Effect of a 3.5% NaCl−10% HCl Corrosive Environment on the Fatigue Behavior of Hot Rolled Aluminum 5083-H111
Abstract
1. Introduction
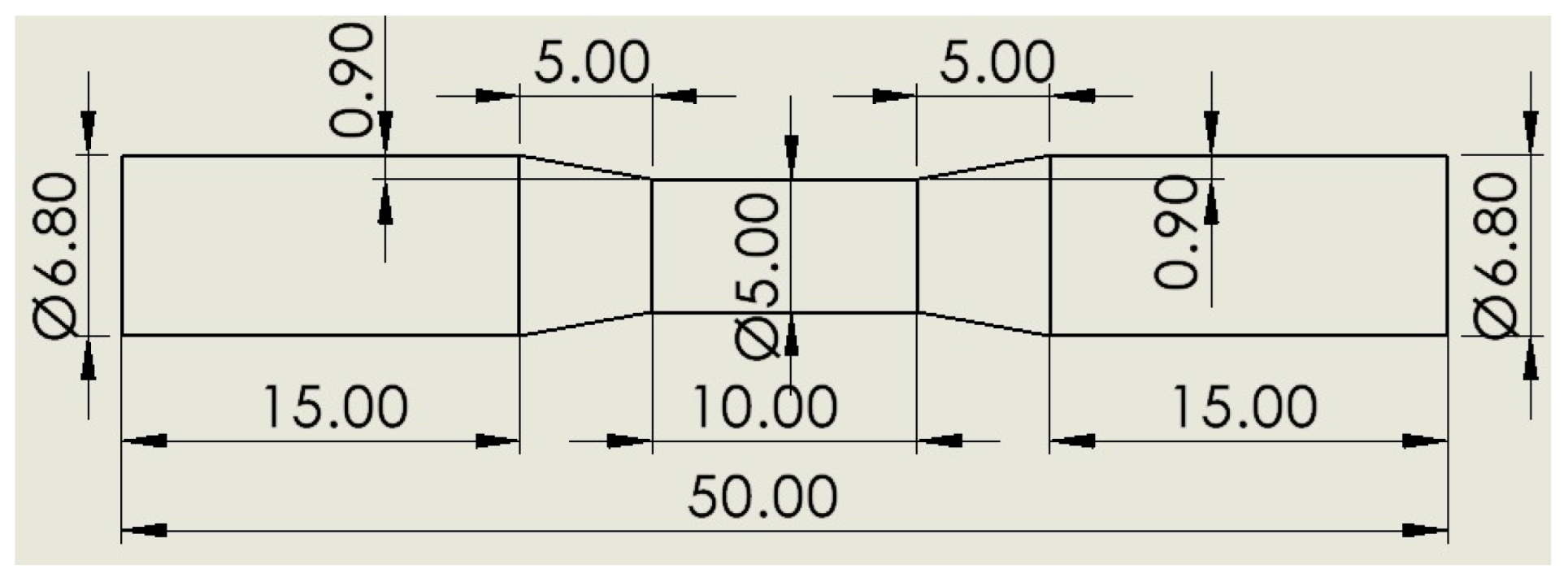
2. Materials and Methods
3. Results and Discussion
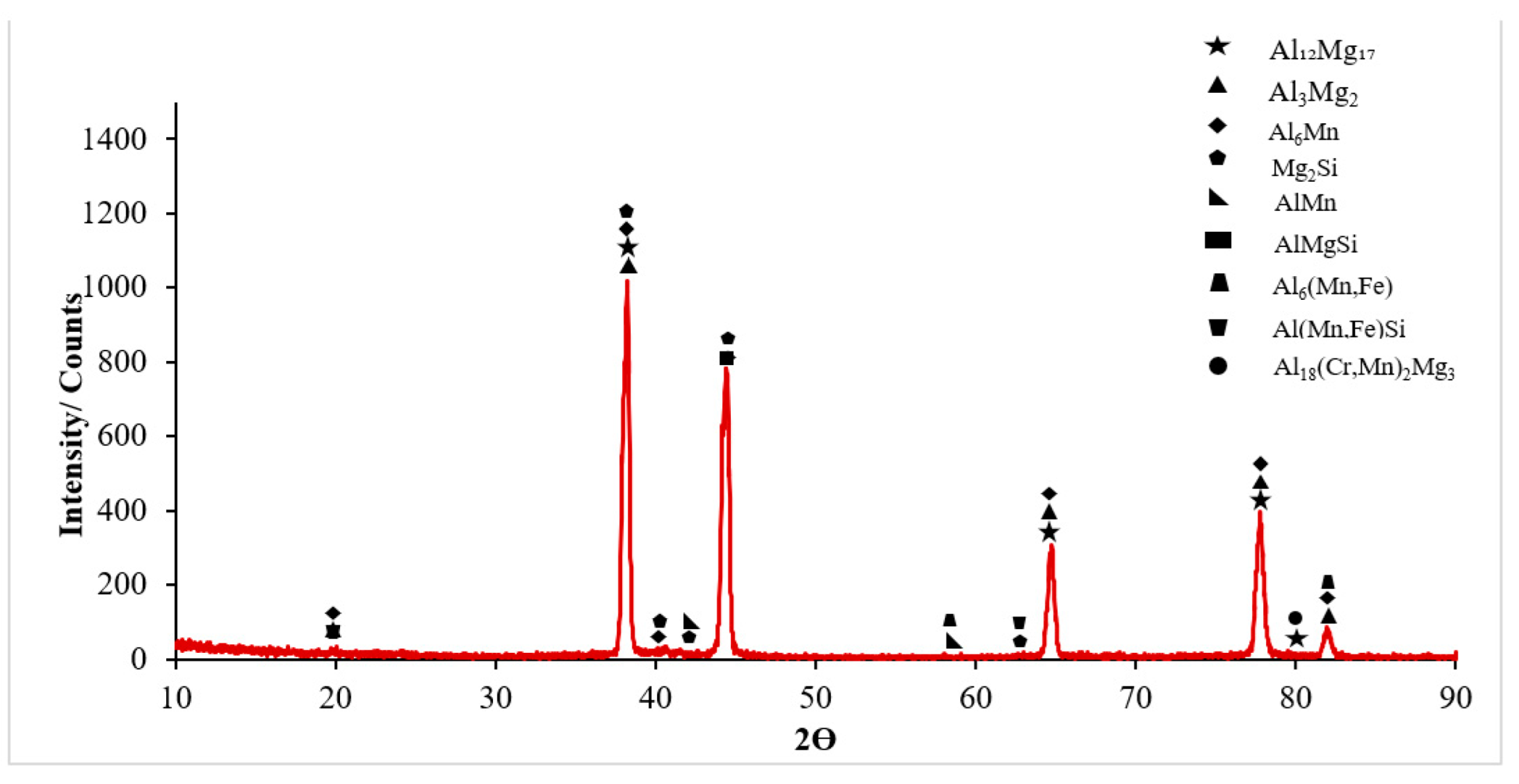
3.1. Microstructural Characterisation
3.2. Hardness Test Results
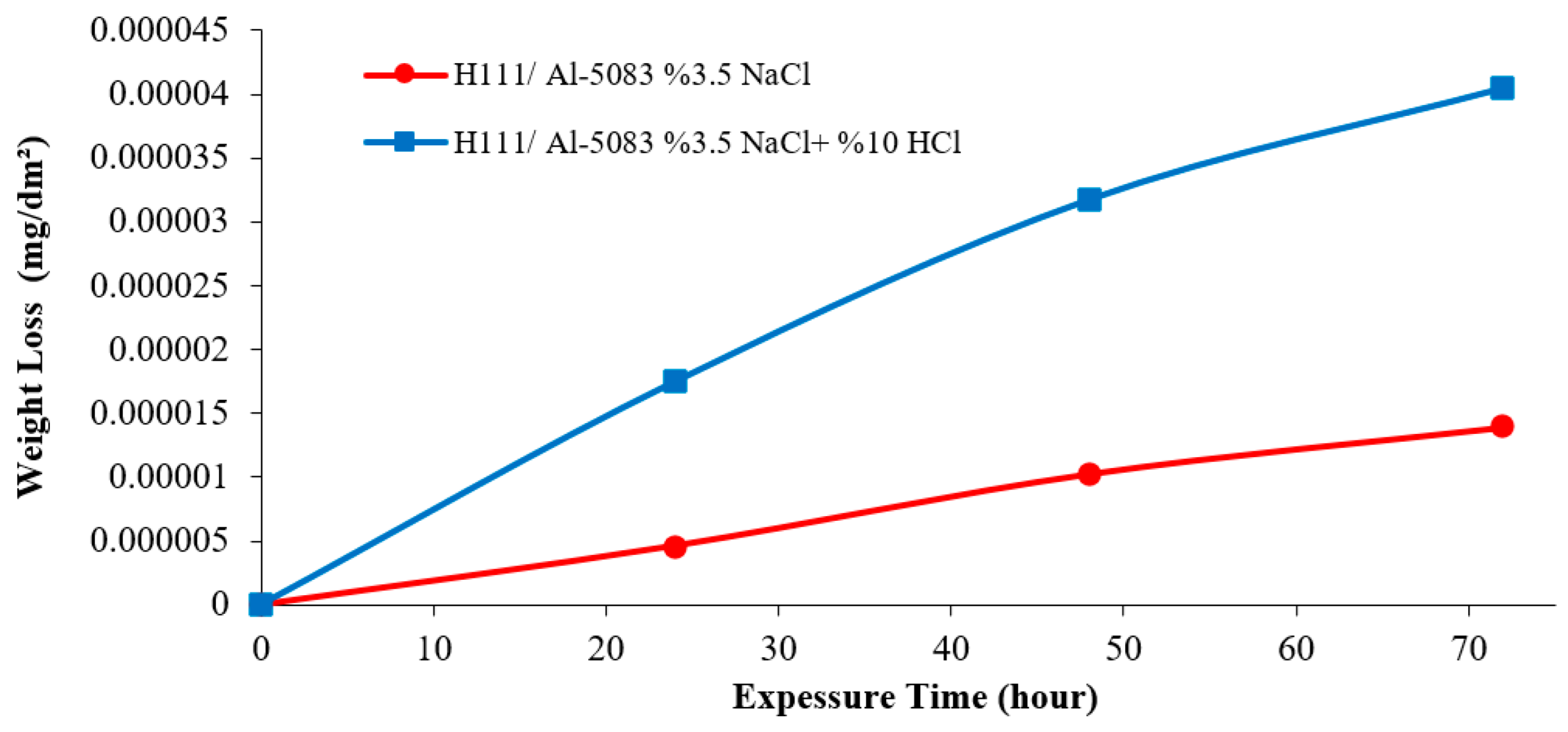
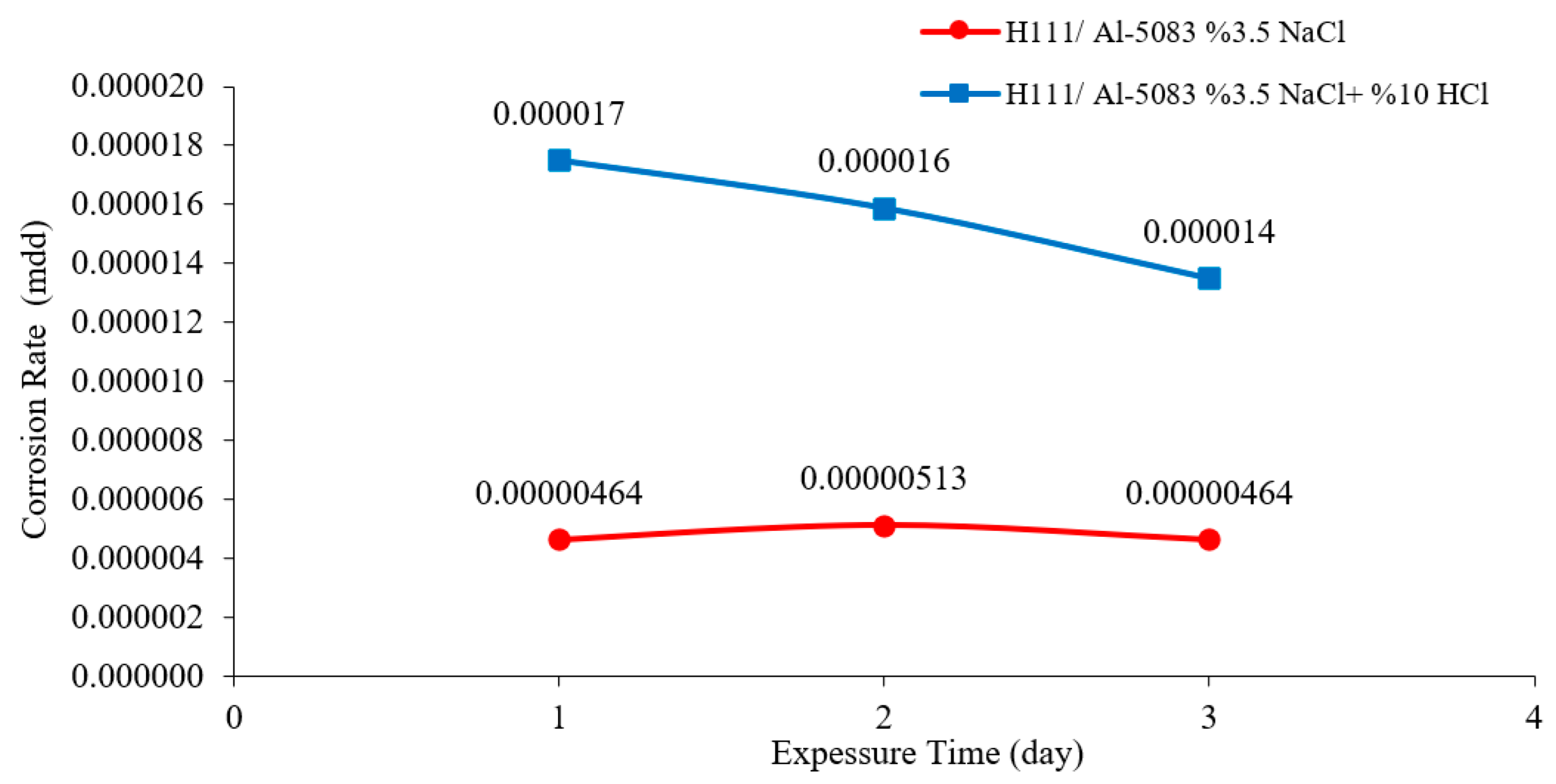
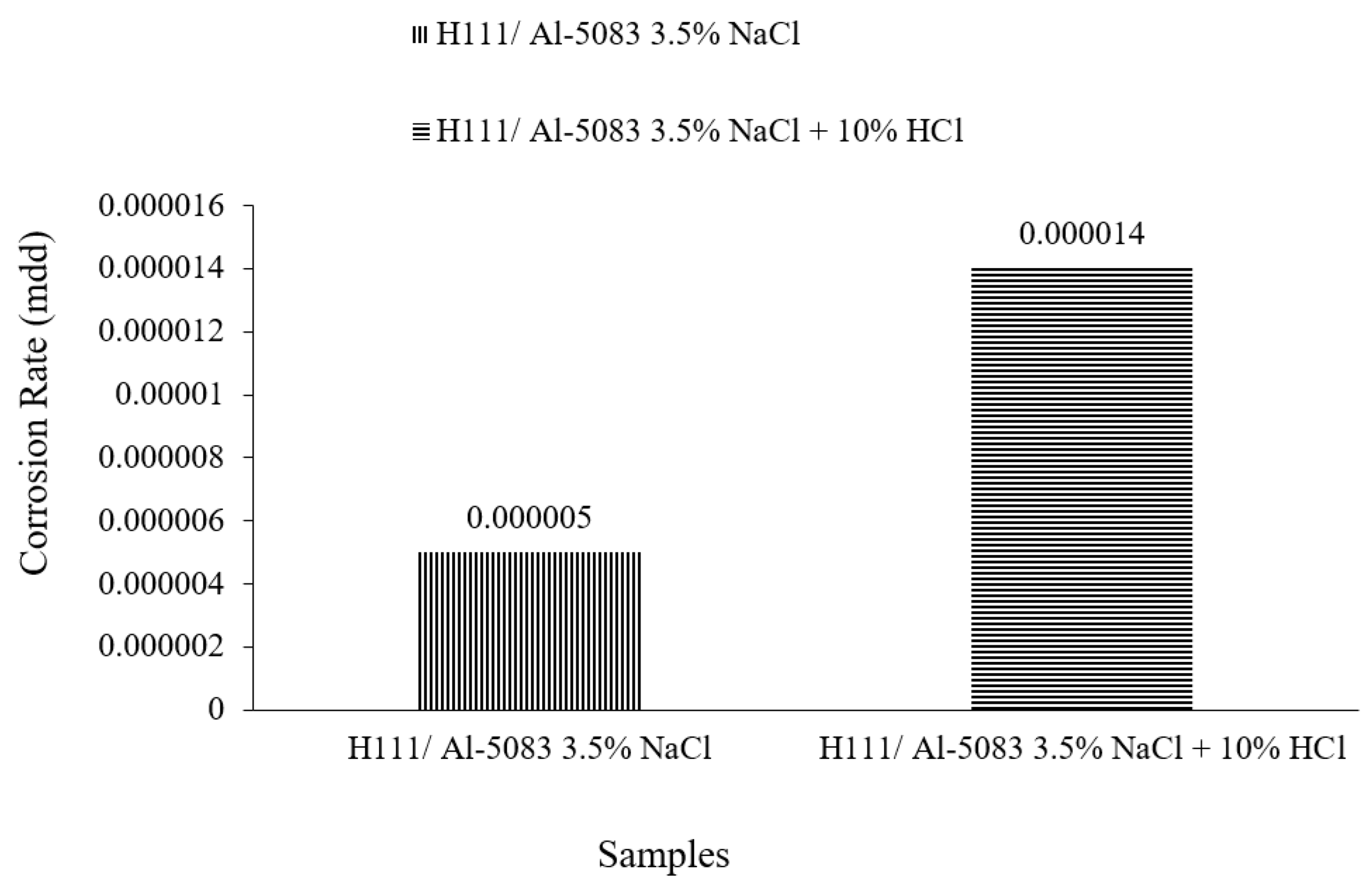
3.3. Immersion Corrosion Test Results
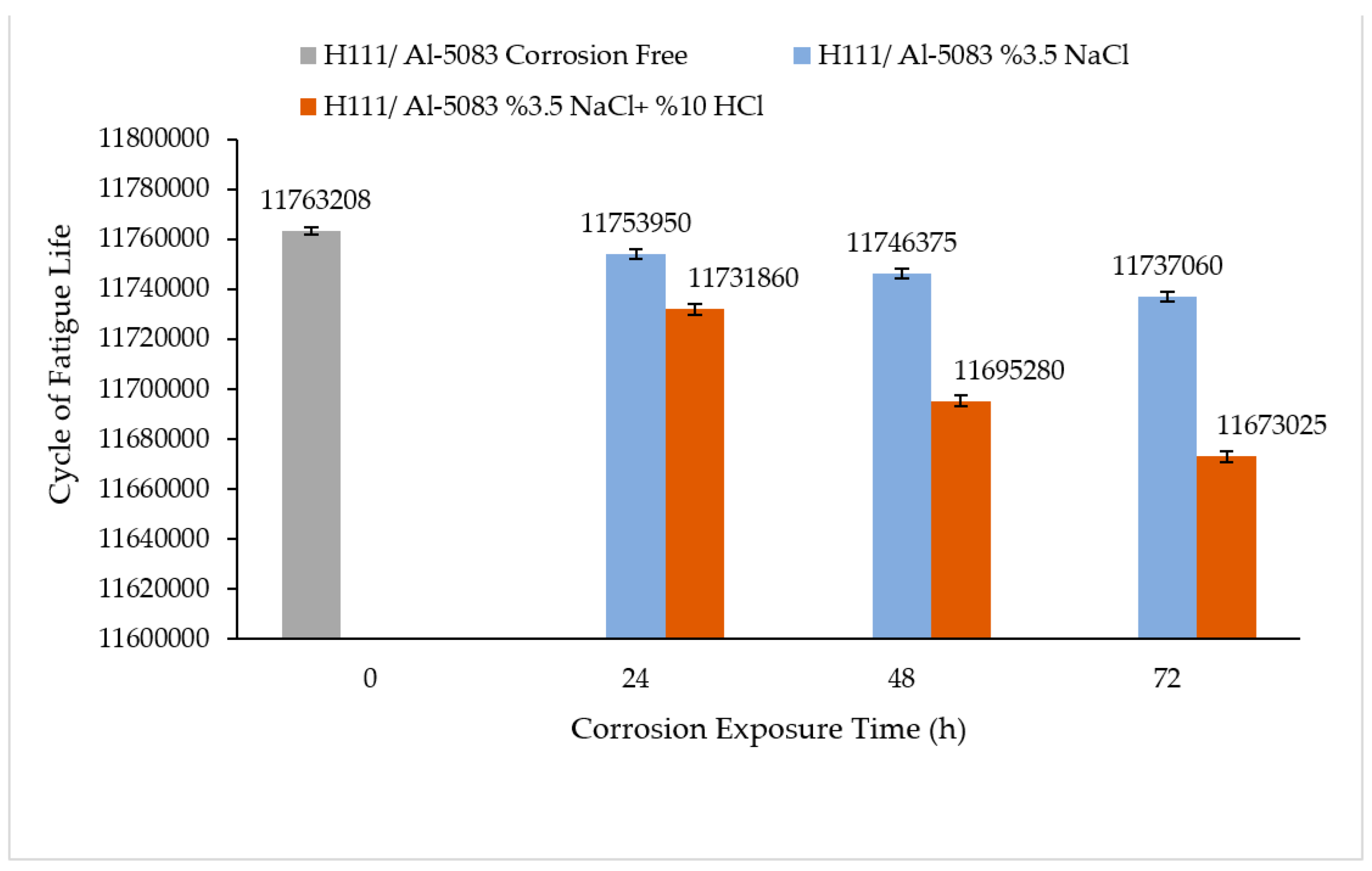
3.4. Rotary Bending Fatigue Test Results
4. Conclusions
- It was seen that the microstructure of the Al5083-H111 material consisted of grains oriented towards the rolling direction. We noticed that intermetallic phases precipitated at the grain boundary and were not evenly distributed.
- The hardness result of the Al5083-H111 material was measured as 68.67 ± 1.84 HB.
- According to the results of the immersion corrosion, while the Al5083 sample was more resistant to corrosion in a 3.5% NaCl environment, it showed less resistant behavior in a 3.5% NaCl + 10% HCl environment. The reason for this is thought to be intergranular corrosion in the material in the 3.5% NaCl + 10% HCl corrosion environment. The sample showed a more stable corrosion behavior after 24 h in a 3.5% NaCl environment. However, the same situation was not valid for a 3.5% NaCl + 10% HCl environment. According to the corrosion rates at the end of 72 h, the presence of the 10% HCl solution increased the corrosion rate by 180%.
- According to the fatigue results, it was observed that the non-corrosive sample showed a better fatigue life than the samples exposed to corrosion. The 3.5% NaCl corrosion fatigue sample, on the other hand, provided better results in the fatigue tests compared with the 3.5% NaCl + 10%HCl corrosion fatigue sample. The stable difference deteriorated after 48 h and the fatigue life of the sample exposed to corrosion in a 3.5% NaCl + 10% HCl environment decreased the most in the following hours compared with the other samples. According to the fatigue rate results, the presence of 10% HCl solution in the corrosion electrolyte reduced the fatigue life by 257%.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Demirci, A.H.; Yiğit, K.; Aydın, H. MgCu2 Partikülleri Içeren Al-Cu Alaşımının Verilen Bir Tribosistem ve Yaşlandırma Şartlarında Aşınma Davranışlarının Belirlenmesi. In Proceedings of the 10. Denizli Malzeme Sempozyumu ve Sergisi Konferansı, Denizli, Türkiye, 14–16 April 2004; pp. 187–193. [Google Scholar]
- Brown, K.R.; Venie, M.S.; Woods, R.A. The Increasing Use of Aluminum in Automotive Applications. JOM 1995, 47, 20–23. [Google Scholar] [CrossRef]
- Stojanovic, B.; Bukvic, M.; Epler, I. Application of Aluminum and Aluminum Alloys in Engineering. Appl. Eng. Lett. 2018, 3, 52–62. [Google Scholar] [CrossRef]
- Santos, M.C.; Machado, A.R.; Sales, W.F.; Barrozo, M.A.S.; Ezugwu, E.O. Machining of Aluminum Alloys: A Review. Int. J. Adv. Manuf. Technol. 2016, 86, 3067–3080. [Google Scholar] [CrossRef]
- Campbell, F.C. Elements of Metallurgy and Engineering Alloys; Campbell, F.C., Ed.; ASM International: Almere, The Netherlands, 2008; ISBN 978-1-62708-251-8. [Google Scholar]
- Kalyoncu, S. Alüminyum Alaşımlarında Alçak Basınçlı Döküm Simülasyonu; Manisa Celal Bayar Üniversitesi: Manisa, Türkiye, 2020. [Google Scholar]
- Mutombo, K.; Du, M. Corrosion Fatigue Behaviour of Aluminium 5083-H111 Welded Using Gas Metal Arc Welding Method. In Arc Welding; InTech Open Access Publishing: London, UK, 2011; pp. 177–218. [Google Scholar]
- Mısırlı, C. 5083 Kalite Alüminyum Alaşımının Homojenize Edilerek Sertlik Değerlerinin Ve Mikroyapılarının İncelenmesi; Trakya Üniversitesi: Edirne, Turkey, 2011. [Google Scholar]
- Sielski, R. Research Needs in Aluminum Structure. Ships Offshore Struct. 2008, 3, 57–65. [Google Scholar] [CrossRef]
- Huskins, E.L.; Cao, B.; Ramesh, K.T. Strengthening Mechanisms in an Al–Mg Alloy. Mater. Sci. Eng. A 2010, 527, 1292–1298. [Google Scholar] [CrossRef]
- Yi, G.; Free, M.L.; Zhu, Y.; Derrick, A.T. Capillarity Effect Controlled Precipitate Growth at the Grain Boundary of Long-Term Aging Al 5083 Alloy. In Proceedings of the Metallurgical and Materials Transactions A: Physical Metallurgy and Materials Science; Springer: Boston, FL, USA, 2014; Volume 45, pp. 4851–4862. [Google Scholar]
- Lean, P.; Gil, L.; Ureña, A. Dissimilar Welds between Unreinforced AA6082 and AA6092/SiC/25p Composite by Pulsed-MIG Arc Welding Using Unreinforced Filler Alloys (Al–5Mg and Al–5Si). J. Mater. Process. Technol. 2003, 143–144, 846–850. [Google Scholar] [CrossRef]
- Czechowski, M. Corrosion Fatigue of Gma Welded Al—Mg Alloy. Adv. Mater. Sci. 2004, 4, 16–24. [Google Scholar]
- Praveen, P.; Yarlagadda, P.K.D.V. Meeting Challenges in Welding of Aluminum Alloys through Pulse Gas Metal Arc Welding. J. Mater. Process. Technol. 2005, 164–165, 1106–1112. [Google Scholar] [CrossRef]
- Olson, D.L.; Siewert, T.A.; Liu, S.; Edwards, G.R. Welding, Brazing, and Soldering; ASM International: Detroit, MI, USA, 2007. [Google Scholar]
- Ralston, K.D.; Birbilis, N.; Weyland, M.; Hutchinson, C.R. The Effect of Precipitate Size on the Yield Strength-Pitting Corrosion Correlation in Al–Cu–Mg Alloys. Acta Mater. 2010, 58, 5941–5948. [Google Scholar] [CrossRef]
- Ishıhara, S.; Saka, S.; Nan, Z.Y.; Goshıma, T.; Sunada, S. Prediction of Corrosion Fatigue Lives of Aluminium Alloy on the Basis of Corrosion Pit Growth Law. Fatigue Fract. Eng. Mater. Struct. 2006, 29, 472–480. [Google Scholar] [CrossRef]
- Rokhlin, S.I.; Kim, J.-Y.; Nagy, H.; Zoofan, B. Effect of Pitting Corrosion on Fatigue Crack Initiation and Fatigue Life. Eng. Fract. Mech. 1999, 62, 425–444. [Google Scholar] [CrossRef]
- Chlıstovsky, R.; Heffernan, P.; Duquesnay, D. Corrosion-Fatigue Behaviour of 7075-T651 Aluminum Alloy Subjected to Periodic Overloads. Int. J. Fatigue 2007, 29, 1941–1949. [Google Scholar] [CrossRef]
- Sharma, M.M.; Tomedi, J.D.; Parks, J.M. A Microscopic Study on the Corrosion Fatigue of Ultra-Fine Grained and Conventional Al-Mg Alloy. Corros. Sci. 2015, 93, 180–190. [Google Scholar] [CrossRef]
- Holtz, R.L.; Pao, P.S.; Bayles, R.A.; Longazel, T.M.; Goswami, R. Corrosion-Fatigue Behavior of Aluminum Alloy 5083-H131 Sensitized at 448 K (175 C). Proc. Metall. Mater. Trans. Phys. Metall. Mater. Sci. 2012, 43, 2839–2849. [Google Scholar] [CrossRef]
- Rudra, A.; Ashiq, M.; Tiwari, J.K.; Das, S.; Dasgupta, R. Study of Processing Map and Effect of Hot Rolling on Mechanical Properties of Aluminum 5083 Alloy. Trans. Indian Inst. Met. 2020, 73, 1809–1826. [Google Scholar] [CrossRef]
- Aballe, A.; Bethencourt, M.; Botana, F.; Cano, M.; Marcos, M. Localized Alkaline Corrosion of Alloy AA5083 in Neutral 3.5% NaCl Solution. Corros. Sci. 2001, 43, 1657–1674. [Google Scholar] [CrossRef]
- Engler, O.; Miller-Jupp, S. Control of Second-Phase Particles in the Al-Mg-Mn Alloy AA 5083. J. Alloys Compd. 2016, 689, 998–1010. [Google Scholar] [CrossRef]
- Zhu, Y.; Cullen, D.A.; Kar, S.; Free, M.L.; Allard, L.F. Evaluation of Al3Mg2 Precipitates and Mn-Rich Phase in Aluminum-Magnesium Alloy Based on Scanning Transmission Electron Microscopy Imaging. Metall. Mater. Trans. A 2012, 43, 4933–4939. [Google Scholar] [CrossRef]
- Goswami, R.; Holtz, R.L. Transmission Electron Microscopic Investigations of Grain Boundary Beta Phase Precipitation in Al 5083 Aged at 373 K (100 °C). Metall. Mater. Trans. A 2013, 44, 1279–1289. [Google Scholar] [CrossRef]
- Li, Y.J.; Zhang, W.Z.; Marthinsen, K. Precipitation Crystallography of Plate-Shaped Al6(Mn,Fe) Dispersoids in AA5182 Alloy. Acta Mater. 2012, 60, 5963–5974. [Google Scholar] [CrossRef]
- Yi, G.; Sun, B.; Poplawsky, J.D.; Zhu, Y.; Free, M.L. Investigation of Pre-Existing Particles in Al 5083 Alloys. J. Alloys Compd. 2018, 740, 461–469. [Google Scholar] [CrossRef]
- Engler, O.; Liu, Z.; Kuhnke, K. Impact of Homogenization on Particles in the Al–Mg–Mn Alloy AA 5454—Experiment and Simulation. J. Alloys Compd. 2013, 560, 111–122. [Google Scholar] [CrossRef]
- Zajac, S.; Hutchinson, B.; Johansson, A.; Gullman, L.-O. Microstructure Control and Extrudability of Al–Mg–Si Alloys Microalloyed with Manganese. Mater. Sci. Technol. 1994, 10, 323–333. [Google Scholar] [CrossRef]
- Flores-V, A.; Sukiennik, M.; Castillejos-E, A.H.; Acosta-G, F.A.; Escobedo-B, J.C. A Kinetic Study on the Nucleation and Growth of the Al8FeMnSi2 Intermetallic Compound for Aluminum Scrap Purification. Intermetallics 1998, 6, 217–227. [Google Scholar] [CrossRef]
- Kılınç, M. 5083 Kalite Al Alaşımının Mekanik Özelliklerine Termomekanik Isıl İşleminin Etkisi; Karabük Üniversitesi: Karabuk, Türkiye, 2021. [Google Scholar]
- Tan, L.; Allen, T.R. Effect of Thermomechanical Treatment on the Corrosion of AA5083. Corros. Sci. 2010, 52, 548–554. [Google Scholar] [CrossRef]
- Yi, G.; Zeng, W.; Poplawsky, J.D.; Cullen, D.A.; Wang, Z.; Free, M.L. Characterizing and Modeling the Precipitation of Mg-Rich Phases in Al 5xxx Alloys Aged at Low Temperatures. J. Mater. Sci. Technol. 2017, 33, 991–1003. [Google Scholar] [CrossRef]
- Yan, J.; Hodge, A.M. Study of β Precipitation and Layer Structure Formation in Al 5083: The Role of Dispersoids and Grain Boundaries. J. Alloys Compd. 2017, 703, 242–250. [Google Scholar] [CrossRef]
- Bethencourt, M.; Botana, F.J.; Calvino, J.J.; Marcos Bárcena, M.; Pérez, J.; Rodríguez, M.A. The Influence of the Surface Distribution of Al6(MnFe) Intermetallic on the Electrochemical Response of AA5083 Aluminium Alloy in NaCl Solutions. Mater. Sci. Forum 1998, 289–292, 567–574. [Google Scholar] [CrossRef]
- Arenas, M.A.; Bethencourt, M.; Botana, F.J.; De Damborenea, J.; Marcos, M. Inhibition of 5083 Aluminium Alloy and Galvanised Steel by Lanthanide Salts. Corros. Sci. 2001, 43, 157–170. [Google Scholar] [CrossRef]
- Szklarska-Smialowska, Z. Pitting Corrosion of Aluminum. Corros. Sci. 1999, 41, 1743–1767. [Google Scholar] [CrossRef]
- Lucas, K.A.; Clarke, H. Corrosion of Aluminum-Based Metal Matrix Composites; Research Studies Press Ltd.: Baldock, UK, 1993. [Google Scholar]
- Speidel, M.O.; Hyatt, M.V.; Fontana, M.G.; Staettle, R.W. Advances in Corrosion Science and Technology; Plenum Press: New York, NY, USA, 1972. [Google Scholar]
- Pidaparti, R.M.; Rao, A.S. Analysis of Pits Induced Stresses Due to Metal Corrosion. Corros. Sci. 2008, 50, 1932–1938. [Google Scholar] [CrossRef]
- Aliravci, C.A.; Pekgüleryüz, M.Ö. Calculation of Phase Diagrams for the Metastable Al-Fe Phases Forming in Direct-Chill (DC)-Cast Aluminum Alloy Ingots. Calphad 1998, 22, 147–155. [Google Scholar] [CrossRef]
- Sharma, M.M.; Ziemian, C.W. Pitting and Stress Corrosion Cracking Susceptibility of Nanostructured Al-Mg Alloys in Natural and Artificial Environments. J. Mater. Eng. Perform. 2008, 17, 870–878. [Google Scholar] [CrossRef]
- Sharma, M.M.; Tomedi, J.D.; Weigley, T.J. Slow Strain Rate Testing and Stress Corrosion Cracking of Ultra-Fine Grained and Conventional Al–Mg Alloy. Mater. Sci. Eng. A 2014, 619, 35–46. [Google Scholar] [CrossRef]
- Sikora, E.; Wei, X.J.; Shaw, B.A. Corrosion Behavior of Nanocrystalline Bulk Al-Mg-Based Alloys. Corrosion 2004, 60, 387–398. [Google Scholar] [CrossRef]

















| Alloy | Fe | Si | Cu | Mn | Mg | Zn | Cr | Ti | Other | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| Al5083 | 0.4 | 0.4 | 0.1 | 0.4–1.0 | 4.0–4.9 | 0.25 | 0.50–0.25 | 0.15 | 0.15 | Balance |
| Mg | Si | S | Cl | Cr | Mn | Fe | Ni | Cu | Zn | Al | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample 5083 | 3.6500 | 0.3486 | 0.0108 | 0.0376 | 0.1218 | 0.5259 | 0.3483 | 0.0126 | 0.069 | 0.0345 | 94.8408 |
| Spectrum | Mg | Al | Si | Cr | Mn | Fe |
|---|---|---|---|---|---|---|
| 1 | 5.19 | 94.81 | 0.00 | 0.00 | 0.00 | 0.00 |
| 2 | 2.06 | 33.29 | 63.76 | 0.28 | 0.61 | 0.00 |
| 3 | 4.62 | 93.34 | 0.92 | 0.16 | 0.81 | 1.06 |
| 4 | 5.16 | 93.68 | 0.50 | 0.22 | 0.45 | 0.00 |
| Spectrum | O | Na | Mg | Al | Si | Cl | Cr | Mn | Fe |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.48 | 0.53 | 5.16 | 92.42 | 0.33 | 0.00 | 0.08 | 0.00 | 0.00 |
| 2 | 1.75 | 0.46 | 0.29 | 2.21 | 94.98 | 0.17 | 0.15 | 0.00 | 0.00 |
| 3 | 6.57 | 0.30 | 4.41 | 86.48 | 0.70 | 0.00 | 0.40 | 0.47 | 0.68 |
| 4 | 7.43 | 0.44 | 4.51 | 86.87 | 0.41 | 0.04 | 0.30 | 0.00 | 0.00 |
| 5 | 6.71 | 0.33 | 3.91 | 75.45 | 12.55 | 0.01 | 0.33 | 0.35 | 0.34 |
| 6 | 3.96 | 0.36 | 0.83 | 10.37 | 81.76 | 0.00 | 0.26 | 1.26 | 1.19 |
| Spectrum | H | O | Na | Mg | Al | Si | Cl | Cr | Mn | Fe |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.04 | 10.77 | 2.05 | 4.38 | 82.13 | 0.11 | 0.00 | 0.00 | 0.00 | 0.51 |
| 2 | 76.00 | 11.48 | 0.05 | 0.46 | 6.16 | 0.45 | 0.00 | 3.67 | 1.73 | 0.00 |
| 3 | 3.96 | 19.09 | 4.62 | 3.43 | 66.13 | 0.59 | 0.69 | 0.58 | 0.00 | 0.91 |
| 4 | 0.20 | 2.20 | 0.38 | 4.84 | 91.97 | 0.29 | 0.12 | 0.00 | 0.00 | 0.00 |
| 5 | 2.29 | 1.79 | 0.30 | 4.46 | 90.02 | 0.00 | 0.13 | 0.00 | 0.75 | 0.27 |
| 6 | 1.42 | 3.84 | 0.98 | 4.05 | 80.41 | 0.11 | 8.54 | 0.12 | 0.17 | 0.35 |
| 7 | 16.77 | 3.43 | 0.54 | 1.49 | 29.39 | 0.00 | 47.68 | 0.00 | 0.42 | 0.28 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elhasslouk, M.M.M.; Esen, İ.; Ahlatcı, H.; Akın, B. Effect of a 3.5% NaCl−10% HCl Corrosive Environment on the Fatigue Behavior of Hot Rolled Aluminum 5083-H111. Materials 2023, 16, 4996. https://doi.org/10.3390/ma16144996
Elhasslouk MMM, Esen İ, Ahlatcı H, Akın B. Effect of a 3.5% NaCl−10% HCl Corrosive Environment on the Fatigue Behavior of Hot Rolled Aluminum 5083-H111. Materials. 2023; 16(14):4996. https://doi.org/10.3390/ma16144996
Chicago/Turabian StyleElhasslouk, Masoud M. M., İsmail Esen, Hayrettin Ahlatcı, and Bengu Akın. 2023. "Effect of a 3.5% NaCl−10% HCl Corrosive Environment on the Fatigue Behavior of Hot Rolled Aluminum 5083-H111" Materials 16, no. 14: 4996. https://doi.org/10.3390/ma16144996
APA StyleElhasslouk, M. M. M., Esen, İ., Ahlatcı, H., & Akın, B. (2023). Effect of a 3.5% NaCl−10% HCl Corrosive Environment on the Fatigue Behavior of Hot Rolled Aluminum 5083-H111. Materials, 16(14), 4996. https://doi.org/10.3390/ma16144996






