1. Introduction
Water pollution is one of the world’s major challenges, especially when it is caused by organic dyes in printing and dyeing wastewater [
1]. The continued accumulation of such pollutants can seriously endanger the safety of water resources. At present, the main methods commonly used to treat dyeing wastewater in China are adsorption, membrane separation, and chemical precipitation. Among them, the adsorption method is considered one of the most effective due to its simple operation and high efficiency in treatment [
2]. However, traditional adsorbents have encountered obstacles in practical applications due to high preparation costs and difficulties in recovery. Therefore, finding a low-cost adsorbent material with excellent adsorption performance that is friendly to the environment is the primary challenge.
Gnanamoorthy et al. [
3] amine-functionalized Bi
2Sn
2O
7/rGO nanocomposites were prepared for the degradation of methylene blue by using thermal decomposition and in situ methods. The experimental results demonstrated that the photocatalytic performance of AF-Bi
2Sn
2O
7/rGO nanocomposites was superior to that of Bi
2Sn
2O
7 and AF-Bi
2Sn
2O
7. Gnanamoorthy et al. [
4] prepared CuNiO
2 and CuNiO
2/rGO nanocomposites by a hydrothermal method, which showed good photocatalytic properties for methylene blue. Bilgic et al. [
5] successfully immobilized BODIP derivatives on APTES-modified sporopollenin microcapsules to obtain a fluorescent microcapsule sensor (Sp-APTES-monoBODIPY) for the selectivity and removal of Cu (II). The experimental results showed that Sp-APTES-monoBODIPY could be used as an ideal adsorbent and sensor material for Cu (II) with a maximum adsorption capacity of 25 mg/g by the complex quasi-secondary kinetic model and Langmuir model. Qu et al. [
6] chose four different commercially available activated carbons to test the adsorption performance of MG. The experimental results showed that the four activated carbons conformed to a quasi-second order kinetic model at the beginning of adsorption; with increasing adsorption, the adsorption followed a quasi-first order equation, and the adsorption mechanism belonged to single-molecule physical adsorption.
Xinjiang is one of China’s largest coal fields, with an annual coal yield of nearly 400 million tons. Coal gangue (CG) is produced during coal preparation and mining, accounting for about 10–15% of the total coal production. The accumulation of CG reduces the utilization rate of available land resources, induces geological disasters, and pollutes the air and water, seriously threatening human health [
7]. For the moment, the overall comprehensive utilization efficiency of CG is relatively low. CG is partially used in building materials [
8], agricultural production [
9], fill reclamation, power generation, and other industries [
10]. Therefore, it is very meaningful to study CG’s comprehensive higher-value utilization as the raw material for constructing a clean, low-carbon, safe, and efficient coal industry system and forming a coal mine development pattern of harmonious coexistence between humans and nature.
CG can be used as the material for molecular sieves because of the high content of Al
2O
3 and SiO
2 [
11]. As the core catalytic material in several crucial processes represented by petroleum refining, molecular sieve materials are essential to resource transformation and environmental protection, playing an irreplaceable role in the sustainable development of the national economy. However, the industrial production of traditional molecular sieve materials is a typical route of high pollution and energy consumption, with a large discharge of three wastes. It is one of the industry’s urgently needed green upgrades in China’s current environmental protection strategy [
12]. After proper treatment, CG can be used as the raw industrial material for cheap molecular sieve adsorbents. Exploring new uses of CG has become a research hotspot.
With corn starch as the pore-forming agent and CG and bauxite as the raw materials, Lu et al. [
13] successfully prepared porous mullite ceramic carriers for filter membranes under a sintering temperature of 1100–1500 °C. They systematically studied the dynamic sintering behavior, phase evolution, shrinkage, porosity and pore radius, gas permeation flux, microstructure, and mechanical properties of the material, providing a new way to utilize CG comprehensively. Jablonska and Siedlecka [
14] carried out adsorption tests on organic substances and heavy metals in industrial wastewater with adsorption materials prepared from CG. The results showed that the adsorption capacity of phenol (phenol concentration up to 10 mg/dm
3) was about 50%, and the adsorption rate of heavy metals Pb (II), Ni (II), and Cu (II) was more than 70%. Jin et al. [
15] prepared a new type of CG-based composite adsorption material with CG as the raw material by the method of alkali activation hydrothermal. By investigating the optimal conditions and reaction mechanisms of product synthesis, they proved that the material had good adsorption performance for Pb
2+. Zhang et al. [
16] studied synthesizing calcium silicate hydrates from CG to remove aqueous solution and Cr (VI) and Cu (II) in aqueous solution. The optimum synthesis parameters were 700 °C, 1 h, and a Ca/Si molar ratio of 1.0. The prepared samples were subjected to quantitative adsorption tests and adsorption mechanism analysis under different conditions, such as temperature, dosage, solution pH, initial metal concentration, and reaction time. The research showed that with CG as the raw material, an environmentally friendly adsorbent calcium silicate hydrate could be prepared, effectively removing metals from aqueous solutions through different mechanisms. Kong et al. [
17] studied the extraction of iron and aluminum from CG. Liang et al. [
18] synthesized NaX zeolite from CG and explored its removal effect and mechanism of Cd
2+ and Cu
2+. However, the synthesis process and conditions were not given. Sun et al. [
19] prepared a ceramsite adsorbent that could adsorb copper ions continuously and efficiently with CG, pulverized coal, and copper slag as the main raw materials, and the maximum adsorption capacity could reach 20.6 mg/g. Bu et al. [
20] and Ge et al. [
21] studied the methods of synthesizing NaY molecular sieves by alkali fusion and hydrothermal with CG as the raw material, respectively. The synthesized NaY had good adsorption performance for lead (Pb
2+), and the removal rate was as high as 100%. After five adsorption/desorption cycles, the removal rate was still over 63.71%. Zhao et al. [
22] prepared a CG-rape straw biochar (CG-RS) composite from CG and rape straw to treat chromium-containing wastewater. The experiment showed that the composite material could effectively remove chromium from wastewater. To sum up, a few scholars have reported that cellulose aerogel was used as a template for the synthesis of zeolite molecular sieves to treat CG in previous studies.
According to commercial zeolite molecular sieves, this paper provides a new method of using cellulose green template and solvent in the synthesis of solid waste CG-based molecular sieves. To realize the resource utilization of wastes, CG is used as the raw material to prepare molecular sieve environmental remediation materials, which greatly reduces the discharge of three wastes and improves the comprehensive utilization of solid wastes. By adjusting the ratio of silicon to aluminum in CG, the CG-based multistage porous molecular sieve material with high crystallinity was successfully synthesized by the method of hydrothermal crystallization-calcination. This study discussed the potential of CG with a high content of Al and Si as the raw material to prepare zeolite molecular sieves, and the optimum technological parameters for the synthesis of zeolite molecular sieves were determined. Through a series of adsorption experiments with different parameters (such as initial solution, pH, etc.), the interaction of the CG-based zeolite molecular sieve in the adsorption of malachite green (MG) in wastewater was determined, and the adsorption kinetics and isothermal model were studied. Finally, the application prospect of CG adsorption in dye-polluted environments was expounded.
2. Experiment Materials and Methods
2.1. Experiment Materials
After crushing the CG (Yankuang Xinjiang Mining Co., Ltd., Zoucheng, China, sulfur ditch coal mine), it was analyzed and SiO2 and Al2O3 were found to be the main chemical components, accounting for 64.49%. They are the basic materials for the preparation of various zeolite molecular sieves, indicating the great potential of CG as the material for synthesizing zeolite molecular sieves. Additionally, CG also contains a certain amount of other metal oxides, such as Fe2O3, CaO, and MgO, which may affect the surface chemical properties and adsorption properties of the material.
Cotton linter cellulose (Xinjiang Aksu cotton linter processing plant, Xinjiang, China); Sodium hydroxide (NaOH, Tianjin Zhiyuan Chemical Reagent Co., Ltd., Tianjin, China); Tetraethyl orthosilicate (TEOS, Tianjin Zhiyuan Chemical Reagent Co., Ltd., Tianjin, China); Sodium aluminate, ammonia, methylamine (NaAlO2, NH3·H2O, CH3NH2, Shanghai McLean Biochemical Technology Co., Ltd., Shanghai, China); and Malachite Green (MG, Tianjin Tianxin Fine Chemical Development Center, Tianjin, China) are all commercially available materials and can be used directly in the sample preparation process.
2.2. Synthesis of ZSM−5/CLCA
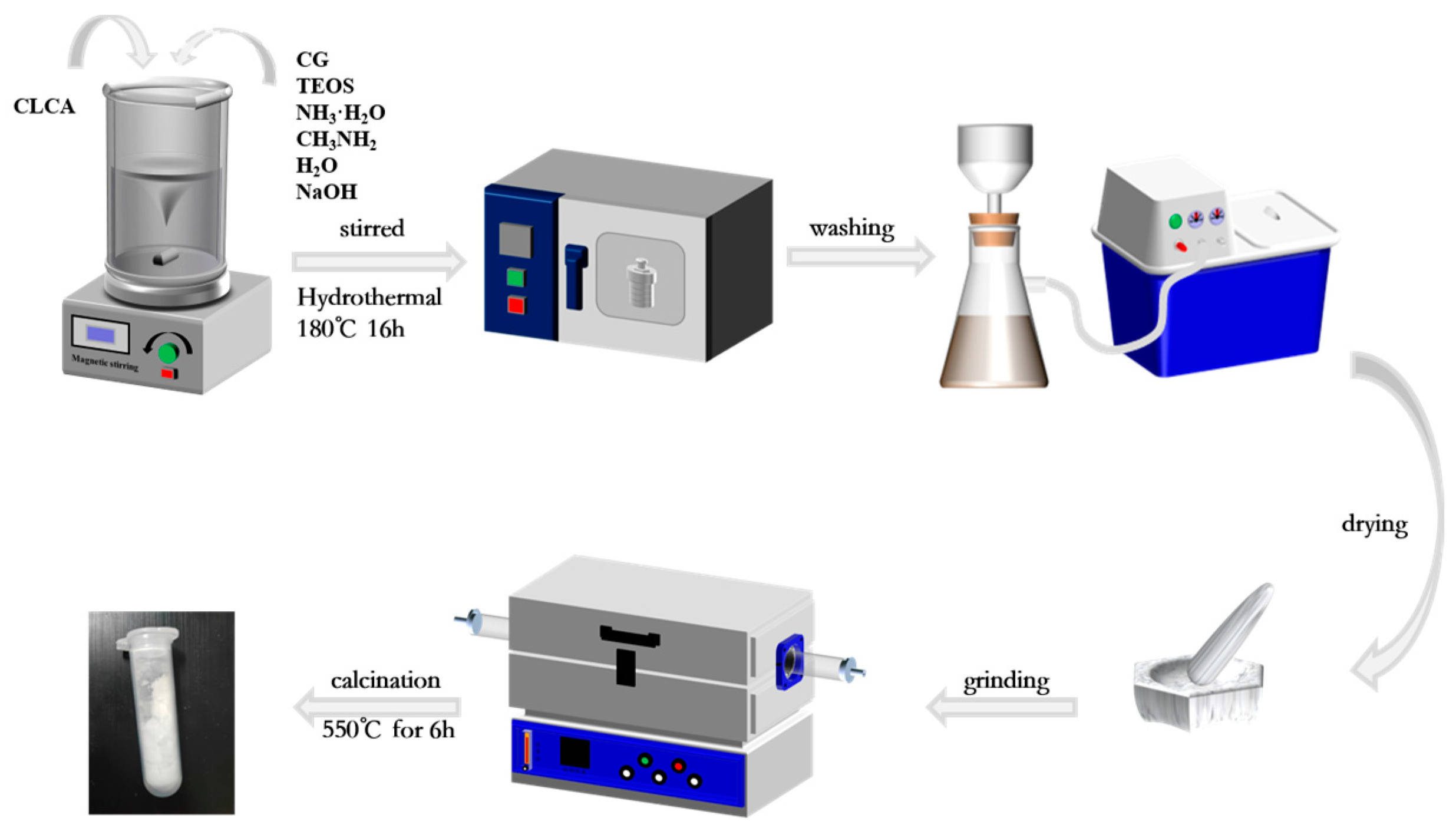
Cellulose aerogels (CLCA) not only have excellent properties such as low density, high porosity, and specific surface area, but the hydroxyl functional groups on their surfaces can interact with silica-aluminum gels and act as structural guides, allowing for changes in the specific surface area and pore volume of the sample. For the reparation of CLCA, 4 g of cellulose powder was dispersed in NaOH/Urea/H2O (7:12:81 by mass) and then placed in a refrigerator for 12 h. The samples were then allowed to thaw at room temperature, washed to neutrality with deionized water, and freeze-dried for 24 h to prepare cellulose aerogel (CLCA).
Reference [
23]. The steps for the synthesis of ZSM−5/CLCA molecular sieves are shown in
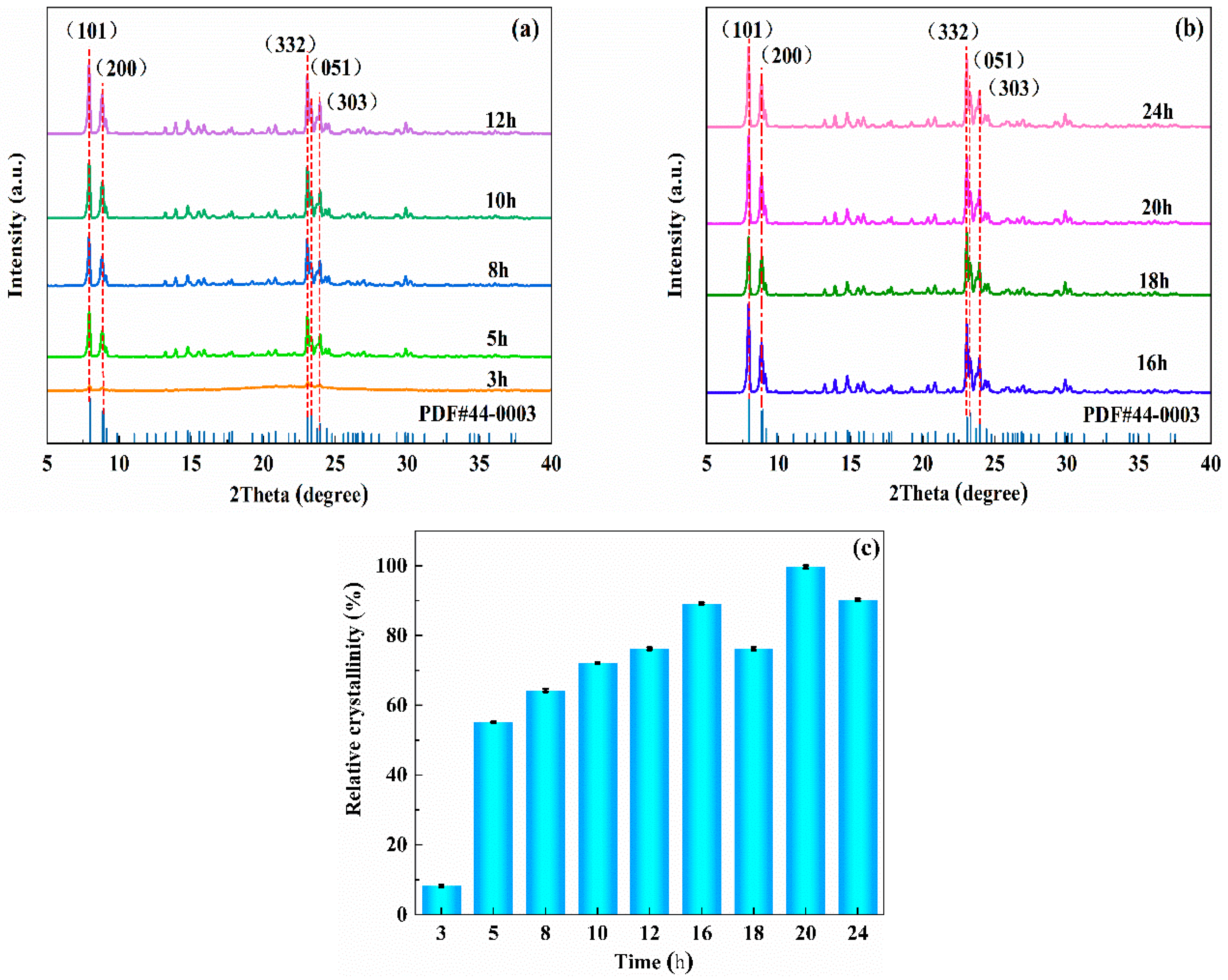
Figure 1. Appropriate amounts of alkali-pretreated CG, deionized water, ammonia, methylamine, and NaOH were weighed, placed into a beaker, and stirred vigorously until the solution was fully mixed. Then, TEOS was added to the mixed solution (the aim was to regulate the molecular silicon-to-aluminum ratio) and stirred at room temperature to form a lotion. Then, the prepared CLCA was added to the above solution and stirred at room temperature to disperse evenly. Then, the lotion was poured into a 100-mL polytetrafluoroethylene reactor, and the reactor was put into an oven. After crystallization at a certain temperature for some time, the reactor was removed from the oven and left to cool naturally. The crystallization solution was removed from the tank and washed to neutralize it with deionized water. Then, it was dried overnight in an oven at a temperature of 60 °C. Finally, the product was ground, heated in a tubular furnace at a rate of 5 °C/min to 550 °C, and baked for 6 h. Then, the template was removed from the crystal, and the resulting sample was named ZSM−5/CLCA.
2.3. Characterization Method
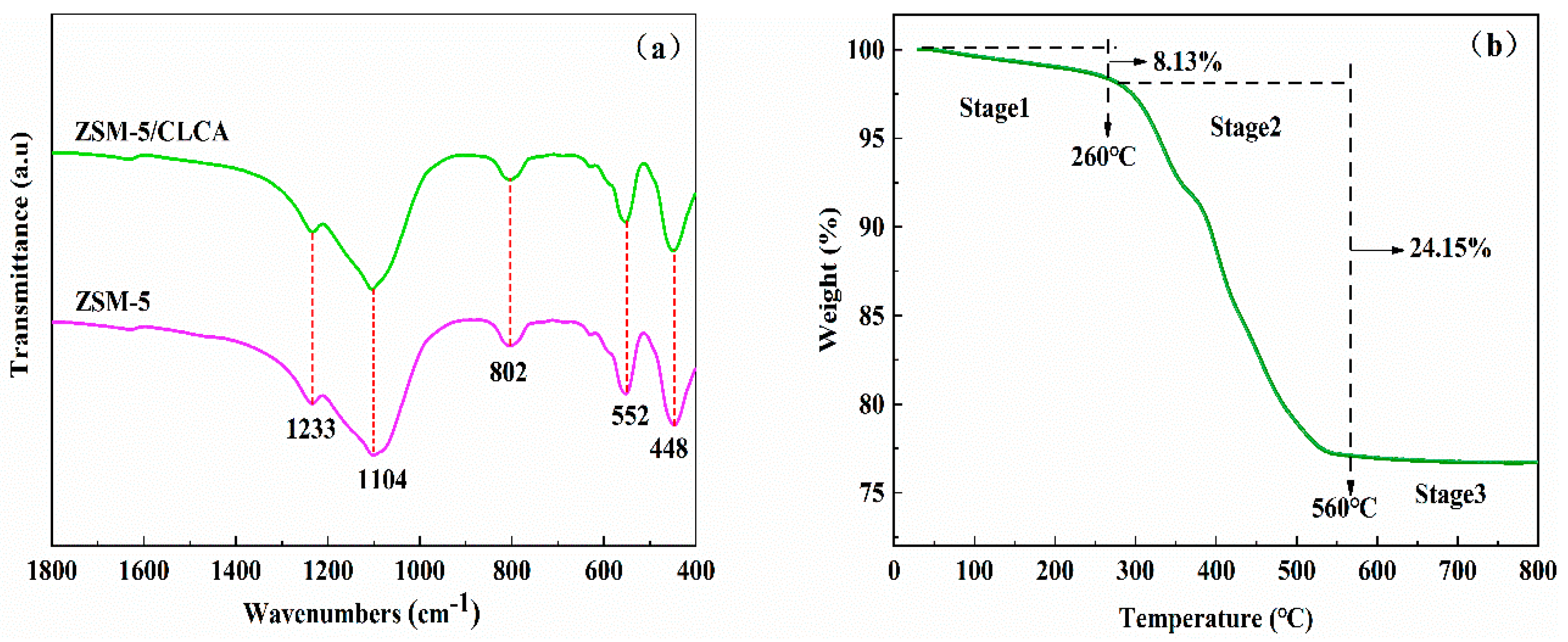
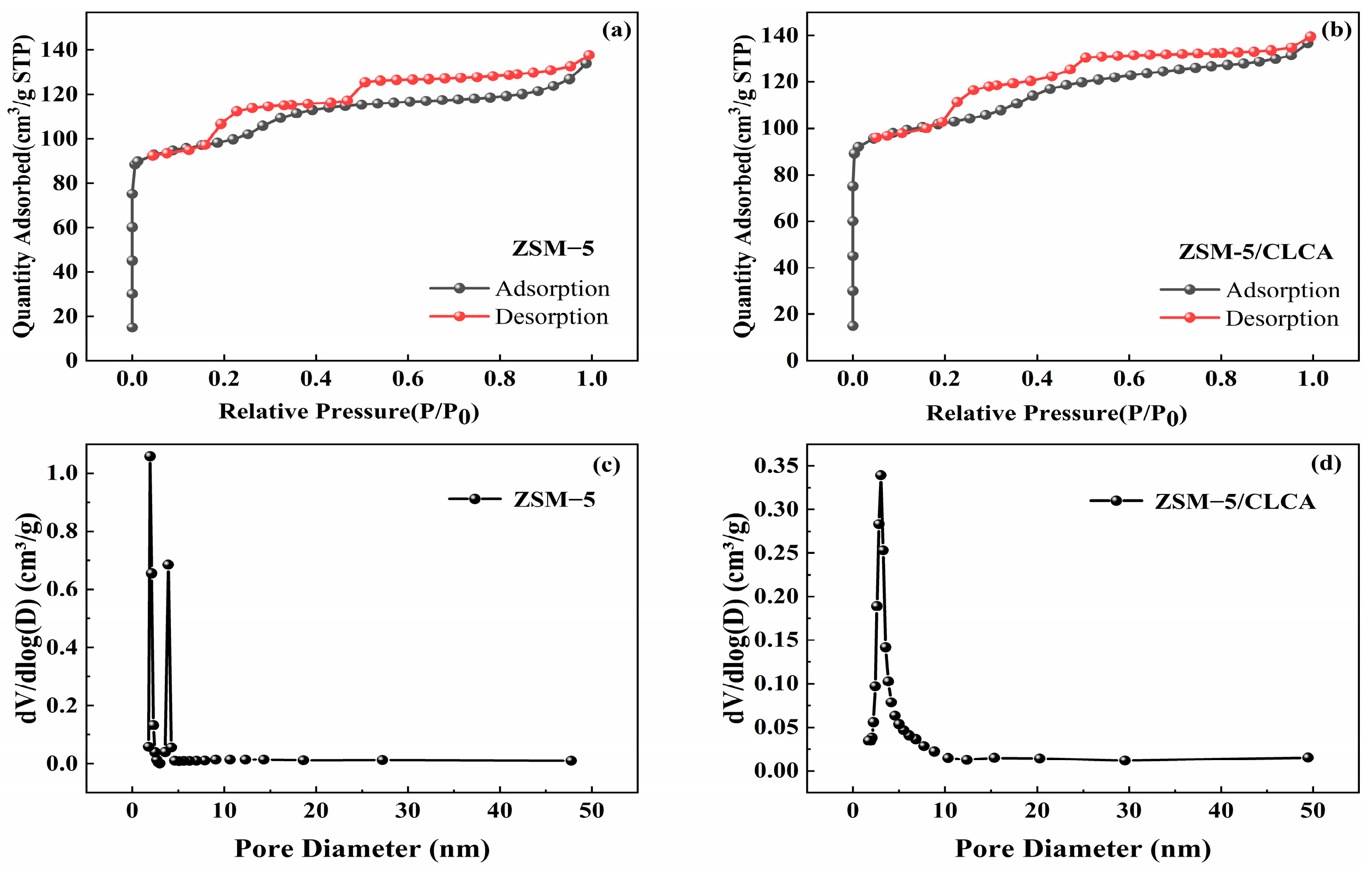
An X-ray diffractometer (XRD, Bruker D8 Advance, Karlsruhe, Germany) was used to analyze the characteristics of the crystal form and the crystallization of the sample. The test conditions are: the sample was powdered (in line with the test requirements, so there was no need to grind before the test), Cu target Kα radiation (λ = 1.5406 Å), scanning angle range of 5~40°, scanning speed of 5°/min (the grinding mentioned in the preparation process was mortar grinding). The purpose was to fully disperse the particles; cellulose aerogel as a template only plays a role in pore-making, and then the templating agent in the crystal needs to be removed by roasting. Therefore, it will not affect the crystallinity of the sample). The appearance of the sample was observed by the scanning electron microscope (SEM, Germany ZSISS Sigma 300, Oberkochen, Germany) test process: A trace sample was directly glued to the conductive adhesive, and the Oxford Quorum SC7620 sputtering coater was used to spray gold for 45 s at 10 mA. Then, the sample morphology was photographed at a 3 kV acceleration voltage. A Fourier transform infrared spectrometer (FT-IR, Bruker VERTEX70, Karlsruhe, Germany) was used to conduct the characterization analysis of the functional groups of the sample. The specific surface area and void distribution of the sample were calculated for the characterization analysis using the automatic specific surface area and porosity analyzer (BET, America Micromerics ASAP 2460, Norcross, GA, USA). The thermal stability of the samples was analyzed using a thermogravimetric analyzer (TG, TA TGA 550, New Castle, DE, USA) under the following conditions: The samples were measured at a temperature increase rate of 10 °C/min from room temperature to 800 °C. The morphology of the molecular sieve was analyzed using a high-power transmission electron microscope (TEM, JEM-2100, Tokyo, Japan). The test was carried out by dispersing the sample powder in anhydrous ethanol, then dropping the suspension onto a copper grid and waiting for the ethanol to evaporate.
2.4. Adsorption Experiment
Next, we explored the adsorption performance of ZSM−5/CLCA (compared with commercial ZSM−5). The adsorption effects of the adsorbent on MG were investigated under different conditions, such as time, initial concentration of MG, temperature, and pH value. After adsorption, the 25 × 0.45 μm organic filter was used as a membrane to separate the adsorbent from the MG solution. The UV-visible spectrophotometer was used to record the absorbance of the solution before and after adsorption. The adsorption capacity and removal rate of the adsorbent from MG were calculated according to Formulas (1) and (2).
Removal rate:
where q
t is the adsorption capacity, unit: mg/g; C
0 represents the initial concentration of MG solution, and C
t represents the concentration of MG solution at time t, unit: mg/L; V is the volume of MG solution, unit: L; W is the mass of adsorbent, unit: g; η represents the removal rate, unit: %.