2.2. Synthetic Protocols
Synthesis of 1,2,4-triazin-6(1H)-ones 6 and 7: An excess Et3N (8.0 mmol, 1.12 mL) was added under inert atmosphere to a suspension of amino ester hydrochloride 12 (1.0 mmol) in dry THF (3.0 mL). Then, a solution of hydrazonoyl bromide 11 (1.1 mmol) in dry THF (3.0 mL) was added, and the stirring was continued overnight (the consumption of 11 was confirmed by TLC). The resulting solution was filtered and the precipitate was washed with Et2O (2 × 4.0 mL). After the filtrates were combined and the solvents were removed under reduced pressure, the crude product 6 or 7 was purified by standard column chromatography (CC). In certain cases of glycine derivatives, the resulting material was additionally recrystallized from hexane-dichloromethane mixtures by the slow evaporation of the solvents.
1-(4-Nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6a): CC (SiO2, CH2Cl2 gradient CH2Cl2/EtOAc 9:1), 216 mg (75%). Colorless solid, m.p. 224–225 °C (CH2Cl2/hexanes). 1H NMR (CDCl3, 600 MHz): δ 4.30 (dbr, J ≈ 1.5 Hz, 2 H, CH2), 5.31 (sbr, H, NH), 7.92, 8.27 (2 dbr, J ≈ 9.2 Hz, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 44.0 (t, CH2), 118.2 (q, 1JC-F = 275.2 Hz, CF3), 123.7, 124.2 (2 d, 4 CH), 137.3 (q, 2JC-F = 37.8 Hz, C(3)), 145.0, 145.5 (2 s, 2 i-C), 158.1 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.6 (s, CF3). IR (neat): ν 3295 (NH), 1685 (C=O), 1584, 1487, 1312, 1144 (CF3), 1059, 854 cm−1. ESI-MS (m/z): 311.1 (100, [M+Na]+), 289.2 (31, [M+H]+). C10H7F3N4O3 (288.0): calcd. C 41.68, H 2.45, N 19.44; found: C 41.50, H 2.46, N 19.61.
1-(3-Nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6b): CC (SiO2, 3% MeOH in CH2Cl2), 248 mg (86%). Yellow crystals, m.p. 144–146 °C (CH2Cl2/hexanes). 1H NMR (CD3OD, 600 MHz): δ 4.19 (s, 2 H, CH2), 4.61 (sbr, 1 H, NH), 7.63 (t, J = 8.2 Hz, 1H), 8.04 (ddd, J = 1.0, 2.2, 8.2 Hz, 1 H), 8.12 (ddd, J = 1.0, 2.2, 8.2 Hz, 1 H), 8.53 (t, J = 2.2 Hz, 1 H). 13C NMR (CD3OD, 151 MHz): δ 44.2 (t, CH2), 119.9 (q, 1JC-F = 274.4 Hz, CF3), 119.8, 121.9, 130.5, 130.8 (4 d, 4 CH), 139.7 (q, 2JC-F = 37.1 Hz, C(3)), 142.6, 149.4 (2 s, 2 i-C), 160.8 (s, C=O). 19F NMR (CD3OD, 565 MHz): δ −72.2 (s, CF3). IR (neat): ν 3290 (NH), 1682 and 1647 (C=O), 1536, 1472, 1349, 1271, 1197–1129 (CF3), 977 cm−1. ESI-MS (m/z): 289.2 (100, [M+H]+). C10H7F3N4O3 (288.0): calcd. C 41.68, H 2.45, N 19.44; found: C 41.44, H 2.36, N 19.48.
4-(3-Trifluoromethyl-4,5-dihydro-6(1H)-oxo-1,2,4-triazin-1-yl)benzonitrile (6c): CC (SiO2, 4% MeOH in CH2Cl2), 247 mg (92%). Colorless solid, m.p. 212–214 °C. 1H NMR (DMSO-d6, 600 MHz): δ 4.17 (s, 2 H, CH2), 7.79, 7.88 (2 dbr, J ≈ 8.8 Hz, 2 H each), 8.67 (sbr, 1 H, NH). 13C NMR (DMSO-d6, 151 MHz): δ 43.0 (t, CH2), 108.1 (s, CN), 118.4 (q, 1JC-F = 275.2 Hz, CF3), 118.7 (s, i-C), 123.8, 132.7 (2 d, 4 CH), 137.4 (q, 2JC-F = 36.2 Hz, C(3)), 143.8 (s, i-C), 159.2 (s, C=O). 19F NMR (DMSO-d6, 565 MHz): δ −69.3 (s, CF3). IR (neat): ν 3261 (NH), 2236 (CN), 1700 and 1681 (C=O), 1334, 1200–1126 (CF3), 1066, 839 cm−1. ESI-MS (m/z): 291.2 (100, [M+Na]+), 269.2 (12, [M+H]+). C11H7F3N4O (268.2): calcd. C 49.26, H 2.63, N 20.89; found: C 49.36, H 2.89, N 20.61.
1-(4-Chlorophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6d): CC (SiO2, 4% MeOH in CH2Cl2), 233 mg (84%). Yellow crystals, m.p. 159–160 °C (CH2Cl2/hexanes). 1H NMR (CDCl3, 600 MHz): δ 4.18 (dbr, J ≈ 1.5 Hz, 2 H, CH2), 5.39 (sbr, 1 H, NH), 7.36–7.38, 7.51–7.53 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 43.8 (t, CH2), 118.2 (q, 1JC-F = 275.2 Hz, CF3), 125.7, 128.9 (2 d, 4 CH), 132.7 (s, i-C), 136.9 (q, 2JC-F = 37.5 Hz, C(3)), 138.5 (s, i-C), 157.8 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.6 (s, CF3). IR (neat): ν 3294 (NH), 1689 and 1655 (C=O), 1491, 1349, 1316, 1194–1133 (CF3), 1085, 828 cm−1. ESI-MS (m/z): 278.2 (100, [M+H]+), 244.2 (16, [M–Cl+H]+). C10H7ClF3N3O (277.6): calcd. C 43.26, H 2.54, N 15.14; found: C 43.30, H 2.53, N 15.24.
1-(2,4-Dichlorophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6e): The crude reaction mixture was additionally refluxed for 2h in order to accomplish the second cyclization step (see the text), and the product was isolated after the standard work-up; CC (SiO2, CH2Cl2/EtOAc 95:5), 289 mg (93%). Colorless solid, m.p. 138–139 °C. 1H NMR (CDCl3, 600 MHz): δ 4.24 (sbr, 2 H, CH2), 5.33 (sbr, 1 H, NH), 7.34, 7.51 (2 mc, 2 H, 1 H). 13C NMR (CDCl3, 151 MHz): δ 43.7 (t, CH2), 118.2 (q, 1JC-F = 275.2 Hz, CF3), 128.3, 130.3, 130.4 (3 d, 3 CH), 133.4, 135.6, 136.3 (3 s, 3 i-C), 136.8 (q, 2JC-F = 37.6 Hz, C(3)), 157.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.5 (s, CF3). IR (neat): ν 3284 (NH), 1685 and 1665 (C=O), 1525, 1402, 1357, 1323, 1189–1129 (CF3), 1103, 1050 cm−1. ESI-MS (m/z): 312.1 (100, [M+H]+). C10H6Cl2F3N3O (312.1): calcd. C 38.49, H 1.94, N 13.47; found: C 38.72, H 2.19, N 13.54.
Amidrazone 13e: The spectroscopically pure sample of intermediate 13e (75 mg, 22%; as ca. 87:13 mixture of E/Z-isomers) was obtained in reaction of 11e with 12a and was isolated from the mother liquor by preparative thin layer chromatography (PTLC; SiO2, petroleum ether/CH2Cl2 3:2) followed by recrystallisation from a hexanes/CH2Cl2 mixture. Colorless solid, m.p. 79–81 °C. 1H NMR (CDCl3, 600 MHz); major isomer: δ 3.82 (s, 3 H, Me), 4.05 (d, J = 5.7 Hz, 2 H), 4.57 (tbr, J ≈ 5.7 Hz, 1 H, NH), 7.20 (dd, J = 2.3, 8.8 Hz, 1 H), 7.21 (sbr, 1 H, NH), 7.29 (d, J = 2.3 Hz, 1 H), 7.39 (d, J = 8.8 Hz, 1 H); diagnostic signals for minor isomer: δ 3.80 (s, 3 H, Me), 4.00 (d, J = 5.2 Hz, 2 H), 4.95 (tbr, J ≈ 5.2 Hz, 1 H, NH), absorptions in the aromatic region could not be seen due to overlap of the signals. 13C NMR (CDCl3, 151 MHz); major isomer: δ 44.8 (t, CH2), 53.0 (q, Me), 116.4 (d, CH), 119.20 (q, 1JC-F = 274.5 Hz, CF3), 119.23, 125.7 (2 s, 2 i-C), 128.2, 128.7 (2 d, 2 CH), 136.7 (q, 2JC-F = 34.9 Hz), 140.4 (s, i-C), 170.8 (s, C=O); diagnostic signals for minor isomer: δ 43.8 (t, CH2), 52.6 (q, Me), 114.1 (d, CH), 117.8, 123.8 (2 s, 2 i-C), 128.0, 128.6 (2 d, 2 CH), 140.7 (s, i-C), 170.4 (s, C=O); absorptions of the CF3 group and of the neighboring C atom could not be found due to overlap and low intensity, respectively.
1-Phenyl-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6f): CC (SiO2, 4% MeOH in CH2Cl2), 192 mg (79%). Colorless crystals, m.p. 146–148 °C (CH2Cl2/hexanes). 1H NMR (CDCl3, 600 MHz): δ 4.13 (dbr, J ≈ 1.6 Hz, 2 H, CH2), 5.39 (sbr, 1 H, NH), 7.28–7.31, 7.40–7.43, 7.52–7.54 (3 m, 1 H, 2 H, 2 H). 13C NMR (CDCl3, 151 MHz): δ 43.8 (t, CH2), 118.3 (q, 1JC-F = 275.1 Hz, CF3), 124.7, 127.5, 128.9 (3 d, 5 CH), 136.7 (q, 2JC-F = 37.6 Hz, C(3)), 140.0 (s, i-C), 157.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.6 (s, CF3). IR (neat): ν 3288 (NH), 1689 and 1655 (C=O), 1495, 1394, 1353, 1320, 1193–1137 (CF3), 1092, 1057 cm−1. ESI-MS (m/z): 244.2 (100, [M+H]+). C10H8F3N3O (243.2): calcd. C 49.39, H 3.32, N 17.28; found: C 49.23, H 3.47, N 17.30.
1-(4-Tolyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6g): CC (SiO2, 5% MeOH in CH2Cl2), 195 mg (76%). Colorless crystals, m.p. 153–154 °C (CH2Cl2/hexanes). 1H NMR (CDCl3, 600 MHz): δ 2.35 (s, 3 H, Me), 4.14 (dbr, J ≈ 1.5 Hz, 2 H, CH2), 5.37 (sbr, 1 H, NH), 7.21, 7.39 (2 dbr, J ≈ 8.2 Hz, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 21.2 (q, Me), 43.8 (t, CH2), 118.3 (q, 1JC-F = 275.0 Hz, CF3), 124.7, 129.5 (2 d, 4 CH), 136.6 (q, 2JC-F = 37.4 Hz, C(3)), 137.45, 137.53 (2 s, 2 i-C), 157.8 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.6 (s, CF3). IR (neat): ν 3243 (NH), 1681 and 1648 (C=O), 1513, 1402, 1357, 1316, 1189–1133 (CF3), 1085, 820 cm−1. ESI-MS (m/z): 258.2 (100, [M+H]+). C11H10F3N3O (257.2): calcd. C 51.37, H 3.92, N 16.34; found: C 51.33, H 3.93, N 16.80.
1-(4-Benzyloxyphenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6h): CC (SiO2, 2% MeOH in CH2Cl2), 286 mg (82%). Light orange crystals, m.p. 130–132 °C (CH2Cl2/hexanes). 1H NMR (CDCl3, 600 MHz): δ 4.15 (dbr, J ≈ 1.4 Hz, 2 H, NCH2), 5.07 (s, 2 H, OCH2), 5.33 (sbr, 1 H, NH), 6.98–7.01, 7.32–7.34, 7.37–7.43 (3 m, 2 H, 1 H, 6 H). 13C NMR (CDCl3, 151 MHz): δ 43.8 (t, NCH2), 70.3 (t, OCH2), 115.1 (d, 2 CH), 118.3 (q, 1JC-F = 275.1 Hz, CF3), 126.3, 127.6, 128.2, 128.7 (4 d, 7 CH), 133.3 (s, i-C), 136.5 (q, 2JC-F = 37.3 Hz, C(3)), 136.8, 157.8, 157.9 (3 s, 2 i-C, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.5 (s, CF3). IR (neat): ν 3276 (NH), 1692–1651 (C=O), 1506, 1349, 1327, 1185–1141 (CF3), 1081, 1051 cm−1. ESI-MS (m/z): 372.3 (100, [M+Na]+), 350.4 (52, [M+H]+). C17H14F3N3O2 (349.3): calcd. C 58.45, H 4.04, N 12.03; found: C 58.23, H 4.12, N 12.20.
(S)-5-Methyl-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6i): CC (SiO2, CH2Cl2), 233 mg (77%). Light orange solid, m.p. 132–133 °C. [α]D20 = −31.7 (c 0.26, CHCl3). 1H NMR (CD3OD, 600 MHz): δ 1.51 (d, J = 6.8 Hz, 3 H, CH3), 4.33 (q, J = 6.8 Hz, 1 H, 5-H), 7.91–7.93, 8.26–8.28 (2 m, 2 H each). 13C NMR (CD3OD, 151 MHz): δ 19.2 (q, Me), 50.8 (d, C(5)), 120.0 (q, 1JC-F = 274.5 Hz, CF3), 124.8, 125.0 (2 d, 4 CH), 139.7 (q, 2JC-F = 37.2 Hz, C(3)), 146.5, 147.0 (2 s, 2 i-C), 164.2 (s, C=O). 19F NMR (CD3OD, 565 MHz): δ −68.5 (s, CF3). IR (neat): ν 3247 (NH), 1685 (C=O), 1588, 1517, 1331, 1197, 1140–1082 (CF3), 855 cm−1. ESI-MS (m/z): 325.2 (100, [M+Na]+). C11H9F3N4O3 (302.2): calcd. C 43.72, H 3.00, N 18.54; found: C 43.60, H 3.17, N 18.66.
(S)-5-(1-Methylethyl)-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6j): CC (SiO2, petroleum ether/EtOAc 4:1), 271 mg (82%). Pale yellow solid, m.p. 143–144 °C. [α]D20 = −213.4 (c 0.27, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 1.02 (d, J = 6.8 Hz, 3 H, CH3), 1.07 (d, J = 7.0 Hz, 3 H, CH3), 2.36–2.44 (m, 1 H), 4.14 (ddbr, J ≈ 2.4, 3.9 Hz, 1 H, 5-H), 5.44 (sbr, 1 H, NH), 7.88–7.90, 8.24–8.27 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 16.7, 18.3 (2 q, 2 Me), 33.2 (d, CHMe2), 59.5 (d, C(5)), 117.4 (q, 1JC-F = 275.4 Hz, CF3), 123.9, 124.2 (2 d, 4 CH), 137.4 (q, 2JC-F = 37.5 Hz, C(3)), 145.3, 145.4 (2 s, 2 i-C), 160.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.8 (s, CF3). IR (neat): ν 3284 (NH), 1689 and 1659 (C=O), 1524, 1495, 1334, 1193, 1148–1111 (CF3), 1081, 850 cm−1. ESI-MS (m/z): 353.2 (100, [M+Na]+), 331.3 (20, [M+H]+). C13H13F3N4O3 (330.3): calcd. C 47.28, H 3.97, N 16.96; found: C 47.38, H 4.12, N 16.83.
(S)-5-(2-Methylpropyl)-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6k): CC (SiO2, petroleum ether/CH2Cl2 1:3), 186 mg (54%). Light orange solid, m.p. 107–108 °C (hexanes). [α]D20 = −190.6 (c 0.23, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 0.98, 1.01 (2 d, J = 6.1 Hz, 3 H each, 2 CH3), 1.69–1.83 (m, 3 H), 4.27 (ddd, J = 2.2, 4.6, 8.4 Hz, 1 H, 5-H), 5.59 (sbr, 1 H, NH), 7.88–7.90, 8.23–8.25 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 21.8, 22.9 (2 q, 2 Me), 24.2 (d, CHMe2), 41.9 (t, CH2), 52.7 (d, C(5)), 118.2 (q, 1JC-F = 275.3 Hz, CF3), 123.6, 124.2 (2 d, 4 CH), 137.4 (q, 2JC-F = 37.5 Hz, C(3)), 145.2, 145.4 (2 s, 2 i-C), 161.7 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.8 (s, CF3). IR (neat): ν 3295 (NH), 1685 and 1659 (C=O), 1521, 1331, 1193–1123 (CF3), 1081, 849 cm−1. ESI-MS (m/z): 345.3 (100, [M+H]+). C14H15F3N4O3 (344.3): calcd. C 48.84, H 4.39, N 16.27; found: C 48.83, H 4.40, N 16.53.
(S)-1-(4-Nitrophenyl)-5-phenyl-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6l): CC (SiO2, CH2Cl2/hexanes 3:1), 211 mg (58%). Pale yellow solid, m.p. 116–117 °C. [α]D20 = +20.7 (c 0.28, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 5.32 (dbr, J ≈ 2.0 Hz, 1 H, 5-H), 5.83 (sbr, 1 H, NH), 7.41–7.46, 7.86–7.89, 8.21–8.24 (3 m, 5 H, 2 H, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 58.2 (d, C(5)), 118.3 (q, 1JC-F = 275.5 Hz, CF3), 123.8, 124.2, 126.8, 129.6, 129.8 (5 d, 9 CH), 136.8 (q, 2JC-F = 37.7 Hz, C(3)), 137.2, 145.2, 145.5 (3 s, 3 i-C), 159.6 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.4 (s, CF3). IR (neat): ν 3291 (NH), 1688 (C=O), 1592, 1517, 1320, 1193–1141 (CF3), 1081, 854 cm−1. ESI-MS (m/z): 387.2 (100, [M+Na]+), 365.4 (20, [M+H]+). C16H11F3N4O3 (364.3): calcd. C 52.75, H 3.04, N 15.38; found: C 52.69, H 3.13, N 15.21.
(S)-5-(Hydroxymethyl)-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6m): CC (SiO2, CH2Cl2/EtOAc 6:1), 239 mg (75%). Yellow solid, m.p. 127–128 °C. [α]D20 = −96.5 (c 0.28, MeCN). 1H NMR (CD3OD, 600 MHz): δ 3.72 (dd, J = 2.8, 11.6 Hz, 1 H, CH2), 4.08 (dd, J = 3.1, 11.6 Hz, 1 H, CH2), 4.33 (pseudo-t, J ≈ 2.9 Hz, 5-H), 7.93–7.95, 8.25–8.27 (2 m, 2 H each). 13C NMR (CD3OD, 151 MHz): δ 57.8 (d, C(5)), 64.7 (t, CH2), 119.9 (q, 1JC-F = 274.5 Hz, CF3), 124.7, 125.2 (2 d, 4 CH), 139.7 (q, 2JC-F = 36.9 Hz, C(3)), 146.5, 147.1 (2 s, 2 i-C), 162.5 (s, C=O). 19F NMR (CD3OD, 565 MHz): δ −72.1 (s, CF3). IR (neat): ν 3411 (OH), 3198 (NH), 1692 and 1659 (C=O), 1513, 1327, 1193–1088 (CF3), 1036 cm−1. ESI-MS (m/z): 341.1 (100, [M+Na]+), 319.2 (13, [M+H]+). C11H9F3N4O4 (318.1): calcd. C 41.52, H 2.85, N 17.61; found: C 41.60, H 3.04, N 17.33.
(S)-5-[2-(Methylthio)ethyl]-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6n): CC (SiO2, petroleum ether/EtOAc 3:1), 329 mg (91%). Thick yellow oil. [α]D20 = −93.0 (c 0.25, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.14 (s, 3 H, Me), 2.15–2.20, 2.32–2.37, 2.68–2.79 (3 m, 1 H, 1 H, 2 H), 4.45 (ddd, J = 1.9, 4.1, 7.9 Hz, 1 H, 5-H), 6.07 (sbr, 1 H, NH), 7.88–7.90, 8.24–8.27 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 15.3 (q, Me), 30.4, 31.2 (2 t, 2 CH2), 54.0 (d, C(5)), 118.2 (q, 1JC-F = 275.5 Hz, CF3), 123.8, 124.2 (2 d, 4 CH), 137.3 (q, 2JC-F = 37.5 Hz, C(3)), 145.2, 145.4 (2 s, 2 i-C), 161.0 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.8 (s, CF3). IR (neat): ν 3302 (NH), 1689 and 1657 (C=O), 1592, 1517, 1327, 1193–1108 (CF3), 1081, 854 cm−1. (-)-ESI-MS (m/z): 360.9 (100, [M–H]−), 350.1 (13). C13H13F3N4O3S (362.1): calcd. C 43.09, H 3.62, N 15.46, S 8.85; found: C 42.99, H 3.73, N 15.67, S 8.78.
Methyl (S)-2-[1-(4-nitrophenyl)-3-trifluoromethyl-6(1H)-oxo-4,5-dihydro-1,2,4-triazin-5-yl]acetate (6o): CC (SiO2, petroleum ether/CH2Cl2 2:1), 335 mg (93%). Yellow solid, m.p. 114–115 °C (hexanes). [α]D20 = −82.0 (c 0.24, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.90 (dd, J = 10.3, 17.6 Hz, 1 H, CH2), 3.24 (dd, J = 2.8, 17.6 Hz, 1 H, CH2), 3.79 (s, 3 H, Me), 4.66 (ddd, J = 1.8, 2.8, 10.3 Hz, 1 H, 5-H), 6.19 (sbr, 1 H, NH), 7.90, 8.26 (2 dbr, J ≈ 9.2 Hz, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 37.1 (t, CH2), 50.8 (d, C(5)), 52.8 (q, Me), 118.1 (q, 1JC-F = 275.4 Hz, CF3), 123.8, 124.2 (2 d, 4 CH), 137.3 (q, 2JC-F = 37.8 Hz, C(3)), 145.0, 145.6 (2 s, 2 i-C), 159.8, 171.6 (2 s, 2 C=O). 19F NMR (CDCl3, 565 MHz): δ −70.8 (s, CF3). IR (neat): ν 3396 (NH), 1715 (C=O), 1681 (C=O), 1588, 1521, 1491, 1390, 1323, 1193–1139 (CF3), 1107, 857 cm−1. ESI-MS (m/z): 383.2 (100, [M+Na]+). C13H11F3N4O5 (360.2): calcd. C 43.34, H 3.08, N 15.55; found: C 43.07, H 2.95, N 15.61.
(S)-5-[(Indol-3-yl)methyl]-1-(4-nitrophenyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one (6p): CC (SiO2, EtOAc), 346 mg (83%). Pale yellow solid, m.p. 140–141 °C. [α]D20 = −143.6 (c 0.24, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 3.24 (dd, J = 9.4, 14.6 Hz, 1 H, CH2), 3.53 (dd, J = 3.4, 14.6 Hz, 1 H, CH2), 4.54 (ddd, J = 1.7, 3.4, 9.4 Hz, 1 H, 5-H), 5.32 (sbr, 1 H, NH), 7.13–7.17, 7.25–7.28 (2 m, 2 H, 1 H), 7.43 (dbr, J ≈ 8.2 Hz, 1 H), 7.63 (dbr, J ≈ 7.9 Hz, 1 H), 7.77–7.80, 8.22–8.24 (2 m, 2 H each) °8.24 (sbr, 1 H, NH). 13C NMR (CDCl3, 151 MHz): δ 30.1 (t, CH2), 54.4 (d, C(5)), 108.5 (s, i-C), 111.7 (d, CH), 118.2 (q, 1JC-F = 275.3 Hz, CF3), 118.6, 120.4, 123.1, 123.8, 123.9, 124.1 (6 d, 8 CH), 126.7, 136.6 (2 s, 2 i-C), 137.0 (q, 2JC-F = 37.6 Hz, C(3)), 145.2, 145.4 (2 s, 2 i-C), 161.39 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −70.6 (s, CF3). IR (neat): ν 3351 (NH), 1689 and 1662 (C=O), 1592, 1517, 1327, 1200–1144 (CF3), 1085, 854 cm−1. (-)-ESI-MS (m/z): 416.0 (88, [M–H]−). C19H14F3N5O3 (417.3): calcd. C 54.68, H 3.38, N 16.78; found: C 54.43, H 3.49, N 16.74.
(S)-5-Phenyl-1-(4-tolyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one ((S)-6q): CC (SiO2, CH2Cl2), 250 mg (75%). Colorless crystals, m.p. 153–154 °C. [α]D20 = +171.6 (c 0.28, CHCl3). 1H NMR (CD3OD, 600 MHz): δ 2.35 (s, 3 H, CH3), 5.28 (sbr, 1 H, NH), 7.22, 7.32 (dbr, J ≈ 8.3 Hz, 2 H each), 7.37–7.40, 7.42–7.45 (2 m, 1 H, 4 H). 13C NMR (CD3OD, 151 MHz): δ 21.1 (q, Me), 58.9 (d, C(5)), 120.1 (q, 1JC-F = 274.4 Hz, CF3), 126.1, 127.9, 129.9, 130.1, 130.2 (5 d, 9 CH), 138.5 (s, i-C), 138.6 (q, 2JC-F = 37.0 Hz, C(3)), 139.4, 140.7 (2 s, 2 i-C), 161.6 (s, C=O). 19F NMR (CD3OD, 565 MHz): δ −72.1 (s, CF3). IR (neat): ν 3336 (NH), 1677 and 1655 (C=O), 1510, 1338, 1189–1144 (CF3), 1084, 820 cm−1. ESI-MS (m/z): 372.1 (57, [M+K]+), 356.1 (47, [M+Na]+), 334.2 (100, [M+H]+). C17H14F3N3O (333.3): calcd. C 61.26, H 4.23, N 12.61; found: C 61.08, H 4.37, N 12.86. A sample of rac-6q (276 mg, 83%) was prepared in an analogous manner starting with hydrazonoyl bromide 11g and racemic methyl phenylglycinate (rac-12e). The obtained 1H and 13C NMR data perfectly matched those obtained for (S)-6q.
(S)-2-(4-Nitrophenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]-triazin-1(2H)-one (7a): CC (SiO2, CH2Cl2), 269 mg (82%). Yellow solid, m.p. 90–92 °C (CH2Cl2/hexanes). [α]D20 = +79.2 (c 0.17, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.08–2.14 (m, 2 H, 7-H2), 2.28–2.34 (m, 1 H, 8-H), 2.46–2.51 (m, 1 H, 8-H), 3.75 (tbr, J ≈ 7.4 Hz, 2 H, 6-H2), 4.17 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 7.92–7.94, 8.24–8.26 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.3 (t, C(8)), 48.8 (q, 4JC-F = 2.3 Hz, C(6)), 58.4 (d, C(8a)), 118.5 (q, 1JC-F = 276.0 Hz, CF3), 123.5, 124.2 (2 d, 4 CH), 138.1 (q, 2JC-F = 36.5 Hz, C(4)), 145.2* (s, 2 i-C), 161.5 (s, C=O); *higher intensity. 19F NMR (CDCl3, 565 MHz): δ −68.6 (s, CF3). IR (neat): ν 1698 (C=O), 1519, 1452, 1344, 1191, 1135 (CF3) cm−1. ESI-MS (m/z): 329.3 (100, [M+H]+). C13H11F3N4O3 (328.2): calcd. C 47.57, H 3.38, N 17.07; found: C 47.78, H 3.48, N 17.19.
(S)-2-(3-Nitrophenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]-triazin-1(2H)-one (7b): CC (SiO2, CH2Cl2), 213 mg (65%). Thick yellow oil. [α]D20 = +27.4 (c 0.15, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.06–2.14 (m, 2 H, 7-H2), 2.27–2.33 (m, 1 H, 8-H), 2.45–2.51 (m, 1 H, 8-H), 3.71–3.77 (m, 2 H, 6-H2), 4.17 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 7.55 (t, J = 8.2 Hz, 1H), 8.01 (ddd, J = 1.0, 2.2, 8.2 Hz, 1 H), 8.10 (ddd, J = 1.0, 2.2, 8.2 Hz, 1 H), 8.56 (t, J = 2.2 Hz, 1 H). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.3 (t, C(8)), 48.8 (q, 4JC-F = 2.4 Hz, C(6)), 58.3 (d, C(8a)), 118.5 (q, 1JC-F = 276.0 Hz, CF3), 119.0, 121.2, 129.4, 129.6 (4 d, 4 CH), 138.0 (q, 2JC-F = 36.5 Hz, C(4)), 141.0, 148.4 (2 s, 2 i-C), 161.3 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.5 (s, CF3). IR (neat): ν 1696 and 1648 (C=O), 1528, 1454, 1349, 1193–1118 (CF3) cm−1. ESI-MS (m/z): 329.3 (64, [M+H]+), 327.3 (100, [M–H]+), 299.2 (36). C13H11F3N4O3 (328.2): calcd. C 47.57, H 3.38, N 17.07; found: C 47.75, H 3.43, N 16.84.
(S)-4-[4-Trifluoromethyl-1(2H)-oxo-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-2-yl]benzonitrile (7c): CC (SiO2, CH2Cl2), 286 mg (93%). Light orange solid, m.p. 103–105 °C. [α]D20 = +68.3 (c 0.16, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.07–2.13 (m, 2 H, 7-H2), 2.26–2.33 (m, 1 H, 8-H), 2.45–2.50 (m, 1 H, 8-H), 3.71–3.76 (m, 2 H, 6-H2), 4.16 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 7.66–7.69, 7.84–7.86 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.3 (t, C(8)), 48.8 (q, 4JC-F = 2.3 Hz, C(6)), 58.4 (d, C(8a)), 109.7 (s, CN), 118.5 (q, 1JC-F = 275.8 Hz, CF3), 118.8 (s, i-C), 123.8, 132.7 (2 d, 4 CH), 138.0 (q, 2JC-F = 36.3 Hz, C(4)), 143.7 (s, i-C), 161.3 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.5 (s, CF3). IR (neat): ν 2223 (CN), 1690 and 1655 (C=O), 1603, 1452, 1315, 1126 (CF3) cm−1. ESI-MS (m/z): 331.3 (29, [M+Na]+), 309.3 (100, [M+H]+). C14H11F3N4O (308.3): calcd. C 54.55, H 3.60, N 18.18; found: C 54.61, H 3.73, N 18.13.
(S)-2-(4-Chlorophenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-1(2H)-one (7d): Reaction time: 2d; CC (SiO2, petroleum ether/EtOAc 4:1), 219 mg (69%). Pale yellow solid, m.p. 86–87 °C. [α]D20 = +35.1 (c 0.19, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.03–2.12 (m, 2 H, 7-H2), 2.24–2.31 (m, 1 H, 8-H), 2.43–2.48 (m, 1 H, 8-H), 3.68–3.75 (m, 2 H, 6-H2), 4.13 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 7.34–7.36, 7.53–7.55 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.4 (t, C(8)), 48.7 (q, 4JC-F = 2.3 Hz, C(6)), 58.3 (d, C(8a)), 118.6 (q, 1JC-F = 275.7 Hz, CF3), 125.5, 128.8 (2 d, 4 CH), 132.3 (s, i-C), 137.6 (q, 2JC-F = 36.2 Hz, C(4)), 138.7 (s, i-C), 160.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.5 (s, CF3). IR (neat): ν 1692 and 1644 (C=O), 1491, 1446, 1331, 1193, 1122 (CF3) cm−1. ESI-MS (m/z): 320.3 (31, [M{37Cl}+H]+), 318.2 (100, [M{35Cl}+H]+). C13H11ClF3N3O (317.7): calcd. C 49.15, H 3.49, N 13.23; found: C 49.22, H 3.68, N 13.10.
(S)-2-(2,4-Dichlorophenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-1(2H)-one (7e): In order to accelerate the second cyclization step, the crude reaction mixture was heated in an oil bath (60 °C) for three days; CC (SiO2, petroleum ether/EtOAc 4:1), 176 mg (50%). Yellow solid, m.p. 107–108 °C. [α]D20 = −6.8 (c 0.13, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.04–2.12 (m, 2 H, 7-H2), 2.25–2.32 (m, 1 H, 8-H), 2.43–2.48 (m, 1 H, 8-H), 3.68–3.78 (m, 2 H, 6-H2), 4.17 (pseudo-t, J ≈ 7.9 Hz, 1 H, 8a-H), 7.32 (mc, 2 H), 7.47 (t, J = 1.2 Hz, 1 H). 13C NMR (CDCl3, 151 MHz): δ 24.0 (t, C(7)), 28.2 (t, C(8)), 48.8 (q, 4JC-F = 2.5 Hz, C(6)), 58.1 (d, C(8a)), 118.5 (q, 1JC-F = 275.7 Hz, CF3), 128.2, 130.2, 130.4 (3 d, 3 CH), 133.3, 135.2, 136.5 (3 s, 3 i-C), 137.6 (q, 2JC-F = 36.2 Hz, C(4)), 161.0 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.4 (s, CF3). IR (neat): ν 1689 and 1640 (C=O), 1480, 1443, 1228, 1189, 1160–1118 (CF3) cm−1. ESI-MS (m/z): 353.5 (24), 352.4 (100). C13H10Cl2F3N3O (352.1): calcd. C 44.34, H 2.86, N 11.93; found: C 44.42, H 2.96, N 12.04.
(S)-2-Phenyl-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-1(2H)-one (7f): CC (SiO2, petroleum ether/EtOAc 4:1), 170 mg (60%). Yellow solid, m.p. 61–63 °C. [α]D20 = +22.6 (c 0.18, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.02–2.11 (m, 2 H, 7-H2), 2.25–2.32 (m, 1 H, 8-H), 2.42–2.47 (m, 1 H, 8-H), 3.67–3.75 (m, 2 H, 6-H2), 4.14 (dd, J = 6.9, 8.8 Hz, 1 H, 8a-H), 7.25–7.28, 7.38–7.41, 7.54–7.56 (3 m, 1 H, 2 H, 2 H). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.4 (t, C(8)), 48.7 (q, 4JC-F = 2.3 Hz, C(6)), 58.2 (d, C(8a)), 118.7 (q, 1JC-F = 275.6 Hz, CF3), 124.5, 127.0, 128.7 (3 d, 5 CH), 137.3 (q, 2JC-F = 36.1 Hz, C(4)), 140.2 (s, i-C), 160.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.4 (s, CF3). IR (neat): ν 1685 and 1640 (C=O), 1457, 1312, 1199, 1133 (CF3), 1102 cm−1. ESI-MS (m/z): 284.2 (32, [M+H]+), 205.2 (100, [M–Ph]+). C13H12F3N3O (283.2): calcd. C 55.12, H 4.27, N 14.84; found: C 55.41, H 4.44, N 14.59.
(S)-2-(4-Tolyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-1(2H)-one (7g): CC (SiO2, petroleum ether/CH2Cl2 1:3), 166 mg (56%). Light gray solid, m.p. 78–80 °C. [α]D20 = +22.7 (c 0.16, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.03–2.11 (m, 2 H, 7-H2), 2.25–2.32 (m, 1 H, 8-H), 2.35 (s, 3 H, Me), 2.42–2.47 (m, 1 H, 8-H), 3.67–3.76 (m, 2 H, 6-H2), 4.13 (dd, J = 6.9, 8.8 Hz, 1 H, 8a-H), 7.18–7.20, 7.39–7.41 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 21.2 (q, Me), 24.1 (t, C(7)), 28.5 (t, C(8)), 48.7 (q, 4JC-F = 2.4 Hz, C(6)), 58.2 (d, C(8a)), 118.7 (q, 1JC-F = 275.6 Hz, CF3), 124.5, 129.4 (2 d, 4 CH), 137.0 (s, i-C), 137.2 (q, 2JC-F = 36.1 Hz, C(4)), 137.7 (s, i-C), 160.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.4 (s, CF3). IR (neat): ν 1687 and 1644 (C=O), 1511, 1444, 1198, 1120 (CF3), 820 cm−1. ESI-MS (m/z): 299.2 (100, [M+2H]+). C14H14F3N3O (297.1): calcd. C 56.56, H 4.75, N 14.14; found: C 56.67, H 4.84, N 13.90.
(S)-2-(4-Benzyloxyphenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]triazin-1(2H)-one (7h): CC (SiO2, petroleum ether/EtOAc 4:1), 183 mg (47%). Colorless solid, m.p. 97–99 °C. [α]D20 = +35.0 (c 0.21, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.02–2.10 (m, 2 H, 7-H2), 2.24–2.31 (m, 1 H, 8-H), 2.42–2.47 (m, 1 H, 8-H), 3.66–3.74 (m, 2 H, 6-H2), 4.13 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 5.07 (s, 2 H, OCH2), 6.97–7.00, 7.31–7.34, 7.37–7.39, 7.41–7.44 (4 m, 2 H, 1 H, 2 H, 4 H). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.5 (t, C(8)), 48.7 (q, 4JC-F = 2.6 Hz, C(6)), 58.2 (d, C(8a)), 70.3 (t, OCH2), 115.0 (d, 2 CH), 118.7 (q, 1JC-F = 275.6 Hz, CF3), 126.1, 127.6, 128.1, 128.7 (4 d, 7 CH), 133.5, 137.0 (2 s, 2 i-C), 137.2 (q, 2JC-F = 36.2 Hz, C(4)), 157.6 (s, i-C), 160.9 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.4 (s, CF3) ppm. IR (neat): ν 1685 and 1644 (C=O), 1506, 1450, 1338, 1241, 1133 (CF3), 1018 cm−1. ESI-MS (m/z): 412.4 (77, [M+Na]+), 390.4 (100, [M+H]+). C20H18F3N3O2 (389.4): calcd. C 61.69, H 4.66, N 10.79; found: C 61.50, H 4.83, N 10.56.
Synthesis of (S)-4-Methyl-5-phenyl-1-(4-tolyl)-3-trifluoromethyl-4,5-dihydro-1,2,4-triazin-6(1H)-one ((S)-6r)): A solution of 1,2,4-triazin-6(1H)-one (S)-6q (0.5 mmol, 167 mg) in anhydrous MeOH (5.0 mL) was added dropwise to a vigorously stirred solution of sodium methoxide (5.0 mmol, 270 mg) in dry MeOH (25 mL) under argon, at room temperature. Then MeI (5.0 mmol, 705 mg) was added to the resulting mixture and the stirring was continued for 24 h. After the solvents were removed in vacuo, the residue was washed with EtOAc (3 × 20 mL). The organic layers were combined, the solvent was removed, and the crude product was purified by CC (SiO2, DCM) to give (S)-6r (101 mg, 58%). Colorless solid, m.p. 92–93 °C. [α]D20 = +5.2 (c 0.24, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.34 (s, 3 H, Me), 3.05 (s, 3 H, NMe), 4.94 (s, 1 H, 5-H), 7.17–7.19, 7.36–7.44 (2 m, 2 H, 7 H). 13C NMR (CDCl3, 151 MHz): δ 21.2 (q, Me), 36.2* (q, JC-F = 3.4 Hz, NMe), 66.0 (d, C(5)), 118.9 (q, 1JC-F = 275.9 Hz, CF3), 124.4, 126.9, 129.36, 129.43, 129.5 (5 d, 9 CH), 135.9 (s), 136.5 (q, 2JC-F = 34.3 Hz, C(3)), 137.1, 137.6 (2 s), 158.6 (s, C=O); *through-space C–F coupling observed. 19F NMR (CDCl3, 565 MHz): δ −66.1 (s, CF3) ppm. IR (neat): ν 1681 and 1655 (C=O), 1513, 1416, 1364, 1238, 1182, 1126 (CF3), 1077 cm−1. ESI-MS (m/z): 370.1 (100, [M+Na]+), 348.1 (80, [M+H]+), 318.2 (94). C18H16F3N3O (347.1): calcd. C 62.24, H 4.64, N 12.10; found: C 61.99, H 4.51, N 12.08. A sample of rac-6r (111 mg, 64%; 0.5 mmol scale) was obtained in an analogous manner starting with rac-6q. Colorless crystals, m.p. 93–95 °C. The NMR spectra (1H and 13C) of rac-6r were in accordance with those of (S)-6r.
Synthesis of 1-(4-tolyl)-3-trifluoromethyl-1,2,4-triazin-6(1H)-one (6s): A mixture of 4,5-dihydro-1,2,4-triazinone 6g (0.5 mmol, 128.5 mg), K3Fe(CN)6 (3.0 mmol, 987 mg), aqueous solution of Na2CO3 (0.5M, 10 mL), and Et4NBr (15 mol%) in CH2Cl2 (10 mL) was vigorously stirred at room temperature for 4 h (monitored on TLC). The resulting mixture was extracted with CH2Cl2 (3 × 10 mL), the combined organic layers were dried over anh. Na2SO4, filtered and the solvents were removed under reduced pressure. The crude product was purified by standard CC (SiO2, CH2Cl2) to give 6s (99 mg, 78%). Colorless solid, m.p. 80–82 °C. 1H NMR (CDCl3, 600 MHz): δ 2.36 (s, 3 H, Me), 7.25, 7.57 (2 dbr, J ≈ 8.3 Hz, 2 H each), 8.47 (s, 1 H, 5-H). 13C NMR (CDCl3, 151 MHz): δ 21.4 (q, Me), 119.2 (q, 1JC-F = 273.6 Hz, CF3), 123.9, 129.9 (2 d, 4 CH), 136.6, 140.2 (2 s, 2 i-C), 140.6 (q, 2JC-F = 39.0 Hz, C(3)), 152.6 (s, C=O), 161.0 (d, C(5)). 19F NMR (CDCl3, 565 MHz): δ −69.7 (s, CF3). IR (neat): ν 1674 (C=O), 1391, 1346, 1156, 1088 (CF3) cm−1. ESI-MS (m/z): 256.1 (100, [M+H]+). C11H8F3N3O (255.1): calcd. C 51.77, H 3.16, N 16.47; found: C 51.59, H 3.23, N 16.43.
General procedure for catalytic hydrogenation reactions: A solution of the corresponding triazinone (7a or 7h, 0.5 mmol) in EtOH (10 mL) was added Pd/C (5.0 mmol), and the resulting mixture was vigorously shaken in the atmosphere of H2 (3 atm) for the required time. The mixture was filtered through Celite, washed with EtOH (5 mL), and the solvents were removed under reduced pressure. The resulting mixture was filtered through a short plug of silica (CC) to give the spectroscopically pure product.
(S)-2-(4-Aminophenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]-triazin-1(2H)-one (7i): Reaction time: 3 h; CC (SiO2, silica was washed with 5% Et3N in EtOAc prior to use; petroleum ether/EtOAc 4:1), 132 mg (89%). Thick light orange oil. [α]D20 = +43.1 (c 0.15, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.03–2.10 (m, 2 H, 7-H2), 2.23–2.32 (m, 1 H, 8-H), 2.41–2.46 (m, 1 H, 8-H), 3.65–3.74 (m, 2 H, 6-H2), 3.70 (sbr, 2 H, NH2), 4.12 (dd, J = 6.9, 8.8 Hz, 1 H, 8a-H), 6.66–6.96, 7.24–7.27 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.5 (t, C(8)), 48.7 (q, 4JC-F = 2.1 Hz, C(6)), 58.2 (d, C(8a)), 115.1 (d, 2 CH), 118.7 (q, 1JC-F = 275.9 Hz, CF3), 126.1 (d, 2 CH), 131.5 (s, i-C), 137.0 (q, 2JC-F = 36.1 Hz, C(4)), 145.6 (s, i-C), 160.8 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.3 (s, CF3). IR (neat): ν 3362 (NH), 1674 and 1633 (C=O), 1513, 1446, 1193, 1140–1095 (CF3) cm−1. ESI-MS (m/z): 299.3 (100, [M+H]+), 279.3 (30).
(S)-2-(4-Hydroxyphenyl)-4-trifluoromethyl-6,7,8,8a-tetrahydropyrrolo [1,2-d][1,2,4]-triazin-1(2H)-one (7j): Reaction time: 16 h; CC (SiO2, hexanes/EtOAc 2:3), 100 mg (67%). Colorless solid, m.p. 94–96 °C. [α]D20 = +23.2 (c 0.18, CHCl3). 1H NMR (CDCl3, 600 MHz): δ 2.02–2.10 (m, 2 H, 7-H2), 2.24–2.31 (m, 1 H, 8-H), 2.42–2.47 (m, 1 H, 8-H), 3.67–3.76 (m, 2 H, 6-H2), 4.15 (dd, J = 6.9, 8.9 Hz, 1 H, 8a-H), 5.69 (sbr, 1 H, OH), 6.74–6.77, 7.28–7.30 (2 m, 2 H each). 13C NMR (CDCl3, 151 MHz): δ 24.1 (t, C(7)), 28.5 (t, C(8)), 48.7 (q, 4JC-F = 2.5 Hz, C(6)), 58.2 (d, C(8a)), 115.8 (d, 2 CH), 118.7 (q, 1JC-F = 275.6 Hz, CF3), 126.5 (d, 2 CH), 133.0 (s, i-C), 137.4 (q, 2JC-F = 36.1 Hz, C(4)), 155.0 (s, i-C), 161.1 (s, C=O). 19F NMR (CDCl3, 565 MHz): δ −68.4 (s, CF3). IR (neat): ν 3317 (OH), 1666 and 1636 (C=O), 1513, 1446, 1341, 1189–1122 (CF3), 835 cm−1. ESI-MS (m/z): 300.2 (100, [M+H]+), 298.2 (59).
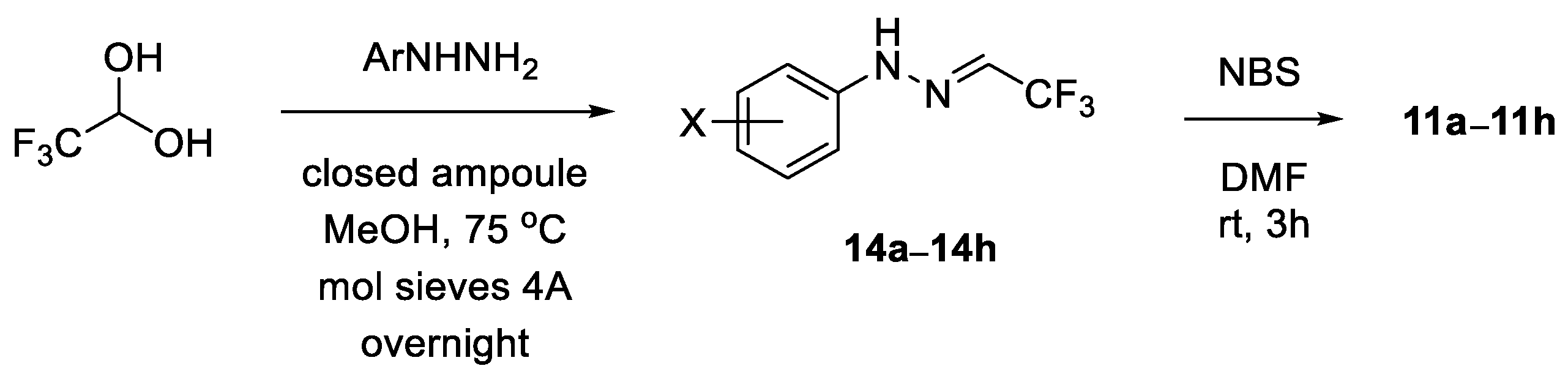
General procedure for synthesis of hydrazonoyl bromides
11: Following the general literature protocol [
25], arylhydrazone
14 (1.0 mmol) was dissolved in dry DMF (3 mL), the solution was cooled to 0 °C, then solid NBS (1.05 mmol, 187 mg) was added and stirring was continued at this temperature. After the starting hydrazone was fully consumed (TLC monitoring, typically ca. 2 h), the resulting mixture was extracted with H
2O/Et
2O 1:1 mixture (20 mL), the organic layer was washed with H
2O (3 × 10 mL), dried over anh. Na
2SO
4, filtered and the solvents were removed in vacuo. Crude products were purified by column chromatography.
N-(3-Nitrophenyl)-trifluoroacetohydrazonoyl bromide (11b): reaction time: 2h; CC (SiO2, petroleum ether/CH2Cl2 3:2), 265 mg (85%). Yellow solid, m.p. 110–111 °C. 1H NMR (CDCl3, 600 MHz): δ 7.49–7.52, 7.87–7.91, 7.99–8.01 (3 m, 2 H, 1 H, 1 H), 8.24 (sbr, 1 H, NH). 13C NMR (CDCl3, 151 MHz): δ 107.1 (q, 2JC-F = 44.0 Hz, =CCF3), 109.2, 117.7 (2 d, 2 CH), 118.2 (q, 1JC-F = 272.1 Hz, CF3), 120.0, 130.7 (2 d, 2 CH), 142.7, 149.4 (2 s, 2 i-C). 19F NMR (CDCl3, 565 MHz): δ −66.6 (s, CF3). IR (neat): ν 3258 (NH), 1614, 1524, 1346, 1304, 1238, 1121–1075 (CF3) cm−1. (-)-ESI-MS (m/z): 311.9 (100, [M{81Br}–H]−), 309.9 (99, [M{79Br}–H]−). C8H5BrF3N3O2 (312.0): calcd. C 30.79, H 1.62, N 13.47; found: C 30.95, H 1.88, N 13.65.
N-(2,4-Dichlorophenyl)-trifluoroacetohydrazonoyl bromide (11e): Reaction time: 3 h; CC (SiO2, hexanes), 332 mg (99%). Thick yellow oil. 1H NMR (CDCl3, 600 MHz): δ 7.24 (dd, J = 2.3, 8.8 Hz, 1 H), 7.35 (d, J = 2.3 Hz, 1 H), 7.42 (d, J = 8.8 Hz, 1 H), 8.52 (sbr, 1 H, NH). 13C NMR (CDCl3, 151 MHz): δ 107.8 (q, 2JC-F = 43.9 Hz, =CCF3), 116.5 (d, CH), 118.3 (q, 1JC-F = 271.9 Hz, CF3), 119.2, 127.8 (2 s, 2 i-C), 128.6, 129.2 (2 d, 2 CH), 136.6 (s, i-C). 19F NMR (CDCl3, 565 MHz): δ −66.6 (s, CF3). IR (neat): ν 3314 (NH), 1595, 1506, 1327, 1282, 1124, 1208, 1133 (CF3), 969 cm−1. (-)-ESI-MS (m/z): 336.7 (38), 335.6 (11), 334.7 (100), 332.8 (63). C8H4BrCl2F3N2 (335.9): calcd. C 28.60, H 1.20, N 8.34; found: C 28.42, H 1.36, N 8.02.
General procedure for synthesis of trifluoroacetaldehyde arylhydrazones
14: Following the literature protocol [
52], a mixture of arylhydrazine hydrochloride (1.0 mmol), excess fluoral hydrate (ca. 3.0 mmol), and freshly activated powdered molecular sieves 4Å (ca. 450 mg) in MeOH (3.5 mL) was heated in a closed ampoule in an oil bath (75 °C) overnight. The solution was cooled to room temperature and filtered through a short pad of Celite, which was washed with several portions of CH
2Cl
2 (4 × 5 mL). The combined organic layers were washed with H
2O (10 mL), then with 5%-aqueous solution of NaHCO
3 (10 mL), and dried over Na
2SO
4. The solid inorganics were filtered off and the solvents were removed under reduced pressure (cold bath). The crude products were purified by standard CC to give spectroscopically pure materials, which were used for the next step without further purification.
Trifluoroacetaldehyde 3-nitrophenylhydrazone (14b): CC (SiO2, petroleum ether/CH2Cl2 3:2), 152 mg (65%). Yellow solid, m.p. 156–158 °C. 1H NMR (CDCl3, 600 MHz): δ 7.08 (qd, JH-H = 1.4 Hz, JH-F = 3.9 Hz, 1 H, =CHCF3), 7.43 (ddd, J = 1.2, 2.2, 8.2 Hz, 1 H), 7.47 (tbr, J ≈ 8.0 Hz, 1 H), 7.83 (ddd, J = 1.2, 2.2, 7.9 Hz, 1 H), 7.91 (t, J = 2.2 Hz, 1 H), 8.18 (sbr, 1 H, NH). 13C NMR (CDCl3, 151 MHz): δ 108.3, 116.9, 119.3 (3 d, 3 CH), 120.7 (q, 1JC-F = 269.8 Hz, CF3), 125.2 (q, 2JC-F = 39.6 Hz, =CCF3), 130.5 (d, CH), 144.0, 149.4 (2 s, 2 i-C). 19F NMR (CDCl3, 565 MHz): δ −65.9 (d, JH-F = 3.9 Hz, CF3). IR (neat): ν 3302 (NH), 1610, 1558, 1342, 1290, 1245, 1118 (CF3), 1074 cm−1. (-)-ESI-MS (m/z): 231.8 (100, [M–H]−). C8H6F3N3O2 (233.0): calcd. C 41.21, H 2.59, N 18.02; found: C 41.21, H 2.72, N 18.03.
Trifluoroacetaldehyde 2,4-dichlorophenylhydrazone (14e): CC (SiO2, petroleum ether/CH2Cl2 3:2), 202 mg (79%). Light orange oil. 1H NMR (CDCl3, 600 MHz): δ 7.12 (qd, JH-H = 1.4 Hz, JH-F = 3.8 Hz, 1 H, =CHCF3), 7.22 (dd, J = 2.3, 8.8 Hz, 1 H), 7.31 (d, J = 2.3 Hz, 1 H), 7.46 (d, J = 8.8 Hz, 1 H), 8.33 (sbr, 1 H, NH). 13C NMR (CDCl3, 151 MHz): δ 115.9 (d, CH), 118.2 (s, i-C), 120.8 (q, 1JC-F = 269.7 Hz, CF3), 125.6 (q, 2JC-F = 39.3 Hz, =CCF3), 126.8 (s, i-C), 128.5, 129.0 (2 d, 2 CH), 137.7 (s, i-C). 19F NMR (CDCl3, 565 MHz): δ −65.9 (d, JH-F = 3.8 Hz, CF3). IR (neat): ν 3354 (NH), 1591, 1521, 1357, 1279, 1234, 1115 (CF3), 1051, 913, 816 cm−1. (-)-ESI-MS (m/z): 254.8 (100, [M–H]−). C8H5Cl2F3N2 (256.0): calcd. C 37.38, H 1.96, N 10.90; found: C 37.36, H 2.20, N 10.97.