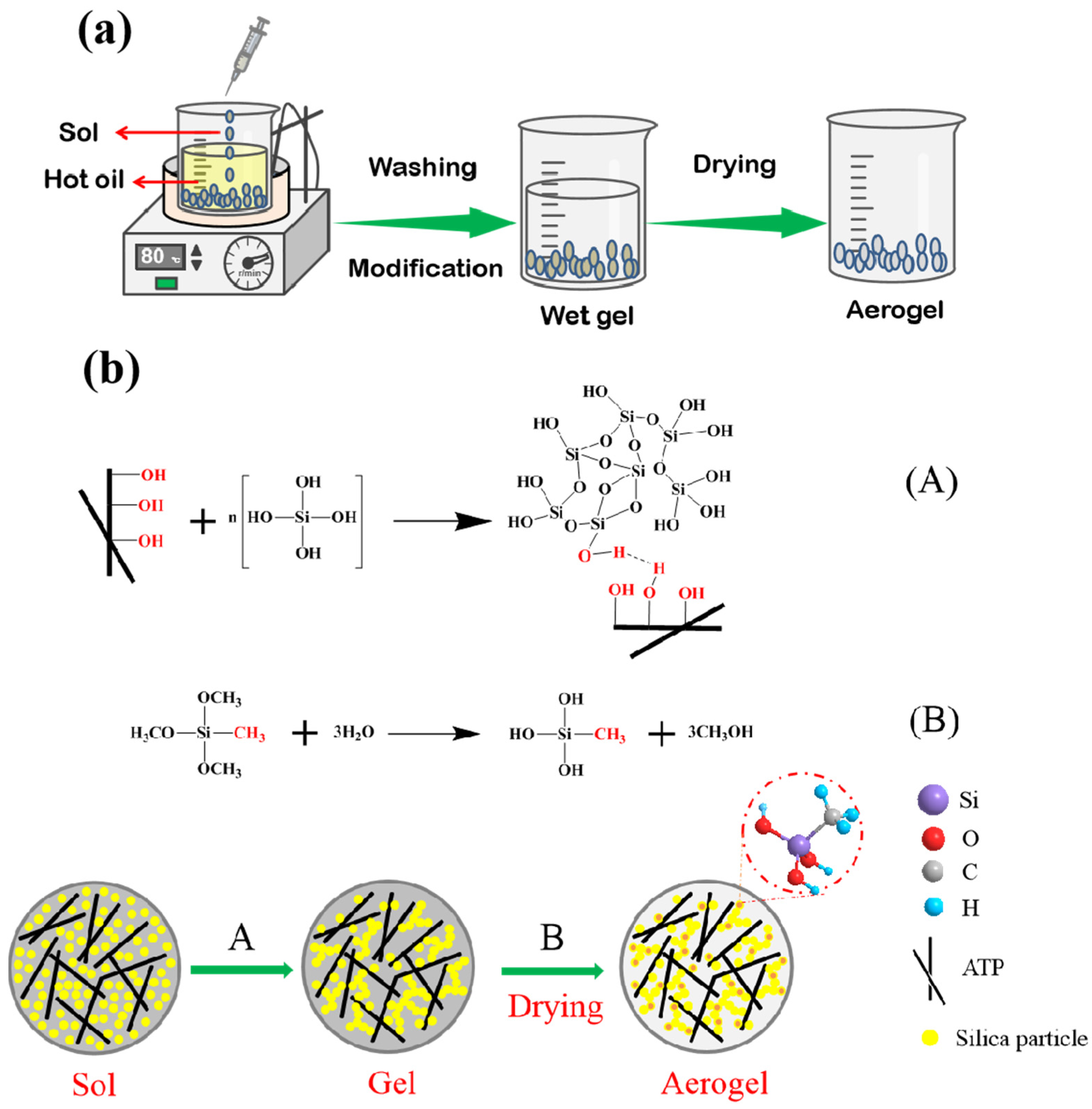
3.2. Effect of Drying Techniques and MTMS Modification on Physical Characteristic and Morphological Properties of Spherical ATP/SiO2 Aerogels
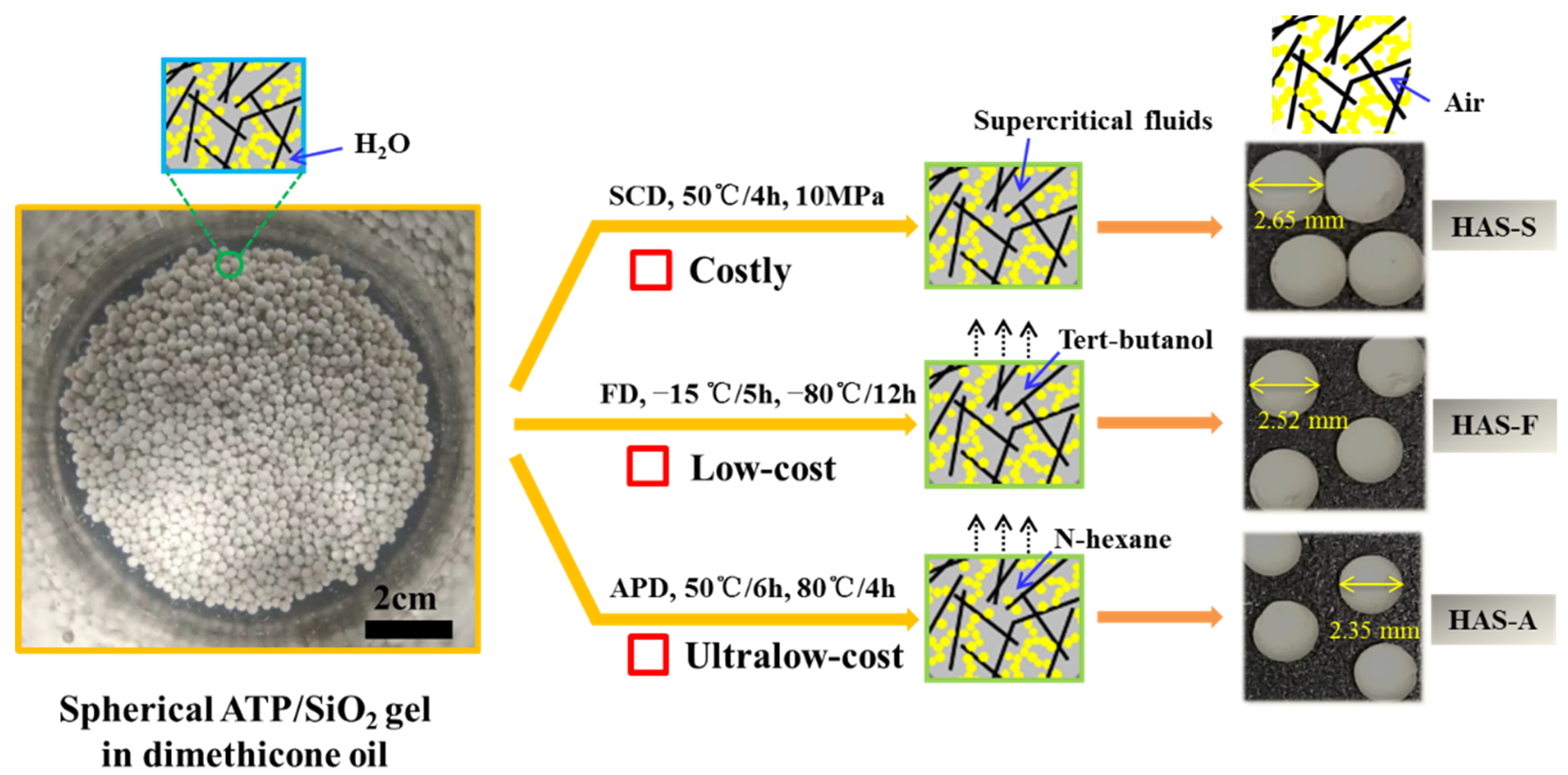
A comparison of the different drying methods for preparing the spherical ATP/SiO
2 aerogels is shown in
Figure 2. All spherical ATP/SiO
2 aerogels exhibited an integrated macroscopic morphology with a diameter of approximately 2.5 ± 0.15 mm (2.65 mm, 2.52 mm, and 2.35 mm for HAS-S, HAS-F, and HAS-A, respectively). By contrast, the shrinkage rate of the HAS-A was larger than the other samples resulting from the drying capillary pressure of the solvent evaporation during the APD process.
Table 1 shows the physical parameters of the ATP and spherical ATP/SiO
2 aerogels prepared by different drying methods. The densities of the ATP/SiO
2 aerogel composites (0.46–0.65 g/cm
3) decreased greatly due to the loose three-dimensional skeleton structure instead of the relatively dense structures of the ATP (2.41 g/cm
3). Meanwhile, the densities of the ATP/SiO
2 aerogels modified by MTMS were larger than the samples without hydrophobic modification, which is attributable to the grafting of CH
3-(SiOH)
3 groups.
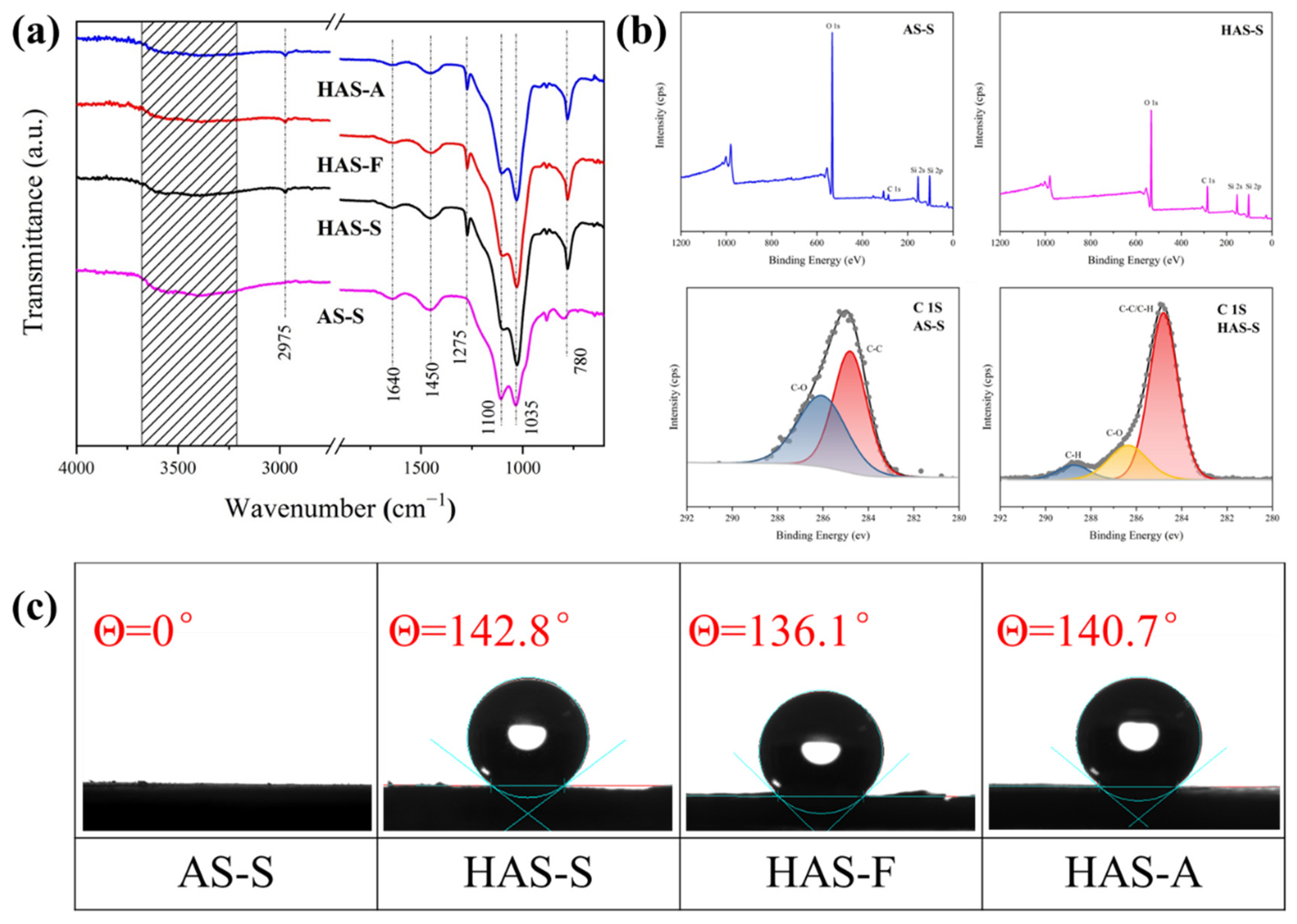
The FT-IR spectra of the as-prepared aerogels are presented in
Figure 3a. A broad transmittance appeared at approximately 3200–3700 cm
−1, which is attributed to the stretching vibrations of -OH groups [
36]. The symmetric stretching and tetrahedra bending vibration of Si-O-Si and Si (or Al)-O appeared at 1035 cm
−1 and 1100 cm
−1 [
37]. The peaks at approximately 1450 cm
−1 and 1640 cm
−1 are assigned to the deformation vibration of the adsorbed water and bound water [
32,
38]. By contrast, there was a new peak at 780 cm
−1, which is normally observed in the hydrophobic group and corresponds to the stretching vibration of -CH
3 in MTMS [
39]. Moreover, all of the modified samples showed the symmetric deformation vibration of Si-C bonds and symmetric stretching vibrations of the terminal -CH
3 groups at 2975 cm
−1 and 1275 cm
−1,which is proof of the presence of Si-CH
3 [
40]. Thus, the FT-IR spectrums have proved that the hydrophobic -CH
3 groups were successfully coupled to the HAS-S, HAS-F, and HAS-A. The XPS measurement (
Figure 3b) was subsequently used to quantify the elemental composition of AS-S and HAS-S. The XPS spectra of AS-S and HAS-S show four peaks at 100, 150, 285, and 531 ev, which are assigned to Si 2s, Si 2p, C 1s, and O 1s, respectively [
39]. Compared with AS-S, the relative strength of the C 1s peak of the HAS-S increased significantly, while that of the O 1s peak became weak. It can be inferred that with the addition of MTMS, the dehydration condensation reaction between the -OH and CH
3-(SiOH)
3 groups resulted in the loss of some O elements and the grafting of -CH
3 groups. The surface wettability of the spherical ATP/SiO
2 aerogels was evaluated by contact angle measurements, which are presented in
Figure 3. Due to the large number of –OH, the unmodified AS-S exhibited obvious hydrophilicity with the water contact angle values of 0°. Due to the existence of numerous -CH
3 groups, the spherical ATP/SiO
2 aerogels obtained by MTMS modification possessed excellent hydrophobic performance, corresponding to the values of 142.8°, 136.1°, and 140.7° for HAS-S, HAS-F, and HAS-A, respectively.
Figure 4 shows the SEM and TEM images of the prepared spherical ATP/SiO
2 aerogels and pure ATP. As shown in
Figure 4e, the pure ATP displays the features of a club-shaped structure with a diameter of approximately 30 nm and partial agglomeration. As shown in
Figure 4a–d, all of the ATP/SiO
2 aerogels exhibited inherent three-dimensional porous network structures, which consisted of club-shaped ATP, SiO
2 nanoparticles, and nanopores. As seen from the magnified SEM images, the unordered nano-scaled ATP did not influence the formation of the porous structure of the ATP/SiO
2 aerogels. Additionally, there were some large pores in the HAS-F and densification phenomenon in the HAS-A, which is detrimental to the specific surface areas. By contrast, the AS-S and HAS-S samples exhibited significant homogeneous pore structures. Furthermore, the TEM images (
Figure 4f) show that the as-prepared ATP/SiO
2 aerogels using different drying methods displayed random interconnected networks of nanometer-sized SiO
2 aerogel particles and club-shaped ATP, with interconnected amorphous silica particles surrounding the ATP. Meanwhile, the ATP could form hydrogen bonds with silica particles during the reaction, which could strengthen the crosslinked structures of the ATP/SiO
2 aerogels and resist the greater capillary pressure during the drying process. Satisfactorily, compared to other aerogel composites strengthened by coarse fibers, the nanoscale club-shaped ATP was conducive to improving the mechanical performance of the prepared spherical ATP/SiO
2 aerogels without destroying the internal pore structures [
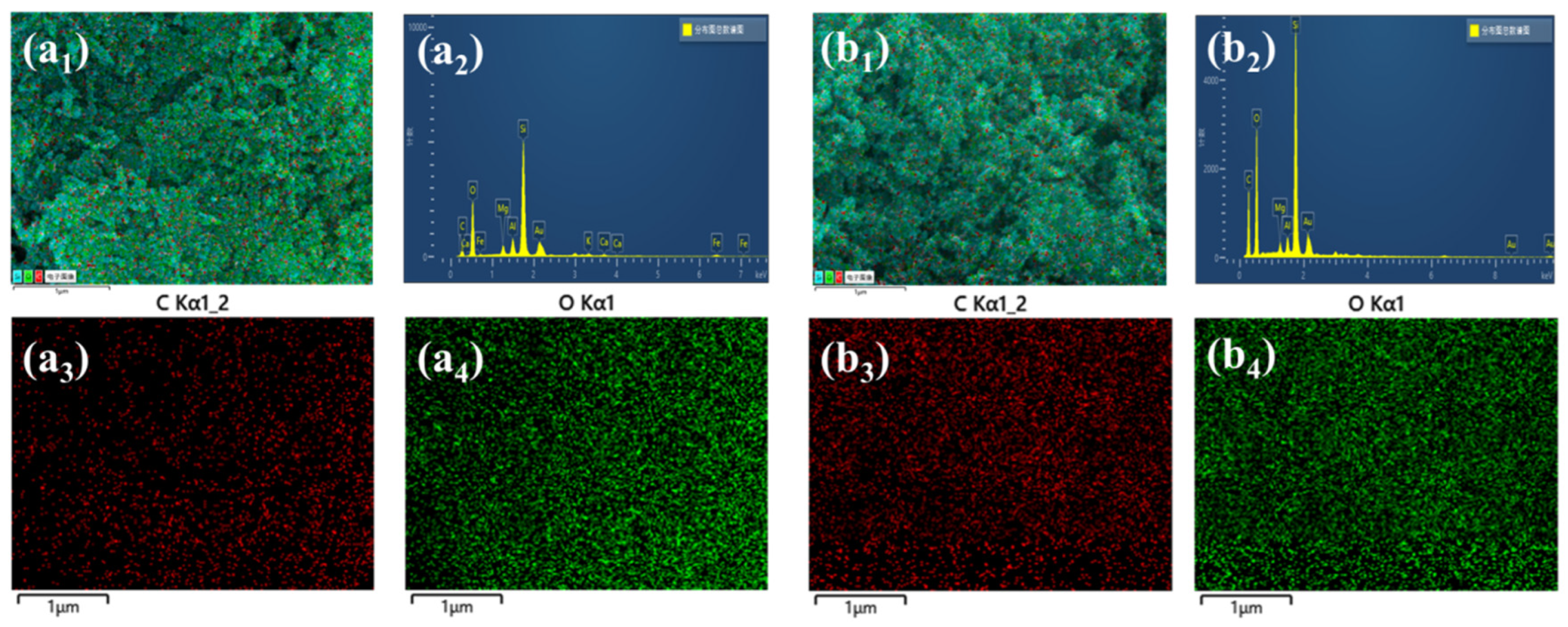
19]. The EDS spectrums of AS-S and HAS-S are presented in
Figure 5, and the concentration of C, O, and Si elements are presented in
Table 2. The apparent concentration of the C element increased from 23.8 wt% (AS-S) to 40.1 wt% (HAS-S). In addition, the homogenous distribution of the C element of HAS-S further proves the successful modification of MTMS.
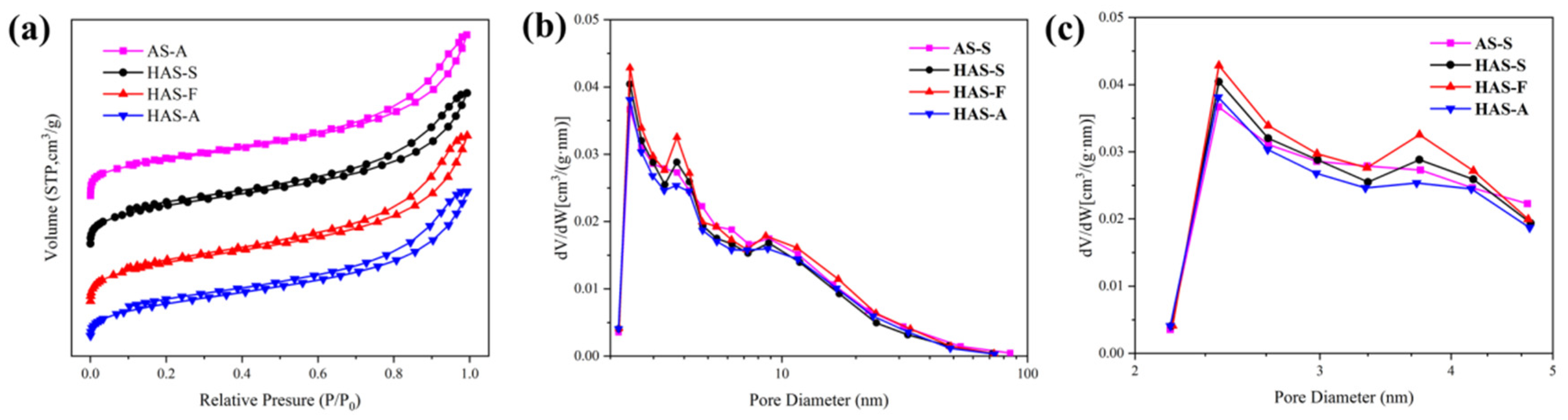
The N
2 adsorption–desorption isotherms and BJH pore size distribution of the spherical ATP/SiO
2 aerogels are described in
Figure 6. All isotherms are Type IV based on the IUPAC classification, reflecting the characteristic of mesoporous materials. The adsorption–desorption curves form closed hysteresis loop, which is generally caused by the capillary condensation in the mesopores. The rapid adsorption process in the low-pressure region (0–0.1) is caused by the micropores inside the aerogel matrix. It is shown in
Table 1 that the specific surface area of the as-prepared ATP/SiO
2 aerogels (with the content of ATP over 60 wt%) was much higher than that of pure ATP, resulting from the improvement in the network skeleton structures of the ATP/SiO
2 aerogel composites. Additionally, the specific surface areas of the aerogels obtained by different treatment techniques showed a decreasing trend (S
SCD > S
FD > S
APD), corresponding to the values of 248.7–267.4 m
2/g, 241.7 m
2/g, and 218.5 m
2/g, respectively. The HAS-A showed the minimum specific surface area because of the particle agglomeration and closed pores appearing during the APD process (shown in
Figure 4d). The HAS-S had higher specific surface areas than AS-S, which can be explained by the fact that the grafting of MTMS improves the hydrophobic property of ATP/SiO
2 aerogel without damaging the nano-porous microstructure. Comparing AS-S to HAS-S, the average pore size and pore volume decreased from 8.3 nm and 0.41 cm
3/g to 7.9 nm and 0.37 cm
3/g, respectively. This is because some organic groups enter the larger pores of spherical ATP/SiO
2 aerogels with the grafting of the CH
3-(SiOH)
3 group, which is favorable to increasing the porosity of HAS-S [
41]. Furthermore, owing to the high capillary pressure of the solvent evaporation and shrinkage of the pore structure in the APD process, the pore volume of the HAS-A decreased significantly in comparison with the other hydrophobic samples. Compared with HAS-S, HAS-F exhibited lower specific surface area and higher pore volume due to the destruction of the original pore structure during the crystallization and sublimation of the solvent. As revealed in
Figure 6b, all spherical ATP/SiO
2 aerogels showed a broad pore size distribution in the range of 2–50 nm. After the hydrophobic modification, the peak of the micropores slightly shifted to higher values (
Figure 6c), which can be explained by the fact that the pores in the aerogel are filled with the hydrolysis products of MTMS [
41]. This indicates that the hydrophobic modification was beneficial to the microporous structure of the aerogels. Therefore, all spherical ATP/SiO
2 aerogels had a good specific surface area and porous network structure, revealing the feasibility of the multipath preparation via the SCD, FD, and APD techniques.
The TG curves of the prepared spherical ATP/SiO
2 aerogels are displayed in
Figure 7, under flowing air. The weight loss rates of the AS-S, HAS-S, HAS-F, and HAS-A were 14.62%, 15.72%, 12.85%, and 13.67%, respectively. The weight loss stage was mainly divided into three parts. The first stage, consisting of the volatilization of the residual solvent and the adsorbed water, appeared at 50–200 °C. In this stage, the weight loss rate of the hydrophobic samples was lower than AS-S (4.51%), which reflects the hydrophobicity of the MTMS modification. Considering the residual solvent, water molecules, and CO
2 during the SCD process, the weight loss rates of AS-S (4.51%) and HAS-S (3.87%) were significantly higher than those of HAS-F (2.59%) and HAS-A (2.17%). In the second stage between 200 °C and 500 °C, the weight loss was assigned to the elimination of the crystal water in the ATP and the condensation of Si-OH. The weight loss in the third stage (over 500 °C) was mainly attributed to the oxidative decomposition of Si-CH
3 and the removal of structural water in the ATP [
27]. It is noted that a characteristic temperature point appeared in the hydrophobic sample at approximately 630 °C, which implies the complete decomposition of the Si-CH
3 group [
42].
3.3. Effect of Heat Treatment on Physical Characteristic and Morphological Properties of HAS-A
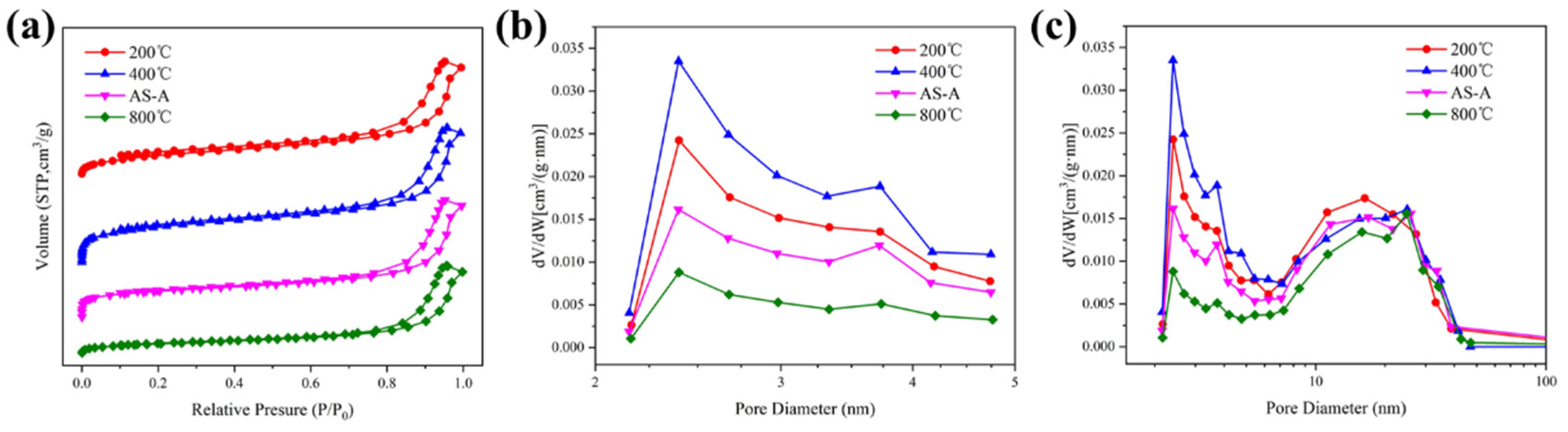
The influence of the heat treatment temperatures on the morphology, microstructure, and pore structure is shown in
Figure 8 and
Figure 9. Heat treatment is a simple and efficient way to modify the crosslinking structure and chemistry of aerogels [
43]. The 600 °C heat-treated HAS-A is denoted as AS-A. As the calcination temperature increased, the appearance of the color of the HAS-A gradually changed from off-white to brick red (
Figure 8a), which is mainly caused by the component of ATP. It is found from
Figure 8b–e that the HAS-A heat treated at 400 °C showed a highly homogeneous porous structure. Due to the pyrolysis of -CH
3 and condensation among -OH, there was a large amount of silica particles aggregated inside the aerogel, and the uniform pore structure was damaged after calcination at 600 °C. A denser silica structure of the as-prepared HAS-A heat treated at 800 °C was clearly observed. All the curves in
Figure 9a are type IV isotherms, suggesting that mesopores are still present in the aerogel despite the high temperature heat treatment. Consequentially, the pore size distributions of the heat-treated samples are shown in
Figure 9b,c. From 200 °C to 400 °C, the peak of the micropores increased and then decreased after 600 °C. This is because at 200–400 °C, the excess organic matter inside the pores of the HAS-A decomposed, resulting in extra pores. At 600–800 °C, the aerogel particles inside the HAS-A will be aggregated and block the pore channels [
44]. The pore structure of the HAS-A at different calcination temperatures is shown in
Table 3. The specific surface area increased dramatically from 231.5 m
2/g to 337.7 m
2/g, with the treatment temperature increasing from 200 °C to 400 °C. Simultaneously, it was observed that the total pore volume changed from 0.40 cm
3/g to 0.43 cm
3/g, while the average pore size changed from 11.3 nm to 10.6 nm. However, owing to the oxidation of -CH
3 groups and condensation between -OH, a further increase in the calcination temperature was not favorable for the specific surface area and total pore volume.
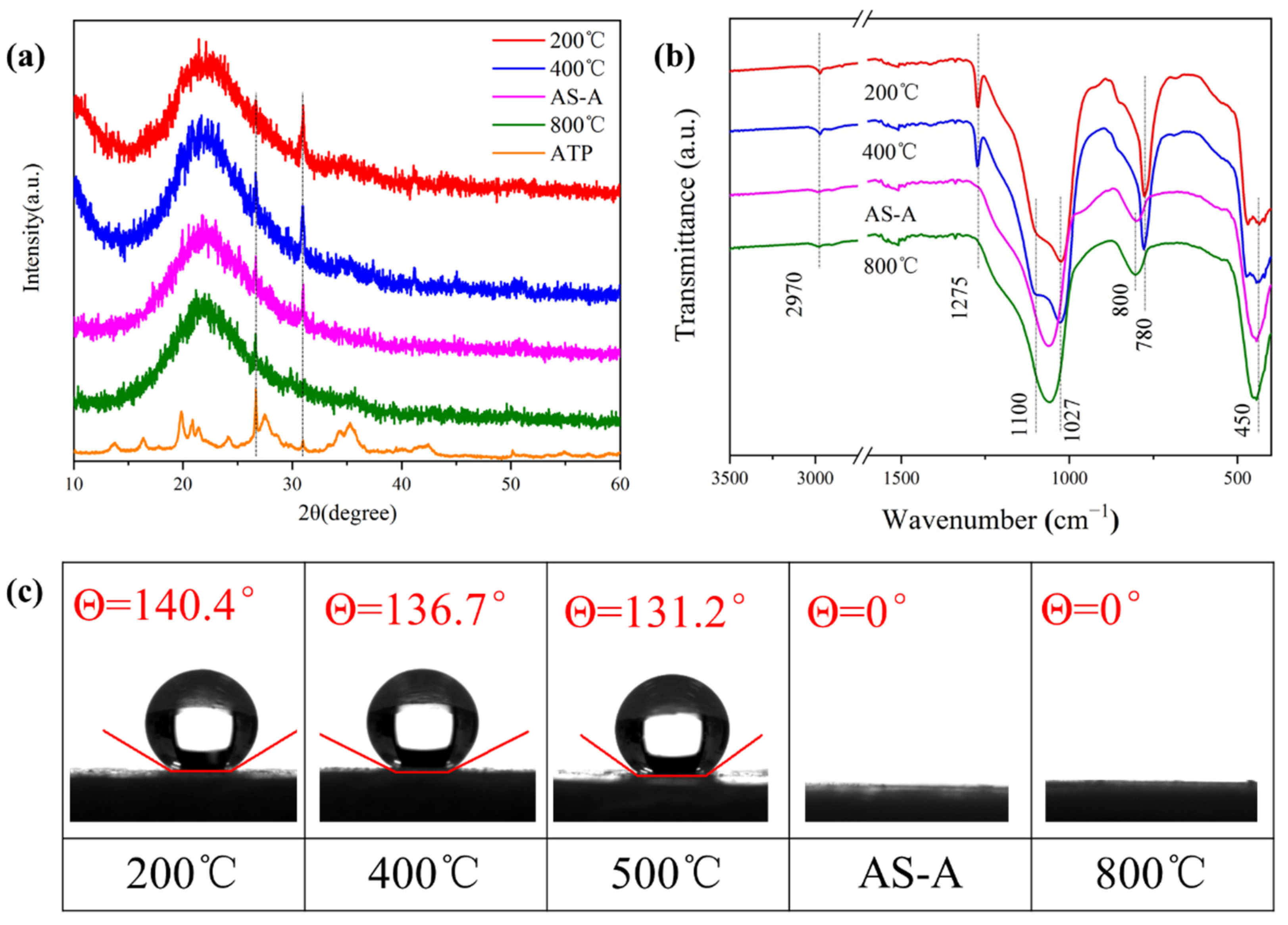
Figure 10a shows the XRD pattern of the heat-treated HAS-A. The characteristic peaks at 2θ = 27.6° and 30.9° were assigned to the (240) and (400) crystal planes of the ATP [
27]. After a 600 °C calcination, the characteristic peak at 30.9° decreased significantly, indicating that the crystal structure of the ATP had been disrupted. After a 800 °C calcination, the characteristic peak at 30.9° gradually disappeared, indicating the complete destruction of the ATP crystal structure. A broad peak at 2θ = 21.5° was clearly identified in all samples, indicating that the silica aerogels preserve their original amorphous structure despite high temperature calcination [
44]. The FT-IR spectra of the HAS-A heat treated at different temperatures are depicted in
Figure 10b. The -CH
3 groups are observed at the absorption peaks of 780 cm
−1, 1275 cm
−1, and 2970 cm
−1. Those bands of the −CH
3 groups disappeared after a 600 °C calcination, indicating the pyrolysis of -CH
3 [
42]. Moreover,
Figure 10c shows the water contact angles of the HAS-A treated at different temperatures. The hydrophobicity of the HAS-A remained stable at 500 °C (200 °C, 400 °C, and 500 °C, corresponding to the values of 140.4°, 136.7°, and 131.2°, respectively), while the HAS-A converted to hydrophilic AS-A completely at 600 °C, with a water contact angle value of 0°, which is in accordance with the representation results in the TG (
Figure 7) and FT-IR (
Figure 10b). The transition from hydrophobic to hydrophilic in the spherical ATP/SiO
2 aerogels is conducive to the adsorption of soluble dyes in aqueous solutions. Therefore, the AS-A were used as adsorbents for studying the adsorption performance.