Recent Advances in Decellularized Extracellular Matrix-Based Bioinks for 3D Bioprinting in Tissue Engineering
Abstract
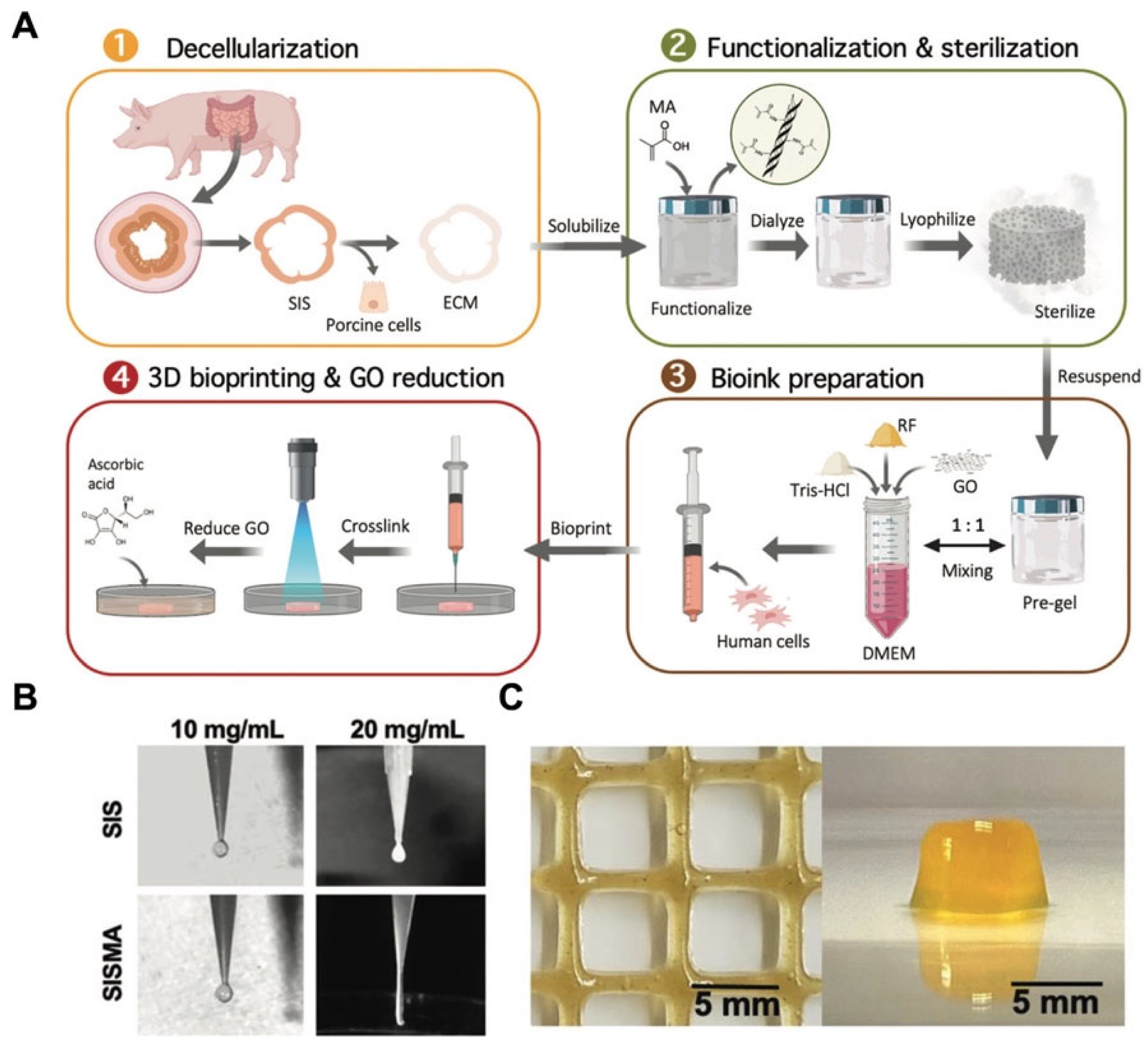
:1. Introduction
2. Bioprinting Technology of Decellularized Extracellular Matrix
2.1. Inkjet-Based Bioprinting
2.2. Laser-Assisted Bioprinting
2.3. Stereolithography-Based Bioprinting
2.4. Extrusion-Based Bioprinting
3. Bioactive Molecules in the Decellularized Extracellular Matrix
4. Construction Methods of Decellularized Extracellular Matrix
4.1. Chemical Methods
4.2. Physical Methods
4.3. Biological Methods
5. The Construction and Modification of Decellularized Extracellular Matrix-Based Bioinks
6. The Applications of Decellularized Extracellular Matrix-Based Bioink for Bioprinting in Tissue Engineering
6.1. Hearts
6.2. Cartilage
6.3. Adipose Tissue
6.4. Skeletal Muscle
6.5. Liver
6.6. Skin
6.7. Cornea
6.8. Brain
6.9. Pancreas
6.10. Trachea
6.11. Blood Vessels
6.12. Tendon
7. Current Challenges and Further Perspectives
7.1. Optimizing the Fabrication Procedure of dECM-Derived Bioinks
7.2. Mechanical Properties
7.3. Long-Term Biosafety of dECM-Derived Bioinks after Implantation
7.4. Drug Discovery and Development
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McMillan, A.; McMillan, N.; Gupta, N.; Kanotra, S.P.; Salem, A.K. 3D Bioprinting in Otolaryngology: A Review. Adv. Healthc. Mater. 2023, 2203268, early view. [Google Scholar] [CrossRef]
- Loukelis, K.; Helal, Z.A.; Mikos, A.G.; Chatzinikolaidou, M. Nanocomposite Bioprinting for Tissue Engineering Applications. Gels 2023, 9, 103. [Google Scholar] [CrossRef] [PubMed]
- Altunbek, M.; Afghah, F.; Caliskan, O.S.; Yoo, J.J.; Koc, B. Design and bioprinting for tissue interfaces. Biofabrication 2023, 15, 022002. [Google Scholar] [CrossRef] [PubMed]
- Sztankovics, D.; Moldvai, D.; Petővári, G.; Gelencsér, R.; Krencz, I.; Raffay, R.; Dankó, T.; Sebestyén, A. 3D bioprinting and the revolution in experimental cancer model systems—A review of developing new models and experiences with in vitro 3D bioprinted breast cancer tissue-mimetic structures. Pathol. Oncol. Res. 2023, 29, 1610996. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, M.; Ma, H.; Chapa-Villarreal, F.A.; Lobo, A.O.; Zhang, Y.S. Stereolithography apparatus and digital light processing-based 3D bioprinting for tissue fabrication. iScience 2023, 26, 106039. [Google Scholar] [CrossRef]
- Raees, S.; Ullah, F.; Javed, F.; Akil, H.M.; Jadoon Khan, M.; Safdar, M.; Din, I.U.; Alotaibi, M.A.; Alharthi, A.I.; Bakht, M.A.; et al. Classification, processing, and applications of bioink and 3D bioprinting: A detailed review. Int. J. Biol. Macromol. 2023, 232, 123476. [Google Scholar] [CrossRef]
- Kang, H.-W.; Lee, S.J.; Ko, I.K.; Kengla, C.; Yoo, J.J.; Atala, A. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat. Biotechnol. 2016, 34, 312–319. [Google Scholar] [CrossRef]
- Wang, Z.; Xiang, L.; Lin, F.; Tang, Y.; Cui, W. 3D bioprinting of emulating homeostasis regulation for regenerative medicine applications. J. Control. Release 2023, 353, 147–165. [Google Scholar] [CrossRef]
- Jo, Y.; Hwang, D.G.; Kim, M.; Yong, U.; Jang, J. Bioprinting-assisted tissue assembly to generate organ substitutes at scale. Trends Biotechnol. 2023, 41, 93–105. [Google Scholar] [CrossRef]
- Kong, Z.; Wang, X. Bioprinting Technologies and Bioinks for Vascular Model Establishment. Int. J. Mol. Sci. 2023, 24, 891. [Google Scholar] [CrossRef]
- Xing, F.; Yin, H.-M.; Zhe, M.; Xie, J.-C.; Duan, X.; Xu, J.-Z.; Xiang, Z.; Li, Z.-M. Nanotopographical 3D-Printed Poly(ε-caprolactone) Scaffolds Enhance Proliferation and Osteogenic Differentiation of Urine-Derived Stem Cells for Bone Regeneration. Pharmaceutics 2022, 14, 1437. [Google Scholar] [CrossRef] [PubMed]
- Mandrycky, C.; Wang, Z.; Kim, K.; Kim, D.H. 3D bioprinting for engineering complex tissues. Biotechnol. Adv. 2016, 34, 422–434. [Google Scholar] [CrossRef] [PubMed]
- Neng, X.; Guohong, S.; Yuling, S.; Yuanjing, X.; Hao, W.; Haiyang, F.; Kerong, D.; Jinwu, W.; Qixin, C. Research Progress of Robot Technology in In situ 3D Bioprinting. Int. J. Bioprint. 2022, 8, 614. [Google Scholar] [CrossRef]
- Mancilla-De-la-Cruz, J.; Rodriguez-Salvador, M.; An, J.; Chua, C.K. Three-Dimensional Printing Technologies for Drug Delivery Applications: Processes, Materials, and Effects. Int. J. Bioprint. 2022, 8, 622. [Google Scholar] [CrossRef] [PubMed]
- Kriegel, A.; Schlosser, C.; Habeck, T.; Dahmen, C.; Götz, H.; Clauder, F.; Armbruster, F.P.; Baranowski, A.; Drees, P.; Rommens, P.M.; et al. Bone Sialoprotein Immobilized in Collagen Type I Enhances Bone Regeneration In vitro and In vivo. Int. J. Bioprint. 2022, 8, 591. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xia, X.; Liu, Z.; Dong, M. The Next Frontier of 3D Bioprinting: Bioactive Materials Functionalized by Bacteria. Small 2023, 19, 2205949. [Google Scholar] [CrossRef] [PubMed]
- Daly, A.C.; Lim, K.S. High resolution lithography 3D bioprinting. Trends Biotechnol. 2023, 41, 262–263. [Google Scholar] [CrossRef]
- Nam, S.Y.; Park, S.-H. ECM Based Bioink for Tissue Mimetic 3D Bioprinting. In Biomimetic Medical Materials: From Nanotechnology to 3D Bioprinting; Noh, I., Ed.; Springer: Singapore, 2018; pp. 335–353. [Google Scholar]
- Hospodiuk, M.; Dey, M.; Sosnoski, D.; Ozbolat, I.T. The bioink: A comprehensive review on bioprintable materials. Biotechnol. Adv. 2017, 35, 217–239. [Google Scholar] [CrossRef]
- Kort-Mascort, J.; Flores-Torres, S.; Peza-Chavez, O.; Jang, J.H.; Pardo, L.A.; Tran, S.D.; Kinsella, J. Decellularized ECM hydrogels: Prior use considerations, applications, and opportunities in tissue engineering and biofabrication. Biomater. Sci. 2023, 11, 400–431. [Google Scholar] [CrossRef]
- Mokhtarinia, K.; Masaeli, E. Post-decellularized printing of cartilage extracellular matrix: Distinction between biomaterial ink and bioink. Biomater. Sci. 2023, 11, 2317–2329. [Google Scholar] [CrossRef]
- Kushige, H.; Amano, Y.; Yagi, H.; Morisaku, T.; Kojima, H.; Satou, A.; Hamada, K.-i.; Kitagawa, Y. Injectable extracellular matrix hydrogels contribute to native cell infiltration in a rat partial nephrectomy model. J. Biomed. Mater. Res. Part B Appl. Biomater. 2023, 111, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Du, L.; Zhang, T.; Chen, J.; Xu, B. Research progress in decellularized extracellular matrix hydrogels for intervertebral disc degeneration. Biomater. Sci. 2023, 11, 1981–1993. [Google Scholar] [CrossRef]
- Shanto, P.C.; Park, S.; Park, M.; Lee, B.-T. Physico-biological evaluation of 3D printed dECM/TOCN/alginate hydrogel based scaffolds for cartilage tissue regeneration. Biomater. Adv. 2023, 145, 213239. [Google Scholar] [CrossRef]
- Kim, B.S.; Das, S.; Jang, J.; Cho, D.W. Decellularized Extracellular Matrix-based Bioinks for Engineering Tissue- and Organ-specific Microenvironments. Chem. Rev. 2020, 120, 10608–10661. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Xu, J.; Meng, L.; Su, Y.; Fang, H.; Liu, J.; Cheng, Y.Y.; Jiang, D.; Nie, Y.; Song, K. 3D bioprinting of dECM/Gel/QCS/nHAp hybrid scaffolds laden with mesenchymal stem cell-derived exosomes to improve angiogenesis and osteogenesis. Biofabrication 2023, 15, 024103. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Ma, Y.; Wang, X.; Yuan, S.; Huo, F.; Yi, G.; Zhang, J.; Yang, B.; Tian, W. A 3D-Bioprinted Functional Module Based on Decellularized Extracellular Matrix Bioink for Periodontal Regeneration. Adv. Sci. 2023, 10, 2205041. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.A.; Sampaio, L.C.; Ferdous, Z.; Gobin, A.S.; Taite, L.J. Decellularized matrices in regenerative medicine. Acta Biomater. 2018, 74, 74–89. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.-Y.; Huang, J.-J.; Liu, Y.; Chen, C.-W.; Qu, G.-W.; Wang, G.-F.; Zhao, Y.; Wu, X.-W.; Ren, J.-A. Extracellular matrix bioink boosts stemness and facilitates transplantation of intestinal organoids as a biosafe Matrigel alternative. Bioeng. Transl. Med. 2023, 8, e10327. [Google Scholar] [CrossRef]
- Jang, J.; Park, H.J.; Kim, S.W.; Kim, H.; Park, J.Y.; Na, S.J.; Kim, H.J.; Park, M.N.; Choi, S.H.; Park, S.H.; et al. 3D printed complex tissue construct using stem cell-laden decellularized extracellular matrix bioinks for cardiac repair. Biomaterials 2017, 112, 264–274. [Google Scholar] [CrossRef]
- Gao, G.; Lee, J.H.; Jang, J.; Lee, D.H.; Kong, J.-S.; Kim, B.S.; Choi, Y.-J.; Jang, W.B.; Hong, Y.J.; Kwon, S.-M.; et al. Tissue Engineered Bio-Blood-Vessels Constructed Using a Tissue-Specific Bioink and 3D Coaxial Cell Printing Technique: A Novel Therapy for Ischemic Disease. Adv. Funct. Mater. 2017, 27, 1700798. [Google Scholar] [CrossRef]
- Choudhury, D.; Tun, H.W.; Wang, T.; Naing, M.W. Organ-Derived Decellularized Extracellular Matrix: A Game Changer for Bioink Manufacturing? Trends Biotechnol. 2018, 36, 787–805. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.A.; Liu, W.; Jimenez, A.; Yang, J.; Akpek, A.; Liu, X.; Pi, Q.; Mu, X.; Hu, N.; Schiffelers, R.M.; et al. 3D Bioprinting: From Benches to Translational Applications. Small 2019, 15, e1805510. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Su, J.; Zhou, S.; Li, J.; Zhang, K. Application of Hydrogels as Three-Dimensional Bioprinting Ink for Tissue Engineering. Gels 2023, 9, 88. [Google Scholar] [CrossRef]
- Samadi, A.; Moammeri, A.; Pourmadadi, M.; Abbasi, P.; Hosseinpour, Z.; Farokh, A.; Shamsabadipour, A.; Heydari, M.; Mohammadi, M.R. Cell Encapsulation and 3D Bioprinting for Therapeutic Cell Transplantation. ACS Biomater. Sci. Eng. 2023, 9, 1862–1890. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, B.; Pei, B.; Chen, J.; Zhou, D.; Peng, J.; Zhang, X.; Jia, W.; Xu, T. Inkjet Bioprinting of Biomaterials. Chem. Rev. 2020, 120, 10793–10833. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Su, H.; Li, M.; Xing, H. Digital light processing-based multi-material bioprinting: Processes, applications, and perspectives. J. Biomed. Mater. Res. Part A 2023, 111, 527–542. [Google Scholar] [CrossRef]
- Xing, F.; Xiang, Z.; Rommens, P.M.; Ritz, U. 3D Bioprinting for Vascularized Tissue-Engineered Bone Fabrication. Materials 2020, 13, 2278. [Google Scholar] [CrossRef]
- Xu, H.; Liu, J.; Shahriar, M.; Xu, C. Investigation of Cell Aggregation on the Printing Performance in Inkjet-Based Bioprinting of Cell-Laden Bioink. Langmuir 2023, 39, 545–555. [Google Scholar] [CrossRef]
- Park, W.; Gao, G.; Cho, D.-W. Tissue-Specific Decellularized Extracellular Matrix Bioinks for Musculoskeletal Tissue Regeneration and Modeling Using 3D Bioprinting Technology. Int. J. Mol. Sci. 2021, 22, 7837. [Google Scholar] [CrossRef]
- Ishack, S.; Khachemoune, A. Future prospects in 3-dimensional (3D) technology and Mohs micrographic surgery. J. Dermatol. Treat. 2022, 33, 2810–2812. [Google Scholar] [CrossRef]
- Gupta, S.; Alrabaiah, H.; Christophe, M.; Rahimi-Gorji, M.; Nadeem, S.; Bit, A. Evaluation of silk-based bioink during pre and post 3D bioprinting: A review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2021, 109, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Angelopoulos, I.; Allenby, M.C.; Lim, M.; Zamorano, M. Engineering inkjet bioprinting processes toward translational therapies. Biotechnol. Bioeng. 2020, 117, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Boix-Lemonche, G.; Nagymihaly, R.M.; Niemi, E.M.; Josifovska, N.; Johansen, S.; Moe, M.C.; Scholz, H.; Petrovski, G. Intra-Corneal Implantation of 3D Bioprinted Scaffolds Containing Mesenchymal Stromal Cells using Femtosecond-Laser-Assisted Intrastromal Keratoplasty. Macromol. Biosci. 2023, 2200422, accepted. [Google Scholar] [CrossRef] [PubMed]
- Yumoto, M.; Hemmi, N.; Sato, N.; Kawashima, Y.; Arikawa, K.; Ide, K.; Hosokawa, M.; Seo, M.; Takeyama, H. Evaluation of the effects of cell-dispensing using an inkjet-based bioprinter on cell integrity by RNA-seq analysis. Sci. Rep. 2020, 10, 7158. [Google Scholar] [CrossRef]
- Yu, J.; Park, S.A.; Kim, W.D.; Ha, T.; Xin, Y.Z.; Lee, J.; Lee, D. Current Advances in 3D Bioprinting Technology and Its Applications for Tissue Engineering. Polymers 2020, 12, 2958. [Google Scholar] [CrossRef]
- Ishack, S.; Lipner, S.R. A Review of 3-Dimensional Skin Bioprinting Techniques: Applications, Approaches, and Trends. Dermatol. Surg. 2020, 46, 1500–1505. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Donderwinkel, I.; Hest, J.; Cameron, N.R.J.P.C. Bio-inks for 3D bioprinting: Recent advances and future prospects. Polym. Chem. 2017, 8, 4451–4471. [Google Scholar] [CrossRef]
- Gu, Z.; Fu, J.; Lin, H.; He, Y. Development of 3D bioprinting: From printing methods to biomedical applications. Asian J. Pharm. Sci. 2020, 15, 529–557. [Google Scholar] [CrossRef]
- Kim, Y.T.; Ahmadianyazdi, A.; Folch, A. A ‘print–pause–print’ protocol for 3D printing microfluidics using multimaterial stereolithography. Nat. Protoc. 2023, 18, 1243–1259. [Google Scholar] [CrossRef]
- Ertugrul, I.; Ulkir, O.; Ersoy, S.; Ragulskis, M. Additive Manufactured Strain Sensor Using Stereolithography Method with Photopolymer Material. Polymers 2023, 15, 991. [Google Scholar] [CrossRef] [PubMed]
- Paruli, E., III; Montagna, V.; García-Soto, M.; Haupt, K.; Gonzato, C. A general photoiniferter approach to the surface functionalization of acrylic and methacrylic structures written by two-photon stereolithography. Nanoscale 2023, 15, 2860–2870. [Google Scholar] [CrossRef]
- Elomaa, L.; Keshi, E.; Sauer, I.M.; Weinhart, M. Development of GelMA/PCL and dECM/PCL resins for 3D printing of acellular in vitro tissue scaffolds by stereolithography. Mater. Sci. Eng. C 2020, 112, 110958. [Google Scholar] [CrossRef] [PubMed]
- Ning, L.; Chen, X. A brief review of extrusion-based tissue scaffold bio-printing. Biotechnol. J. 2017, 12, 1600671. [Google Scholar] [CrossRef]
- Unagolla, J.M.; Jayasuriya, A.C. Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl. Mater. Today 2020, 18, 100479. [Google Scholar] [CrossRef] [PubMed]
- Jongprasitkul, H.; Turunen, S.; Parihar, V.S.; Kellomäki, M. Sequential Cross-linking of Gallic Acid-Functionalized GelMA-Based Bioinks with Enhanced Printability for Extrusion-Based 3D Bioprinting. Biomacromolecules 2023, 24, 502–514. [Google Scholar] [CrossRef]
- Flores-Torres, S.; Jiang, T.; Kort-Mascort, J.; Yang, Y.; Peza-Chavez, O.; Pal, S.; Mainolfi, A.; Pardo, L.A.; Ferri, L.; Bertos, N.; et al. Constructing 3D In Vitro Models of Heterocellular Solid Tumors and Stromal Tissues Using Extrusion-Based Bioprinting. ACS Biomater. Sci. Eng. 2023, 9, 542–561. [Google Scholar] [CrossRef]
- Yang, J.; Li, Z.; Li, S.; Zhang, Q.; Zhou, X.; He, C. Tunable metacrylated silk fibroin-based hybrid bioinks for the bioprinting of tissue engineering scaffolds. Biomater. Sci. 2023, 11, 1895–1909. [Google Scholar] [CrossRef]
- Alarçin, E.; İzbudak, B.; Yüce Erarslan, E.; Domingo, S.; Tutar, R.; Titi, K.; Kocaaga, B.; Guner, F.S.; Bal-Öztürk, A. Optimization of methacrylated gelatin /layered double hydroxides nanocomposite cell-laden hydrogel bioinks with high printability for 3D extrusion bioprinting. J. Biomed. Mater. Res. Part A 2023, 111, 209–223. [Google Scholar] [CrossRef]
- Ghahri, T.; Salehi, Z.; Aghajanpour, S.; Eslaminejad, M.B.; Kalantari, N.; Akrami, M.; Dinarvand, R.; Jang, H.L.; Esfandyari-Manesh, M. Development of osteon-like scaffold-cell construct by quadruple coaxial extrusion-based 3D bioprinting of nanocomposite hydrogel. Biomater. Adv. 2023, 145, 213254. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Hong, H.; Hu, R.; Liu, J.; Liu, C. Decellularized extracellular matrix scaffolds: Recent trends and emerging strategies in tissue engineering. Bioact. Mater. 2022, 10, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Hoshiba, T. Decellularized Extracellular Matrix for Cancer Research. Materials 2019, 12, 1311. [Google Scholar] [CrossRef] [PubMed]
- Dhan Da Yuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S.J.I.J.o.P.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 609–618. [Google Scholar]
- Hubmacher, D.; Apte, S.S. The biology of the extracellular matrix: Novel insights. Curr. Opin. Rheumatol. 2013, 25, 65–70. [Google Scholar] [CrossRef]
- Derakhshanfar, S.; Mbeleck, R.; Xu, K.; Zhang, X.; Zhong, W.; Xing, M. 3D bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact. Mater. 2018, 3, 144–156. [Google Scholar] [CrossRef]
- Heran, W.; Xin, L.; Qi, G.; Xiongfei, Z. Vascularized organ bioprinting: From strategy to paradigm. Cell Prolif. 2023, e13453, early view. [Google Scholar] [CrossRef]
- Dzobo, K.; Motaung, K.; Adesida, A. Recent Trends in Decellularized Extracellular Matrix Bioinks for 3D Printing: An Updated Review. Int. J. Mol. Sci. 2019, 20, 4628. [Google Scholar] [CrossRef]
- Wang, X.; Ansari, A.; Pierre, V.; Young, K.; Kothapalli, C.R.; von Recum, H.A.; Senyo, S.E. Injectable Extracellular Matrix Microparticles Promote Heart Regeneration in Mice with Post-ischemic Heart Injury. Adv. Healthc. Mater. 2022, 11, 2102265. [Google Scholar] [CrossRef]
- Behre, A.; Tashman, J.W.; Dikyol, C.; Shiwarski, D.J.; Crum, R.J.; Johnson, S.A.; Kommeri, R.; Hussey, G.S.; Badylak, S.F.; Feinberg, A.W. 3D Bioprinted Patient-Specific Extracellular Matrix Scaffolds for Soft Tissue Defects. Adv. Healthc. Mater. 2022, 11, 2200866. [Google Scholar] [CrossRef]
- Neishabouri, A.; Soltani Khaboushan, A.; Daghigh, F.; Kajbafzadeh, A.-M.; Majidi Zolbin, M. Decellularization in Tissue Engineering and Regenerative Medicine: Evaluation, Modification, and Application Methods. Front. Bioeng. Biotechnol. 2022, 10, 805299. [Google Scholar] [CrossRef]
- Hoshiba, T. A decellularized extracellular matrix derived from keratinocytes can suppress cellular senescence induced by replicative and oxidative stresses. Biomater. Sci. 2022, 10, 6828–6835. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Park, Y.; Choi, Y.-m.; Yong, U.; Kang, B.; Shin, W.; Min, S.; Kim, H.J.; Jang, J. A Bioprinted Tubular Intestine Model Using a Colon-Specific Extracellular Matrix Bioink. Adv. Healthc. Mater. 2022, 11, 2101768. [Google Scholar] [CrossRef] [PubMed]
- Sarmin, A.M.; El Moussaid, N.; Suntornnond, R.; Tyler, E.J.; Kim, Y.-H.; Di Cio, S.; Megone, W.V.; Pearce, O.; Gautrot, J.E.; Dawson, J.; et al. Multi-Scale Analysis of the Composition, Structure, and Function of Decellularized Extracellular Matrix for Human Skin and Wound Healing Models. Biomolecules 2022, 12, 837. [Google Scholar] [CrossRef]
- Di Francesco, D.; Bertani, F.; Fusaro, L.; Clemente, N.; Carton, F.; Talmon, M.; Fresu, L.G.; Boccafoschi, F. Regenerative Potential of A Bovine ECM-Derived Hydrogel for Biomedical Applications. Biomolecules 2022, 12, 1222. [Google Scholar] [CrossRef]
- Gao, C.; Fu, L.; Yu, Y.; Zhang, X.; Yang, X.; Cai, Q. Strategy of a cell-derived extracellular matrix for the construction of an osteochondral interlayer. Biomater. Sci. 2022, 10, 6472–6485. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Pei, M.; Li, Q.; Zhang, Y. Decellularized extracellular matrix mediates tissue construction and regeneration. Front. Med. 2022, 16, 56–82. [Google Scholar] [CrossRef]
- Niklason, L.E. Understanding the Extracellular Matrix to Enhance Stem Cell-Based Tissue Regeneration. Cell Stem Cell 2018, 22, 302–305. [Google Scholar] [CrossRef]
- Das, S.; Ge, G.; Lee, J.Y.; Jang, J.; Cho, D.W. Decellularized Tissue Matrix-based 3D Tissue Modeling. In Biofabrication and 3D Tissue Modeling; Royal Society of Chemistry: Cambridge, UK, 2019; Chapter 7. [Google Scholar]
- Lee, S.; Lee, H.S.; Chung, J.J.; Kim, S.H.; Park, J.W.; Lee, K.; Jung, Y. Enhanced Regeneration of Vascularized Adipose Tissue with Dual 3D-Printed Elastic Polymer/dECM Hydrogel Complex. Int. J. Mol. Sci. 2021, 22, 2886. [Google Scholar] [CrossRef]
- Salti, H.; Kramer, L.; Nelz, S.-C.; Lorenz, M.; Breitrück, A.; Hofrichter, J.; Frank, M.; Schulz, K.; Mitzner, S.; Wasserkort, R. Decellularization of precision-cut kidney slices—Application of physical and chemical methods. Biomed. Mater. 2023, 18, 025004. [Google Scholar] [CrossRef]
- van Tienderen, G.S.; Conboy, J.; Muntz, I.; Willemse, J.; Tieleman, J.; Monfils, K.; Schurink, I.J.; Demmers, J.A.A.; Doukas, M.; Koenderink, G.H.; et al. Tumor decellularization reveals proteomic and mechanical characteristics of the extracellular matrix of primary liver cancer. Biomater. Adv. 2023, 146, 213289. [Google Scholar] [CrossRef]
- Sevastianov, V.I.; Basok, Y.B.; Grigoriev, A.M.; Nemets, E.A.; Kirillova, A.D.; Kirsanova, L.A.; Lazhko, A.E.; Subbot, A.; Kravchik, M.V.; Khesuani, Y.D.; et al. Decellularization of cartilage microparticles: Effects of temperature, supercritical carbon dioxide and ultrasound on biochemical, mechanical, and biological properties. J. Biomed. Mater. Res. Part A 2023, 111, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Hoshiba, T.; Yunoki, S. Comparison of decellularization protocols for cultured cell-derived extracellular matrix—Effects on decellularization efficacy, extracellular matrix retention, and cell functions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2023, 111, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Biehl, A.; Gracioso Martins, A.M.; Davis, Z.G.; Sze, D.; Collins, L.; Mora-Navarro, C.; Fisher, M.B.; Freytes, D.O. Towards a standardized multi-tissue decellularization protocol for the derivation of extracellular matrix materials. Biomater. Sci. 2023, 11, 641–654. [Google Scholar] [CrossRef]
- Jambar Nooshin, B.; Tayebi, T.; Babajani, A.; Khani, M.M.; Niknejad, H. Effects of Different Perfusing Routes through The Portal Vein, Hepatic Vein, and Biliary Duct on Whole Rat Liver Decellularization. Cell J. 2023, 25, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Zubarevich, A.; Osswald, A.; Amanov, L.; Arjomandi Rad, A.; Schmack, B.; Ruhparwar, A.; Weymann, A. Development and evaluation of a novel combined perfusion decellularization heart-lung model for tissue engineering of bioartificial heart-lung scaffolds. Artif. Organs 2023, 47, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Mansour, R.N.; Karimizade, A.; Enderami, S.E.; Abasi, M.; Talebpour Amiri, F.; Jafarirad, A.; Mellati, A. The effect of source animal age, decellularization protocol, and sterilization method on bovine acellular dermal matrix as a scaffold for wound healing and skin regeneration. Artif. Organs 2023, 47, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Yuhan, J.; Yu, H.; Zhang, B.; Huang, K.; Zhu, L. Decellularized Extracellular Matrix for Remodeling Bioengineering Organoid’s Microenvironment. Small 2023, 2207752, early view. [Google Scholar] [CrossRef]
- Hodde, J.; Hiles, M. Virus safety of a porcine-derived medical device: Evaluation of a viral inactivation method. Biotechnol. Bioeng. 2002, 79, 211–216. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Y.; Qiao, W.; Shi, J.; Qiu, X.; Dong, N. Application of decellularized vascular matrix in small-diameter vascular grafts. Front. Bioeng. Biotechnol. 2023, 10, 1081233. [Google Scholar] [CrossRef]
- Xiao, H.; Chen, X.; Liu, X.; Wen, G.; Yu, Y. Recent advances in decellularized biomaterials for wound healing. Mater. Today Bio. 2023, 19, 100589. [Google Scholar] [CrossRef]
- Lim, L.Y.; Ding, S.S.L.; Muthukumaran, P.; Teoh, S.H.; Koh, Y.; Teo, A.K.K. Tissue engineering of decellularized pancreas scaffolds for regenerative medicine in diabetes. Acta Biomater. 2023, 157, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Mammana, M.; Zambello, G.; Contran, M.; Parnigotto, P.P.; Macchi, V.; Conconi, M.T.; Rea, F.; De Caro, R.; et al. Preclinical and clinical orthotopic transplantation of decellularized/engineered tracheal scaffolds: A systematic literature review. J. Tissue Eng. 2023, 14, 20417314231151826. [Google Scholar] [CrossRef] [PubMed]
- Kasravi, M.; Ahmadi, A.; Babajani, A.; Mazloomnejad, R.; Hatamnejad, M.R.; Shariatzadeh, S.; Bahrami, S.; Niknejad, H. Immunogenicity of decellularized extracellular matrix scaffolds: A bottleneck in tissue engineering and regenerative medicine. Biomater. Res. 2023, 27, 10. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; He, L.; Ye, Z.; Wei, Z.; Ao, J. Decellularized matrix for repairing intervertebral disc degeneration: Fabrication methods, applications and animal models. Mater. Today Bio. 2023, 18, 100523. [Google Scholar] [CrossRef]
- Kanda, H.; Oya, K.; Irisawa, T.; Wahyudiono; Goto, M. Tensile Strength of Porcine Aorta Decellularized with Liquefied Dimethyl Ether and DNase. ACS Omega 2022, 7, 34449–34453. [Google Scholar] [CrossRef] [PubMed]
- Long, J.; Qin, Z.; Chen, G.; Song, B.; Zhang, Z. Decellularized extracellular matrix (d-ECM): The key role of the inflammatory process in pre-regeneration after implantation. Biomater. Sci. 2023, 11, 1215–1235. [Google Scholar] [CrossRef]
- Moffat, D.; Ye, K.; Jin, S. Decellularization for the retention of tissue niches. J. Tissue Eng. 2022, 13, 20417314221101151. [Google Scholar] [CrossRef]
- García-Gareta, E.; Pérez, M.Á.; García-Aznar, J.M. Decellularization of tumours: A new frontier in tissue engineering. J. Tissue Eng. 2022, 13, 20417314221091682. [Google Scholar] [CrossRef]
- Tan, Y.H.; Helms, H.R.; Nakayama, K.H. Decellularization Strategies for Regenerating Cardiac and Skeletal Muscle Tissues. Front. Bioeng. Biotechnol. 2022, 10, 831300. [Google Scholar] [CrossRef]
- Krishnan, A.; Wang, H.; MacArthur, J.W. Applications of Tissue Decellularization Techniques in Ventricular Myocardial Biofabrication. Front. Bioeng. Biotechnol. 2022, 10, 802283. [Google Scholar] [CrossRef]
- Whitehead, K.M.; Hendricks, H.K.L.; Cakir, S.N.; Brás, L.E.d.C. ECM roles and biomechanics in cardiac tissue decellularization. Am. J. Physiol.-Heart Circ. Physiol. 2022, 323, H585–H596. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.M.; Silva, I.V.; Eisenhut, A.R.; Bionda, N.; Duarte, A.R.C.; Oliveira, A.L. Contributions of supercritical fluid technology for advancing decellularization and postprocessing of viable biological materials. Mater. Horiz. 2022, 9, 864–891. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chan, V.; Corridon, P.R. Decellularized blood vessel development: Current state-of-the-art and future directions. Front. Bioeng. Biotechnol. 2022, 10, 1400. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Jiang, W.; Huang, F.; Song, F.; Zhang, J.; Zhao, H. Recent Advances in Liver Engineering With Decellularized Scaffold. Front. Bioeng. Biotechnol. 2022, 10, 831477. [Google Scholar] [CrossRef] [PubMed]
- Naso, F.; Gandaglia, A. Can Heart Valve Decellularization Be Standardized? A Review of the Parameters Used for the Quality Control of Decellularization Processes. Front. Bioeng. Biotechnol. 2022, 10, 216. [Google Scholar] [CrossRef]
- Parameshwar, P.K.; Sagrillo-Fagundes, L.; Azevedo Portilho, N.; Pastor, W.A.; Vaillancourt, C.; Moraes, C. Engineered models for placental toxicology: Emerging approaches based on tissue decellularization. Reprod. Toxicol. 2022, 112, 148–159. [Google Scholar] [CrossRef]
- Wang, J.K.; Luo, B.; Guneta, V.; Li, L.; Foo, S.E.M.; Dai, Y.; Tan, T.T.Y.; Tan, N.S.; Choong, C.; Wong, M.T.C. Supercritical carbon dioxide extracted extracellular matrix material from adipose tissue. Mater. Sci. Eng. C 2017, 75, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, D.-J.; Srinivasan, P.; Yen, K.-C.; Yeh, Y.-C.; Chen, Y.-J.; Wang, H.-C.; Tarng, Y.-W. Protocols for the preparation and characterization of decellularized tissue and organ scaffolds for tissue engineering. BioTechniques 2021, 70, 107–115. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Lee, H.-S.; Hsieh, D.-J.; Periasamy, S.; Yeh, Y.-C.; Lai, Y.-P.; Tarng, Y.-W. 3D composite engineered using supercritical CO2 decellularized porcine cartilage scaffold, chondrocytes, and PRP: Role in articular cartilage regeneration. J. Tissue Eng. Regen. Med. 2021, 15, 163–175. [Google Scholar] [CrossRef]
- Gentilin, E.; D’Angelo, E.; Agostini, M.; Astolfi, L. Decellularized normal and cancer tissues as tools for cancer research. Cancer Gene Ther. 2022, 29, 879–888. [Google Scholar] [CrossRef]
- Sung, S.-Y.; Lin, Y.-W.; Wu, C.-C.; Lin, C.-Y.; Hsu, P.-S.; Periasamy, S.; Nagarajan, B.; Hsieh, D.-J.; Tsai, Y.-T.; Tsai, C.-S.; et al. Supercritical carbon dioxide-decellularized arteries exhibit physiologic-like vessel regeneration following xenotransplantation in rats. Biomater. Sci. 2023, 11, 2566–2580. [Google Scholar] [CrossRef] [PubMed]
- Giang, N.N.; Trinh, X.-T.; Han, J.; Chien, P.N.; Lee, J.; Noh, S.-R.; Shin, Y.; Nam, S.-Y.; Heo, C.-Y. Effective decellularization of human skin tissue for regenerative medicine by supercritical carbon dioxide technique. J. Tissue Eng. Regen. Med. 2022, 16, 1196–1207. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-S.; Wu, Y.-C.; Huang, S.-H.; Chen, Y.-C.; Srinivasan, P.; Hsieh, D.-J.; Yeh, Y.-C.; Lai, Y.-P.; Lin, Y.-N. A novel 3D histotypic cartilage construct engineered by supercritical carbon dioxide decellularized porcine nasal cartilage graft and chondrocytes exhibited chondrogenic capability in vitro. Int. J. Med. Sci. 2021, 18, 2217–2227. [Google Scholar] [CrossRef]
- Sawada, K.; Terada, D.; Yamaoka, T.; Kitamura, S.; Fujisato, T.J.J.o.C.T. Cell removal with supercritical carbon dioxide for acellular artificial tissue. J. Chem. Technol. Biotechnol. 2010, 83, 943–949. [Google Scholar] [CrossRef]
- Funamoto, S.; Nam, K.; Kimura, T.; Murakoshi, A.; Hashimoto, Y.; Niwaya, K.; Kitamura, S.; Fujisato, T.; Kishida, A. The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials 2010, 31, 3590–3595. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, Y.; Funamoto, S.; Sasaki, S.; Honda, T.; Hattori, S.; Nam, K.; Kimura, T.; Mochizuki, M.; Fujisato, T.; Kobayashi, H.; et al. Preparation and characterization of decellularized cornea using high-hydrostatic pressurization for corneal tissue engineering. Biomaterials 2010, 31, 3941–3948. [Google Scholar] [CrossRef]
- Seo, Y.; Jung, Y.; Kim, S.H. Decellularized heart ECM hydrogel using supercritical carbon dioxide for improved angiogenesis. Acta Biomater. 2018, 67, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Philips, C.; Terrie, L.; Thorrez, L. Decellularized skeletal muscle: A versatile biomaterial in tissue engineering and regenerative medicine. Biomaterials 2022, 283, 121436. [Google Scholar] [CrossRef]
- Zhu, D.; Jiang, Z.; Li, N.; Wang, X.; Ren, L.; Ye, Y.; Pan, Y.; Yang, G. Insights into the use of genetically modified decellularized biomaterials for tissue engineering and regenerative medicine. Adv. Drug Deliv. Rev. 2022, 188, 114413. [Google Scholar] [CrossRef]
- Marques-Magalhães, Â.; Cruz, T.; Costa, Â.M.; Estêvão, D.; Rios, E.; Canão, P.A.; Velho, S.; Carneiro, F.; Oliveira, M.J.; Cardoso, A.P. Decellularized Colorectal Cancer Matrices as Bioactive Scaffolds for Studying Tumor-Stroma Interactions. Cancers 2022, 14, 359. [Google Scholar] [CrossRef]
- Kim, B.-S.; Kim, J.-U.; So, K.-H.; Hwang, N.S. Supercritical Fluid-Based Decellularization Technologies for Regenerative Medicine Applications. Macromol. Biosci. 2021, 21, 2100160. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Zhang, Y.; Zhou, L.; Sun, Z.; Zheng, J.; Chen, Y.; Dai, Y. Development of a porcine bladder acellular matrix with well-preserved extracellular bioactive factors for tissue engineering. Tissue Eng. Part C Methods 2010, 16, 1201–1211. [Google Scholar] [CrossRef] [PubMed]
- Maghsoudlou, P.; Georgiades, F.; Smith, H.; Milan, A.; Shangaris, P.; Urbani, L.; Loukogeorgakis, S.P.; Lombardi, B.; Mazza, G.; Hagen, C.; et al. Optimization of Liver Decellularization Maintains Extracellular Matrix Micro-Architecture and Composition Predisposing to Effective Cell Seeding. PLoS ONE 2016, 11, e0155324. [Google Scholar] [CrossRef] [PubMed]
- Gillies, A.R.; Smith, L.R.; Lieber, R.L.; Varghese, S. Method for decellularizing skeletal muscle without detergents or proteolytic enzymes. Tissue Eng. Part C Methods 2011, 17, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Nouri Barkestani, M.; Naserian, S.; Uzan, G.; Shamdani, S. Post-decellularization techniques ameliorate cartilage decellularization process for tissue engineering applications. J. Tissue Eng. 2021, 12, 2041731420983562. [Google Scholar] [CrossRef] [PubMed]
- Capella-Monsonís, H.; Zeugolis, D.I. Decellularized xenografts in regenerative medicine: From processing to clinical application. Xenotransplantation 2021, 28, e12683. [Google Scholar] [CrossRef]
- Khajavi, M.; Hashemi, M.; Kalalinia, F. Recent advances in optimization of liver decellularization procedures used for liver regeneration. Life Sci. 2021, 281, 119801. [Google Scholar] [CrossRef]
- Asadi, M.; Khalili, M.; Lotfi, H.; Vaghefi Moghaddam, S.; Zarghami, N.; André, H.; Alizadeh, E. Liver bioengineering: Recent trends/advances in decellularization and cell sheet technologies towards translation into the clinic. Life Sci. 2021, 276, 119373. [Google Scholar] [CrossRef]
- Li, J.; Narayanan, K.; Zhang, Y.; Hill, R.C.; He, F.; Hansen, K.C.; Pei, M. Role of lineage-specific matrix in stem cell chondrogenesis. Biomaterials 2020, 231, 119681. [Google Scholar] [CrossRef]
- Beachley, V.Z.; Wolf, M.T.; Sadtler, K.; Manda, S.S.; Jacobs, H.; Blatchley, M.R.; Bader, J.S.; Pandey, A.; Pardoll, D.; Elisseeff, J.H. Tissue matrix arrays for high-throughput screening and systems analysis of cell function. Nat. Methods 2015, 12, 1197–1204. [Google Scholar] [CrossRef]
- Shin, Y.J.; Shafranek, R.T.; Tsui, J.H.; Walcott, J.; Nelson, A.; Kim, D.H. 3D bioprinting of mechanically tuned bioinks derived from cardiac decellularized extracellular matrix. Acta Biomater. 2021, 119, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Rueda-Gensini, L.; Serna, J.A.; Cifuentes, J.; Cruz, J.C.; Muñoz-Camargo, C. Graphene Oxide-Embedded Extracellular Matrix-Derived Hydrogel as a Multiresponsive Platform for 3D Bioprinting Applications. Int. J. Bioprint. 2021, 7, 353. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Meng, Z.; He, J.; Mao, M.; Li, X.; Chen, P.; Fan, J.; Li, D. Embedded bioprinting for designer 3D tissue constructs with complex structural organization. Acta Biomater. 2022, 140, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Galarraga, J.H.; Kwon, M.Y.; Lee, H.; Burdick, J.A. Gallol-derived ECM-mimetic adhesive bioinks exhibiting temporal shear-thinning and stabilization behavior. Acta Biomater. 2019, 95, 165–175. [Google Scholar] [CrossRef]
- Fu, Z.; Naghieh, S.; Xu, C.; Wang, C.; Sun, W.; Chen, X. Printability in extrusion bioprinting. Biofabrication 2021, 13, 033001. [Google Scholar] [CrossRef]
- Liu, W.; Heinrich, M.A.; Zhou, Y.; Akpek, A.; Hu, N.; Liu, X.; Guan, X.; Zhong, Z.; Jin, X.; Khademhosseini, A.; et al. Extrusion Bioprinting of Shear-Thinning Gelatin Methacryloyl Bioinks. Adv. Healthc. Mater. 2017, 6, 1601451. [Google Scholar] [CrossRef]
- Sobreiro-Almeida, R.; Gómez-Florit, M.; Quinteira, R.; Reis, R.L.; Gomes, M.E.; Neves, N.M. Decellularized kidney extracellular matrix bioinks recapitulate renal 3D microenvironmentin vitro. Biofabrication 2021, 13, 045006. [Google Scholar] [CrossRef]
- Athirasala, A.; Tahayeri, A.; Thrivikraman, G.; França, C.M.; Monteiro, N.; Tran, V.; Ferracane, J.; Bertassoni, L.E. A dentin-derived hydrogel bioink for 3D bioprinting of cell laden scaffolds for regenerative dentistry. Biofabrication 2018, 10, 024101. [Google Scholar] [CrossRef]
- Mendibil, U.; Ruiz-Hernandez, R.; Retegi-Carrion, S.; Garcia-Urquia, N.; Olalde-Graells, B.; Abarrategi, A. Tissue-Specific Decellularization Methods: Rationale and Strategies to Achieve Regenerative Compounds. Int. J. Mol. Sci. 2020, 21, 5447. [Google Scholar] [CrossRef]
- Pati, F.; Jang, J.; Ha, D.H.; Won Kim, S.; Rhie, J.W.; Shim, J.H.; Kim, D.H.; Cho, D.W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 2014, 5, 3935. [Google Scholar] [CrossRef]
- Jang, J.; Kim, T.G.; Kim, B.S.; Kim, S.W.; Kwon, S.M.; Cho, D.W. Tailoring mechanical properties of decellularized extracellular matrix bioink by vitamin B2-induced photo-crosslinking. Acta Biomater. 2016, 33, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Ma, X.; Zhu, W.; Wang, P.; Miller, K.L.; Stupin, J.; Koroleva-Maharajh, A.; Hairabedian, A.; Chen, S. Scanningless and continuous 3D bioprinting of human tissues with decellularized extracellular matrix. Biomaterials 2019, 194, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Kim, S.W.; Choi, Y.J.; Lee, S.; Lee, S.H.; Kong, J.S.; Park, H.J.; Cho, D.W.; Jang, J. Decellularized extracellular matrix bioinks and the external stimuli to enhance cardiac tissue development in vitro. Acta Biomater. 2019, 95, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.S.; Kim, B.K.; Lee, J.; Min, B.H.; Park, S.H. Development of Printable Natural Cartilage Matrix Bioink for 3D Printing of Irregular Tissue Shape. Tissue Eng. Regen. Med. 2018, 15, 155–162. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Luo, C.; Zhai, C.; Li, Z.; Zhang, Y.; Yuan, T.; Dong, S.; Zhang, J.; Fan, W. Crosslinker-free silk/decellularized extracellular matrix porous bioink for 3D bioprinting-based cartilage tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 118, 111388. [Google Scholar] [CrossRef]
- Chae, S.; Lee, S.S.; Choi, Y.J.; Hong, D.H.; Gao, G.; Wang, J.H.; Cho, D.W. 3D cell-printing of biocompatible and functional meniscus constructs using meniscus-derived bioink. Biomaterials 2021, 267, 120466. [Google Scholar] [CrossRef]
- Lu, J.; Huang, J.; Jin, J.; Xie, C.; Xue, B.; Lai, J.; Cheng, B.; Li, L.; Jiang, Q. The Design and Characterization of a Strong Bio-Ink for Meniscus Regeneration. Int. J. Bioprint. 2022, 8, 600. [Google Scholar] [CrossRef]
- Ahn, M.; Cho, W.-W.; Kim, B.S.; Cho, D.-W. Engineering Densely Packed Adipose Tissue via Environmentally Controlled In-Bath 3D Bioprinting. Adv. Funct. Mater. 2022, 32, 2200203. [Google Scholar] [CrossRef]
- Del Amo, C.; Fernández-San Argimiro, X.; Cascajo-Castresana, M.; Perez-Valle, A.; Madarieta, I.; Olalde, B.; Andia, I. Wound-Microenvironment Engineering through Advanced-Dressing Bioprinting. Int. J. Mol. Sci. 2022, 23, 2836. [Google Scholar] [CrossRef]
- Yu, H.W.; Kim, B.S.; Lee, J.Y.; Lee, K.; Ahn, M.; Jang, J.; Cho, D.-W.; Choi, J.Y. Tissue printing for engineering transplantable human parathyroid patch to improve parathyroid engraftment, integration, and hormone secretion in vivo. Biofabrication 2021, 13, 035033. [Google Scholar] [CrossRef]
- Choi, Y.J.; Jun, Y.J.; Kim, D.Y.; Yi, H.G.; Chae, S.H.; Kang, J.; Lee, J.; Gao, G.; Kong, J.S.; Jang, J.; et al. A 3D cell printed muscle construct with tissue-derived bioink for the treatment of volumetric muscle loss. Biomaterials 2019, 206, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Lee, H.; Lee, J.; Atala, A.; Yoo, J.J.; Lee, S.J.; Kim, G.H. Efficient myotube formation in 3D bioprinted tissue construct by biochemical and topographical cues. Biomaterials 2020, 230, 119632. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Han, W.; Kim, H.; Ha, D.-H.; Jang, J.; Kim, B.S.; Cho, D.-W. Development of Liver Decellularized Extracellular Matrix Bioink for Three-Dimensional Cell Printing-Based Liver Tissue Engineering. Biomacromolecules 2017, 18, 1229–1237. [Google Scholar] [CrossRef]
- Ma, X.; Yu, C.; Wang, P.; Xu, W.; Wan, X.; Lai, C.S.E.; Liu, J.; Koroleva-Maharajh, A.; Chen, S. Rapid 3D bioprinting of decellularized extracellular matrix with regionally varied mechanical properties and biomimetic microarchitecture. Biomaterials 2018, 185, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Kwon, Y.W.; Kong, J.-S.; Park, G.T.; Gao, G.; Han, W.; Kim, M.-B.; Lee, H.; Kim, J.H.; Cho, D.-W. 3D cell printing of in vitro stabilized skin model and in vivo pre-vascularized skin patch using tissue-specific extracellular matrix bioink: A step towards advanced skin tissue engineering. Biomaterials 2018, 168, 38–53. [Google Scholar] [CrossRef] [PubMed]
- Won, J.Y.; Lee, M.H.; Kim, M.J.; Min, K.H.; Ahn, G.; Han, J.S.; Jin, S.; Yun, W.S.; Shim, J.H. A potential dermal substitute using decellularized dermis extracellular matrix derived bio-ink. Artif. Cells Nanomed. Biotechnol. 2019, 47, 644–649. [Google Scholar] [CrossRef]
- Ahn, G.; Min, K.H.; Kim, C.; Lee, J.S.; Kang, D.; Won, J.Y.; Cho, D.W.; Kim, J.Y.; Jin, S.; Yun, W.S.; et al. Precise stacking of decellularized extracellular matrix based 3D cell-laden constructs by a 3D cell printing system equipped with heating modules. Sci. Rep. 2017, 7, 8624. [Google Scholar] [CrossRef]
- Hong, H.; Kim, H.; Han, S.J.; Jang, J.; Kim, H.K.; Cho, D.W.; Kim, D.S. Compressed collagen intermixed with cornea-derived decellularized extracellular matrix providing mechanical and biochemical niches for corneal stroma analogue. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 103, 109837. [Google Scholar] [CrossRef]
- Kim, H.; Park, M.-N.; Kim, J.; Jang, J.; Kim, H.-K.; Cho, D.-W. Characterization of cornea-specific bioink: High transparency, improved in vivo safety. J. Tissue Eng. 2019, 10, 2041731418823382. [Google Scholar] [CrossRef]
- Yi, H.G.; Jeong, Y.H.; Kim, Y.; Choi, Y.J.; Moon, H.E.; Park, S.H.; Kang, K.S.; Bae, M.; Jang, J.; Youn, H.; et al. A bioprinted human-glioblastoma-on-a-chip for the identification of patient-specific responses to chemoradiotherapy. Nat. Biomed. Eng. 2019, 3, 509–519. [Google Scholar] [CrossRef]
- Kim, J.; Shim, I.K.; Hwang, D.G.; Lee, Y.N.; Kim, M.; Kim, H.; Kim, S.W.; Lee, S.; Kim, S.C.; Cho, D.W.; et al. 3D cell printing of islet-laden pancreatic tissue-derived extracellular matrix bioink constructs for enhancing pancreatic functions. J. Mater. Chem. B 2019, 7, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, M.; Hwang, D.G.; Shim, I.K.; Kim, S.C.; Jang, J. Pancreatic Tissue-Derived Extracellular Matrix Bioink for Printing 3D Cell-Laden Pancreatic Tissue Constructs. J. Vis. Exp. 2019, 154, e60434. [Google Scholar] [CrossRef]
- Park, J.Y.; Ryu, H.; Lee, B.; Ha, D.H.; Ahn, M.; Kim, S.; Kim, J.Y.; Jeon, N.L.; Cho, D.W. Development of a functional airway-on-a-chip by 3D cell printing. Biofabrication 2018, 11, 015002. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, J.Y.; Nam, I.C.; Ahn, M.; Lee, J.Y.; Choi, S.H.; Kim, S.W.; Cho, D.W. A rational tissue engineering strategy based on three-dimensional (3D) printing for extensive circumferential tracheal reconstruction. Biomaterials 2018, 185, 276–283. [Google Scholar] [CrossRef]
- Gao, G.; Park, J.Y.; Kim, B.S.; Jang, J.; Cho, D.-W. Coaxial Cell Printing of Freestanding, Perfusable, and Functional In Vitro Vascular Models for Recapitulation of Native Vascular Endothelium Pathophysiology. Adv. Healthc. Mater. 2018, 7, 1801102. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, G.; Lin, H.; Shang, Y.; Liu, N.; Zhen, Y.; An, Y. Cartilage 3D bioprinting for rhinoplasty using adipose-derived stem cells as seed cells: Review and recent advances. Cell Prolif. 2023, 56, e13417. [Google Scholar] [CrossRef]
- Sang, S.; Mao, X.; Cao, Y.; Liu, Z.; Shen, Z.; Li, M.; Jia, W.; Guo, Z.; Wang, Z.; Xiang, C.; et al. 3D Bioprinting Using Synovium-Derived MSC-Laden Photo-Cross-Linked ECM Bioink for Cartilage Regeneration. ACS Appl. Mater. Interfaces 2023, 15, 8895–8913. [Google Scholar] [CrossRef]
- Pati, F.; Ha, D.H.; Jang, J.; Han, H.H.; Rhie, J.W.; Cho, D.W. Biomimetic 3D tissue printing for soft tissue regeneration. Biomaterials 2015, 62, 164–175. [Google Scholar] [CrossRef]
- Cui, Q.; Zhang, D.; Kong, D.; Tang, J.; Liao, X.; Yang, Q.; Ren, J.; Gong, Y.; Wu, G. Co-transplantation with adipose-derived cells to improve parathyroid transplantation in a mice model. Stem Cell Res. Ther. 2020, 11, 200. [Google Scholar] [CrossRef]
- Kakuta, T.; Sawada, K.; Kanai, G.; Tatsumi, R.; Miyakogawa, T.; Ishida, M.; Nakazawa, R.; Fukagawa, M. Parathyroid hormone-producing cells exist in adipose tissues surrounding the parathyroid glands in hemodialysis patients with secondary hyperparathyroidism. Sci. Rep. 2020, 10, 3290. [Google Scholar] [CrossRef]
- Charbe, N.B.; Tambuwala, M.; Palakurthi, S.S.; Warokar, A.; Hromić-Jahjefendić, A.; Bakshi, H.; Zacconi, F.; Mishra, V.; Khadse, S.; Aljabali, A.A.; et al. Biomedical applications of three-dimensional bioprinted craniofacial tissue engineering. Bioeng. Transl. Med. 2023, 8, e10333. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Kim, T.G.; Jeong, J.; Yi, H.G.; Park, J.W.; Hwang, W.; Cho, D.W. 3D Cell Printing of Functional Skeletal Muscle Constructs Using Skeletal Muscle-Derived Bioink. Adv. Healthc. Mater. 2016, 5, 2636–2645. [Google Scholar] [CrossRef] [PubMed]
- Skardal, A.; Devarasetty, M.; Kang, H.W.; Mead, I.; Bishop, C.; Shupe, T.; Lee, S.J.; Jackson, J.; Yoo, J.; Soker, S.; et al. A hydrogel bioink toolkit for mimicking native tissue biochemical and mechanical properties in bioprinted tissue constructs. Acta Biomater. 2015, 25, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Baltazar, T.; Jiang, B.; Moncayo, A.; Merola, J.; Albanna, M.Z.; Saltzman, W.M.; Pober, J.S. 3D bioprinting of an implantable xeno-free vascularized human skin graft. Bioeng. Transl. Med. 2023, 8, e10324. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Sun, L.; Liu, Z.; Shen, Z.; Cao, Y.; Han, L.; Sang, S.; Wang, J. 3D bioprinting of heterogeneous tissue-engineered skin containing human dermal fibroblasts and keratinocytes. Biomater. Sci. 2023, 11, 2461–2477. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Gao, G.; Kim, J.Y.; Cho, D.-W. 3D Cell Printing of Perfusable Vascularized Human Skin Equivalent Composed of Epidermis, Dermis, and Hypodermis for Better Structural Recapitulation of Native Skin. Adv. Healthc. Mater. 2019, 8, 1801019. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Ahn, M.; Cho, W.-W.; Gao, G.; Jang, J.; Cho, D.-W. Engineering of diseased human skin equivalent using 3D cell printing for representing pathophysiological hallmarks of type 2 diabetes in vitro. Biomaterials 2021, 272, 120776. [Google Scholar] [CrossRef]
- Orash Mahmoud Salehi, A.; Heidari-Keshel, S.; Poursamar, S.A.; Zarrabi, A.; Sefat, F.; Mamidi, N.; Behrouz, M.J.; Rafienia, M. Bioprinted Membranes for Corneal Tissue Engineering: A Review. Pharmaceutics 2022, 14, 2797. [Google Scholar] [CrossRef]
- Kostenko, A.; Swioklo, S.; Connon, C.J. Alginate in corneal tissue engineering. Biomed. Mater. 2022, 17, 022004. [Google Scholar] [CrossRef]
- Chameettachal, S.; Puranik, C.J.; Veluthedathu, M.N.; Chalil, N.B.; John, R.; Pati, F. Thickening of Ectatic Cornea through Regeneration Using Decellularized Corneal Matrix Injectable Hydrogel: A Strategic Advancement to Mitigate Corneal Ectasia. ACS Appl. Bio. Mater. 2021, 4, 7300–7313. [Google Scholar] [CrossRef]
- Tian, L.; Ma, J.; Li, W.; Zhang, X.; Gao, X. Microfiber Fabricated via Microfluidic Spinning toward Tissue Engineering Applications. Macromol. Biosci. 2023, 23, 2200429. [Google Scholar] [CrossRef] [PubMed]
- Khalili, M.; Asadi, M.; Kahroba, H.; Soleyman, M.R.; Andre, H.; Alizadeh, E. Corneal endothelium tissue engineering: An evolution of signaling molecules, cells, and scaffolds toward 3D bioprinting and cell sheets. J. Cell Physiol. 2020, 236, 3275–3303. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, K.-P.; Kim, H.; Park, S.; Wijesinghe, R.E.; Lee, J.; Han, S.; Lee, S.; Kim, P.; Cho, D.-W.; et al. Biocompatibility evaluation of bioprinted decellularized collagen sheet implanted in vivo cornea using swept-source optical coherence tomography. J. Biophotonics 2019, 12, e201900098. [Google Scholar] [CrossRef] [PubMed]
- Soetedjo, A.A.P.; Lee, J.M.; Lau, H.H.; Goh, G.L.; An, J.; Koh, Y.; Yeong, W.Y.; Teo, A.K.K. Tissue engineering and 3D printing of bioartificial pancreas for regenerative medicine in diabetes. Trends Endocrinol. Metab. 2021, 32, 609–622. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, M.A.; Campbell-Thompson, M.; Kusmartseva, I.; Kaestner, K.H. Organisation of the human pancreas in health and in diabetes. Diabetologia 2020, 63, 1966–1973. [Google Scholar] [CrossRef]
- Gupta, P.; Mandal, B.B. Silk biomaterials for vascular tissue engineering applications. Acta Biomater. 2021, 134, 79–106. [Google Scholar] [CrossRef]
- Xu, Y.; Hu, Y.; Liu, C.; Yao, H.; Liu, B.; Mi, S. A Novel Strategy for Creating Tissue-Engineered Biomimetic Blood Vessels Using 3D Bioprinting Technology. Materials 2018, 11, 1581. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, X.; Huang, S.; Liu, Y.; Fu, B.S.-C.; Mak, K.K.-L.; Blocki, A.M.; Yung, P.S.-H.; Tuan, R.S.; Ker, D.F.E. Engineering multi-tissue units for regenerative Medicine: Bone-tendon-muscle units of the rotator cuff. Biomaterials 2021, 272, 120789. [Google Scholar] [CrossRef]
- Chae, S.; Sun, Y.; Choi, Y.-J.; Ha, D.-H.; Jeon, I.; Cho, D.-W. 3D cell-printing of tendon-bone interface using tissue-derived extracellular matrix bioinks for chronic rotator cuff repair. Biofabrication 2021, 13, 035005. [Google Scholar] [CrossRef]
- Hernandez, M.J.; Yakutis, G.E.; Zelus, E.I.; Hill, R.C.; Dzieciatkowska, M.; Hansen, K.C.; Christman, K.L. Manufacturing considerations for producing and assessing decellularized extracellular matrix hydrogels. Methods 2020, 171, 20–27. [Google Scholar] [CrossRef]
- Kim, M.K.; Jeong, W.; Lee, S.M.; Kim, J.B.; Jin, S.; Kang, H.-W. Decellularized extracellular matrix-based bio-ink with enhanced 3D printability and mechanical properties. Biofabrication 2020, 12, 025003. [Google Scholar] [CrossRef]
- Xing, F.; Li, L.; Zhou, C.; Long, C.; Wu, L.; Lei, H.; Kong, Q.; Fan, Y.; Xiang, Z.; Zhang, X. Regulation and Directing Stem Cell Fate by Tissue Engineering Functional Microenvironments: Scaffold Physical and Chemical Cues. Stem Cells Int. 2019, 2019, 2180925. [Google Scholar] [CrossRef]
- Xing, F.; Li, S.; Yin, D.; Xie, J.; Rommens, P.M.; Xiang, Z.; Liu, M.; Ritz, U. Recent progress in Mg-based alloys as a novel bioabsorbable biomaterials for orthopedic applications. J. Magnes. Alloy. 2022, 10, 1428–1456. [Google Scholar] [CrossRef]
- Yeleswarapu, S.; Chameettachal, S.; Bera, A.; Pati, F. Tissue-Specific Bioink from Xenogeneic Sources for 3D Bioprinting of Tissue Constructs. In Xenotransplantation-Comprehensive Study; IntechOpen: Rijeka, Croatia, 2019. [Google Scholar]
- Diaz, M.D.; Christman, K.L. Injectable Hydrogels to Treat Myocardial Infarction. In Cardiovascular Regenerative Medicine: Tissue Engineering and Clinical Applications; Serpooshan, V., Wu, S.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 185–206. [Google Scholar]
- Traverse, J.H.; Henry, T.D.; Dib, N.; Patel, A.N.; Pepine, C.; Schaer, G.L.; DeQuach, J.A.; Kinsey, A.M.; Chamberlin, P.; Christman, K.L. First-in-Man Study of a Cardiac Extracellular Matrix Hydrogel in Early and Late Myocardial Infarction Patients. JACC Basic Transl. Sci. 2019, 4, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.H.; Zhu, X.Z.; Guo, P.Y.; Rose, P.; Liu, X.T.; Liu, X.; Zhu, Y.Z. Recent updates in click and computational chemistry for drug discovery and development. Front. Chem. 2023, 11, 1114970. [Google Scholar] [CrossRef]
- Kostidis, S.; Sánchez-López, E.; Giera, M. Lipidomics analysis in drug discovery and development. Curr. Opin. Chem. Biol. 2023, 72, 102256. [Google Scholar] [CrossRef] [PubMed]
- Shyam, R.; Reddy, L.V.; Palaniappan, A. Fabrication and Characterization Techniques of In Vitro 3D Tissue Models. Int. J. Mol. Sci. 2023, 24, 1912. [Google Scholar] [CrossRef]
- Whitehouse, C.; Corbett, N.; Brownlees, J. 3D models of neurodegeneration: Implementation in drug discovery. Trends Pharmacol. Sci. 2023, 44, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Datta, P.; Ayan, B.; Ozbolat, V.; Sosnoski, D.; Ozbolat, I.T. 3D bioprinting for drug discovery and development in pharmaceutics. Acta Biomater. 2017, 57, 26–46. [Google Scholar] [CrossRef] [PubMed]
- Youhanna, S.; Kemas, A.M.; Preiss, L.; Zhou, Y.; Shen, J.X.; Cakal, S.D.; Paqualini, F.S.; Goparaju, S.K.; Shafagh, R.Z.; Lind, J.U.; et al. Organotypic and Microphysiological Human Tissue Models for Drug Discovery and Development—Current State-of-the-Art and Future Perspectives. Pharmacol. Rev. 2022, 74, 141–206. [Google Scholar] [CrossRef]









| Bioink Composition | Seed Cells | Biological Factors | Source of dECM | Applications | References |
|---|---|---|---|---|---|
| Decellularized heart tissue | Rat myoblast cells | - | Porcine heart | Heart tissue regeneration | [142] |
| Decellularized heart tissue | Cardiac progenitor cells | VEGF | Left ventricle from the complete porcine heart | Hydrogel patch for cardiac repair | [30] |
| Decellularized heart tissue | Human cardiac progenitor cells | Vitamin B2 | Heart tissue from a 6-month-old Korean domestic pig | In vitro fabrication of engineered tissue | [143] |
| Decellularized heart tissue | Human iPSCs | - | Heart left ventricles from Yorkshire pigs | Fabrication of patient-specific tissue model | [144] |
| Decellularized heart tissue | Neonatal rat cardiomyocytes | - | Heart tissues from 6-month-old Korean domestic pigs | Engineered heart tissue | [145] |
| Decellularized cartilage tissue | Human inferior turbinate-tissue-derived MSCs | - | Porcine cartilage tissue | Cartilage tissue regeneration | [142] |
| Decellularized cartilage tissue and silk fibroin | Rabbit bone-marrow-derived MSCs | - | Porcine articular cartilage | Developing tissue substitutes with irregular shape | [146] |
| Decellularized cartilage tissue and silk fibroin | Rabbit bone-marrow-derived MSCs | TGF-β3 | Articular cartilage tissue from female goats | Cartilage regeneration | [147] |
| Decellularized menisci and polyurethane and polycaprolactone polymers | Human bone-marrow-derived MSC | - | Porcine lateral and medial menisci | Meniscus regeneration | [148] |
| Decellularized menisci and poly(vinyl alcohol) | - | - | Rabbit menisci | Meniscus regeneration | [149] |
| Decellularized adipose tissue | Human adipose-derived stem cells | - | Porcine adipose | Adipose tissue regeneration | [142] |
| Decellularized adipose tissue and alginate | Human subcutaneous preadipocyte cells | - | Human adipose | Engineering densely packed adipose tissue | [150] |
| Decellularized adipose tissue and plasma | Human dermal fibroblasts | - | Porcine adipose | Tissue substitutes with optimal microenvironment | [151] |
| Decellularized adipose tissue | Minced parathyroid glands | - | Porcine adipose | Maintaining biological functions of parathyroid glands | [152] |
| Decellularized skeletal muscle | Human skeletal muscle cells | - | Porcine tibialis anterior muscles and descending aortas | Volumetric muscle loss treatment | [153] |
| Methacrylate decellularized skeletal muscle | C2C12 cells | - | Lower limb muscle from adult Yorkshire porcine | Muscle regeneration | [154] |
| Decellularized liver tissue | HepG2 cells and human MSCs | - | Porcine liver tissue | Liver in vitro models for transplantation and drug screening | [155] |
| Decellularized liver tissue | Human iPSCs | - | Liver from three-month-old healthy Yorkshire pigs | Fabrication of patient-specific tissue substitutes | [144] |
| Decellularized liver tissue and collagen I | HepG2 cells | - | Porcine liver from Yorkshire pigs | Disease mechanism exploration and drug screening | [156] |
| Decellularized skin tissue | Human neonatal epidermal keratinocytes, human adipose-derived MSCs | - | Porcine skin tissue | Skin regeneration | [157] |
| Decellularized skin tissue | Human dermal fibroblasts | - | Porcine skin tissue | Dermal substitute | [158] |
| Decellularized skin tissue | Mouse fibroblasts | - | Native skin tissues from a Korean domestic pig | Establishing a 3D cell printing process | [159] |
| Decellularized cornea and collagen I | Human corneal keratocytes | - | Bovine eyeballs | Cornea substitutes | [160] |
| Decellularized cornea | - | - | Bovine eyeballs | Artificial corneas | [161] |
| Decellularized brain | Glioblastoma cells and endothelial cells | - | Cephalic parts of market pigs | In vitro disease model | [162] |
| Decellularized pancreatic tissues | Rat islets and endothelial cells | - | Porcine pancreatic tissue | Fabricating 3D pancreatic tissue constructs | [163] |
| Decellularized pancreatic tissues | - | - | Porcine pancreatic tissue | Pancreatic tissue substitutes | [164] |
| Decellularized tracheal mucosa | Endothelial cells and fibroblasts | - | Porcine tracheal mucosa | Functional airway-on-a-chip | [165] |
| Decellularized trachea | Human inferior turbinate MSCs | - | Porcine trachea | Tracheal reconstruction | [166] |
| Decellularized vascular tissue | Endothelial progenitor cells | - | Porcine descending aortas of pigs | Therapy for ischemic disease | [31] |
| Decellularized vascular tissue | Endothelial cells | - | Fresh porcine aortic tissue | In vitro vascular models | [167] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhe, M.; Wu, X.; Yu, P.; Xu, J.; Liu, M.; Yang, G.; Xiang, Z.; Xing, F.; Ritz, U. Recent Advances in Decellularized Extracellular Matrix-Based Bioinks for 3D Bioprinting in Tissue Engineering. Materials 2023, 16, 3197. https://doi.org/10.3390/ma16083197
Zhe M, Wu X, Yu P, Xu J, Liu M, Yang G, Xiang Z, Xing F, Ritz U. Recent Advances in Decellularized Extracellular Matrix-Based Bioinks for 3D Bioprinting in Tissue Engineering. Materials. 2023; 16(8):3197. https://doi.org/10.3390/ma16083197
Chicago/Turabian StyleZhe, Man, Xinyu Wu, Peiyun Yu, Jiawei Xu, Ming Liu, Guang Yang, Zhou Xiang, Fei Xing, and Ulrike Ritz. 2023. "Recent Advances in Decellularized Extracellular Matrix-Based Bioinks for 3D Bioprinting in Tissue Engineering" Materials 16, no. 8: 3197. https://doi.org/10.3390/ma16083197








