Fabrication and Arc Erosion Behavior of Ag-SnO2-ZnO Electrical Contact Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of Ag-SnO2-ZnO Composite Powders
2.2. Production of Ag-SnO2-ZnO Composite Samples
2.3. Characterization
2.4. Electrical Contact Test
3. Results
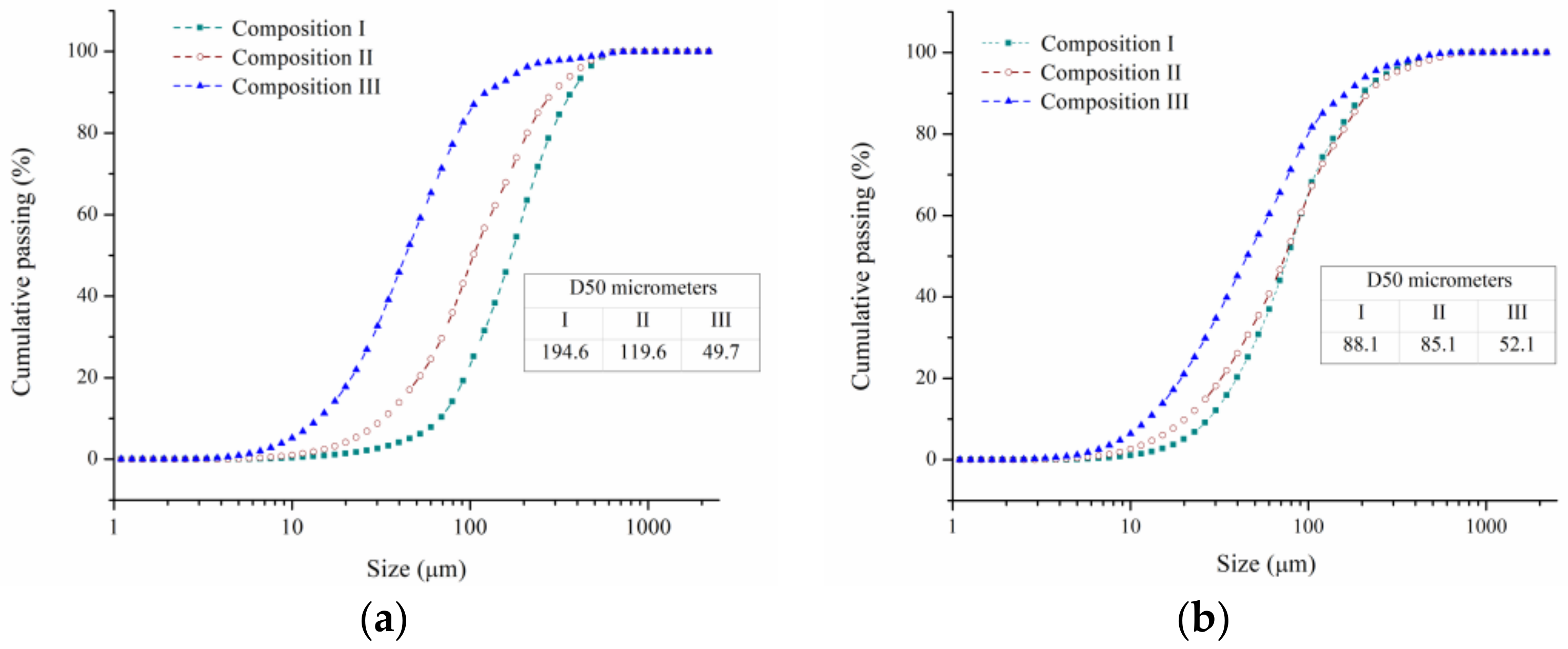
3.1. Morphological and Microstructural Examination of the Powders
3.2. Physical and Microstructural Characterization of the Sintered Samples
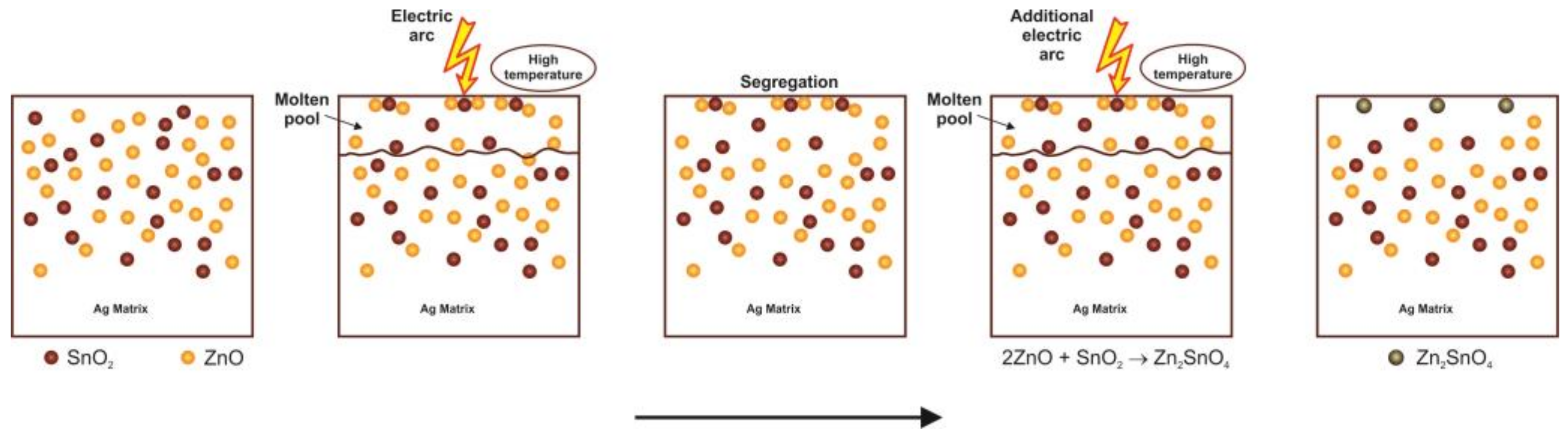
3.3. Arc Erosion Behavior of Ag-SnO2-ZnO
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ćosović, V.; Ćosović, A.; Talijan, N.; Živković, D.; Živković, Ž. State of the art and challenges in development of electrical contact materials in the light of the RoHS directive. Sci. Sinter. 2012, 44, 245–253. [Google Scholar] [CrossRef]
- Talijan, N. Electrical contact materials based on silver. Zaštita Mater. 2011, 52, 173–180. [Google Scholar]
- Wu, C.; Yi, D.; Wen, W.; Li, S.; Zhou, J. Influence of alloy components on arc erosion morphology of Ag/MeO electrical contact materials. Trans. Nonferrous Met. Soc. 2016, 26, 185–195. [Google Scholar] [CrossRef]
- Wojtasik, K.; Missol, W. PM helps develop cadmium-free electrical contacts. Met. Powder Rep. 2004, 59, 34–39. [Google Scholar] [CrossRef]
- Manhart, H.; Rieder, W. Erosion behavior and erodibility of Ag/CdO and Ag/SnO2 contacts under AC3 and AC4 test conditions. IEEE Trans. Compon. Hybr. 1990, 13, 56–64. [Google Scholar] [CrossRef]
- Shou Shen, Y.; Gould, L.; Swann, S. DTA and TGA studies of four Ag-MeO electrical contact materials. IEEE Trans. Compon. Hybr. 1985, 8, 352–358. [Google Scholar] [CrossRef]
- Kubono, T. Sticking Trouble Rate on Cd-Added Ag-Based Contacts. Electron. Commun. Jpn. 1995, 78, 246–253. [Google Scholar] [CrossRef]
- Peana, M.; Pelucelli, A.; Chasapis, C.; Perlepes, S.; Bekiari, V.; Medici, S.; Zoroddu, M. Biological effects of human exposure to environmental cadmium. Biomolecules 2023, 13, 36. [Google Scholar] [CrossRef]
- Chen, X.; Wang, Z.; Zhu, G.; Ding, X.; Jin, T. The references level of cadmium intake for renal dysfunction in a Chinese population. Sci. Rep. 2018, 8, 9011. [Google Scholar] [CrossRef]
- Slade, P. Electrical Contacts, Principles and Applications, 2nd ed.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2013; p. 883. [Google Scholar]
- Li, G.; Fang, X.; Feng, W.; Liu, J. In situ formation and doping of Ag/SnO2 electrical contact materials. J. Alloys Compd. 2017, 716, 106–111. [Google Scholar] [CrossRef]
- Chen, J.; Jia, H.; Zhu, G.; Chen, S.; Li, A. Observation on AgSnO2In2O3 molten bridge phenomena under different currents. Indian J. Eng. Mater. Sci. 2017, 24, 369–376. [Google Scholar]
- Wang, Z.; Wang, Y. Impact of convection-diffusion and flow-path interactions on the dynamic evolution of microstructure: Arc erosion behavior of Ag-SnO2 contact materials. J. Alloys Compd. 2019, 774, 1046–1058. [Google Scholar] [CrossRef]
- Liu, X.; Wu, S.; Chu, P.; Chung, C.; Zheng, J.; Li, S. Effects of coating process on the characteristics of Ag-SnO2 contact materials. Mater. Chem. Phys. 2006, 98, 477–480. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; Du, J.; Meng, F. Influence of rare earth on the wetting ability of AgSnO2 contact material. Rare Met. Mater. Eng. 2014, 43, 1846–1849. [Google Scholar] [CrossRef]
- Cao, M.; Feng, Y.; Wang, L.; Zhao, T.; Zhao, H.; Zhou, Z. Effect of La2Sn2O7 content on Ag-La2Sn2O7/SnO2 arc erosion behavior and mechanism. J. Rare Earth 2022, 40, 1488–1498. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, J.; An, L.; Wang, H. Preparation and study of nano-Ag/SnO2 electrical contact material doped with titanium element. Rare Met. Mater. Eng. 2014, 43, 1566–1570. [Google Scholar] [CrossRef]
- Wu, C.; Yi, D.; Goto, S.; Xu, C.; Weng, W.; Zhou, J.; Xu, G. Oxidation of Ag-Sn–Sb alloy powders. Mater. Corros. 2012, 63, 522–530. [Google Scholar] [CrossRef]
- Wu, C.; Yuan, M.; Zhao, C.; Wu, O.; Huang, R.; Xu, G. Oxidation kinetics characteristics of Ag-5.08Sn-3.14Sb alloy powders in different oxidizing atmosphere. Corros. Sci. 2022, 199, 110167. [Google Scholar] [CrossRef]
- Rapp, R. The transition from internal to external oxidation and the formation of interruption bands in silver-indium alloys. Acta Metall. Mater. 1961, 9, 730–741. [Google Scholar] [CrossRef]
- Wathanyu, K.; Rojananan, S. Microstructures and hardness of silver alloys after internal oxidation process. Adv. Mater. Res. 2011, 194–196, 1217–1220. [Google Scholar] [CrossRef]
- Schimmel, G.; Rettenmayr, M.; Kempf, B.; Fischer-Buehner, J. Study on the microstructure of internally oxidized Ag-Sn–In alloys. Oxid. Met. 2008, 70, 25–38. [Google Scholar] [CrossRef]
- Wingert, P.; Brecher, C.; Kim, H.; Kang, S. Electrical contact material of Ag, SnO2, GeO2 and ln2O3. U.S. Patent 4,817,695, 4 April 1989. [Google Scholar]
- Verma, P.; Pandey, O.; Verma, A. Influence of metal oxides on the arc erosion behavior of silver metal oxides electrical contact materials. J. Mater. Sci. Technol. 2004, 20, 49–52. [Google Scholar]
- Gavriliu, S.; Lungu, M.; Enescu, E.; Nitu, S.; Patroi, D. A comparative study concerning the obtaining and using of some Ag-Cd, Ag-ZnO and Ag-SnO2 sintered electrical contact materials. Optoelectron. Adv. Mater. Rapid Commun. 2009, 58, 688–692. [Google Scholar]
- Li, G.; Cui, H.; Chen, J.; Fang, X.; Feng, W.; Liu, J. Formation and effects of CuO nanoparticles on Ag/SnO2 electrical contact materials. J. Alloys Compd. 2017, 696, 1228–1234. [Google Scholar] [CrossRef]
- Wang, J.; Qiu, H.; Si, F.; Liu, S.; Matsumoto, N. Preparation of Ag/SnO2 contact material by chemical deposition. Defect Diffus. Forum. 2019, 394, 21–25. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Yang, M.; Ding, B.; Yang, Z. Observation of arc discharging process of nanocomposite Ag-SnO2 and La-doped Ag-SnO2 contact with a high-speed camera. Mater. Sci. Eng. B 2006, 131, 230–234. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, S.H.; Chen, J.L.; Mao, Y.; Xie, M.; Sun, D. Preparation of rod-like SnO2 powder and its application in Ag-SnO2 electrical contact materials. Mater. Res. Innov. 2015, 19, S152–S156. [Google Scholar] [CrossRef]
- Li, G.; Ma, Y.; Feng, W.; Zhang, C.; Yan, Z.; Fang, X.; Tian, R. Effects of the CuO additive nanoparticles on the internal strain homogenization and microstructure evolution in Ag-SnO2 composites. Mater. Charact. 2022, 192, 112212. [Google Scholar] [CrossRef]
- Han, X.; Wang, Z.; Li, G.; Feng, W.; Fang, X. Interfacial thermal stress relief in Ag-SnO2 composites by in situ formation of CuO nanoparticles additive on SnO2. Ceram Int. 2022, 48, 16638–16648. [Google Scholar] [CrossRef]
- Liu, S.; Sun, O.; Wang, J.; Guo, M.; Hou, H. Exploration of the influence mechanism of La doping on the arc erosion resistance of Ag/SnO2 contact materials by a laser-simulated arc. J. Mater. Eng. Perform. 2021, 30, 7577–7583. [Google Scholar] [CrossRef]
- Zhang, J.; Mu, C.; Shen, T.; Qi, G. Study on the milling processes and sintering properties of Ag/SnO2 electrical contact material with different micro-structured SnO2 powders. J. Sol-Gel Sci. Technol. 2023, 106, 444–454. [Google Scholar] [CrossRef]
- Zhu, H.; Yang, D.; Yu, G.; Zhang, H.; Jin, D.; Yao, K. Hydrothermal synthesis of Zn2SnO4 nanorods in the diameter regime of sub-5 nm and their properties. J. Phys. Chem. B 2006, 110, 7631–7634. [Google Scholar] [CrossRef]
- Mihaiu, S.; Toader, A.; Atkinson, I.; Mocioiu, O.; Hornoiu, C.; Teodorescu, V.; Zaharescu, M. Advanced ceramics in the SnO2-ZnO binary system. Ceram. Int. 2015, 41, 4936–4945. [Google Scholar] [CrossRef]
- Huai, Z.; Chen, J.; Qi, C.; Sun, B.; Liu, S.; Song, H.; He, J. Sintering densification behavior of ZnO–SnO2 binary ceramic targets. J. Am. Ceram. Soc. 2023, 106, 259–273. [Google Scholar] [CrossRef]
- Choi, K.; Koo, H.; Kim, T.; Kim, H. Antireflective ZnSnO/Ag bilayer-based transparent source and drain electrodes for transparent thin film transistors. Appl. Phys. Lett. 2012, 100, 263505. [Google Scholar] [CrossRef]
- Manière, C.; Saccardo, E.; Lee, G.; McKittrick, J.; Molinari, A.; Olevsky, E. Swelling negation during sintering of silver: An experimental and theoretical approach. Results Phys. 2018, 11, 79–84. [Google Scholar] [CrossRef]
- Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Lutterotti, L. MAUD—Materials Analysis Using Diffraction. Available online: http://maud.radiographema.eu/ (accessed on 20 December 2022).
- Nowik, K.; Zybała, R.; Oksiuta, Z. Formation and microstructural evolution of ferritic ODS steel powders during mechanical alloying. Materials 2023, 16, 765. [Google Scholar] [CrossRef]
- Allalhesabi, S.; Manafi, S.; Borhani, E. The structural and mechanical properties of Al-2.5%wt. B4C metal matrix nano composite fabricated by mechanical alloying. Mech. Adv. Compos. Struct. 2015, 2, 39–44. [Google Scholar] [CrossRef]
- Ivetić, T. Zinc-Tin-Oxide-based porous ceramics: Structure, preparation and properties. In Recent Advances in Porous Ceramics; Al-Naib, U., Ed.; IntechOpen: London, UK, 2018; pp. 80–81. [Google Scholar]
- Guzmán, D.; Rivera, O.; Aguilar, C.; Ordoñez, S.; Martínez, C.; Serafini, D.; Paula, R. Mechanical alloying and subsequent heat treatment of Ag-Zn powders. Trans. Nonferrous Met. Soc. China 2013, 23, 2071–2078. [Google Scholar] [CrossRef]
- King, H. Quantitative size-factors for metallic solid solutions. J. Mater. Sci. 1966, 1, 79–90. [Google Scholar] [CrossRef]
- Ecker, J.; Holzer, J.C.; Krill, C.E.; Johnson, W.L. Structural and thermodynamic properties of nanocrystalline FCC metals prepared by mechanical attrition. J. Mater. Res. 1992, 7, 1751–1761. [Google Scholar] [CrossRef]
- Shigenori Hori, S.; Tai, H.; Kawaguchi, S. Effect of trace elements on recrystallization temperature of silver. J. Soc. Mater. Sci. Jpn. 1981, 30, 556–561. [Google Scholar] [CrossRef]
- Rhodes, W. Agglomerate and particle size effects on sintering yttria-stabilized zirconia. J. Am. Ceram. Soc. 1981, 64, 19–22. [Google Scholar] [CrossRef]
- Maki, K.; Ito, Y.; Matsunaga, H.; Mori, H. Solid-solution copper alloys with high strength and high electrical conductivity. Scr. Mater. 2013, 68, 777–780. [Google Scholar] [CrossRef]
- Ertugrul, O.; Park, H.; Onel, K.; Willert-Porada, M. Effect of particle size and heating rate in microwave sintering of 316L stainless steel. Powder Technol. 2013, 253, 703–709. [Google Scholar] [CrossRef]
- Wathanyu, K.; Rojananan, S. Microstructure, hardness and electrical conductivity of silver-metal oxides electrical contact materials. Adv. Mater. Res. 2012, 486, 529–532. [Google Scholar] [CrossRef]
- Talijan, N.; Ćosović, V.; Ćosović, A.; Živković, D. Processing and properties of silver-metal oxide electrical contact materials. Metall. Mater. Eng. 2012, 18, 259–272. [Google Scholar]
- Kumar, S.; Senthil, S.; Parameshwaran, R.; Rathanasamy, R. Fabrication of a novel silver-based electrical contact composites and easement of its mechanical and electrical properties. Arch. Metall. Mater. 2021, 66, 1087–1094. [Google Scholar] [CrossRef]
- Wang, Q.; Chung, Y. Encyclopedia of Tribology; Springer Science + Business Media: New York, NY, USA, 2013; pp. 101–104. [Google Scholar] [CrossRef]
- Güler, O.; Varol, T.; Alver, U.; Biyik, S. The wear and arc erosion behavior of novel copper based functionally graded electrical contact materials fabricated by hot pressing assisted electroless plating. Adv. Powder Technol. 2021, 32, 2873–2890. [Google Scholar] [CrossRef]
- Wu, C.; Zhao, Q.; Li, N.; Wang, H.; Yi, D.; Weng, W. Influence of fabrication technology on arc erosion of Ag/10SnO2 electrical contact materials. J. Alloys Compd. 2018, 766, 161–177. [Google Scholar] [CrossRef]
- Wu, C.; Yi, D.; Weng, W.; Li, S.; Zhou, J.; Zheng, F. Arc erosion behavior of Ag/Ni electrical contact materials. Mater. Des. 2015, 85, 511–519. [Google Scholar] [CrossRef]
- Huang, X.; Feng, Y.; Qian, G.; Liu, K. Erosion behavior of Ti3AlC2 cathode under atmosphere air arc. J. Alloys Compd. 2017, 727, 419–427. [Google Scholar] [CrossRef]












Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzmán, D.; González, F.; Muranda, D.; Aguilar, C.; Guzmán, A.; Soliz, Á.; Lozada, L.; Iturriza, I.; Castro, F. Fabrication and Arc Erosion Behavior of Ag-SnO2-ZnO Electrical Contact Materials. Materials 2023, 16, 3618. https://doi.org/10.3390/ma16103618
Guzmán D, González F, Muranda D, Aguilar C, Guzmán A, Soliz Á, Lozada L, Iturriza I, Castro F. Fabrication and Arc Erosion Behavior of Ag-SnO2-ZnO Electrical Contact Materials. Materials. 2023; 16(10):3618. https://doi.org/10.3390/ma16103618
Chicago/Turabian StyleGuzmán, Danny, Felipe González, Diego Muranda, Claudio Aguilar, Alexis Guzmán, Álvaro Soliz, Lorena Lozada, Iñigo Iturriza, and Felipe Castro. 2023. "Fabrication and Arc Erosion Behavior of Ag-SnO2-ZnO Electrical Contact Materials" Materials 16, no. 10: 3618. https://doi.org/10.3390/ma16103618








