.3390/ma15082726
Chicago/Turabian StyleCheng, Yin, Yuan Sun, Yizhou Zhou, Shiyang Wang, Jie Meng, Nan Cao, and Wanpeng Shi. 2022. "Study on Morphology and Chemical States of Surface Active Layer of Th-W Cathode" Materials 15, no. 8: 2726. https://doi.org/10.3390/ma15082726






