Synthesis and Characterization of Polyaniline Emeraldine Salt (PANI-ES) Colloids Using Potato Starch as a Stabilizer to Enhance the Physicochemical Properties and Processability
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Polyaniline-Starch Particles (PANI-ES/Starch)
2.3. Characterizations of the Materials
2.4. Membrane Preparation
2.5. Characterization of the Membranes
3. Results and Discussion
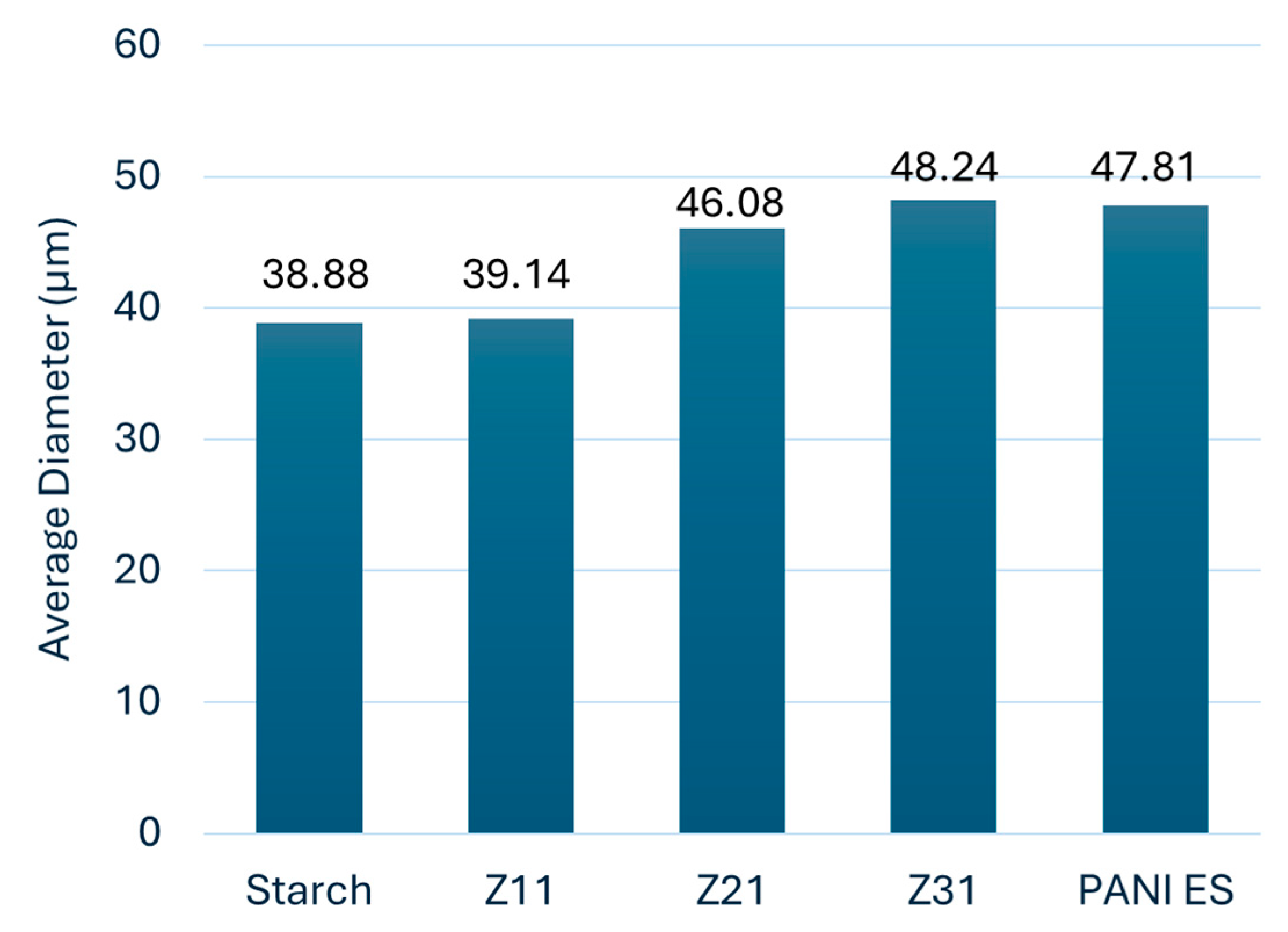
3.1. Particle Morphology and Dimensions
3.2. Solubility
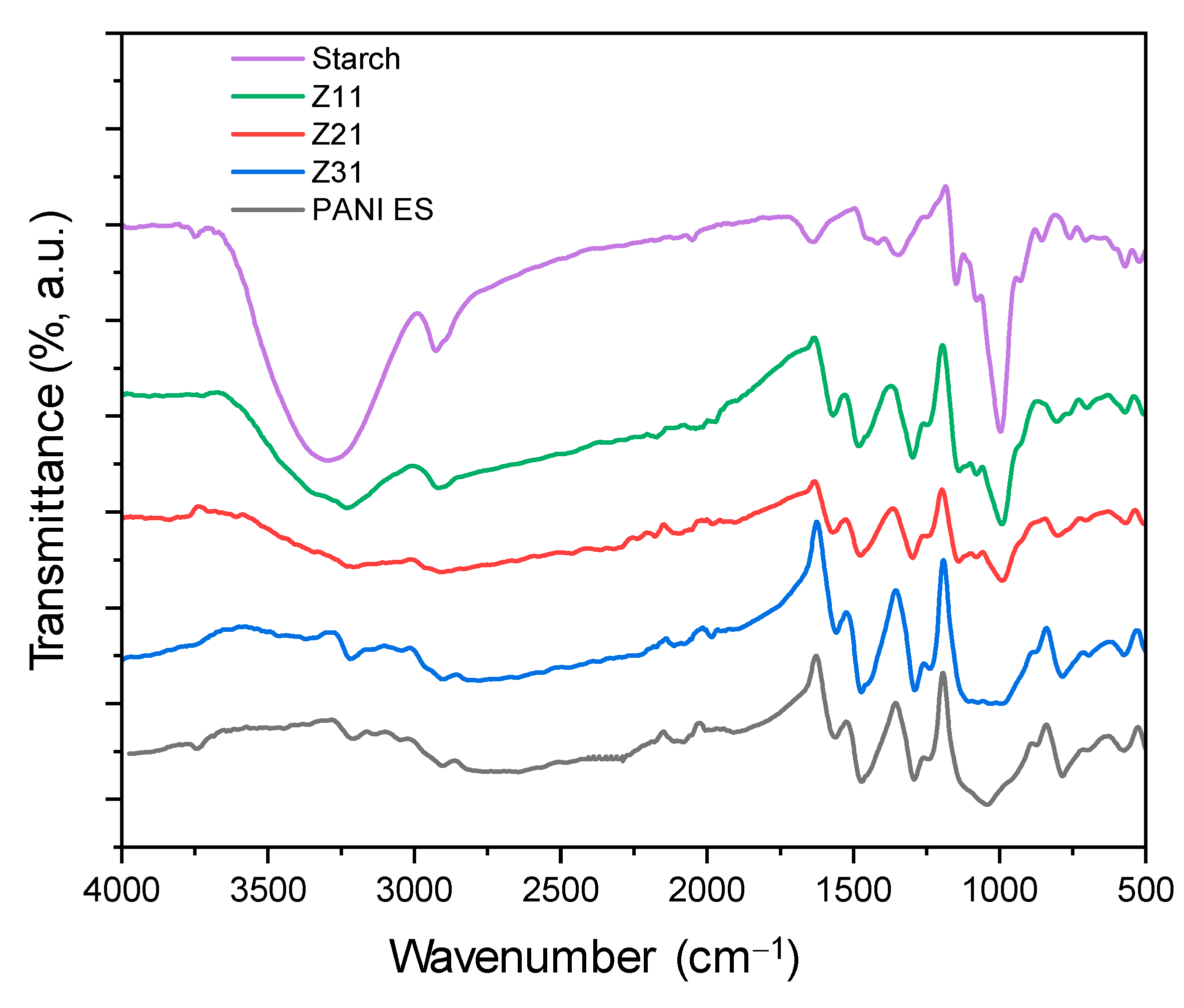
3.3. FTIR-ATR
3.4. UV-Vis
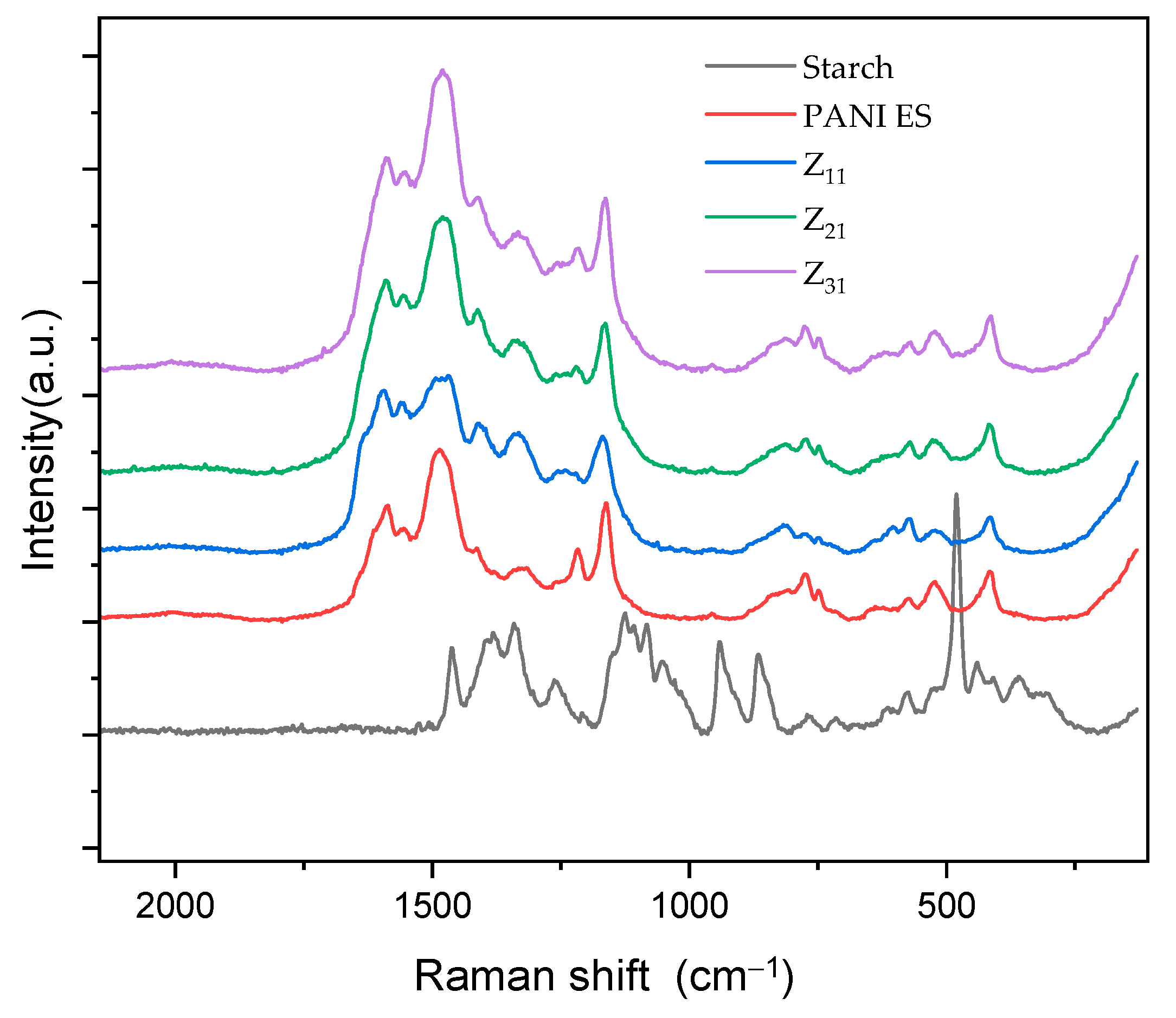
3.5. RAMAN
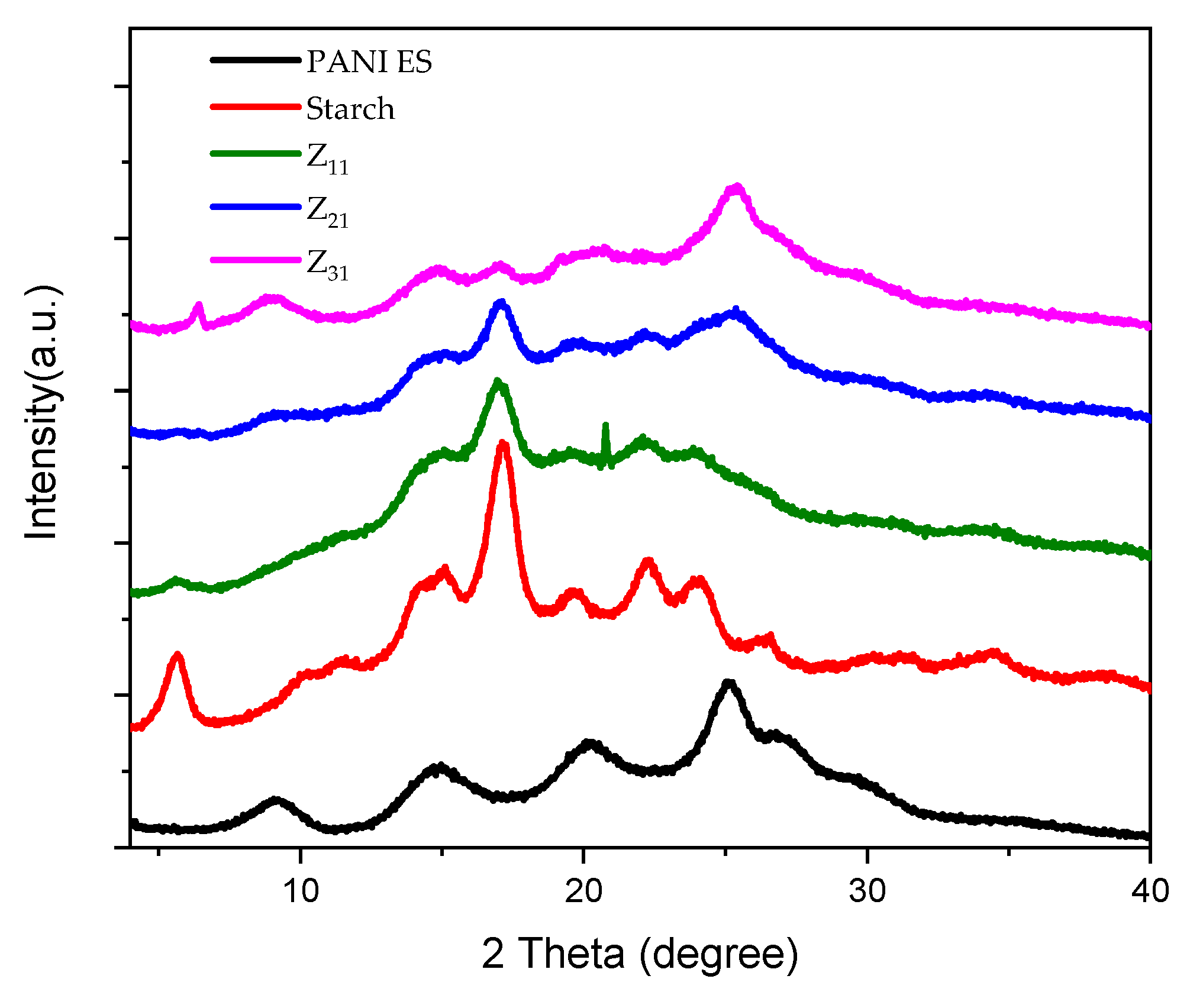
3.6. X-ray Diffraction Pattern (XRD)
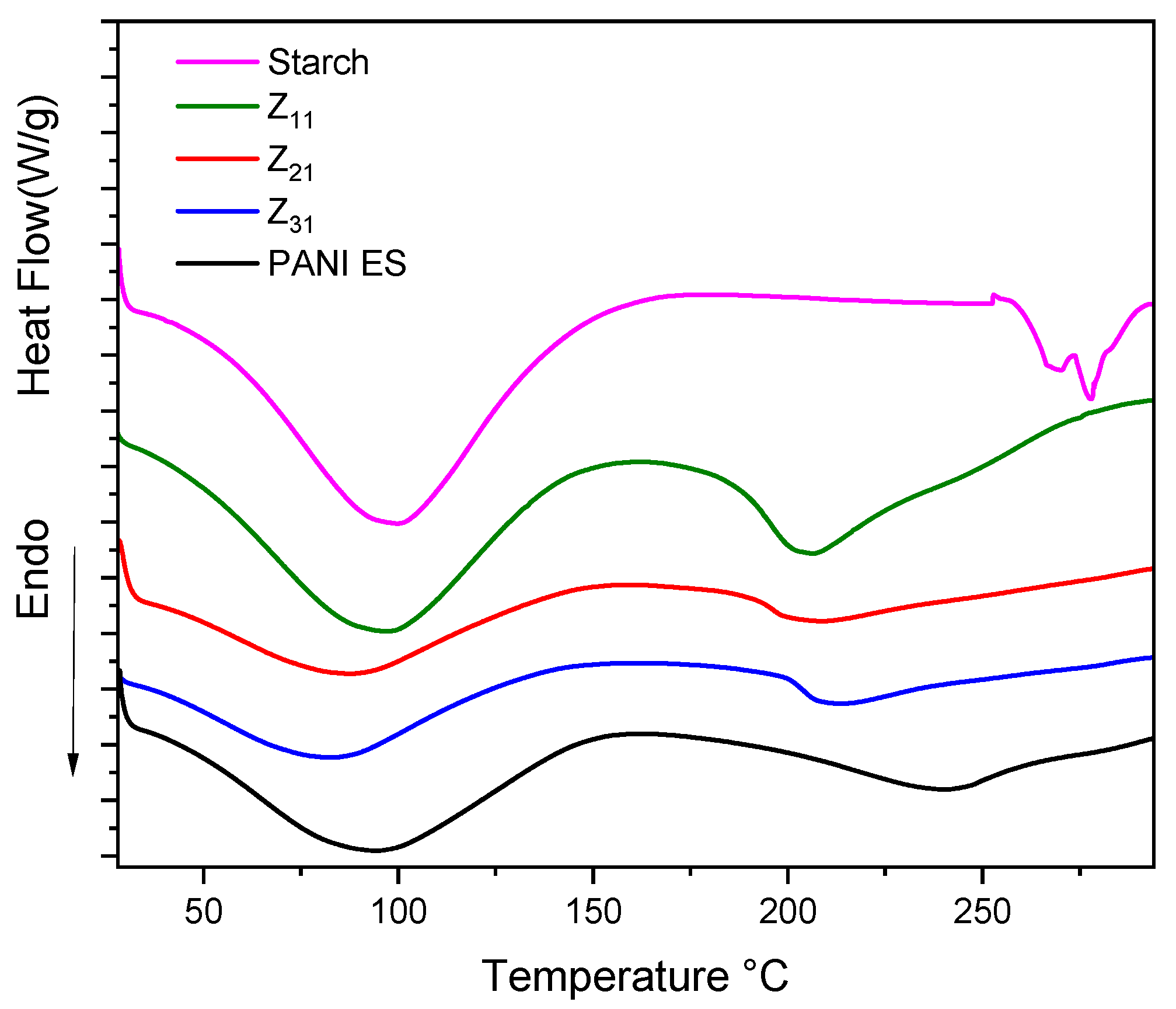
3.7. Thermal Characterization
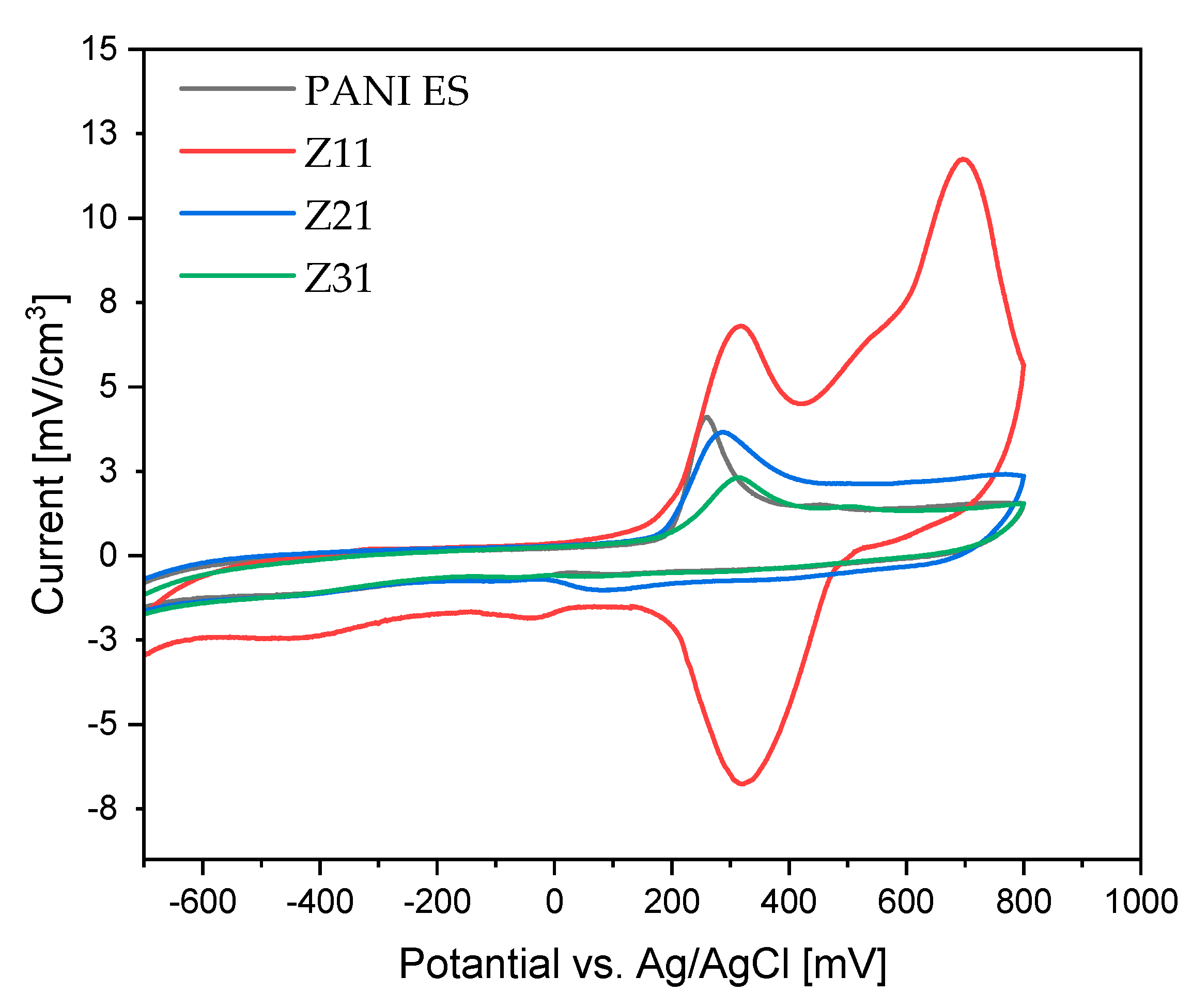
3.8. Cyclic Voltammetry


3.9. Embedding of PANI-ES/Starch into a Polymeric Membrane
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marcilla, R.; Ochoteco, E.; Pozo-Gonzalo, C.; Grande, H.; Pomposo, J.A.; Mecerreyes, D. New Organic Dispersions of Conducting Polymers Using Polymeric Ionic Liquids as Stabilizers. Macromol. Rapid Commun. 2005, 26, 1122–1126. [Google Scholar] [CrossRef]
- Cawdery, N.; Obey, T.M.; Vincent, B. Colloidal Dispersions of Electrically Conducting Polypyrrole Particles in Various Media. J. Chem. Soc. Chem. Commun. 1988, 17, 1189–1190. [Google Scholar] [CrossRef]
- Digar, M.L.; Bhattacharyya, S.N.; Mandal, B.M. Conducting Polypyrrole Particles Dispersible in Both Aqueous and Non-Aqueous Media. J. Chem. Soc. Chem. Commun. 1992, 1, 18–20. [Google Scholar] [CrossRef]
- Stejskal, J.; Sapurina, I. Polyaniline: Thin Films and Colloidal Dispersions (IUPAC Technical Report). Pure Appl. Chem. 2005, 77, 815–826. [Google Scholar] [CrossRef]
- Lu, Y.; Pich, A.; Adler, H.P. Synthesis and Characterization of Polypyrrole Dispersions Prepared with Different Dopants. Macromol. Symp. 2004, 210, 411–417. [Google Scholar] [CrossRef]
- Mandal, T.K.; Mandal, B.M. Ethylhydroxyethylcellulose Stabilized Polypyrrole Dispersions. Polymer 1995, 36, 1911–1913. [Google Scholar] [CrossRef]
- Kwon, J.-Y.; Kim, E.-Y.; Kim, H.-D. Preparation and Properties of Waterborne-Polyurethane Coating Materials Containing Conductive Polyaniline. Macromol. Res. 2004, 12, 303–310. [Google Scholar] [CrossRef]
- Bjorklund, R.B.; Liedberg, B. Electrically Conducting Composites of Colloidal Polypyrrole and Methylcellulose. J. Chem. Soc. Chem. Commun. 1986, 16, 1293–1295. [Google Scholar] [CrossRef]
- Stejskal, J.; Kratochvíl, P.; Helmstedt, M. Polyaniline Dispersions. 5. Poly(Vinyl Alcohol) and Poly(N-Vinylpyrrolidone) as Steric Stabilizers. Langmuir 1996, 12, 3389–3392. [Google Scholar] [CrossRef]
- Stejskal, J.; Kratochvíl, P.; Armes, S.P.; Lascelles, S.F.; Riede, A.; Helmstedt, M.; Prokeš, J.; Křivka, I. Polyaniline Dispersions. 6. Stabilization by Colloidal Silica Particles. Macromolecules 1996, 29, 6814–6819. [Google Scholar] [CrossRef]
- Armes, S.P.; Miller, J.F.; Vincent, B. Aqueous Dispersions of Electrically Conducting Monodisperse Polypyrrole Particles. J. Colloid Interface Sci. 1987, 118, 410–416. [Google Scholar] [CrossRef]
- Ohsawa, T.; Kabata, T.; Kimura, O.; Nakajima, S.; Nishihara, H.; Yoshino, K. Non-Linear Electric Properties of Polyaniline Doped with Organic Acceptors. Synth. Met. 1993, 57, 4842–4847. [Google Scholar] [CrossRef]
- Subathira, A.; Meyyappan, R. Inhibition of Corrosion of Steel Alloy Using Polyaniline Conducting Polymer Coatings. Int. J. Chem. Sci. 2010, 8, 2563–2574. [Google Scholar]
- Li, Y.; Ying, B.; Hong, L.; Yang, M. Water-Soluble Polyaniline and Its Composite with Poly(Vinyl Alcohol) for Humidity Sensing. Synth. Met. 2010, 160, 455–461. [Google Scholar] [CrossRef]
- Shannon, K.; Fernandez, J.E. Preparation and Properties of Water-Soluble, Poly(Styrenesulfonic Acid)-Doped Polyaniline. J. Chem. Soc. Chem. Commun. 1994, 5, 643–644. [Google Scholar] [CrossRef]
- Tallman, D.E.; Wallace, G.G. Preparation and Preliminary Characterization of a Poly(4-Vinylpyridine) Complex of a Water-Soluble Polyaniline. Synth. Met. 1997, 90, 13–18. [Google Scholar] [CrossRef]
- Winnik, M.; Lukas, R.; Chen, W.; Furlong, P. Studies of the Dispersion Polymerisation of Methyl Methacrylate in Nonaqueous Media. Makromol. Chem. Macromol. Symp. 1987, 10–11, 483–501. [Google Scholar]
- Arenas, M.; Sánchez, G.; Martínez-Álvarez, O.; Castaño, V. Electrical and Morphological Properties of Polyaniline–Polyvinyl Alcohol in Situ Nanocomposites. Compos. Part B Eng. 2014, 56, 857–861. [Google Scholar] [CrossRef]
- Riede, A.; Helmstedt, M.; Riede, V.; Stejskal, J. Polyaniline Dispersions. 9. Dynamic Light Scattering Study of Particle Formation Using Different Stabilizers. Langmuir 1998, 14, 6767–6771. [Google Scholar] [CrossRef]
- Skotheim, T.A.; Reynolds, J. Conjugated Polymers: Theory, Synthesis, Properties, and Characterization; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Eisazadeh, H.; Gilmore, K.; Hodgson, A.; Spinks, G.; Wallace, G. Electrochemical Production of Conducting Polymer Colloids. Colloids Surf. A Physicochem. Eng. Asp. 1995, 103, 281–288. [Google Scholar] [CrossRef]
- Bober, P.; Humpolíček, P.; Syrový, T.; Capáková, Z.; Syrová, L.; Hromádková, J.; Stejskal, J. Biological Properties of Printable Polyaniline and Polyaniline–Silver Colloidal Dispersions Stabilized by Gelatin. Synth. Met. 2017, 232, 52–59. [Google Scholar] [CrossRef]
- Eisazadeh, H.; Spinks, G.; Wallace, G. Electrochemical Production of Polypyrrole Colloids. Polymer 1994, 35, 3801–3803. [Google Scholar] [CrossRef]
- Kašpárková, V.; Jasenská, D.; Capáková, Z.; Maráková, N.; Stejskal, J.; Bober, P.; Lehocký, M.; Humpolíček, P. Polyaniline Colloids Stabilized with Bioactive Polysaccharides: Non-Cytotoxic Antibacterial Materials. Carbohydr. Polym. 2019, 219, 423–430. [Google Scholar] [CrossRef]
- Cruz-Silva, R.; Arizmendi, L.; Del-Angel, M.; Romero-Garcia, J. pH-and Thermosensitive Polyaniline Colloidal Particles Prepared by Enzymatic Polymerization. Langmuir 2007, 23, 8–12. [Google Scholar] [CrossRef]
- Cruz-Silva, R.; Escamilla, A.; Nicho, M.; Padron, G.; Ledezma-Perez, A.; Arias-Marin, E.; Moggio, I.; Romero-Garcia, J. Enzymatic Synthesis of pH-Responsive Polyaniline Colloids by Using Chitosan as Steric Stabilizer. Eur. Polym. J. 2007, 43, 3471–3479. [Google Scholar] [CrossRef]
- Chattopadhyay, D.; Mandal, B.M. Methyl Cellulose Stabilized Polyaniline Dispersions. Langmuir 1996, 12, 1585–1588. [Google Scholar] [CrossRef]
- Luong, N.D.; Korhonen, J.T.; Soininen, A.J.; Ruokolainen, J.; Johansson, L.-S.; Seppälä, J. Processable Polyaniline Suspensions through in Situ Polymerization onto Nanocellulose. Eur. Polym. J. 2013, 49, 335–344. [Google Scholar] [CrossRef]
- Amarnath, C.A.; Venkatesan, N.; Doble, M.; Sawant, S.N. Water Dispersible Ag@ Polyaniline-Pectin as Supercapacitor Electrode for Physiological Environment. J. Mater. Chem. B 2014, 2, 5012–5019. [Google Scholar] [CrossRef]
- Djellali, S.; Touati, A.; Kebaili, M.; Sahraoui, R. Synthesis of Polyaniline/Pectin Biocomposite and Its Efficiency as Adsorbent for Methylene Blue Removal. In Recent Advances in Environmental Science from the Euro-Mediterranean and Surrounding Regions, 2nd ed.; Springer International Publishing: Berlin/Heidelberg, Germany, 2021. [Google Scholar]
- Aridi, N.; Sapuan, S.; Zainudin, E.; AL-Oqla, F.M. Mechanical and Morphin ological Properties of Injection-Molded Rice Husk Polypropylene Composites. Int. J. Polym. Anal. Charact. 2016, 21, 305–313. [Google Scholar] [CrossRef]
- Schirp, A.; Barrio, A. Fire Retardancy of Polypropylene Composites Reinforced with Rice Husks: From Oxygen Index Measurements and Cone Calorimetry to Large-scale Single-burning-item Tests. J. Appl. Polym. Sci. 2018, 135, 46654. [Google Scholar] [CrossRef]
- Salasinska, K.; Barczewski, M.; Górny, R.; Kloziński, A. Evaluation of Highly Filled Epoxy Composites Modified with Walnut Shell Waste Filler. Polym. Bull. 2018, 75, 2511–2528. [Google Scholar] [CrossRef]
- Singh, T.; Gangil, B.; Patnaik, A.; Biswas, D.; Fekete, G. Agriculture Waste Reinforced Corn Starch-Based Biocomposites: Effect of Rice Husk/Walnut Shell on Physicomechanical, Biodegradable and Thermal Properties. Mater. Res. Express 2019, 6, 045702. [Google Scholar] [CrossRef]
- Jiménez, A.; Fabra, M.J.; Talens, P.; Chiralt, A. Physical Properties and Antioxidant Capacity of Starch–Sodium Caseinate Films Containing Lipids. J. Food Eng. 2013, 116, 695–702. [Google Scholar] [CrossRef]
- Yan, Q.; Hou, H.; Guo, P.; Dong, H. Effects of Extrusion and Glycerol Content on Properties of Oxidized and Acetylated Corn Starch-Based Films. Carbohydr. Polym. 2012, 87, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Azmi, N.S.; Kadir Bahsa, R.; Othman, S.H.; Mohammed, M.A.P. Characterization of Antioxidant Tapioca Starch/Polyaniline Composites Film Prepared Using Solution Casting Method. Food Res. 2019, 3, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.D.; Fradinho, P.; Rodríguez, P.; Falqué, E.; Santos, V.; Domínguez, H. Biorefinery Concept for Discarded Potatoes: Recovery of Starch and Bioactive Compounds. J. Food Eng. 2020, 275, 109886. [Google Scholar] [CrossRef]
- Saikia, J.P.; Banerjee, S.; Konwar, B.K.; Kumar, A. Biocompatible Novel Starch/Polyaniline Composites: Characterization, Anti-Cytotoxicity and Antioxidant Activity. Colloids Surf. B Biointerfaces 2010, 81, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Lukasiewicz, M.; Ptaszek, P.; Ptaszek, A.; Bednarz, S. Polyaniline–Starch Blends: Synthesis, Rheological, and Electrical Properties. Starch-Stärke 2014, 66, 583–594. [Google Scholar] [CrossRef]
- Pandi, N.; Sonawane, S.H.; Gumfekar, S.P.; Kola, A.K.; Borse, P.H.; Ambade, S.B.; Guptha, S.; Ashokkumar, M. Electrochemical Performance of Starch-Polyaniline Nanocomposites Synthesized by Sonochemical Process Intensification. J. Renew. Mater. 2019, 7, 1279. [Google Scholar] [CrossRef]
- Janaki, V.; Vijayaraghavan, K.; Oh, B.-T.; Lee, K.-J.; Muthuchelian, K.; Ramasamy, A.; Kamala-Kannan, S. Starch/Polyaniline Nanocomposite for Enhanced Removal of Reactive Dyes from Synthetic Effluent. Carbohydr. Polym. 2012, 90, 1437–1444. [Google Scholar] [CrossRef]
- Gautam, V.; Srivastava, A.; Singh, K.P.; Yadav, V.L. Preparation and Characterization of Polyaniline, Multiwall Carbon Nanotubes, and Starch Bionanocomposite Material for Potential Bioanalytical Applications. Polym. Compos. 2017, 38, 496–506. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Saadat, Y.; Abdolbaghi, S.; Afshar-Taromi, F.; Hosseinzadeh, A. Shape of the Particles Produced by Seeded Dispersion Polymerization of Styrene. Colloid J. 2014, 76, 104–112. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Shin, C.H.; Han, S. Dispersion Polymerization of Polystyrene Particles Using Alcohol as Reaction Medium. Nanoscale Res. Lett. 2016, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- Amalina, A.N.; Suendo, V.; Reza, M.; Milana, P.; Sunarya, R.R.; Adhika, D.R.; Tanuwijaya, V.V. Preparation of Polyaniline Emeraldine Salt for Conducting-Polymer-Activated Counter Electrode in Dye Sensitized Solar Cell (DSSC) Using Rapid-Mixing Polymerization at Various Temperature. Bull. Chem. React. Eng. Catal. 2019, 14, 521–528. [Google Scholar] [CrossRef]
- Rai, R.; Roether, J.A.; Boccaccini, A.R. Polyaniline Based Polymers in Tissue Engineering Applications: A Review. Prog. Biomed. Eng. 2022, 4, 042004. [Google Scholar] [CrossRef]
- Nazarzadeh, Z.E.; Najafi, M.P.; Azariyan, E.; Sharifian, I. Conductive and Biodegradable Polyaniline/Starch Blends and Their Composites with Polystyrene. Iran. Polym. J. 2011, 20, 319–328. [Google Scholar]
- Russo, F.; Tiecco, M.; Galiano, F.; Mancuso, R.; Gabriele, B.; Figoli, A. Launching Deep Eutectic Solvents (DESs) and Natural Deep Eutectic Solvents (NADESs), in Combination with Different Harmless Co-Solvents, for the Preparation of More Sustainable Membranes. J. Membr. Sci. 2022, 649, 120387. [Google Scholar] [CrossRef]
- Boudjelida, S.; Djellali, S.; Ferkous, H.; Benguerba, Y.; Chikouche, I.; Carraro, M. Physicochemical Properties and Atomic-Scale Interactions in Polyaniline (Emeraldine Base)/Starch Bio-Based Composites: Experimental and Computational Investigations. Polymers 2022, 14, 1505. [Google Scholar] [CrossRef] [PubMed]
- Sulimenko, T.; Stejskal, J.; Křivka, I.; Prokeš, J. Conductivity of Colloidal Polyaniline Dispersions. Eur. Polym. J. 2001, 37, 219–226. [Google Scholar] [CrossRef]
- Do Nascimento, G.M.; Silva, C.H.; Temperini, M.L. Spectroscopic Characterization of the Structural Changes of Polyaniline Nanofibers after Heating. Polym. Degrad. Stab. 2008, 93, 291–297. [Google Scholar] [CrossRef]
- Furukawa, Y.; Ueda, F.; Hyodo, Y.; Harada, I.; Nakajima, T.; Kawagoe, T. Vibrational Spectra and Structure of Polyaniline. Macromolecules 1988, 21, 1297–1305. [Google Scholar] [CrossRef]
- Lee, D.; Char, K. Thermal Degradation Behavior of Polyaniline in Polyaniline/Na+-Montmorillonite Nanocomposites. Polym. Degrad. Stab. 2002, 75, 555–560. [Google Scholar] [CrossRef]
- Hou, X.; Zhou, Y.; Liu, Y.; Wang, L.; Wang, J. Coaxial Electrospun Flexible PANI//PU Fibers as Highly Sensitive pH Wearable Sensor. J. Mater. Sci. 2020, 55, 16033–16047. [Google Scholar] [CrossRef]
- Gautam, V.; Srivastava, A.; Singh, K.P.; Yadav, V.L. Vibrational and Gravimetric Analysis of Polyaniline/Polysaccharide Composite Materials. Polym. Sci. Ser. A 2016, 58, 206–219. [Google Scholar] [CrossRef]
- Laska, J.; Widlarz, J. Spectroscopic and Structural Characterization of Low Molecular Weight Fractions of Polyaniline. Polymer 2005, 46, 1485–1495. [Google Scholar] [CrossRef]
- Masters, J.; Sun, Y.; MacDiarmid, A.; Epstein, A. Polyaniline: Allowed Oxidation States. Synth. Met. 1991, 41, 715–718. [Google Scholar] [CrossRef]
- Šeděnková, I.; Trchová, M.; Stejskal, J. Thermal Degradation of Polyaniline Films Prepared in Solutions of Strong and Weak Acids and in Water–FTIR and Raman Spectroscopic Studies. Polym. Degrad. Stab. 2008, 93, 2147–2157. [Google Scholar] [CrossRef]
- Ćirić-Marjanović, G.; Trchová, M.; Stejskal, J. The Chemical Oxidative Polymerization of Aniline in Water: Raman Spectroscopy. J. Raman Spectrosc. Int. J. Orig. Work. Asp. Raman Spectrosc. Incl. High. Order Process. Brillouin Rayleigh Scatt. 2008, 39, 1375–1387. [Google Scholar] [CrossRef]
- Izumi, C.M.; Brito, H.F.; Ferreira, A.M.D.; Constantino, V.R.; Temperini, M.L. Spectroscopic Investigation of the Interactions between Emeraldine Base Polyaniline and Eu (III) Ions. Synth. Met. 2009, 159, 377–384. [Google Scholar] [CrossRef]
- do Nascimento, G.M.; Silva, C.H.; Izumi, C.M.; Temperini, M.L. The Role of Cross-Linking Structures to the Formation of One-Dimensional Nano-Organized Polyaniline and Their Raman Fingerprint. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2008, 71, 869–875. [Google Scholar] [CrossRef]
- Bober, P.; Trchová, M.; Prokeš, J.; Varga, M.; Stejskal, J. Polyaniline–Silver Composites Prepared by the Oxidation of Aniline with Silver Nitrate in Solutions of Sulfonic Acids. Electrochim. Acta 2011, 56, 3580–3585. [Google Scholar] [CrossRef]
- Mažeikienė, R.; Niaura, G.; Malinauskas, A. A Comparative Raman Spectroelectrochemical Study of Selected Polyaniline Derivatives in a pH-Neutral Solution. Synth. Met. 2010, 160, 1060–1064. [Google Scholar] [CrossRef]
- Hao, Q.; Lei, W.; Xia, X.; Yan, Z.; Yang, X.; Lu, L.; Wang, X. Exchange of Counter Anions in Electropolymerized Polyaniline Films. Electrochim. Acta 2010, 55, 632–640. [Google Scholar] [CrossRef]
- Abdullah, H.S. Electrochemical Polymerization and Raman Study of Polypyrrole and Polyaniline Thin Films. Int. J. Phys. Sci 2012, 7, 5468–5476. [Google Scholar]
- Liu, R.; Qiu, H.; Zong, H.; Fang, C. Fabrication and Characterization of Composite Containing HCl-Doped Polyaniline and Fe Nanoparticles. J. Nanomater. 2012, 2012, 674104. [Google Scholar] [CrossRef]
- Colonna, P.; Buleon, A.; Merciar, C. Starch: Properties and Potential; Galliard, T., Ed.; Wiley: Hoboken, NJ, USA, 1987. [Google Scholar]
- Doumeng, M.; Makhlouf, L.; Berthet, F.; Marsan, O.; Delbé, K.; Denape, J.; Chabert, F. A Comparative Study of the Crystallinity of Polyetheretherketone by Using Density, DSC, XRD, and Raman Spectroscopy Techniques. Polym. Test. 2021, 93, 106878. [Google Scholar] [CrossRef]
- Wei, Y.; Jang, G.-W.; Hsueh, K.F.; Scherr, E.M.; MacDiarmid, A.G.; Epstein, A.J. Thermal Transitions and Mechanical Properties of Films of Chemically Prepared Polyaniline. Polymer 1992, 33, 314–322. [Google Scholar] [CrossRef]
- Ding, L.; Wang, X.; Gregory, R. Thermal Properties of Chemically Synthesized Polyaniline (EB) Powder. Synth. Met. 1999, 104, 73–78. [Google Scholar] [CrossRef]
- Hejna, A.; Lenża, J.; Formela, K.; Korol, J. Studies on the Combined Impact of Starch Source and Multiple Processing on Selected Properties of Thermoplastic Starch/Ethylene-Vinyl Acetate Blends. J. Polym. Environ. 2019, 27, 1112–1126. [Google Scholar] [CrossRef]
- Song, E.; Choi, J.-W. Conducting Polyaniline Nanowire and Its Applications in Chemiresistive Sensing. Nanomaterials 2013, 3, 498–523. [Google Scholar] [CrossRef]
- Huang, W.-S.; Humphrey, B.D.; MacDiarmid, A.G. Polyaniline, a Novel Conducting Polymer. Morphology and Chemistry of Its Oxidation and Reduction in Aqueous Electrolytes. J. Chem. Soc. Faraday Trans. Phys. Chem. Condens. Phases 1986, 82, 2385–2400. [Google Scholar] [CrossRef]
- Yu, J.; Boudjelida, S.; Galiano, F.; Figoli, A.; Bonchio, M.; Carraro, M. Porous Polymeric Membranes Doped with Halloysite Nanotubes and Oxygenic Polyoxometalates. Adv. Mater. Interfaces 2022, 9, 2102152. [Google Scholar] [CrossRef]
- Russo, F.; Galiano, F.; Pedace, F.; Aricò, F.; Figoli, A. Dimethyl Isosorbide As a Green Solvent for Sustainable Ultrafiltration and Microfiltration Membrane Preparation. ACS Sustain. Chem. Eng. 2020, 8, 659–668. [Google Scholar] [CrossRef]
- Wang, H.H.; Jung, J.T.; Kim, J.F.; Kim, S.; Drioli, E.; Lee, Y.M. A Novel Green Solvent Alternative for Polymeric Membrane Preparation via Nonsolvent-Induced Phase Separation (NIPS). J. Membr. Sci. 2019, 574, 44–54. [Google Scholar] [CrossRef]
- Yang, L.; Wu, S.; Chen, J.P. Modification of Activated Carbon by Polyaniline for Enhanced Adsorption of Aqueous Arsenate. Ind. Eng. Chem. Res. 2007, 46, 2133–2140. [Google Scholar] [CrossRef]
- Sharma, N.; Singh, A.; Kumar, N.; Tiwari, A.; Lal, M.; Arya, S. A review on polyaniline and its composites: From synthesis to properties and progressive applications. J. Mater. Sci. 2024, 59, 6206–6244. [Google Scholar] [CrossRef]
- Gualandi, I.; Tessarolo, M.; Mariani, F.; Possanzini, L.; Scavetta, E.; Fraboni, B. Textile Chemical Sensors Based on Conductive Polymers for the Analysis of Sweat. Polymers 2021, 13, 894. [Google Scholar] [CrossRef]
- Li, Z.; Gong, L. Research Progress on Applications of Polyaniline (PANI) for Electrochemical Energy Storage and Conversion. Materials 2020, 13, 548. [Google Scholar] [CrossRef]




| Aniline: Starch wt ratio | 1:1 | 2:1 | 3:1 |
| Codes | Z11 | Z21 | Z31 |
| PES (wt.%) | Z11 (wt.%) | PVP (wt.%) | PEG (wt.%) | NMP (wt.%) | |
|---|---|---|---|---|---|
| PES-0 | 12 | 0 | 5 | 35 | 48 |
| PES-1 | 12 | 1 | 5 | 35 | 47 |
| Product | 2θ (°) | hkl | d (Å) |
|---|---|---|---|
| PANI-ES | 14.9 | (011) 010 | 5.95 |
| 20.2 | (020) 100 | 4.40 | |
| 25.2 | (200) 111 | 3.53 | |
| Starch | 5.7 | (001) | 15.51 |
| 11.4 | (111) | 7.76 | |
| 15.0 | (140) | 5.89 | |
| 17.3 | (131) | 5.14 | |
| 19.6 | (103) | 4.53 | |
| 22.2 | (113) | 4.00 | |
| 24.1 | (132) | 3.69 | |
| 26.6 | (142) | 3.35 |
| Material | Degree of Crystallinity (%) |
|---|---|
| Starch | 37.0 |
| Z11 | 35.4 |
| Z21 | 33.6 |
| Z31 | 22.8 |
| PANI-ES | 19.6 |
| Material | 1st Peak | 2nd Peak | ||||
|---|---|---|---|---|---|---|
| Peak (°C) | Onset (°C) | ∆H (j/g) | Peak (°C) | Onset (°C) | ∆H (j/g) | |
| Z11 | 95.9 | 42.7 | 234.5 | 207.8 | 186.7 | 103.6 |
| Z21 | 84.4 | 37.8 | 287.3 | 217.6 | 199.9 | 95.2 |
| Z31 | 98.9 | 45.8 | 247.6 | 205.4 | 168.4 | 212.9 |
| PANI-ES | 98.1 | 35.3 | 275.1 | 218.4 | 199.6 | 23.3 |
| 293.5 | 281.2 | 1.1 | ||||
| Starch | 100.0 | 50.5 | 439.7 | 266.4 | 261.7 | 9.1 |
| 278.1 | 273.8 | 15.8 | ||||
| Products | Ox 1 | Ox 2 | Red 1 | Red 2 |
|---|---|---|---|---|
| Z11 | 6.80 | 11.75 | −6.77 | −1.80 |
| Z21 | 3.63 | 2.41 | −0.69 | −0.77 |
| Z31 | 2.30 | 1.47 | −0.47 | −0.64 |
| PANI-ES | 4.11 | 1.50 | −0.37 | −0.69 |
| Z11 Content in Membranes | WCA (Top Surface) (°) | Thickness (µm) | Porosity (%) | Mean Flow Pore Size Diameter (MFD) (µm) |
|---|---|---|---|---|
| 0% | 72 ± 3 | 153 ± 6 | 91 ± 0 | 0.9 |
| 1% | 75 ± 2 | 228 ± 7 | 90 ± 0 | 0.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boudjelida, S.; Li, X.; Djellali, S.; Chiappetta, G.; Russo, F.; Figoli, A.; Carraro, M. Synthesis and Characterization of Polyaniline Emeraldine Salt (PANI-ES) Colloids Using Potato Starch as a Stabilizer to Enhance the Physicochemical Properties and Processability. Materials 2024, 17, 2941. https://doi.org/10.3390/ma17122941
Boudjelida S, Li X, Djellali S, Chiappetta G, Russo F, Figoli A, Carraro M. Synthesis and Characterization of Polyaniline Emeraldine Salt (PANI-ES) Colloids Using Potato Starch as a Stabilizer to Enhance the Physicochemical Properties and Processability. Materials. 2024; 17(12):2941. https://doi.org/10.3390/ma17122941
Chicago/Turabian StyleBoudjelida, Soufiane, Xue Li, Souad Djellali, Giampiero Chiappetta, Francesca Russo, Alberto Figoli, and Mauro Carraro. 2024. "Synthesis and Characterization of Polyaniline Emeraldine Salt (PANI-ES) Colloids Using Potato Starch as a Stabilizer to Enhance the Physicochemical Properties and Processability" Materials 17, no. 12: 2941. https://doi.org/10.3390/ma17122941
APA StyleBoudjelida, S., Li, X., Djellali, S., Chiappetta, G., Russo, F., Figoli, A., & Carraro, M. (2024). Synthesis and Characterization of Polyaniline Emeraldine Salt (PANI-ES) Colloids Using Potato Starch as a Stabilizer to Enhance the Physicochemical Properties and Processability. Materials, 17(12), 2941. https://doi.org/10.3390/ma17122941











