3.1. Properties of Concrete Made of Calcium Silicate Aggregate
The normal concrete was made from CEM I 42.5R cement at 320 kg/m
3 without chemical additives. A total of 1 + 6 (reference + tested) different aggregate mixes were used to produce concrete with the following proportions in
Table 6.
A constant consistency was aimed for across the seven different mixes. The consistency was determined according to EN 12350-2 [
30]. The density of the concrete mix was determined according to EN 12350-6 [
31]. The compressive strength of the concrete was tested according to EN 12390-3 [
32]. For testing the frost resistance of the concrete, the surface delamination method used in Estonia, Sweden, and Finland was applied. In Estonia, the method for determining frost resistance is described in national standard EVS 814 [
33].
The water demand of the concrete increased due to the higher water absorption of calcium silicate aggregate (~10%) compared to the water absorption of limestone aggregate (~2%). The density of the concrete mix decreased with the increase in the proportion of calcium silicate aggregate in the concrete due to the increase in water content and the lower bulk density of calcium silicate aggregate (~960 kg/m3) compared to the bulk density of limestone aggregate (~1380 kg/m3).
3.1.1. Compressive Strength
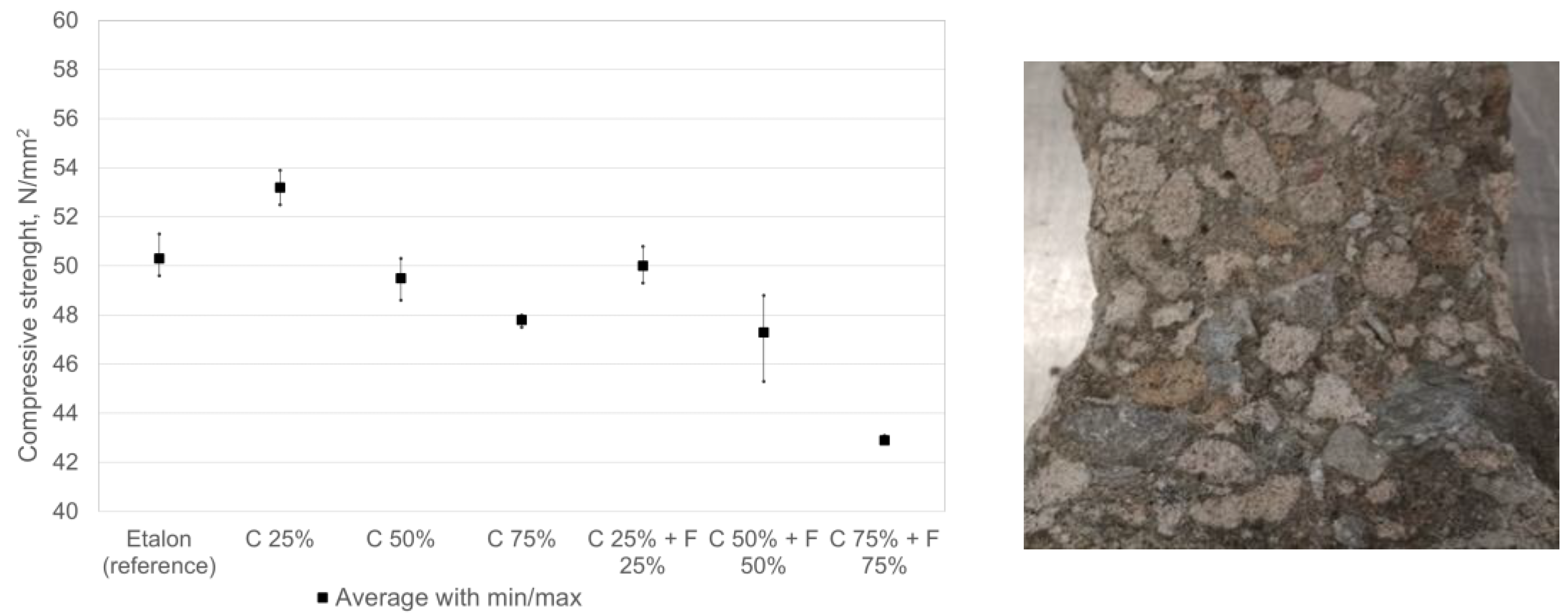
The compressive strengths of the concrete were determined at 28 days of age. The concrete specimens were cured for 1 day in the mold and 27 days in water at (20 ± 2) °C. For the average compressive strengths of the concretes, obtained as the average of the three parallel specimens, see
Figure 4. The texture of concrete is dense and the grains of limestone as well as calcium silicate aggregate are well bonded to the cement mortar.
The results in the graph show that recycled calcium silica aggregate does not significantly affect the strength properties of concrete but a tendency to lower strengths with higher levels of replaced aggregate can be detected. It is possible to use silicate aggregate as a concrete aggregate, replacing primary concrete aggregates at substitution levels of up to 50%. The increase in compressive strength with up to 25% replacement can be explained by the higher bond strength of the cement due to the rough and porous surface of the CCSS [
34], also additional chemical bonding might appear. Further increase (50% and 75%) lowers the compressive strength due to lower density (related to strength) of calcium silicate aggregate (~960 kg/m
3) compared to that of limestone (~1380 kg/m
3). More than 50% substitution is not desirable, as it will lead to increased water demand and a stiffer concrete mix structure, which will lead to problems in pouring and compaction due to loss of workability and reduced service life.
3.1.2. Frost Resistance
Frost resistance tests of concrete were carried out in accordance with EVS 814 [
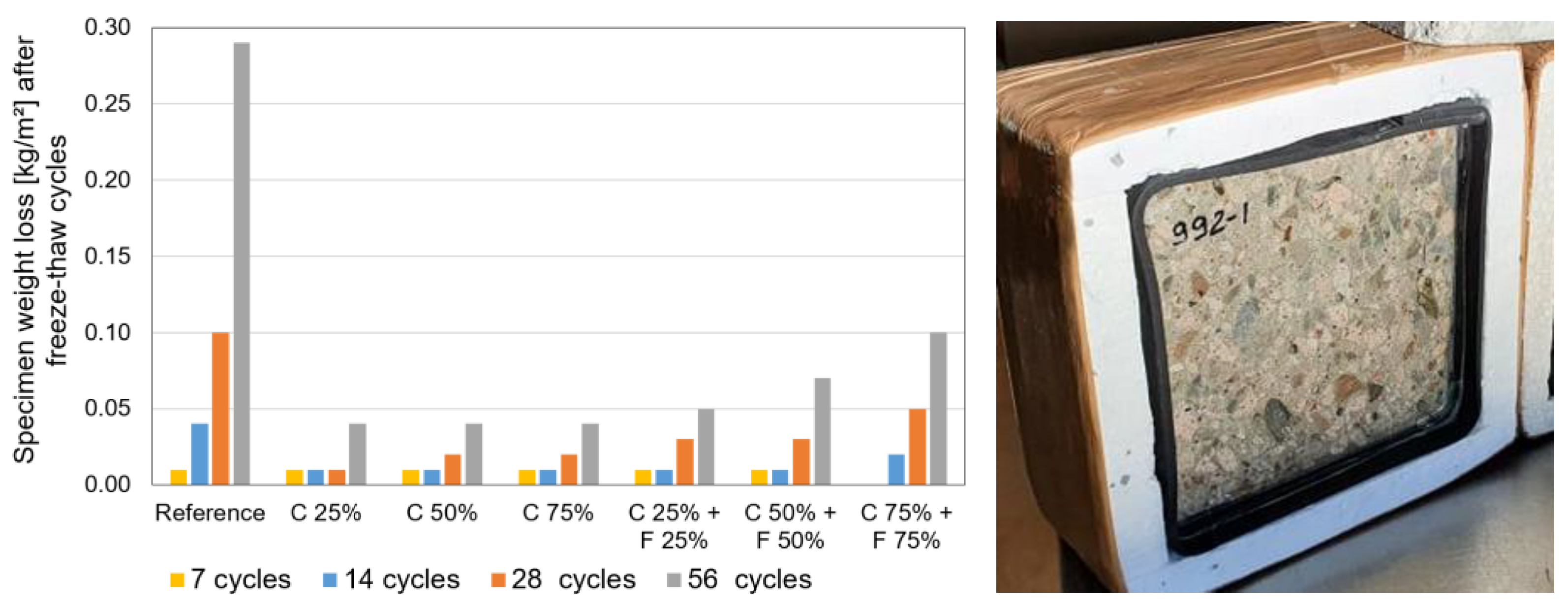
33]. Freezing and thawing of the test specimens was carried out according to the standard in a climate chamber with forced air circulation. The freezing agent on the surface to be exposed to the test was a 3 mm thick layer of distilled water. The duration of one freeze-thaw cycle was 24 h. After the number of cycles of 7, 14, 28 and 56, the loss in mass of each test specimen was determined, and the total amount of crushed material, Σ M (g), and the total loss in mass per unit area, Σ S [kg/m
2], were calculated. The average mass loss per unit area of the two test specimens is given in
Figure 5.
Although the average mass loss of recycled calcium silica aggregates (22–24%) in the frost resistance test according to EN 1367-1 was ~10 times higher than the corresponding property of primary limestone aggregates (2.2% in
Table 2), the frost resistance tests of both whole silicate bricks (through comparison of compressive strength) and concrete made of silicate aggregates show that the use of silicate aggregates as an aggregate for concrete does not significantly impair the frost resistance of concrete. The values given in
Figure 5 can be compared with the compliance criteria of EVS 814 [
33], which are also given after 56 cycles with values ranging from 0.10 to 1.0 kg/m
2 (depending on the frost resistance class and service life). It can be concluded that all concretes made with partial replacement of aggregates can be used in EN 206 [
23] environmental class XF1 and XF3 (vertical and horizontal concrete surfaces exposed to rain and freezing). Concrete made of secondary (recycled calcium silicate brick masonry) aggregate had lower mass loss compared to the reference concrete made with conventional primary aggregates limestone and sand.
Experimental results on calcium silicate aggregate concretes show that the partial use of calcium silicate (both coarse and fine) aggregate from demolished buildings is realistic and very promising circular options for the future circular construction even in frost resistant concrete.
3.2. Properties of Mortar Made of Calcium Silicate Aggregate
Table 4 shows that replacing natural sand with lower-density recycled crushed calcium silicate sand (CCSS) also reduces the bulk density of the aggregate, which is probably going to affect the density and strength of the hardened mortar.
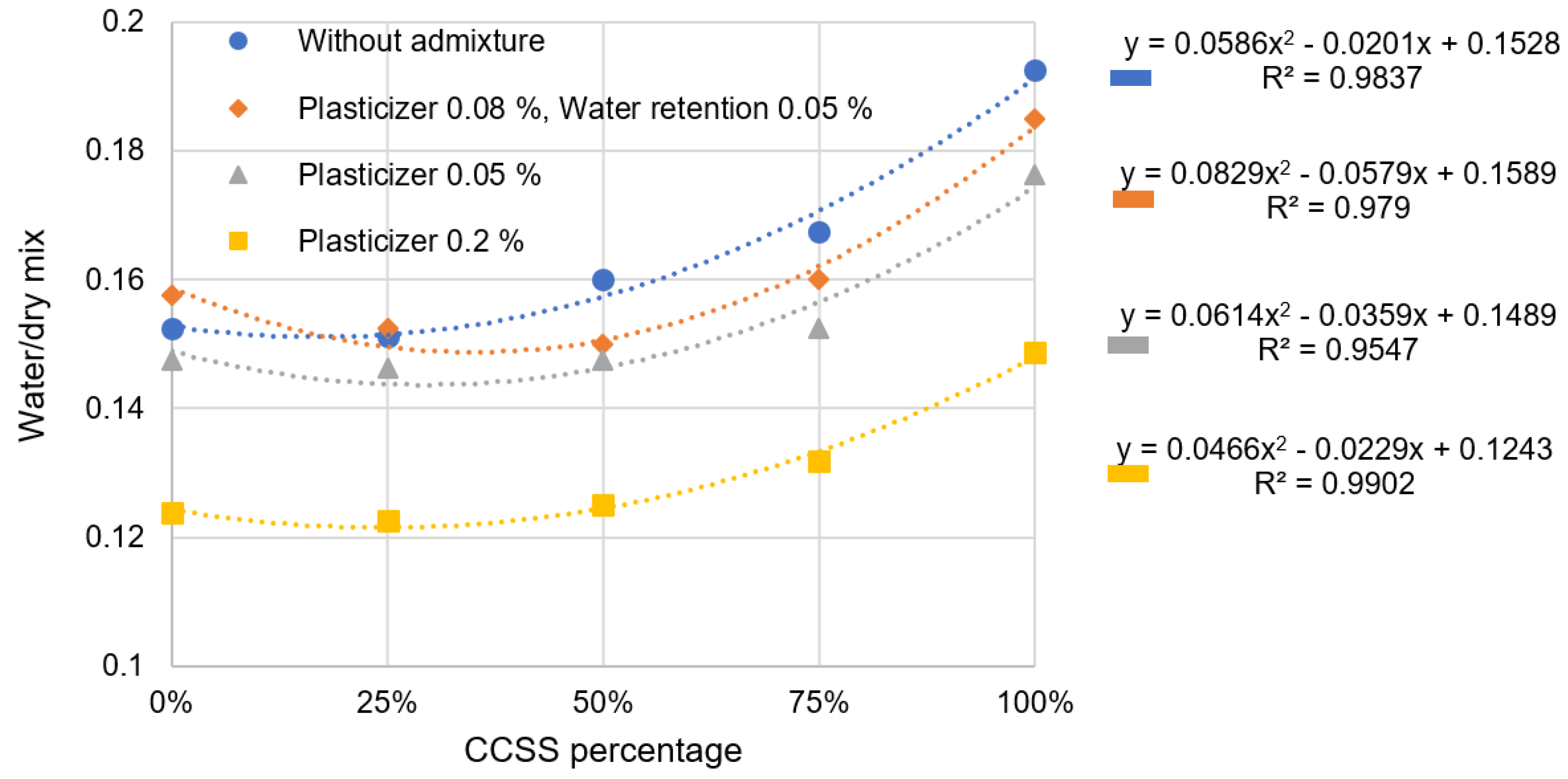
Figure 6 shows a clear trend that the more CCSS used in a mixture, the higher its water demand. This trendline can be seen in the water demand of each mixture and is consistent with the number of admixtures. Based on dry mixes without additives, the water demand of a 100% CCSS blend is about 21% higher than that of a 0% CCSS mix. This means that a mixture with only CCSS requires 1/5 times more water than a mixture with natural sand. The same result was obtained by Huang [
35], Ceylan [
36], Omur [
37], and Fang [
38], who recycled different types of crushed aggregates, which were porous and highly water-absorbent.
Comparing 0% and 25% CCSS mixes, 0% CCSS mixes have a slightly higher water demand than 25% CCSS mixes—about 1%, which is statistically irrelevant. The reduction in water demand is more pronounced in mixtures with 0.08% plasticizer and 0.05% water retention additive. Naturally, the water demand decreases with the plasticizer. It is remarkable that the effect increases with higher CCSS amounts.
The increase in water demand can be attributed to a number of different factors. Several different factors can be expected:
The water absorption of CCSS is several times higher than that of natural silica sand typically used in masonry mortars.
The CCSS grain is more angular, so more water is needed to reduce its cohesiveness.
CCSS has a high proportion of fines in its granular composition, which increases the geometrical area of the sand grains and the water demand of the mixture.
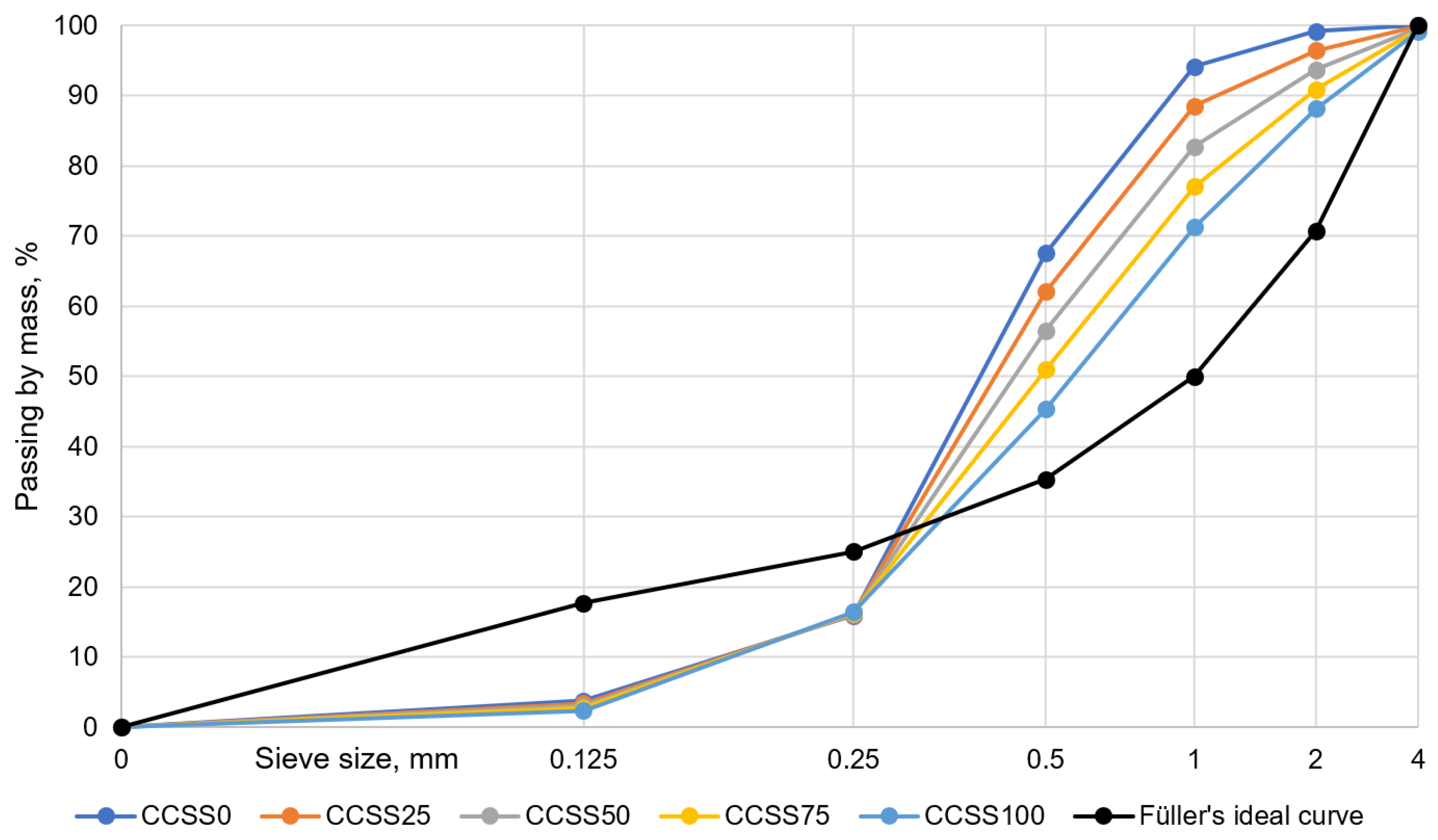
CCSS was used in mixtures in non-fractionated (all-in) form (shown as “CCSS
0” in
Table 4). Comparing the grain size compositions of natural sand and CCSS, the natural sand has a small amount of the 1–4 mm fraction. In contrast, the CCSS has almost 5 times as many 1–4 mm fractions. In the tested natural sand, ~30% of the weight consists of particles larger than 0.5 mm and more than half of the weight is between 0.25 and 0.5 mm. This is well reflected in the line placement and the difference in slope in the
Figure 7. In CCSS, the slope is lower and the sieve curve lower, and in natural sand, the slope is higher and the sieve curve higher.
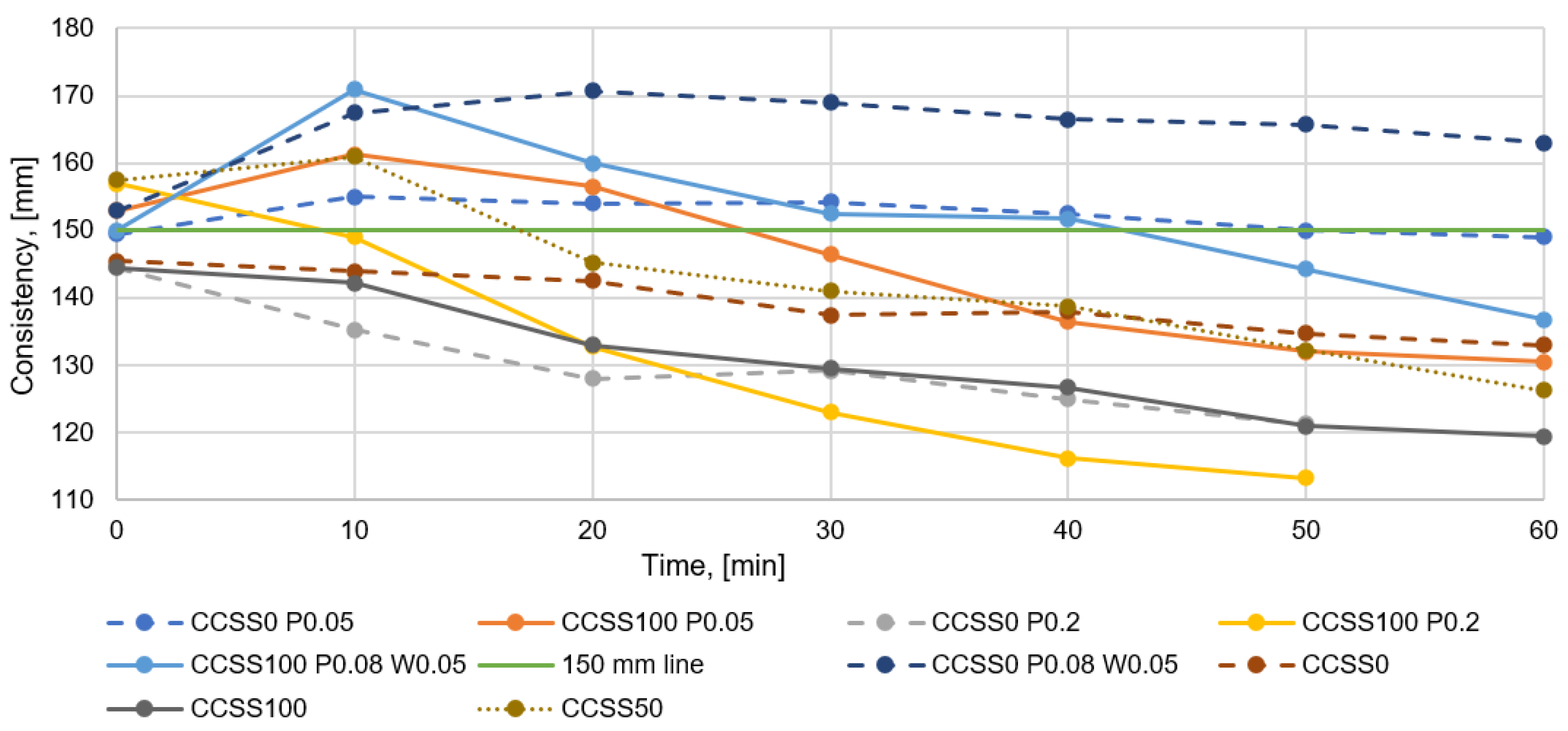
Analyzing
Figure 8, all the mixtures tested without admixtures lose consistency over time, which is the expected result. The more CCSS is used in the dry mix, the faster the consistency of the mix disappears. This is due to the high water absorption of CCSS, which absorbs the free water in the mix over time. Exceptionally, if 50% CCSS is used, the consistency will increase in the first 10 min and then decrease in a similar way to other mixes. A small initial increase in consistency is also observed in other tests. The anomaly in the current test may be due to an excessively high initial consistency of the mixture of 158 mm (by default, the study aimed for consistency of 150 mm ± 10 mm). There is a larger amount of water in the mix which cannot all be absorbed by the CCSS and the consistency is higher over time.
Mixtures without admixtures give a good indication of the effect of the CCSS proportion on the consistency of the mix. In order to obtain further confirmation of the issue of whether increasing the proportion of CCSS in a mixture leads to a faster loss of consistency, it is necessary to compare the effect of the proportion of CCSS in mixtures with admixtures on consistency.
Examining the results obtained when using the minimum amount of plasticizer (
Figure 8), a trendline can also be seen that the more CCSS is used in the mix, the faster the consistency of the mix is lost. However, some differences can be seen where, for example, with the use of a minimum amount of plasticizer, the consistency of all the mixtures tested increases slightly during the first ten minutes and then starts to decrease. The reason why the consistency may have increased in the first minutes is related to the solubility time of the admixture in the mix. If the admixture does not dissolve quickly enough in the mix, the initial consistency on the flow table may change in later testing. Nevertheless, it can be seen that replacing natural silica sand with CCSS leads to a faster loss of consistency in the mixture.
At the maximum use of the plasticizer (
Figure 8), the trend lines are more similar to the mixtures without admixture, where it can be seen that mixes with 75% and 100% CCSS lose their consistency faster. Mixes made with the maximum amount of plasticizer lose consistency more quickly because there is less water in the mix for CCSS to absorb.
Regarding mixes where 0.08% plasticizer and 0.05% water retention admixture were used, a similar trend is also visible for the previously mentioned blends, where the consistency of the mix decreased faster with increasing CCSS content. Overall, it can be seen that the water retention admixture acts as a water retainer in the fresh mix and the CCSS does not absorb excessive amounts of water. The loss of consistency of CCSS can be achieved similar to, and even better than, that of the etalon mix in the algepio mix. Similar to the minimum amount of plasticizer, a trend is also seen where the consistency of the combined mixture increases in the first 10–30 min and then starts to decrease. This may also be due to the solubility time of the admixtures, as observed with the minimum amount of plasticizer.
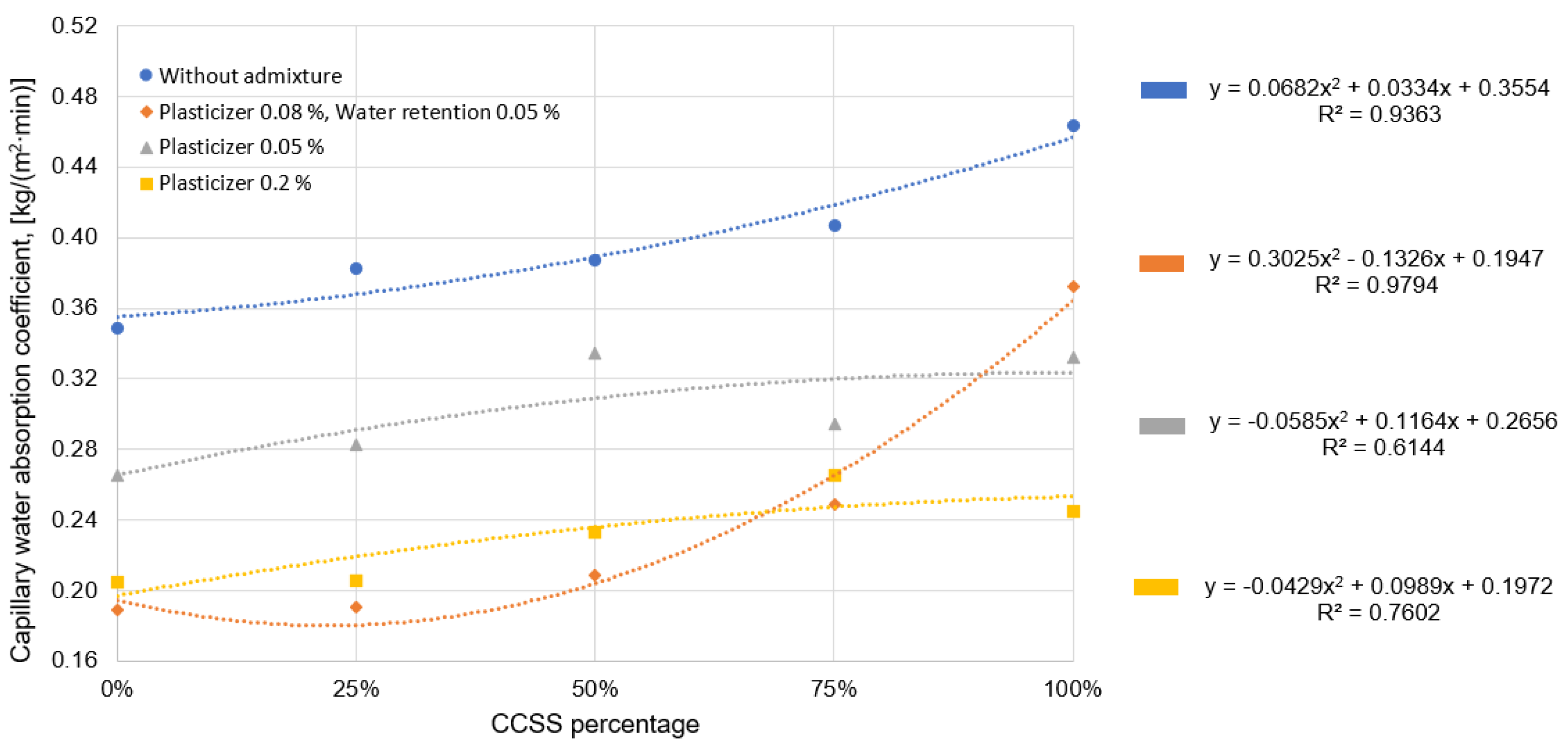
Figure 9 shows the general trend that the more CCSS is used in the mixture, the higher the water absorption of the mortar. CCSS with higher water absorption increases the water absorption of mortar. As in the case of high water absorption of CCSS, the slags used in the Omur study [
37] had high water absorption, which also increased the water absorption of the hardened mortar. The water absorption in mortar is also increased by voids in the mixture due to large fractions of silica sand. Omur [
37] came to a similar conclusion in his study, where he found that more angular aggregate grains create more voids in the mix and increase the water absorption of the hardened mortar.
Comparing mortar with 0% silica sand to mortar with 100% CCSS, the difference in water absorption is almost 25%. The difference in water absorption decreases with increasing the amount of plasticizer, but is still 16% higher for 100% CCSS mortar than for 0% CCSS mortar.
There is a significant difference in water absorption between 0.08% plasticizer and 0.05% water retention additive, where the difference in water absorption is almost 50% for a mixture made with natural sand only and a mixture made with 100% CCSS. The large difference may be since the 100% CCSS mix did not compact sufficiently during molding (
Figure 10).
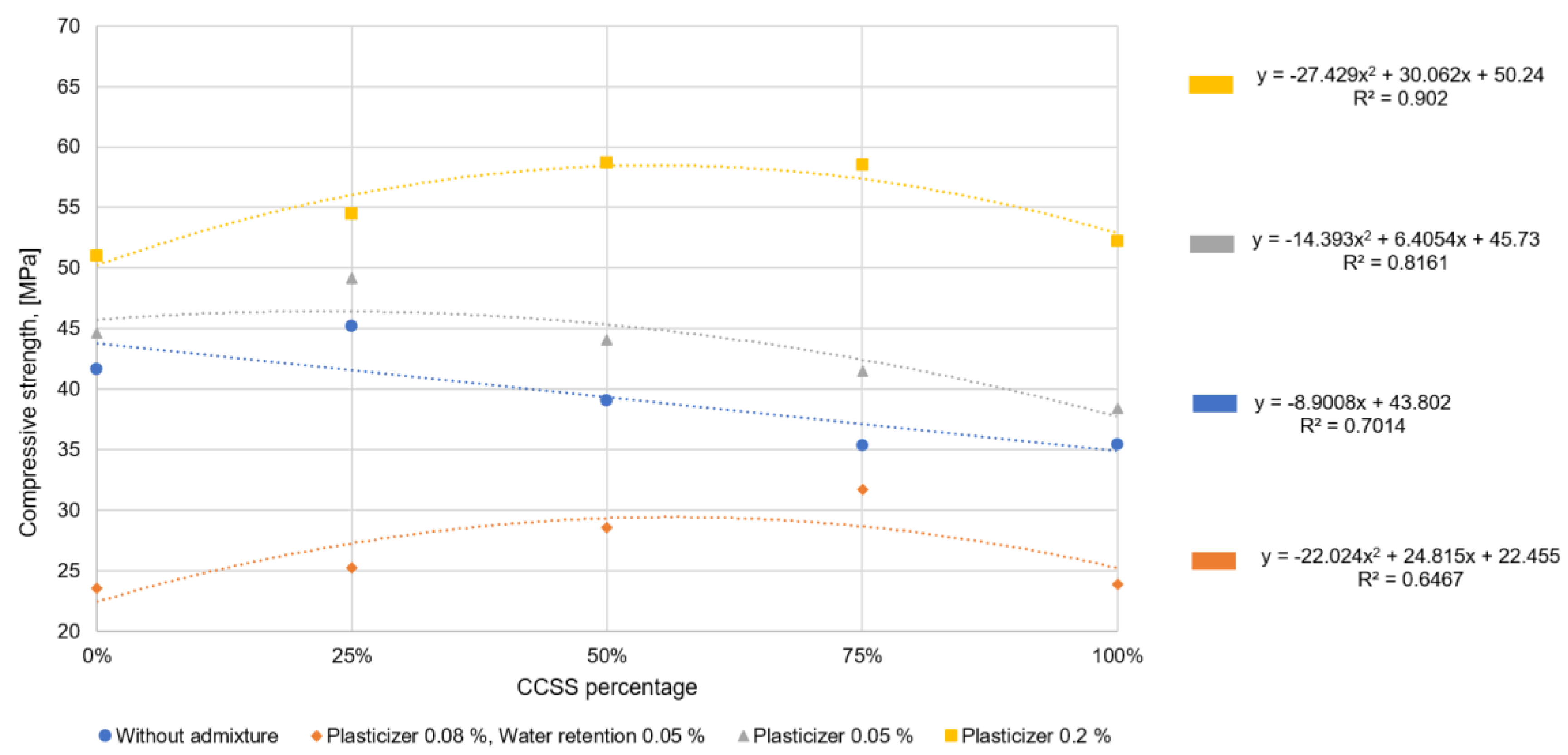
The addition of a small amount of CCSS does not increase the water demand and does not significantly reduce the mortar density. The compressive strength, however, increases with all additive dosages. This is probably related to pozzolanic reactions. At higher CCSS dosages, the density of the hardened mortar decreases significantly and the water/solids factor increases, which leads to a decrease in strength. Similar results have been obtained in studies [
39,
40] where crushed and ground glass has been used as a recycled aggregate.