Enhanced Desulfurization by Tannin Extract Absorption Assisted by Binuclear Sulfonated Phthalocyanine Cobalt Polymer: Performance and Mechanism
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Chemicals
2.2. Batch Absorption Experiments
2.3. Characterization Methods
3. Results and Discussion
3.1. Optimization of Reaction Parameters
3.2. Effect of Total Alkalinity and Na2CO3 Content
3.3. Effect of Content of TE
3.4. Effect of Content of NaVO3
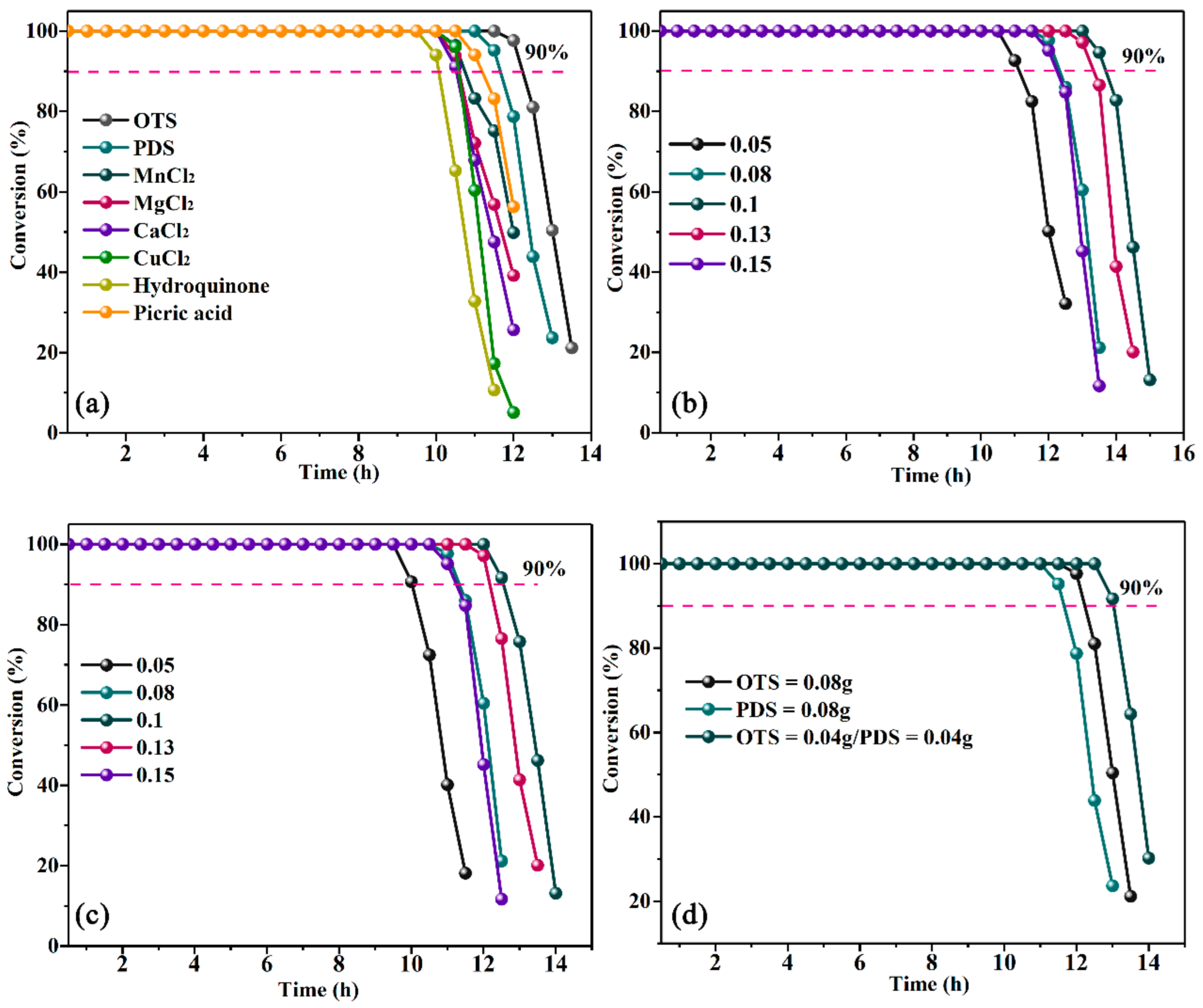
3.5. TE Technology Combined with OTS and PDS
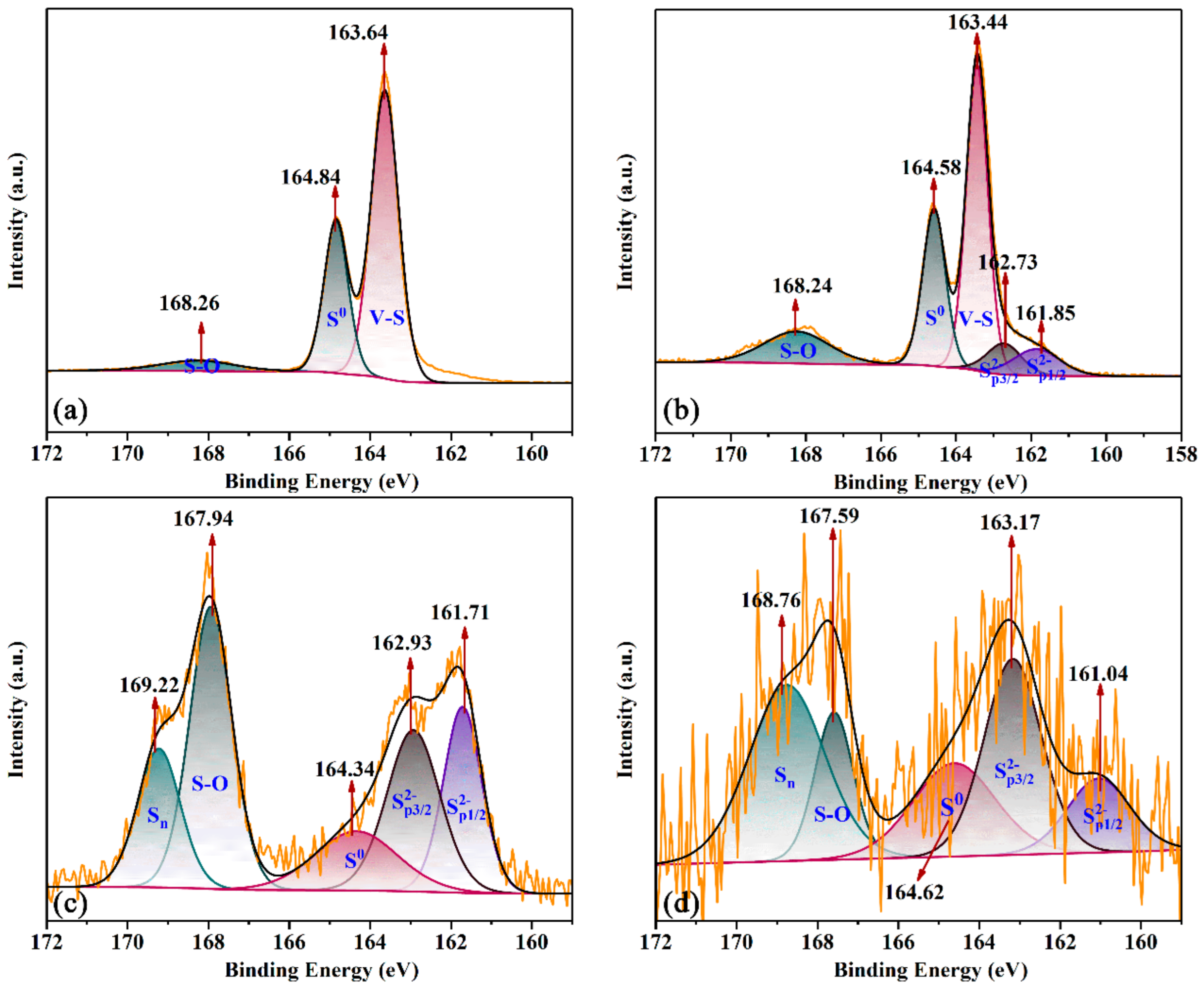
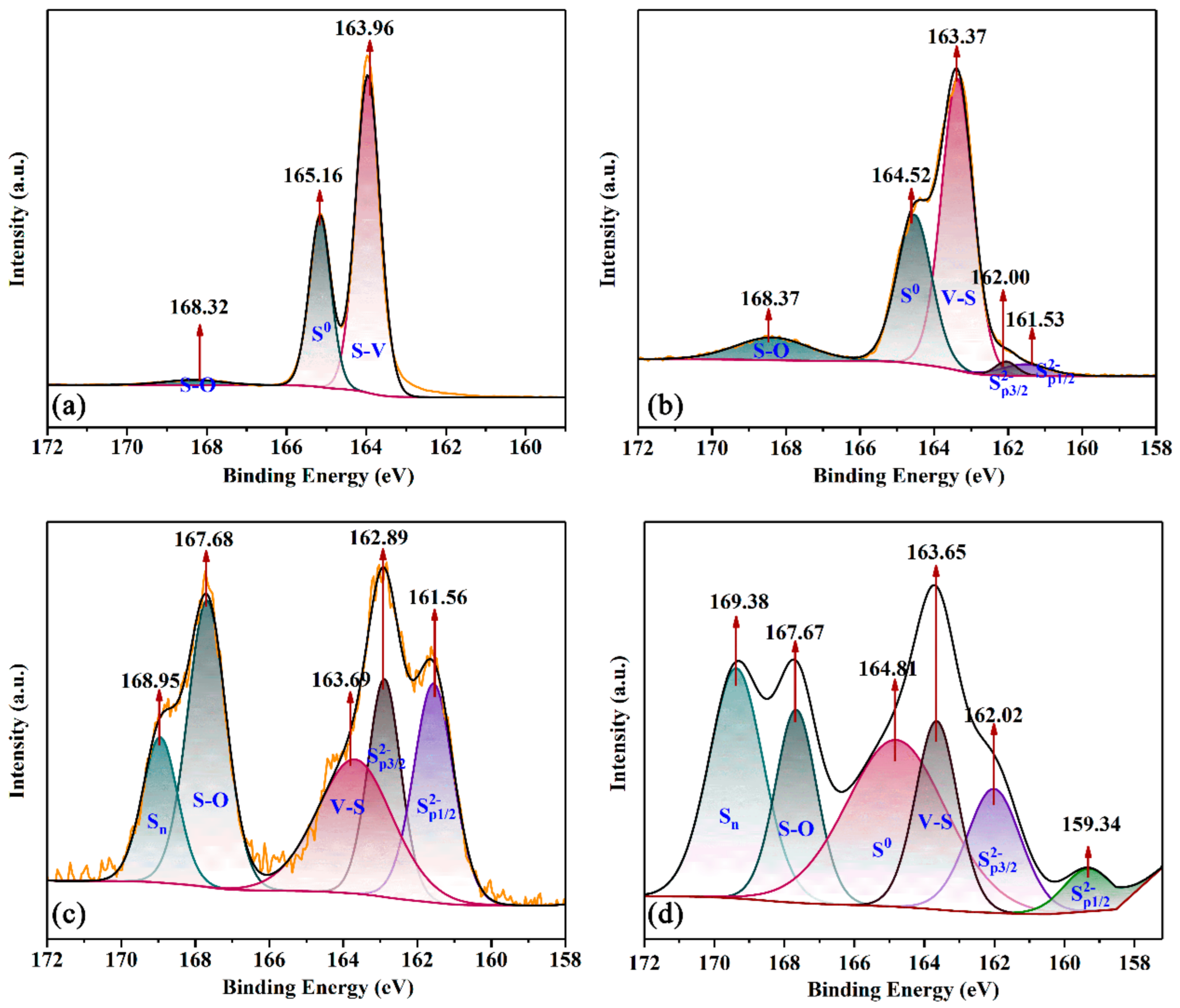
3.6. Insights into the Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dong, Q.; Wang, C.; Peng, S. A Many-Objective Optimization for an Eco-Efficient Flue Gas Desulfurization Process Using a Surrogate-Assisted Evolutionary Algorithm. Sustainability 2021, 13, 9015. [Google Scholar] [CrossRef]
- Razzaq, R.; Li, C.; Zhang, S. Coke oven gas: Availability, properties, purification, and utilization in China. Fuel 2013, 113, 287–299. [Google Scholar] [CrossRef]
- Isobaev, M.D.; Mingboev, S.A.; Davlatnazarova, M.D. Dynamics of Coke-Oven Gas Formation from Brown Coal under Changes in Process Conditions. Solid Fuel Chem. 2019, 53, 248–251. [Google Scholar] [CrossRef]
- Qiao, J. Technology development and industry application progress of desulfurization and denitrification from coke oven flue gas. Nat. Gas Chem. Ind. 2020, 45, 130–134. [Google Scholar]
- Zhizhe, Y. Hydrogen production from coke oven gas and development. Pet. Refin. Eng. 2014, 44, 17–19. [Google Scholar]
- Wei, F.; Zhang, X.; Liao, J. Desulfurization mechanism of an excellent Cu/ZnO sorbent for ultra-deep removal of thiophene in simulated coke oven gas. Chem. Eng. J. 2022, 44, 2–14. [Google Scholar] [CrossRef]
- Donatti, A.F.; Soriano, R.N.; Sabino, J.P.; Branco, L.G.S. Involvement of endogenous hydrogen sulfide (H2S) in the rostral ventrolateral medulla (RVLM) in hypoxia-induced hypothermia. Brain Res. Bull. 2014, 108, 94–99. [Google Scholar] [CrossRef]
- Ndou, M.; Matjie, R.; Mahlamvana, F.; Neomagus, H.; Bunt, J.; Mphahlele, K. Effect of the V5+ to V4+ Molar Ratio on H2S Absorption and Conversion to Elemental Sulfur. Ind. Eng. Chem. Res. 2021, 60, 1505–1516. [Google Scholar] [CrossRef]
- Gao, Z.Q.; Niu, Y.X.; Ling, K.C.; Shen, J. A Study on the Reaction Between HS- and V(V) in Tannin Extract Desulfurization Technology. Energy Sources Part A Recovery Util. Environ. Eff. 2011, 33, 1661–1668. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, X.; Liu, Z.; Yang, X.; Yang, X.J. Recovery of high purity elemental sulfur from coal syngas by liquid redox catalytic process using tannin extracts. J. Clean. Prod. 2017, 142, 3204–3211. [Google Scholar] [CrossRef]
- Jiao, W.; Yang, P.; Qi, G.; Liu, Y. Selective absorption of H2S with High CO2 concentration in mixture in a rotating packed bed. Chem. Eng. Process. -Process Intensif. 2018, 129, 142–147. [Google Scholar] [CrossRef]
- Gao, S.; Li, J. A combination desulfurization method for diesel fuel: Oxidation by ionic liquid with extraction by solvent. Fuel 2018, 224, 545–551. [Google Scholar] [CrossRef]
- Zhang, X.; Dou, G.; Wang, Z. Selective oxidation of H2S over V2O5 supported on CeO2-intercalated Laponite clay catalysts. Catal. Sci. Technol. 2013, 3, 2778. [Google Scholar] [CrossRef]
- Kelsall, G.H.; Thompson, I.; Francis, P.A. Redox chemistry of H2S oxidation by the British Gas Stretford process part IV: V-S-H2O thermodynamics and aqueous vanadium (v) reduction in alkaline solutions. J. Appl. Electrochem. 1993, 23, 417–426. [Google Scholar] [CrossRef]
- Rozan, T.F.; Theberge, S.M.; Luther, G. Quantifying elemental sulfur (S0), bisulfide (HS−) and polysulfides (Sx2−) using a voltammetric method. Anal. Chim. Acta 2000, 415, 175–184. [Google Scholar] [CrossRef]
- Niu, Y.; Ling, K.C.; Sheng, Q.T.; Zhang, W.S.; Shen, J. Thermodynamic Analysis on the Formation of Sulfur-Containing Byproducts in Tannin Extract Desulfurization Technology. Energy Sources Part A Recovery Util. Environ. Eff. 2013, 35, 880–890. [Google Scholar] [CrossRef]
- Yin, M.; Yun, Z.; Fan, F. Insights into the mechanism of low-temperature H2S oxidation over Zn–Cu/Al2O3 catalyst. Chemosphere 2022, 291, 133105. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Wu, H.; Ji, B. Application of Desulfurizing Agent OTS in Tannin Extract Gas Desulphurization Process. New Technol. New Process 2008, 1, 81–83. [Google Scholar]
- Li, J.; Zhang, S.; Jin, Y. Primary study on digestive methane desulfurization (H2S) of sewage treatment plants. Environ. Eng. 2006, 24, 43–46. [Google Scholar]
- Zhang, Z.; Wang, J.; Li, W.; Wang, M.; Qiao, W.; Long, D.; Ling, L. Millimeter-sized mesoporous carbon spheres for highly efficient catalytic oxidation of hydrogen sulfide at room temperature. Carbon 2016, 96, 608–615. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Fan, H. Highly porous copper oxide sorbent for H2S capture at ambient temperature. Fuel 2017, 209, 329–338. [Google Scholar] [CrossRef]
- Bukhtiyarova, G.A.; Bukhtiyarov, V.I.; Sakaeva, N.S. XPS study of the silica-supported Fe-containing catalysts for deep or partial H2S oxidation. J. Mol. Catal. A Chem. 2000, 158, 251–255. [Google Scholar] [CrossRef]
- Falco, G.D.; Montagnaro, F.; Balsamo, M. Synergic effect of Zn and Cu oxides dispersed on activated carbon during reactive adsorption of H2S at room temperature. Microporous Mesoporous Mater. 2018, 257, 135–146. [Google Scholar] [CrossRef]
- Feng, L.L.; Li, G.D.; Liu, Y. Carbon-Armored Co9S8 Nanoparticles as All-pH Efficient and Durable H2-Evolving Electrocatalysts. ACS Appl. Mater. Interfaces 2015, 7, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Huang, J.; Feng, L. 3D self-assembled VS4 microspheres with high pseudocapacitance as highly efficient anodes for Na-ion batteries. Nanoscale 2018, 10, 21671–21680. [Google Scholar] [CrossRef]
- Dan, Y.; Cao, L.; Huang, J.; Koji, K. Generation of Ni3S2 nanorod arrays with high-density bridging S22− by introducing a small amount of Na3VO4·12H2O for superior hydrogen evolution reaction. Nanoscale 2020, 12, 2063–2070. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Chen, H.; Hao, X.; Li, K.; Sun, X.; Li, Y.; Ning, P. Enhanced Desulfurization by Tannin Extract Absorption Assisted by Binuclear Sulfonated Phthalocyanine Cobalt Polymer: Performance and Mechanism. Materials 2023, 16, 2343. https://doi.org/10.3390/ma16062343
Wang B, Chen H, Hao X, Li K, Sun X, Li Y, Ning P. Enhanced Desulfurization by Tannin Extract Absorption Assisted by Binuclear Sulfonated Phthalocyanine Cobalt Polymer: Performance and Mechanism. Materials. 2023; 16(6):2343. https://doi.org/10.3390/ma16062343
Chicago/Turabian StyleWang, Bing, Huanyu Chen, Xingguang Hao, Kai Li, Xin Sun, Yuan Li, and Ping Ning. 2023. "Enhanced Desulfurization by Tannin Extract Absorption Assisted by Binuclear Sulfonated Phthalocyanine Cobalt Polymer: Performance and Mechanism" Materials 16, no. 6: 2343. https://doi.org/10.3390/ma16062343
APA StyleWang, B., Chen, H., Hao, X., Li, K., Sun, X., Li, Y., & Ning, P. (2023). Enhanced Desulfurization by Tannin Extract Absorption Assisted by Binuclear Sulfonated Phthalocyanine Cobalt Polymer: Performance and Mechanism. Materials, 16(6), 2343. https://doi.org/10.3390/ma16062343




